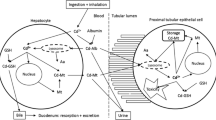
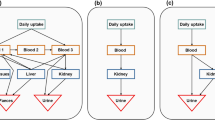
Abstract
In much of the world, currently employed upper limits of tolerable intake and acceptable excretion of cadmium (Cd) (ECd/Ecr) are 0.83 µg/kg body weight/day and 5.24 µg/g creatinine, respectively. These figures were derived from a risk assessment model that interpreted β2-microglobulin (β2MG) excretion > 300 μg/g creatinine as a “critical” endpoint. However, current evidence suggests that Cd accumulation reduces glomerular filtration rate at values of ECd/Ecr much lower than 5.24 µg/g creatinine. Low ECd/Ecr has also been associated with increased risks of kidney disease, type 2 diabetes, osteoporosis, cancer, and other disorders. These associations have cast considerable doubt on conventional guidelines. The goals of this paper are to evaluate whether these guidelines are low enough to minimize associated health risks reliably, and indeed whether permissible intake of a cumulative toxin like Cd is a valid concept. We highlight sources and levels of Cd in the human diet and review absorption, distribution, kidney accumulation, and excretion of the metal. We present evidence for the following propositions: excreted Cd emanates from injured tubular epithelial cells of the kidney; Cd excretion is a manifestation of current tissue injury; reduction of present and future exposure to environmental Cd cannot mitigate injury in progress; and Cd excretion is optimally expressed as a function of creatinine clearance rather than creatinine excretion. We comprehensively review the adverse health effects of Cd and urine and blood Cd levels at which adverse effects have been observed. The cumulative nature of Cd toxicity and the susceptibility of multiple organs to toxicity at low body burdens raise serious doubt that guidelines concerning permissible intake of Cd can be meaningful.




Similar content being viewed by others
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
Abbreviations
- BMD:
-
A benchmark dose is a dose level, derived from an estimated dose-response curve, associated with a specified change in response. The benchmark response can be set at 0.1%, 1%, 5%, or 10% incidence
- BMDL:
-
Benchmark dose limit is a lower confidence limit for the BMD, computed to quantify the statistical variation toward the conservative end of health protection. A BMDL5 can be interpreted as a dose for which a response is likely to be smaller than 5%
- JECFA:
-
Joint Food and Agriculture Organization and World Health Organization (FAO/WHO) expert committee on food additives and contaminants
- NOAEL:
-
No-observed-adverse-effect level is the highest experimental dose level for which the response does not significantly differ from the response in the control group
- PTWI:
-
Provisional tolerable weekly intake is an estimate of the amount of a chemical with no known biological function that can be ingested weekly over a lifetime without appreciable health risk
- RfD:
-
Reference dose is an estimate, with uncertainty of an order of magnitude, of a daily exposure over a lifetime that is likely to be without appreciable risk of adverse effects
- TMI:
-
Tolerable monthly intake
- TDS:
-
Total diet study
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- β2MG:
-
Beta2-microglobulin
- Ca:
-
Calcium
- Cd:
-
Cadmium
- CKD-EPI:
-
Chronic kidney disease epidemiology collaboration
- CdMT:
-
Cadmium–metallothionein complex
- CdPC:
-
Cadmium–phytochelatin complex
- Co:
-
Cobalt
- Cu:
-
Copper
- DMT1:
-
Divalent metal transporter 1
- eGFR:
-
Estimated glomerular filtration rate, units of mL/min/1.73 m2
- GSH:
-
Glutathione
- KIM1:
-
Kidney injury molecule 1
- MCU:
-
Mitochondrial calcium uniporter
- mtDNA:
-
Mitochondrial DNA
- Mn:
-
Manganese
- MT:
-
Metallothionein
- NAFLD:
-
Non-alcoholic fatty liver disease
- NAG:
-
N-acetyl-β-D-glucosaminidase
- NASH:
-
Non-alcoholic steatohepatitis
- NGAL:
-
Neutrophil gelatinase-associated lipocalin
- OCTs:
-
Organic cation transporters
- PC:
-
Phytochelatin
- TfR1:
-
Transferrin receptor protein 1
- TRVP5:
-
Transient receptor potential vanilloid 5
- Zn:
-
Zinc
- ZIP8:
-
Zrt- and Irt-like protein 8
- ZIP14:
-
Zrt- and Irt-like protein 14
- Ccr :
-
Creatinine clearance, units of volume/time
- Ex/Ccr :
-
Excretion rate of x per volume of filtrate, units of mass/volume
- [x]u :
-
Urinary concentration of x
- [x]p :
-
Plasma concentration of x
- [x]b :
-
Blood concentration of x
- Vu :
-
Urine flow rate, units of volume/time
References
Adams SV, Passarelli MN, Newcomb PA (2012) Cadmium exposure and cancer mortality in the third national health and nutrition examination survey cohort. Occup Environ Med 69(2):153–156
Adams SV, Barrick B, Christopher EP et al (2015) Genetic variation in metallothionein and metal-regulatory transcription factor 1 in relation to urinary cadmium, copper, and zinc. Toxicol Appl Pharmacol 289(3):381–388
Ajjimaporn A, Botsford T, Garrett SH et al (2012) ZIP8 expression in human proximal tubule cells, human urothelial cells transformed by Cd+2 and As+3 and in specimens of normal human urothelium and urothelial cancer. Cancer Cell Int 12(1):16
Akerstrom M, Barregard L, Lundh T et al (2013) The relationship between cadmium in kidney and cadmium in urine and blood in an environmentally exposed population. Toxicol Appl Pharmacol 268(3):286–293
Allen JL, Oberdorster G, Morris-Schaffer K et al (2017) Developmental neurotoxicity of inhaled ambient ultrafine particle air pollution: parallels with neuropathological and behavioral features of autism and other neurodevelopmental disorders. Neurotoxicology 59:140–154
Amegah AK, Sewor C, Jaakkola JJK (2021) Cadmium exposure and risk of adverse pregnancy and birth outcomes: a systematic review and dose-response meta-analysis of cohort and cohort-based case-control studies. J Expo Sci Environ Epidemiol 31(2):299–317
Aoshima K (1987) Epidemiology of renal tubular dysfunction in the inhabitants of a cadmium-polluted area in the Jinzu River basin in Toyama Prefecture. Tohoku J Exp Med 152(2):151–172
Aramjoo H, Arab-Zozani M, Feyzi A et al (2022) The association between environmental cadmium exposure, blood pressure, and hypertension: a systematic review and meta-analysis. Environ Sci Pollut Res Int. https://doi.org/10.1007/s11356-021-17777-9
Asefi Y, Mahmoudabad AG, Sezavar AH et al (2022) Association between maternal cadmium exposure and preterm birth: a meta-analysis. Int J Environ Health Res 32(3):628–637
Bao QJ, Zhao K, Guo Y et al (2022) Environmental toxic metal contaminants and risk of stroke: a systematic review and meta-analysis. Environ Sci Pollut Res Int. https://doi.org/10.1007/s11356-022-18866-z
Barregard L, Fabricius-Lagging E, Lundh T et al (2010) Cadmium, mercury, and lead in kidney cortex of living kidney donors: impact of different exposure sources. Environ Res 110(1):47–54
Barregard L, Sallsten G, Lundh T et al (2022) Low-level exposure to lead, cadmium and mercury, and histopathological findings in kidney biopsies. Environ Res 211:113119
Benbrahim-Tallaa L, Tokar EJ, Diwan BA et al (2009) Cadmium malignantly transforms normal human breast epithelial cells into a basal-like phenotype. Environ Health Perspect 117(12):1847–1852
Bendell LI (2010) Cadmium in shellfish: the British Columbia, Canada experience–a mini-review. Toxicol Lett 198(1):7–12
Bernard A, Goret A, Buchet JP et al (1980) Significance of cadmium levels in blood and urine during long-term exposure of rats to cadmium. J Toxicol Environ Health 6(1):175–184
Bernard A, Thielemans N, Roels H et al (1995) Association between NAG-B and cadmium in urine with no evidence of a threshold. Occup Environ Med 52(3):177–180
Boonprasert K, Ruengweerayut R, Aunpad R et al (2012) Expression of metallothionein isoforms in peripheral blood leukocytes from Thai population residing in cadmium-contaminated areas. Environ Toxicol Pharmacol 34(3):935–940
Boonprasert K, Satarug S, Morais C et al (2016) The stress response of human proximal tubule cells to cadmium involves up-regulation of haemoxygenase 1 and metallothionein but not cytochrome P450 enzymes. Toxicol Lett 249:5–14
Branca JJV, Maresca M, Morucci G et al (2019) Effects of cadmium on ZO-1 tight junction integrity of the blood brain barrier. Int J Mol Sci 20(23):6010
Branca JJV, Fiorillo C, Carrino D et al (2020) Cadmium-induced oxidative stress: focus on the central nervous system. Antioxidants (basel) 9(6):492
Buha A, Đukić-Ćosić D, Ćurčić M et al (2020) Emerging links between cadmium exposure and insulin resistance: human, animal, and cell study data. Toxics 8(3):63
Buser MC, Ingber SZ, Raines N et al (2016) Urinary and blood cadmium and lead and kidney function: NHANES 2007–2012. Int J Hyg Environ Health 219(3):261–267
Calafat AM (2012) The U.S. national health and nutrition examination survey and human exposure to environmental chemicals. Int J Hyg Environ Health 215(2):99–101
Callan A, Hinwood A, Devine A (2014) Metals in commonly eaten groceries in Western Australia: a market basket survey and dietary assessment. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 31(12):1968–1981
Camaschella C (2015) Iron-deficiency anemia. N Engl J Med 372(19):1832–1843
Capel ID, Pinnock MH, Dorrell HM et al (1981) Comparison of concentrations of some trace, bulk, and toxic metals in the hair of normal and dyslexic children. Clin Chem 27(6):879–881
Chan HM, Tamura Y, Cherian MG et al (1993a) Pregnancy-associated changes in plasma metallothionein concentration and renal cadmium accumulation in rats. Proc Soc Exp Biol Med 202(4):420–427
Chan HM, Zhu LF, Zhong R et al (1993b) Nephrotoxicity in rats following liver transplantation from cadmium-exposed rats. Toxicol Appl Pharmacol 123(1):89–96
Chen Z, Myers R, Wei T et al (2014) Placental transfer and concentrations of cadmium, mercury, lead, and selenium in mothers, newborns, and young children. J Expo Sci Environ Epidemiol 24(5):537–544
Chen C, Xun P, Nishijo M et al (2015) Cadmium exposure and risk of pancreatic cancer: a meta-analysis of prospective cohort studies and case-control studies among individuals without occupational exposure history. Environ Sci Pollut Res Int 22(22):17465–17474
Chen C, Xun P, Nishijo M et al (2016a) Cadmium exposure and risk of lung cancer: a meta-analysis of cohort and case-control studies among general and occupational populations. J Expo Sci Environ Epidemiol 26(5):437–444
Chen C, Xun P, Nishijo M et al (2016b) Cadmium exposure and risk of prostate cancer: a meta-analysis of cohort and case-control studies among the general and occupational populations. Sci Rep 6:25814
Chen L, Sun Q, Peng S et al (2022) Associations of blood and urinary heavy metals with rheumatoid arthritis risk among adults in NHANES, 1999-2018. Chemosphere 289:133147
Cheng X, Niu Y, Ding Q et al (2016) Cadmium exposure and risk of any fracture: a PRISMA-compliant systematic review and meta-analysis. Medicine (baltimore) 95(10):e2932
Choi YH, Hu H, Mukherjee B et al (2012) Environmental cadmium and lead exposures and hearing loss in U.S. adults: the National Health and Nutrition Examination Survey, 1999 to 2004. Environ Health Perspect 120(11):1544–1550
Choudhury H, Harvey T, Thayer WC et al (2001) Urinary cadmium elimination as a biomarker of exposure for evaluating a cadmium dietary exposure–biokinetics model. J Toxicol Environ Health A 63(5):321–350
Chowdhury R, Ramond A, O’Keeffe LM et al (2018) Environmental toxic metal contaminants and risk of cardiovascular disease: systematic review and meta-analysis. BMJ 362:k3310
Christensen EI, Birn H, Storm T et al (2012) Endocytic receptors in the renal proximal tubule. Physiology (bethesda) 27(4):223–236
Chung S, Chung JH, Kim SJ et al (2014) Blood lead and cadmium levels and renal function in Korean adults. Clin Exp Nephrol 18(5):726–734
Ciarimboli G, Ludwig T, Lang D et al (2005) Cisplatin nephrotoxicity is critically mediated via the human organic cation transporter 2. Am J Pathol 167(6):1477–1484
Ciesielski T, Weuve J, Bellinger DC et al (2012) Cadmium exposure and neurodevelopmental outcomes in U.S. children. Environ Health Perspect 120(5):758–763
Ciesielski T, Bellinger DC, Schwartz J et al (2013) Associations between cadmium exposure and neurocognitive test scores in a cross-sectional study of US adults. Environ Health 12:13
Cirovic A, Cirovic A (2021) Iron deficiency as promoter of heavy metals-induced acute myeloid leukemia. Leuk Res 112:106755
Codex Alimentarius (2014) CODEX STAN 193–1995, general standard for contaminants and toxins in food and feed. Available at http://www.fao.org/fileadmin/user_upload/livestockgov/documents/1_CXS_193e.pdf
Copes R, Clark NA, Rideout K et al (2008) Uptake of cadmium from Pacific oysters (Crassostrea gigas) in British Columbia oyster growers. Environ Res 107(2):160–169
Costa LG, Aschner M, Vitalone A et al (2004) Developmental neuropathology of environmental agents. Annu Rev Pharmacol Toxicol 44:87–110
Crinnion WJ (2010) The CDC fourth national report on human exposure to environmental chemicals: what it tells us about our toxic burden and how it assists environmental medicine physicians. Altern Med Rev 15(2):101–109
Da Cunha RS, Azevedo CAB, Falconi CA et al (2022) The interplay between uremic toxins and albumin, membrane transporters and drug interaction. Toxins (basel) 14(3):177
De Nicola L, Zoccali C (2016) Chronic kidney disease prevalence in the general population: heterogeneity and concerns. Nephrol Dial Transplant 31(3):331–335
DelRaso NJ, Foy BD, Gearhart JM et al (2003) Cadmium uptake kinetics in rat hepatocytes: correction for albumin binding. Toxicol Sci 72(1):19–30
Diamond GL, Thayer WC, Choudhury H (2003) Pharmacokinetics/pharmacodynamics (PK/PD) modeling of risks of kidney toxicity from exposure to cadmium: estimates of dietary risks in the U.S. population. J Toxicol Environ Health A 66(22):2141–2164
Edwards JR, Prozialeck WC (2009) Cadmium, diabetes and chronic kidney disease. Toxicol Appl Pharmacol 238(3):289–293
EFSA (2009) European food safety agency, cadmium in food - scientific opinion of the panel on contaminants in the food chain. EFSA J 7(3):980
EFSA (2011) European food safety agency, statement on tolerable weekly intake for cadmium. EFSA J 9(2):1975
Egan SK, Bolger PM, Clark Dewitt Carrington CD (2007) Update of US FDA’s total diet study food list and diets. J Expo Sci Environ Epidemiol 17(6):573–582
Elinder CG, Lind B, Kjellstorm T et al (1976) Cadmium in kidney cortex, liver and pancreas from Swedish autopsies: estimation of biological half time in kidney cortex, considering calorie intake and smoking habits. Arch Environ Health 31(6):292–302
Elinder CG, Kjellstorm T, Lind B et al (1978) Cadmium concentrations in human liver, blood, and bile: comparison with a metabolic model. Environ Res 17(2):236–241
Faroon O, Ashizawa A, Wright S et al (2012) Toxicological profile for cadmium. Agency for Toxic Substances and Disease Registry, Atlanta, GA, USA
Fels J, Scharner B, Zarbock R et al (2019) Cadmium complexed with β2-microglubulin, albumin and lipocalin-2 rather than metallothionein cause megalin:cubilin dependent toxicity of the renal proximal tubule. Int J Mol Sci 20(10):2379
Feng CX, Cao J, Bendell L (2011) Exploring spatial and temporal variations of cadmium concentrations in pacific oysters from British Columbia. Biometrics 67(3):1142–1152
Feng X, Zhou R, Jiang Q et al (2022) Analysis of cadmium accumulation in community adults and its correlation with low-grade albuminuria. Sci Total Environ 834:155210
Ferraro PM, Costanzi S, Naticchia A et al (2010) Low level exposure to cadmium increases the risk of chronic kidney disease: analysis of the NHANES 1999–2006. BMC Public Health 10:304
Filippini T, Wise LA, Vinceti M (2022) Cadmium exposure and risk of diabetes and prediabetes: a systematic review and dose-response meta-analysis. Environ Int 158:106920
Finley JW (1999) Manganese absorption and retention by young women is associated with serum ferritin concentration. Am J Clin Nutr 70(1):37–43
Fitzgerald R, Olsen A, Nguyen J et al (2020) Pancreatic islets accumulate cadmium in a rodent model of cadmium-induced hyperglycemia. Int J Mol Sci 22(1):360
Flanagan PR, McLellan JS, Haist J et al (1978) Increased dietary cadmium absorption in mice and human subjects with iron deficiency. Gastroenterology 74(5 Pt 1):841–846
Flannery BM, Schaefer HR, Middleton KB (2022) A scoping review of infant and children health effects associated with cadmium exposure. Regul Toxicol Pharmacol 131:105155
Fleming RE, Bacon BR (2005) Orchestration of iron homeostasis. N Engl J Med 352(17):1741–1744
Fransson MN, Barregard L, Sallsten G et al (2014) Physiologically-based toxicokinetic model for cadmium using Markov-chain Monte Carlo analysis of concentrations in blood, urine, and kidney cortex from living kidney donors. Toxicol Sci 141(2):365–376
Fujishiro H, Himeno S (2019) New insights into the roles of ZIP8, a cadmium and manganese transporter, and its relation to human diseases. Biol Pharm Bull 42(7):1076–1082
Fujishiro H, Hamao S, Tanaka R et al (2017) Concentration-dependent roles of DMT1 and ZIP14 in cadmium absorption in Caco-2 cells. J Toxicol Sci 42(5):559–567
Fujita Y, ElBelbasi HI, Min KS et al (1993) Fate of cadmium bound to phytochelatin in rats. Res Commun Chem Pathol Pharmacol 82(3):357–365
Gallagher CM, Kovach JS, Meliker JR (2008) Urinary cadmium and osteoporosis in U.S. Women >or= 50 years of age: NHANES 1988–1994 and 1999–2004. Environ Health Perspect 116(10):1338–1343
Gallagher CM, Chen JJ, Kovach JS (2010) Environmental cadmium and breast cancer risk. Aging (albany NY) 2(11):804–814
Garner R, Levallois P (2016) Cadmium levels and sources of exposure among Canadian adults. Health Rep 27(2):10–18
Garrett SH, Sens MA, Todd JH et al (1999) Expression of MT-3 protein in the human kidney. Toxicol Lett 105(3):207–214
Gasser M, Lenglet S, Bararpour N et al (2022) Cadmium acute exposure induces metabolic and transcriptomic perturbations in human mature adipocytes. Toxicology 470:153153
Ghasemi A (2021) Uric acid-induced pancreatic β-cell dysfunction. BMC Endocr Disord 21(1):24
Glassock RJ, Warnock DG, Delanaye P (2017) The global burden of chronic kidney disease: estimates, variability and pitfalls. Nat Rev Nephrol 13(2):104–114
Gobe G, Crane D (2010) Mitochondria, reactive oxygen species and cadmium toxicity in the kidney. Toxicol Lett 198(1):49–55
Gonzalez-Nahm S, Nihlani K, House JS et al (2020) Associations between maternal cadmium exposure with risk of preterm birth and low birth weight: effect of Mediterranean diet adherence on affected prenatal outcomes. Toxics 8(4):90
Grau-Perez M, Pichler G, Galan-Chilet I et al (2017) Urine cadmium levels and albuminuria in a general population from Spain: a gene-environment interaction analysis. Environ Int 106:27–36
Guo S, Frazer DM, Anderson GJ (2016) Iron homeostasis: transport, metabolism, and regulation. Curr Opin Clin Nutr Metab Care 19(4):276–281
Guo ZL, Wang JY, Gong LL et al (2018) Association between cadmium exposure and urolithiasis risk: a systematic review and meta-analysis. Medicine (baltimore) 97(1):e9460
Guo FF, Hu ZY, Li BY et al (2019) Evaluation of the association between urinary cadmium levels below threshold limits and the risk of diabetes mellitus: a dose-response meta-analysis. Environ Sci Pollut Res Int 26(19):19272–19281
Gustin K, Tofail F, Vahter M et al (2018) Cadmium exposure and cognitive abilities and behavior at 10 years of age: a prospective cohort study. Environ Int 113:259–268
Haswell-Elkins M, McGrath V, Moore M et al (2007a) Exploring potential dietary contributions including traditional seafood and other determinants of urinary cadmium levels among indigenous women of a Torres Strait Island (Australia). J Expo Sci Environ Epidemiol 17(3):298–306
Haswell-Elkins M, Imray P, Satarug S et al (2007b) Urinary excretion of cadmium among Torres Strait Islanders (Australia) at risk of elevated dietary exposure through traditional foods. J Expo Sci Environ Epidemiol 17(4):372–377
Haswell-Elkins M, Satarug S, O’Rourke P et al (2008) Striking association between urinary cadmium level and albuminuria among Torres Strait Islander people with diabetes. Environ Res 106(3):379–383
Hauptman M, Woolf AD (2017) Childhood ingestions of environmental toxins: what are the risks? Pediatr Ann 46(12):e466–e471
Hecht EM, Arheart KL, Lee DJ et al (2016) Interrelation of cadmium, smoking, and cardiovascular disease (from the National Health and Nutrition Examination Survey). Am J Cardiol 118(2):204–209
Hecht EM, Arheart KL, Lee DJ, Hennekens CH, Hlaing WM (2019) Interrelationships of cadmium, smoking, and angina in the national health and nutrition examination survey, a cross-sectional study. Cardiology 141(4):177–182
Hennigar SR, Kelley AM, McClung JP (2016) Metallothionein and zinc transporter expression in circulating human blood cells as biomarkers of zinc status: a systematic review. Adv Nutr 7(4):735–746
Heymsfield SB, Arteaga C, McManus C et al (1983) Measurement of muscle mass in humans: validity of the 24-hour urinary creatinine method. Am J Clin Nutr 37(3):478–494
Hong D, Min JY, Min KB (2021) Association between cadmium exposure and liver function in adults in the United States: a cross-sectional study. J Prev Med Public Health 54(6):471–480
Horiguchi H, Oguma E, Sasaki S et al (2004) Comprehensive study of the effects of age, iron deficiency, diabetes mellitus, and cadmium burden on dietary cadmium absorption in cadmium-exposed female Japanese farmers. Toxicol Appl Pharmacol 196(1):114–123
Horiguchi H, Aoshima K, Oguma E (2010) Latest status of cadmium accumulation and its effects on kidneys, bone, and erythropoiesis in inhabitants of the formerly cadmium-polluted Jinzu River Basin in Toyama, Japan, after restoration of rice paddies. Int Arch Occup Environ Health 83(8):953–970
Horiguchi H, Oguma E, Sasaki S et al (2020) Exposure assessment of cadmium in female farmers in cadmium-polluted areas in Northern Japan. Toxics 8(2):44
Huang C-H, Wang C-W, Chen H-C et al (2022) Gender difference in the associations among heavy metals with red blood cell hemogram. Int J Environ Res Public Health 19(1):189
Hübner C, Haase H (2021) Interactions of zinc- and redox-signaling pathways. Redox Biol 41:101916
Huff J, Lunn RM, Waalkes MP et al (2007) Cadmium-induced cancers in animals and in humans. Int J Occup Environ Health 13(2):202–212
Hyder O, Chung M, Cosgrove D et al (2013) Cadmium exposure and liver disease among US adults. J Gastrointest Surg 17(7):1265–1273
IARC (1993) Cadmium and cadmium compounds. IARC Monogr Eval Carcinog Risks Hum 58:119–238
Ibrahimou B, Sun N, Azim SI et al (2021) Interaction between chronic bronchitis and blood cadmium levels on the prevalence of myocardial infarction in US adults. J Occup Environ Med 63(12):1087–1092
Ishizaki M, Suwazono Y, Kido T et al (2015) Estimation of biological half-life of urinary cadmium in inhabitants after cessation of environmental cadmium pollution using a mixed linear model. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 32(8):1273–1276
Jacobo-Estrada T, Santoyo-Sánchez M, Thévenod F et al (2017) Cadmium handling, toxicity and molecular targets involved during pregnancy: lessons from experimental models. Int J Mol Sci 18(7):1590
Jacquet A, Cottet-Rousselle C, Arnaud J et al (2018) Mitochondrial morphology and function of the pancreatic β-cells INS-1 model upon chronic exposure to sub-lethal cadmium doses. Toxics 6(2):20
Jalili C, Kazemi M, Cheng H et al (2021) Associations between exposure to heavy metals and the risk of chronic kidney disease: a systematic review and meta-analysis. Crit Rev Toxicol 51(2):165–182
Järup L, Rogenfelt A, Elinder CG et al (1983) Biological half-time of cadmium in the blood of workers after cessation of exposure. Scand J Work Environ Health 9(4):327–331
JECFA (2010) Joint FAO/WHO Expert committee on food additives, seventy-third meeting, Geneva, 8–17 June 2010. Summary and conclusions. JECFA/73/SC. Geneva, Switzerland: Food and Agriculture Organization of the United Nations; World Health Organization. Available at http://www.who.int/foodsafety/publications/chem/summary73.pdf
Jin R, Zhu X, Shrubsole MJ et al (2018) Associations of renal function with urinary excretion of metals: evidence from NHANES 2003–2012. Environ Int 121(Pt 2):1355–1362
Jing Y, Liu LZ, Jiang Y et al (2012) Cadmium increases HIF-1 and VEGF expression through ROS, ERK, and AKT signaling pathways and induces malignant transformation of human bronchial epithelial cells. Toxicol Sci 125(1):10–19
Jomova K, Valko M (2011) Advances in metal-induced oxidative stress and human disease. Toxicol 283(2–3):65–87
Jorge-Nebert LF, Gálvez-Peralta M, Figueroa JL et al (2015) Comparing gene expression during cadmium uptake and distribution: untreated versus oral Cd-treated wild-type and ZIP14 knockout mice. Toxicol Sci 143(1):26–35
Jouybari L, Saei Ghare Naz M, Sanagoo A et al (2018) Toxic elements as biomarkers for breast cancer: a meta-analysis study. Cancer Manag Res 10:69–79
Jung W, Kim Y, Lihm H et al (2019) Associations between blood lead, cadmium, and mercury levels with hyperuricemia in the Korean general population: a retrospective analysis of population-based nationally representative data. Int J Rheum Dis 22(8):1435–1444
Kalantar-Zadeh K, Jafar TH, Nitsch D et al (2021) Chronic kidney disease. Lancet 398(10302):786–802
Kang MY, Cho SH, Lim YH et al (2013) Effects of environmental cadmium exposure on liver function in adults. Occup Environ Med 70(4):268–273
Kawada T, Shinmyo RR, Suzuki S (1992) Urinary cadmium and N-acetyl-beta-D-glucosaminidase excretion of inhabitants living in a cadmium-polluted area. Int Arch Occup Environ Health 63(8):541–546
Kikuchi Y, Nomiyama T, Kumagai N et al (2003) Uptake of cadmium in meals from the digestive tract of young non-smoking Japanese female volunteers. J Occup Health 45(1):43–52
Kim MH, Zhao D, Cho J et al (2016) Cadmium exposure and age-related macular degeneration. J Expo Sci Environ Epidemiol 26(2):214–218
Kim K, Melough MM, Vance TM et al (2018) Dietary cadmium intake and sources in the US. Nutrients 11(1):2
Kim H, Lee J, Woo HD et al (2019) Association between dietary cadmium intake and early gastric cancer risk in a Korean population: a case-control study. Eur J Nutr 58(8):3255–3266
Kim DW, Ock J, Moon K-W et al (2021) Association between Pb, Cd, and Hg exposure and liver injury among Korean adults. Int J Environ Res Public Health 18(13):6783
Kippler M, Lönnerdal B, Goessler W et al (2009) Cadmium interacts with the transport of essential micronutrients in the mammary gland - a study in rural Bangladeshi women. Toxicology 257(1–2):64–69
Kippler M, Hoque AM, Raqib R et al (2010) Accumulation of cadmium in human placenta interacts with the transport of micronutrients to the fetus. Toxicol Lett 192(2):162–168
Kjellström T, Nordberg GF (1978) A kinetic model of cadmium metabolism in the human being. Environ Res 16(1–3):248–269
Knez M, Graham RD, Welch RM et al (2017) New perspectives on the regulation of iron absorption via cellular zinc concentrations in humans. Crit Rev Food Sci Nutr 57(10):2128–2143
Kondaiah P, Yaduvanshi PS, Sharp PA et al (2019) Iron and zinc homeostasis and interactions: does enteric zinc excretion cross-talk with intestinal iron absorption? Nutrients 11(8):1885
Kovacs G, Danko T, Bergeron MJ et al (2011) Heavy metal cations permeate the TRPV6 epithelial cation channel. Cell Calcium 49(1):43–55
Kovacs G, Montalbetti N, Franz MC et al (2013) Human TRPV5 and TRPV6: key players in cadmium and zinc toxicity. Cell Calcium 54(4):276–286
Koyama H, Satoh H, Suzuki S et al (1992) Increased urinary cadmium excretion and its relationship to urinary N-acetyl-beta-D-glucosaminidase activity in smokers. Arch Toxicol 66(8):598–601
Krężel A, Maret W (2017) The functions of metamorphic metallothioneins in zinc and copper metabolism. Int J Mol Sci 18(6):1237
Krężel A, Maret W (2021) The bioinorganic chemistry of mammalian metallothioneins. Chem Rev 121(23):14594–14648
Krueger WS, Wade TJ (2016) Elevated blood lead and cadmium levels associated with chronic infections among non-smokers in a cross-sectional analysis of NHANES data. Environ Health 15:16
Kruzynski GM (2004) Cadmium in oysters and scallops: the BC experience. Toxicol Lett 148(3):159–169
Langelueddecke C, Roussa E, Fenton RA et al (2012) Lipocalin-2 (24p3/neutrophil gelatinase-associated lipocalin (NGAL) receptor is expressed in distal nephron and mediates protein endocytosis. J Biol Chem 287(1):159–169
Langelueddecke C, Roussa E, Fenton RA et al (2013) Expression and function of the lipocalin-2 (24p3/NGAL) receptor in rodent and human intestinal epithelia. PLoS ONE 8(8):e71586
Langelueddecke C, Lee WK, Thevenod F (2014) Differential transcytosis and toxicity of the hNGAL receptor ligands cadmium-metallothionein and cadmium-phytochelatin in colon-like Caco-2 cells: implications for cadmium toxicity. Toxicol Lett 226(2):228–235
Larsson SC, Wolk A (2016) Urinary cadmium and mortality from all causes, cancer and cardiovascular disease in the general population: systematic review and meta-analysis of cohort studies. Int J Epidemiol 45(3):782–791
Larsson SC, Orsini N, Wolk A (2015) Urinary cadmium concentration and risk of breast cancer: a systematic review and dose-response meta-analysis. Am J Epidemiol 182(5):375–380
Leconte S, Rousselle C, Bodin L et al (2021) Refinement of health-based guidance values for cadmium in the French population based on modelling. Toxicol Lett 340:43–51
Lee WK, Thévenod F (2020) Cell organelles as targets of mammalian cadmium toxicity. Arch Toxicol 94(4):1017–1049
Lee J, Oh S, Kang H et al (2020) Environment-wide association study of CKD. Clin J Am Soc Nephrol 15(6):766–775
Levey AS, Inker LA, Matsushita K et al (2014) GFR decline as an end point for clinical trials in CKD: a scientific workshop sponsored by the national kidney foundation and the US food and drug administration. Am J Kidney Dis 64(6):821–835
Li H, Wang Z, Fu Z et al (2018) Associations between blood cadmium levels and cognitive function in a cross-sectional study of US adults aged 60 years or older. BMJ Open 8(4):e020533
Li D, Lin H, Zhang M et al (2021) Urine cadmium as a risk factor for osteoporosis and osteopenia: a meta-analysis. Front Med (lausanne) 8:648902
Lim JA, Kwon HJ, Ha M et al (2015) Korean research project on the integrated exposure assessment of hazardous substances for food safety. Environ Health Toxicol 30:e2015004
Lin YS, Caffrey JL, Lin JW et al (2013) Increased risk of cancer mortality associated with cadmium exposures in older Americans with low zinc intake. J Toxicol Environ Health A 76(1):1–15
Lin YS, Ho WC, Caffrey JL et al (2014) Low serum zinc is associated with elevated risk of cadmium nephrotoxicity. Environ Res 134:33–38
Lin J, Zhang F, Lei Y (2016) Dietary intake and urinary level of cadmium and breast cancer risk: a meta-analysis. Cancer Epidemiol 42:101–107
Liu J, Qu W, Kadiiska MB (2009) Role of oxidative stress in cadmium toxicity and carcinogenesis. Toxicol Appl Pharmacol 238(3):209–214
Liu Z, Cai L, Liu Y et al (2019) Association between prenatal cadmium exposure and cognitive development of offspring: a systematic review. Environ Pollut 254(Pt B):113081
Lou M, Garay R, Alda JO (1991) Cadmium uptake through the anion exchanger in human red blood cells. J Physiol 443:123–136
Lu J, He Y, Cui L et al (2020) Hyperuricemia predisposes to the onset of diabetes via promoting pancreatic β-cell death in uricase-deficient male mice. Diabetes 69(6):1149–1163
Ma C, Iwai-Shimada M, Nakayama SF et al (2021) Association of prenatal exposure to cadmium with neurodevelopment in children at 2 years of age: the Japan environment and children’s study. Environ Int 156:106762
Madrigal JM, Ricardo AC, Persky V et al (2019) Associations between blood cadmium concentration and kidney function in the U.S. population: impact of sex, diabetes and hypertension. Environ Res 169:180–188
Marín S, Pardo O, Báguena R et al (2017) Dietary exposure to trace elements and health risk assessment in the region of Valencia, Spain: a total diet study. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 34(2):228–240
Martins AC, Almeida Lopes ACB, Urbano MR et al (2021) An updated systematic review on the association between Cd exposure, blood pressure and hypertension. Ecotoxicol Environ Saf 208:111636
McElroy JA, Hunter MI (2019) Cadmium: a new risk factor for endometrial cancer? Expert Rev Anticancer Ther 19(5):355–358
Meltzer HM, Brantsaeter AL, Borch-Iohnsen B et al (2010) Low iron stores are related to higher blood concentrations of manganese, cobalt and cadmium in non-smoking, Norwegian women in the HUNT 2 study. Environ Res 110(5):497–504
Menke A, Muntner P, Silbergeld EK et al (2009) Cadmium levels in urine and mortality among U.S. adults. Environ Health Perspect 117(2):190–196
Min KB, Lee KJ, Park JB et al (2012) Lead and cadmium levels and balance and vestibular dysfunction among adult participants in the national health and nutrition examination survey (NHANES) 1999–2004. Environ Health Perspect 120(3):413–417
Misra RR, Hochadel JF, Smith GT (1996) Evidence that nitric oxide enhances cadmium toxicity by displacing the metal from metallothionein. Chem Res Toxicol 9(1):326–332
Moffett DB, Mumtaz MM, Sullivan DW Jr et al (2022) Chapter 13, general considerations of dose-effect and dose-response relationships. In: Nordberg G, Costa M (eds) Handbook on the toxicology of metals (Fifth Edition), volume I: general considerations. Elsevier, pp 299–317
Mortensen ME, Wong LY, Osterloh JD (2011) Smoking status and urine cadmium above levels associated with subclinical renal effects in U.S. adults without chronic kidney disease. Int J Hyg Environ Health 214(4):305–310
Moulis JM, Nahoui-Zarouri I, Lénon M et al (2021) Low-level cadmium doses do not jeopardize the insulin secretion pathway of β-cell models until the onset of cell death. J Trace Elem Med Biol 68:126834
Myong J-P, Kim H-R, Baker D et al (2012) Blood cadmium and moderate-to-severe glomerular dysfunction in Korean adults: analysis of KNHANES 2005–2008 data. Int Arch Occup Environ Health 85(8):885–893
Navar LG, Maddox DA, Munger KA (2020) Chapter 3, the renal circulations and glomerular filtration. Brenner and Rector’s the kidney, 11th edn. Elsevier, Philadelphia, USA, pp 80–114
Navas-Acien A, Tellez-Plaza M, Guallar E et al (2009) Blood cadmium and lead and chronic kidney disease in US adults: a joint analysis. Am J Epidemiol 170(9):1156–1164
Nawrot TS, Martens DS, Hara A et al (2015) Association of total cancer and lung cancer with environmental exposure to cadmium: the meta-analytical evidence. Cancer Causes Control 26(9):1281–1288
Nguyen HD, Oh H, Hoang NHM et al (2022) Environmental science and pollution research role of heavy metal concentrations and vitamin intake from food in depression: a national cross-sectional study (2009–2017). Environ Sci Pollut Res Int 29(3):4574–4586
Nielsen R, Christensen EI, Birn H (2016) Megalin and cubilin in proximal tubule protein reabsorption: from experimental models to human disease. Kidney Int 89(1):58–67
Nishijo M, Nogawa K, Suwazono Y et al (2020) Lifetime cadmium exposure and mortality for renal diseases in residents of the cadmium-polluted Kakehashi River Basin in Japan. Toxics 8(4):81
Nishito Y, Kambe T (2018) Absorption mechanisms of iron, copper, and zinc: an overview. J Nutr Sci Vitaminol (tokyo) 64(1):1–7
Nogawa K, Sakurai M, Ishizaki M et al (2017) Threshold limit values of the cadmium concentration in rice in the development of itai-itai disease using benchmark dose analysis. J Appl Toxicol 37(8):962–966
Nogawa K, Suwazono Y, Nishijo M et al (2018a) Increase of lifetime cadmium intake dose-dependently increased all cause of mortality in female inhabitants of the cadmium-polluted Jinzu River Basin, Toyama, Japan. Environ Res 164:379–384
Nogawa K, Suwazono Y, Nishijo M et al (2018b) Relationship between mortality and rice cadmium concentration in inhabitants of the polluted Jinzu River basin, Toyama, Japan: a 26-year follow-up. J Appl Toxicol 38(6):855–861
Nomiyama K, Foulkes EC (1977) Reabsorption of filtered cadmium-metallothionein in the rabbit kidney. Proc Soc Exp Biol Med 156(1):97–99
Olsén L, Lind PM, Lind L (2012) Gender differences for associations between circulating levels of metals and coronary risk in the elderly. Int J Hyg Environ Health 215(3):411–417
Olszowski T, Baranowska-Bosiacka I, Rębacz-Maron E et al (2016) Cadmium concentration in mother’s blood, milk, and newborn’s blood and its correlation with fatty acids, anthropometric characteristics, and mother’s smoking status. Biol Trace Elem Res 174(1):8–20
Onodera A, Tani M, Michigami T et al (2012) Role of megalin and the soluble form of its ligand RAP in Cd-metallothionein endocytosis and Cd-metallothionein-induced nephrotoxicity in vivo. Toxicol Lett 212(2):91–96
Ougier E, Fiore K, Rousselle C et al (2021) Burden of osteoporosis and costs associated with human biomonitored cadmium exposure in three European countries: France, Spain and Belgium. Int J Hyg Environ Health 234:113747
Park J, Kim Y (2021) Associations of blood heavy metals with uric acid in the Korean general population: analysis of data from the 2016–2017 Korean national health and nutrition examination survey. Biol Trace Elem Res 199(1):102–112
Park E, Kim J, Kim B et al (2021) Association between environmental exposure to cadmium and risk of suspected non-alcoholic fatty liver disease. Chemosphere 266:128947
Peng Q, Bakulski KM, Nan B et al (2017) Cadmium and Alzheimer’s disease mortality in U.S. adults: updated evidence with a urinary biomarker and extended follow-up time. Environ Res 157:44–51
Peng Y, Li Z, Yang X et al (2020) Relation between cadmium body burden and cognitive function in older men: a cross-sectional study in China. Chemosphere 250:126535
Pennemans V, De Winter LM, Elke Munters E et al (2011) The association between urinary kidney injury molecule 1 and urinary cadmium in elderly during long-term, low-dose cadmium exposure: a pilot study. Environ Health 10:77
Petering DH (2017) Reactions of the Zn proteome with Cd2+ and other xenobiotics: trafficking and toxicity. Chem Res Toxicol 30(1):189–202
Petering DH, Mahim A (2017) Proteomic high affinity Zn (2+) trafficking: where does metallothionein fit in? Int J Mol Sci 18(6):1289
Phelps KR, Gosmanova EO (2020) A generic method for analysis of plasma concentrations. Clin Nephrol 94(1):43–49
Phelps KR, Stote KS, Mason D (2014) Use of sevelamer to examine the role of intraluminal phosphate in the pathogenesis of secondary hyperparathyroidism. Clin Nephrol 82(3):191–201
Pouillot R, Santillana Farakos S, Van Doren JM (2022) Modeling the risk of low bone mass and osteoporosis as a function of urinary cadmium in U.S adults aged 50–79 years. Environ Res 212(Pt B):113315
Price RG (1992) The role of NAG (N-acetyl-β-D-glucosaminidase) in the diagnosis of kidney disease including the monitoring of nephrotoxicity. Clin Nephrol 38(Suppl 1):S14–S19
Prozialeck WC, Edwards JR (2010) Early biomarkers of cadmium exposure and nephrotoxicity. Biometals 23(5):793–809
Prozialeck WC, Vaidya VS, Liu J et al (2007) Kidney injury molecule-1 is an early biomarker of cadmium nephrotoxicity. Kidney Int 72(8):985–993
Prozialeck WC, Edwards JR, Vaidya VS et al (2009) Preclinical evaluation of novel urinary biomarkers of cadmium nephrotoxicity. Toxicol Appl Pharmacol 238(3):301–305
Qu W, Tokar EJ, Kim AJ et al (2012) Chronic cadmium exposure in vitro causes acquisition of multiple tumor cell characteristics in human pancreatic epithelial cells. Environ Health Perspect 120(9):1265–1271
Quraishi SM, Adams SV, Shafer M et al (2016) Urinary cadmium and estimated dietary cadmium in the women’s health initiative. J Expo Sci Environ Epidemiol 26(3):303–308
Rahman HH, Niemann D, Munson-McGee SH (2021) Association of albumin to creatinine ratio with urinary arsenic and metal exposure: evidence from NHANES 2015–2016. Int Urol Nephrol. https://doi.org/10.1007/s11255-021-03018-y
Rai A, Maurya SK, Khare P et al (2010) Characterization of developmental neurotoxicity of As, Cd, and Pb mixture: synergistic action of metal mixture in glial and neuronal functions. Toxicol Sci 118(2):586–601
Rebelo FM, Caldas ED (2016) Arsenic, lead, mercury and cadmium: toxicity, levels in breast milk and the risks for breastfed infants. Environ Res 151:671–688
Rezapour M, Rezapour HA, Chegeni M et al (2021) Exposure to cadmium and head and neck cancers: a meta-analysis of observational studies. Rev Environ Health 36(4):577–584
Rocca A, Fanchon E, Moulis, J-M (2020) Theoretical modeling of oral glucose tolerance tests guides the interpretation of the impact of perinatal cadmium exposure on the offspring's glucose homeostasis. Toxics 8(2):30
Roels HA, Lauwerys RR, Buchet JP et al (1989) Health significance of cadmium induced renal dysfunction: a five year follow up. Br J Ind Med 46(11):755–764
Rokadia HK, Agarwal S (2013) Serum heavy metals and obstructive lung disease: results from the national health and nutrition examination survey. Chest 143(2):388–397
Ruangyuttikarn W, Panyamoon A, Nambunmee K et al (2013) Use of the kidney injury molecule-1 as a biomarker for early detection of renal tubular dysfunction in a population chronically exposed to cadmium in the environment. Springerplus 17(2):533
Rubino FM (2015) Toxicity of glutathione-binding metals: a review of targets and mechanisms. Toxics 3(1):20–62
Ruczaj A, Brzóska MM (2022) Environmental exposure of the general population to cadmium as a risk factor of the damage to the nervous system: a critical review of current data. J Appl Toxicol. https://doi.org/10.1002/jat.4322
Ruiz P, Fowler BA, Osterloh JD et al (2010a) Physiologically based pharmacokinetic (PBPK) tool kit for environmental pollutants–metals. SAR QSAR Environ Res 21(7–8):603–618
Ruiz P, Mumtaz M, Osterloh J et al (2010b) Interpreting NHANES biomonitoring data, cadmium. Toxicol Lett 198(1):44–48
Ruiz P, Ray M, Fisher J et al (2011) Development of a human physiologically based pharmacokinetic (PBPK) toolkit for environmental pollutants. Int J Mol Sci 12(11):7469–7480
Sabolić I, Breljak D, Skarica M et al (2010) Role of metallothionein in cadmium traffic and toxicity in kidneys and other mammalian organs. Biometals 23(5):897–926
Sabolić I, Skarica M, Ljubojevic M et al (2018) Expression and immunolocalization of metallothioneins MT1, MT2 and MT3 in rat nephron. J Trace Elem Med Biol 46:62–75
Sakamoto M, Chan HM, Domingo JL et al (2012) Changes in body burden of mercury, lead, arsenic, cadmium and selenium in infants during early lactation in comparison with placental transfer. Ecotoxicol Environ Saf 84:179–184
Salcedo-Bellido I, Gómez-Peña C, Pérez-Carrascosa FM et al (2021) Adipose tissue cadmium concentrations as a potential risk factor for insulin resistance and future type 2 diabetes mellitus in GraMo adult cohort. Sci Total Environ 780:146359
Sarmiento-Ortega VE, Moroni-González D, Díaz A et al (2021) Oral subacute exposure to cadmium LOAEL dose induces insulin resistance and impairment of the hormonal and metabolic liver-adipose axis in Wistar rats. Biol Trace Elem Res. https://doi.org/10.1007/s12011-021-03027-z
Sasaki N, Carpenter DO (2022) Associations between metal exposures and cognitive function in American older adults. Int J Environ Res Public Health 19(4):2327
Satarug S (2018) Dietary cadmium intake and its effects on kidneys. Toxics 6(1):15
Satarug S, Moore MR (2012) Emerging roles of cadmium and heme oxygenase in type-2 diabetes and cancer susceptibility. Tohoku J Exp Med 228(4):267–288
Satarug S, Phelps KR (2021) Chapter 14, cadmium exposure and toxicity. In: Bagchi M, Bagchi D (eds) Metal toxicology. CRC Press
Satarug S, Baker JR, Reilly PEB et al (2000a) Evidence for a synergistic interaction between cadmium and endotoxin toxicity and for nitric oxide and cadmium displacement of metals in the kidney. Nitric Oxide 4(4):431–440
Satarug S, Haswell-Elkins MR, Moore MR (2000b) Safe levels of cadmium intake to prevent renal toxicity in human subjects. Br J Nutr 84(6):791–802
Satarug S, Baker JR, Reilly PE et al (2001) Changes in zinc and copper homeostasis in human livers and kidneys associated with exposure to environmental cadmium. Hum Exp Toxicol 20(4):205–213
Satarug S, Baker JR, Reilly PE et al (2002) Cadmium levels in the lung, liver, kidney cortex, and urine samples from Australians without occupational exposure to metals. Arch Environ Health 57(1):69–77
Satarug S, Baker JR, Urbenjapol S et al (2003) A global perspective on cadmium pollution and toxicity in non-occupationally exposed population. Toxicol Lett 137(1–2):65–83
Satarug S, Ujjin P, Vanavanitkun Y et al (2004) Influence of body iron store status and cigarette smoking on cadmium body burden of healthy Thai women and men. Toxicol Lett 148(3):177–185
Satarug S, Garrett SH, Sens MA et al (2010) Cadmium, environmental exposure, and health outcomes. Environ Health Perspect 118(2):182–190
Satarug S, Swaddiwudhipong W, Ruangyuttikarn W et al (2013) Modeling cadmium exposures in low- and high-exposure areas in Thailand. Environ Health Perspect 121(5):531–536
Satarug S, Vesey DA, Gobe GC (2017a) Health risk assessment of dietary cadmium intake: do current guidelines indicate how much is safe? Environ Health Perspect 125(3):284–288
Satarug S, Vesey DA, Gobe GC (2017b) Current health risk assessment practice for dietary cadmium: data from different countries. Food Chem Toxicol 106(Pt A):430–445
Satarug S, Vesey DA, Ruangyuttikarn W et al (2019) The source and pathophysiologic significance of excreted cadmium. Toxics 7(4):55
Satarug S, Gobe CG, Vesey DA et al (2020) Cadmium and lead exposure, nephrotoxicity, and mortality. Toxics 8(4):86
Satarug S, Garrett SH, Somji S et al (2021a) Aberrant expression of ZIP and ZnT zinc transporters in UROtsa cells transformed malignant cells by cadmium. Stresses 1(2):78–89
Satarug S, Vesey DA, Gobe GC (2021b) The evolving role for zinc and zinc transporters in cadmium tolerance and urothelial cancer. Stresses 1(2):105–118
Satarug S, Vesey DA, Nishijo M et al (2021c) The effect of cadmium on GFR is clarified by normalization of excretion rates to creatinine clearance. Int J Mol Sci 22(4):1762
Satarug S, Đorđević AB, Yimthiang S et al (2022) The NOAEL equivalent of environmental cadmium exposure associated with GFR reduction and chronic kidney disease. Toxics 10(10):614
Satir S (2022) The relationship between oral cancer and cadmium: a review. Mol Biol Rep 49(3):2413–2419
Schildroth S, Friedman A, Bauer JA et al (2022) Associations of a metal mixture with iron status in U.S. adolescents: evidence from the national health and nutrition examination survey. New Dir Child Adolesc Dev. https://doi.org/10.1002/cad.20457
Schneider SN, Liu Z, Wang B et al (2014) Oral cadmium in mice carrying 5 versus 2 copies of the Slc39a8 gene: comparison of uptake, distribution, metal content, and toxicity. Int J Toxicol 33(1):14–20
Schwartz GG, Il’yasova D, Ivanova A (2003) Urinary cadmium, impaired fasting glucose, and diabetes in the NHANES III. Diabetes Care 26(2):468–470
Schwarz MA, Lindtner O, Blume K et al (2014) Cadmium exposure from food: the German LExUKon project. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 31(6):1038–1051
Scinicariello F, Buser MC (2015) Blood cadmium and depressive symptoms in young adults (aged 20–39 years). Psychol Med 45(4):807–815
Scinicariello F, Abadin HG, Murray HE (2011) Association of low-level blood lead and blood pressure in NHANES 1999–2006. Environ Res 111(8):1249–1257
Sens DA, Park S, Gurel V et al (2004) Inorganic cadmium- and arsenite-induced malignant transformation of human bladder urothelial cells. Toxicol Sci 79(1):56–63
Shaikh ZA, Hirayama K (1979) Metallothionein in the extracellular fluids as an index of cadmium toxicity. Environ Health Perspect 28:267–271
Shargorodsky J, Curhan SG, Henderson E et al (2011) Heavy metals exposure and hearing loss in US adolescents. Arch Otolaryngol Head Neck Surg 137(12):1183–1189
Shi P, Yan H, Fan X et al (2021) A benchmark dose analysis for urinary cadmium and type 2 diabetes mellitus. Environ Pollut 273:116519
Silver MK, Lozoff B, Meeker JD (2013) Blood cadmium is elevated in iron deficient U.S. children: a cross-sectional study. Environ Health 12:117
Smith CP, Lee WK, Haley M et al (2019) Proximal tubule transferrin uptake is modulated by cellular iron and mediated by apical membrane megalin-cubilin complex and transferrin receptor 1. J Biol Chem 294(17):7025–7036
Song JK, Luo H, Yin XH et al (2015) Association between cadmium exposure and renal cancer risk: a meta-analysis of observational studies. Sci Rep 5:17976
Soodvilai S, Nantavishit J, Muanprasat C et al (2011) Renal organic cation transporters mediated cadmium-induced nephrotoxicity. Toxicol Lett 204(1):38–42
Suh YJ, Lee JE, Lee DH et al (2016) Prevalence and relationships of iron deficiency anemia with blood cadmium and vitamin D levels in Korean women. J Korean Med Sci 31(1):25–32
Sun H, Wang D, Zhou Z et al (2016) Association of cadmium in urine and blood with age in a general population with low environmental exposure. Chemosphere 156:392–397
Sun H, Wang N, Chen C et al (2017) Cadmium exposure and its association with serum uric acid and hyperuricemia. Sci Rep 7(1):550
Sunderman FW Jr (2001) Nasal toxicity, carcinogenicity, and olfactory uptake of metals. Ann Clin Lab Sci 31(1):3–24
Suomi J, Valsta L, Tuominen P (2021) Dietary heavy metal exposure among Finnish adults in 2007 and in 2012. Int J Environ Res Public Health 18(20):10581
Suwazono Y, Kido T, Nakagawa H et al (2009) Biological half-life of cadmium in the urine of inhabitants after cessation of cadmium exposure. Biomarkers 14(2):77–81
Świetlik R, Trojanowska M (2022) Chemical fractionation in environmental studies of potentially toxic particulate-bound elements in urban air: a critical review. Toxics 10(3):124
Taylor CM, Golding J, Emond AM (2014) Lead, cadmium and mercury levels in pregnancy: the need for international consensus on levels of concern. J Dev Orig Health Dis 5(1):16–30
Tellez-Plaza M, Navas-Acien A, Crainiceanu CM et al (2010) Cadmium and peripheral arterial disease: gender differences in the 1999–2004 US national health and nutrition examination survey. Am J Epidemiol 172(6):671–681
Tellez-Plaza M, Navas-Acien A, Caldwell KL et al (2012a) Reduction in cadmium exposure in the United States population, 1988–2008: the contribution of declining smoking rates. Environ Health Perspect 120(2):204–209
Tellez-Plaza M, Navas-Acien A, Menke A et al (2012b) Cadmium exposure and all-cause and cardiovascular mortality in the U.S. general population. Environ Health Perspect 120(7):1017–1022
Thatcher RW, Lester ML, McAlaster R et al (1982) Effects of low levels of cadmium and lead on cognitive functioning in children. Arch Environ Health 37(3):159–166
Thatcher RW, McAlaster R, Lester ML (1984) Evoked potentials related to hair cadmium and lead in children. Ann N Y Acad Sci 425:384–390
Thévenod F, Fels J, Lee WK et al (2019) Channels, transporters and receptors for cadmium and cadmium complexes in eukaryotic cells: myths and facts. Biometals 32(3):469–489
Thévenod F, Lee WK, Garrick MD (2020) Iron and cadmium entry into renal mitochondria: physiological and toxicological implications. Front Cell Dev Biol 8:848
Thomas LD, Hodgson S, Nieuwenhuijsen M et al (2009) Early kidney damage in a population exposed to cadmium and other heavy metals. Environ Health Perspect 117(2):181–184
Tian X, Xue B, Wang B et al (2022) Physical activity reduces the role of blood cadmium on depression: a cross-sectional analysis with NHANES data. Environ Pollut 304:119211
Treviño S, Waalkes MP, Flores Hernández JA et al (2015) Chronic cadmium exposure in rats produces pancreatic impairment and insulin resistance in multiple peripheral tissues. Arch Biochem Biophys 583:27–35
Tsai HJ, Hung CH, Wang CW et al (2021a) Associations among heavy metals and proteinuria and chronic kidney disease. Diagnostics (basel) 11(2):282
Tsai K-F, Hsu P-C, Lee C-T et al (2021b) Association between enzyme-linked immunosorbent assay-measured kidney injury markers and urinary cadmium levels in chronic kidney disease. J Clin Med 11(1):156
Tsukahara T, Ezaki T, Moriguchi J et al (2003) Rice as the most influential source of cadmium intake among general Japanese population. Sci Total Environ 305:41–51
Vacchi-Suzzi C, Kruse D, Harrington J et al (2016) Is urinary cadmium a biomarker of long-term exposure in humans? A review. Curr Environ Health Rep 3:450–458
Valko M, Jomova K, Rhodes CJ et al (2016) Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Arch Toxicol 90(1):1–37
Van Maele-Fabry G, Lombaert N, Lison D (2016) Dietary exposure to cadmium and risk of breast cancer in postmenopausal women: a systematic review and meta-analysis. Environ Int 86:1–13
Vašák M, Meloni G (2017) Mammalian metallothionein-3: new functional and structural insights. Int J Mol Sci 18(6):1117
Vogt R, Bennett D, Cassady D (2012) Cancer and non-cancer health effects from food contaminant exposures for children and adults in California: a risk assessment. Environ Health 11:83
Waalkes MP (2003) Cadmium carcinogenesis. Mutat Res 533(1–2):107–120
Waalkes MP, Rehm S (1994) Chronic toxic and carcinogenic effects of cadmium chloride in male DBA/2NCr and NFS/NCr mice: strain-dependent association with tumors of the hematopoietic system, injection site, liver, and lung. Fundam Appl Toxicol 23(1):21–31
Wallia A, Allen NB, Badon S et al (2014) Association between urinary cadmium levels and prediabetes in the NHANES 2005–2010 population. Int J Hyg Environ Health 217(8):854–860
Wang D, Sun H, Wu Y et al (2016) Tubular and glomerular kidney effects in the Chinese general population with low environmental cadmium exposure. Chemosphere 147:3–8
Wang H, Zhang L, Abel GM et al (2018) Cadmium exposure impairs cognition and olfactory memory in male C57BL/6 mice. Toxicol Sci 161(1):87–102
Watanabe T, Kataoka Y, Hayashi K et al (2022) Dietary exposure of the Japanese general population to elements: total diet study 2013–2018. Food Saf (tokyo) 10(3):83–101
Weaver VM, Kim NS, Jaar BG et al (2011) Associations of low-level urine cadmium with kidney function in lead workers. Occup Environ Med 68(4):250–256
Wei J, Gao J, Cen K (2019) Levels of eight heavy metals and health risk assessment considering food consumption by China’s residents based on the 5th China total diet study. Sci Total Environ 689:1141–1148
Wolf C, Strenziok R, Kyriakopoulos A (2009) Elevated metallothionein-bound cadmium concentrations in urine from bladder carcinoma patients, investigated by size exclusion chromatography-inductively coupled plasma mass spectrometry. Anal Chim Acta 631(2):218–222
Wong C, Roberts SM, Saab IN (2022) Review of regulatory reference values and background levels for heavy metals in the human diet. Regul Toxicol Pharmacol 130:105122
Wu EW, Schaumberg DA, Park SK (2014) Environmental cadmium and lead exposures and age-related macular degeneration in U.S. adults: the national health and nutrition examination survey 2005 to 2008. Environ Res 133:178–184
Xiao G, Liu Y, Dong KF et al (2020) Regional characteristics of cadmium intake in adult residents from the 4th and 5th Chinese total diet study. Environ Sci Pollut Res Int 27(4):3850–3857
Xiao L, Li W, Zhu C et al (2021) Cadmium exposure, fasting blood glucose changes, and type 2 diabetes mellitus: a longitudinal prospective study in China. Environ Res 192:110259
Xu Z, Weng Z, Liang J et al (2022) Association between urinary cadmium concentrations and liver function in adolescents. Environ Sci Pollut Res Int 29(26):39768–39776
Yan L-J, Allen DC (2021) Cadmium-induced kidney injury: oxidative damage as a unifying mechanism. Biomolecules 11(11):1575
Yang H, Shu Y (2015) Cadmium transporters in the kidney and cadmium-induced nephrotoxicity. Int J Mol Sci 16(1):1484–1494
Yimthiang S, Pouyfung P, Khamphaya T et al (2022) Effects of environmental exposure to cadmium and lead on the risks of diabetes and kidney dysfunction. Int J Environ Res Public Health 19(4):2259
Yorita Christensen KL (2013) Metals in blood and urine, and thyroid function among adults in the United States 2007–2008. Int J Hyg Environ Health 216(6):624–632
Zalups RK (2000) Evidence for basolateral uptake of cadmium in the kidneys of rats. Toxicol Appl Pharmacol 164(1):15–23
Zavala-Guevara IP, Ortega-Romero MS, Narváez-Morales J et al (2021) Increased endocytosis of cadmium-metallothionein through the 24p3 receptor in an in vivo model with reduced proximal tubular activity. Int J Mol Sci 22(14):7262
Zeng A, Li S, Zhou Y et al (2022) Association between low-level blood cadmium exposure and hyperuricemia in the American general population: a cross-sectional study. Biol Trace Elem Res 200(2):560–567
Zhang L, Zhu Y, Hao R et al (2016) Cadmium levels in tissue and plasma as a risk factor for prostate carcinoma: a meta-analysis. Biol Trace Elem Res 172(1):86–92
Zhu XJ, Wang JJ, Mao JH et al (2019) Relationships of cadmium, lead, and mercury levels with albuminuria in US adults: results from the national health and nutrition examination survey database, 2009–2012. Am J Epidemiol 188(7):1281–1287
Acknowledgements
This work was supported with resources of the Kidney Disease Research Collaborative, and the Department of Nephrology, Princess Alexandra Hospital. It was also supported with resources of the Stratton Veterans Affairs Medical Center, Albany, NY, USA. Opinions expressed herein are those of the authors and do not represent the official position of the United States Department of Veterans Affairs.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no actual or potential competing financial interests.
Additional information
This review is dedicated to the late Professor Michael R. Moore, who was Director of the National Research Centre for Environmental Toxicology (EnTox), University of Queensland, between 1994 and 2009. He was instrumental in establishing toxicology research on heavy metals in Australia, and he was an inspiration to all who worked in this field.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Satarug, S., Vesey, D.A., Gobe, G.C. et al. Estimation of health risks associated with dietary cadmium exposure. Arch Toxicol 97, 329–358 (2023). https://doi.org/10.1007/s00204-022-03432-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-022-03432-w