Abstract
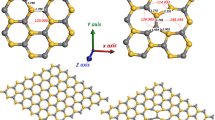
Porous silicon carbide offers a great potential as a sensor material for applications in medicine and energetics; however, the theoretical chemical characterization of its surface is almost nonexistent, and a correct understanding of its chemical properties could lead to the development of better applications of this nanostructure. Hence, a study of the effects of different passivation agents on the structure and electronic properties of porous silicon carbide by means of density functional theory and the supercell technique was developed. The porous structures were modeled by removing columns of atoms of an otherwise perfect SiC crystal in the [001] direction, so that the porous structure exhibits a surface exclusively composed of Si atoms (Si-rich) using different surface passivation agents, such as hydrogen (H), fluoride (F) and chloride (Cl). The results demonstrate that all of the passivation schemes exhibit an irregular band gap energy evolution due to a hybridization change of the surface. The structural analysis shows a great dependence of the bond characteristics on the electronegativity of the bonded atoms, and all of the structural and electronic changes could be explained due to steric effects. These results could be important in the characterization of pSiC because they provide insight into the most stable surface configurations and their electronic structures.





Similar content being viewed by others
References
Garralaga Rojas E, Hensen J, Baur C, Brendel R (2011) Sintering and reorganization of electrochemically etched mesoporous germanium layers in various atmospheres. Sol Energy Mater Sol Cells 95(1):292–295. doi:10.1016/j.solmat.2010.04.042
Lima FAS, Vasconcelos IF, Lira-Cantu M (2015) Electrochemically synthesized mesoporous thin films of ZnO for highly efficient dye sensitized solar cells. Ceram Int 41(8):9314–9320. doi:10.1016/j.ceramint.2015.03.271
Ge M, Fang X, Rong J, Zhou C (2013) Review of porous silicon preparation and its application for lithium-ion battery anodes. Nanotechnology 24(42):422001. doi:10.1088/0957-4484/24/42/422001
Kim M, Oh I, Kim J (2015) Effects of different electrolytes on the electrochemical and dynamic behavior of electric double layer capacitors based on a porous silicon carbide electrode. Phys Chem Chem Phys 17(25):16367–16374. doi:10.1039/C5CP01728A
Lee S-H, Yun S-M, Kim S, Park S-J, Lee Y-S (2010) Characterization of nanoporous β-SiC fiber complex prepared by electrospinning and carbothermal reduction. Res Chem Intermed 36(6–7):731–742. doi:10.1007/s11164-010-0175-9
Naderi N, Hashim MR, Saron KMA, Rouhi J (2013) Enhanced optical performance of electrochemically etched porous silicon carbide. Semicond Sci Technol 28(2):025011. doi:10.1088/0268-1242/28/2/025011
Kim K-S, Chung G-S (2011) Fast response hydrogen sensors based on palladium and platinum/porous 3C-SiC Schottky diodes. Sens Actuators, B 160(1):1232–1236. doi:10.1016/j.snb.2011.09.054
Naderi N, Hashim MR (2013) Visible-blind ultraviolet photodetectors on porous silicon carbide substrates. Mater Res Bull 48(6):2406–2408. doi:10.1016/j.materresbull.2013.02.078
Kim K-S, Chung G-S (2011) Characterization of porous cubic silicon carbide deposited with Pd and Pt nanoparticles as a hydrogen sensor. Sens Actuators, B 157(2):482–487. doi:10.1016/j.snb.2011.05.004
Trejo A, Cruz-Irisson M (2013) Computational modeling of the size effects on the optical vibrational modes of H-terminated Ge nanostructures. Molecules 18(4):4776. doi:10.3390/molecules18044776
Calvino M, Trejo A, Iturrios MI, Crisóstomo MC, Carvajal E, Cruz-Irisson M (2014) DFT study of the electronic structure of cubic-SiC nanopores with a C-terminated surface. Journal of Nanomaterials 2014:7. doi:10.1155/2014/471351
Shin W, Seo W, Takai O, Koumoto K (1998) Surface chemistry of porous silicon carbide. J Electron Mater 27(4):304–307. doi:10.1007/s11664-998-0405-8
Shishkin Y, Choyke WJ, Devaty RP (2004) Photoelectrochemical etching of n-type 4H silicon carbide. J Appl Phys 96(4):2311–2322. doi:10.1063/1.1768612
Fang C, Föll H, Carstensen J (2006) Electrochemical pore etching in germanium. J Electroanal Chem 589(2):259–288. doi:10.1016/j.jelechem.2006.02.021
Miranda A, Trejo A, Canadell E, Rurali R, Cruz-Irisson M (2012) Interconnection effects on the electronic and optical properties of Ge nanostructures: a semi-empirical approach. Physica E 44(7–8):1230–1235. doi:10.1016/j.physe.2012.01.017
Hammer B, Hansen LB, Nørskov JK (1999) Improved adsorption energetics within density-functional theory using revised Perdew–Burke–Ernzerhof functionals. Phys Rev B 59(11):7413–7421. doi:10.1103/PhysRevB.59.7413
Hamann DR, Schlüter M, Chiang C (1979) Norm-conserving pseudopotentials. Phys Rev Lett 43(20):1494–1497. doi:10.1103/PhysRevLett.43.1494
Clark SJ, Segall MD, Pickard CJ, Hasnip PJ, Probert MI, Refson K, Payne MC (2005) First principles methods using CASTEP. Z Kristallogr 220:567–570. doi:10.1524/zkri.220.5.567.65075
Pfrommer BG, Côté M, Louie SG, Cohen ML (1997) Relaxation of crystals with the quasi-Newton method. J Comput Phys 131(1):233–240. doi:10.1006/jcph.1996.5612
Monkhorst HJ, Pack JD (1976) Special points for Brillouin-zone integrations. Phys Rev B 13(12):5188–5192. doi:10.1103/PhysRevB.13.5188
Tossell JA (1987) Quantum mechanical studies of Si–O and Si–F bonds in molecules and minerals. Phys Chem Miner 14(4):320–326. doi:10.1007/BF00309804
Shaik S, Danovich D, Silvi B, Lauvergnat DL, Hiberty PC (2005) Charge-shift bonding—a class of electron-pair bonds that emerges from valence bond theory and is supported by the electron localization function approach. Chem Eur J 11(21):6358–6371. doi:10.1002/chem.200500265
Lommerse JPM, Stone AJ, Taylor R, Allen FH (1996) The nature and geometry of intermolecular interactions between halogens and oxygen or nitrogen. J Am Chem Soc 118(13):3108–3116. doi:10.1021/ja953281x
Gillespie RJ, Robinson EA (2005) Models of molecular geometry. Chem Soc Rev 34(5):396–407. doi:10.1039/B405359C
Wang W, Wong N-B, Zheng W, Tian A (2004) Theoretical study on the blueshifting halogen bond. J Phys Chem A 108(10):1799–1805. doi:10.1021/jp036769q
Lauvergnat D, Hiberty PC, Danovich D, Shaik S (1996) Comparison of C–Cl and Si–Cl bonds. A valence bond study. J Phys Chem 100(14):5715–5720. doi:10.1021/jp960145l
Lambert JB, Wang G-T (1988) Participation of beta carbon-silicon bonds in the development of positive charge in five-membered rings. J Phys Org Chem 1(3):169–178. doi:10.1002/poc.610010307
Acknowledgments
This work was partially supported by Project 252749 from CONACyT and multidisciplinary Projects SIP-IPN 2014-1640 y 2014-1641 from InstitutoPolitécnico Nacional.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published as part of the special collection of articles “CHITEL 2015 - Torino - Italy”.
Rights and permissions
About this article
Cite this article
Calvino, M., Trejo, A., Crisóstomo, M.C. et al. Modeling the effects of Si-X (X = F, Cl) bonds on the chemical and electronic properties of Si-surface terminated porous 3C-SiC. Theor Chem Acc 135, 104 (2016). https://doi.org/10.1007/s00214-016-1861-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-016-1861-5