Abstract
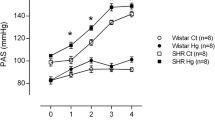
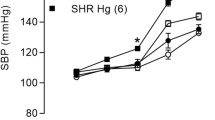
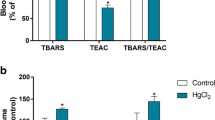
Increased risk of hypertension after methylmercury (MeHg) exposure has been suggested. However, the underlying mechanisms are not well explored. In this paper, we have analyzed whether sub-chronic exposure to MeHg increases systolic blood pressure even at very low levels. In addition, we analyzed if the methylmercury-induced hypertension is associated with a decreased plasmatic nitric oxide levels and with a dysregulation of the activities of the antioxidant enzymes superoxide dismutase (SOD) and catalase (CAT), as well as the levels of MDA and glutathione. For this study, Wistar rats were treated with methylmercury chloride (100 μg/kg per day) or vehicle. Total treatment time was 100 days. Malondialdehyde (MDA) and circulating NOx levels and superoxide dismutase (SOD) and catalase (CAT) activities were determined in plasma, whereas glutathione levels were determined in erythrocytes. Our results show that long-term treatment at a low level of MeHg affected systolic blood pressure, increasing and reducing the levels of plasmatic MDA and NOx, respectively. However, the activity of SOD did not decrease in the MeHg exposed group when compared to the control. We found a negative correlation between plasmatic nitrite/nitrate (NOx) levels and systolic blood pressure (r = −0.67; P = 0.001), and a positive correlation between MDA and systolic blood pressure (r = 0.61; P = 0.03), thus suggesting increased inhibition of NO formation with the increase of hypertension. In conclusion, long-term exposure to a low dose of MeHg increases the systolic pressure and is associated, at least in part, with increased production of ROS as judged by increased production of malondialdehyde and depressed NO availability.




Similar content being viewed by others
References
Abdel-Hamid HA, Fahmy FC, Sharaf IA (2001) Influence of free radicals on cardiovascular risk due to occupational exposure to mercury. J Egypt Public Health Assoc 76:53–69
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. doi:10.1016/S0076-6879(84)05016-3
Ali SF, LeBel CP, Bondy SC (1992) Reactive oxygen species formation as a biomarker of methylmercury and trimethyltin neurotoxicity. Neurotoxicology 13:637–648
Ariza ME, Bijur GN, Williams MV (1998) Lead and mercury metagenesis: role of H2O2, superoxide dismutase, and xanthine oxidase. Environ Mol Mutagen 31:352–361. doi:10.1002/(SICI)1098-2280(1998)31:4<352::AID-EM8>3.0.CO;2-K
Aschner M, Yao CP, Allen JW, Tan KH (2000) Methylmercury alters glutamate transport in astrocytes. Neurochem Int 37:199–206. doi:10.1016/S0197-0186(00)00023-1
Aschner M, Syversen T, Souza DO, Rocha JBT, Farina M (2007) Involvement of glutamate and reactive oxygen species in methylmercury neurotoxicity. Braz J Med Biol Res 40:285–291. doi:10.1590/S0100-879X2007000300001
Barbosa F, Sertorio JTC, Gerlach RF, Tanus-Santos JE (2006a) Clinical evidence for lead-induced inhibition of nitric oxide formation. Arch Toxicol 80:811–816. doi:10.1007/s00204-006-0111-3
Barbosa F, Sandrim VC, Uzuelli JA, Gerlach RF, Tanus-Santos JE (2006b) eNOS genotype-dependent correlation between whole blood lead and plasma nitric oxide products concentrations. Nitric Oxide 14:58–64. doi:10.1016/j.niox.2005.09.007
Carvalho MC, Franco JL, Ghizonia H, Kobusa K, Nazaria EM, Rocha JBT, Nogueira CW, Dafred AL, Müllera YMR, Farina M (2007) Effects of 2,3-dimercapto-1-propanesulfonic acid (DMPS) on methylmercury-induced locomotor deficits and cerebellar toxicity in mice. Toxicology 239:195–203. doi:10.1016/j.tox.2007.07.009
Castoldi AF, Coccini T, Manzo L (2003) Neurotoxic and molecular effects of methylmercury in humans. Rev Environ Health 18:19–31
Cengel A, Sahinarslan A (2006) Nitric oxide and cardiovascular system. Anadolu Kardiyol Derg 6:364–368
Chisolm GM, Steinberg D (2000) The oxidative modification hypothesis of atherogenesis: an overview. Free Radic Biol Med 28:1815–1826. doi:10.1016/S0891-5849(00)00344-0
Clarkson TW (1997) The toxicology of mercury. Crit Rev Clin Lab Sci 34:369–403. doi:10.3109/10408369708998098
Clarkson TW (2002) The three modern faces of mercury. Environ Health Perspect 110:11–23
Clarkson TW, Magos L, Myers GJ (2003) The toxicology of mercury current exposures and clinical manifestations. N Engl J Med 349:1731–1737. doi:10.1056/NEJMra022471
Dietrich MO, Mantese CE, dos Anjos G, Souza DO, Farina M (2005) Motor impairment induced by oral exposure to methylmercury in adult mice. Environ Toxicol Pharmacol 19:169–175. doi:10.1016/j.etap.2004.07.004
Dutczak WJ, Ballatori N (1994) Transport of the glutathione–methylmercury complex across liver canalicular membranes on reduced glutathione carriers. J Biol Chem 269:9746–9751
Ehara S, Ueda M, Naruko T, Haze K, Itoh A, Otsuka M, Komatsu R, Matsuo T, Itabe H, Takano T, Tsukamoto Y, Yoshiyama M, Takeuchi K, Yoshikawa J, Becker AE (2001) Elevated levels of oxidized low density lipoprotein show a positive relationship with the severity of acute coronary syndromes. Circulation 103:1955–1960
Ellis G, Adatia I, Yazdanpanah M, Makela SK (1998) Nitrite and nitrate analyses: a clinical biochemistry perspective. Clin Biochem 31:195–220. doi:10.1016/S0009-9120(98)00015-0
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77. doi:10.1016/0003-9861(59)90090-6
Esterbauer H, Schaur RJ, Zollner H (1991) Chemistry and biochemistry of 4-hydroxynoneal malondialdehyde and related aldehydes. Free Radic Biol Med 11:81–128. doi:10.1016/0891-5849(91)90192-6
Fadini PS, Jardim WF (2001) Is the Negro River Basin (Amazon) impacted by naturally occurring mercury? Sci Total Environ 275:71–82. doi:10.1016/S0048-9697(00)00855-X
Fillion M, Mergler D, Passos CJS, Larribe F, Lemire M, Guimarães JRD (2006) A preliminary study of mercury exposure and blood pressure in the Brazilian Amazon. Environ Health 5:29–37. doi:10.1186/1476-069X-5-29
Grotto D, Santa Maria LD, Boeira S, Valentini J, Charão MF, Moro AM, Nascimento PC, Pomblum VJ, Garcia SC (2007) Rapid quantification of malondialdehyde in plasma by high performance liquid chromatography–visible detection. J Pharm Biomed Anal 43:619–624. doi:10.1016/j.jpba.2006.07.030
Grotto D, Barcelos GRM, Valentini J, Antunes MGA, Angeli JPA, Garcia SC, Barbosa F (2009) Low levels of methylmercury induce DNA damage in rats: protective effects of selenium. Arch Toxicol 83:249–254. doi:10.1007/s00204-008-0353-3
Guallar E, Sanz-Gallardo MI, van’t Veer P, Bode P, Aro A, Gómez-Aracena J, Kark JD, Riemersma RA, Martín-Moreno JM, Kok FJ (2002) Mercury, fish oils, and the risk of myocardial infarction. N Engl J Med 347:1747–1754. doi:10.1056/NEJMoa020157
Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97:1634–1658. doi:10.1111/j.1471-4159.2006.03907.x
Halliwell B, Chirico S (1993) Lipid peroxidation: its mechanism, measurement, and significance. Am J Clin Nutr 57:715–725
Halliwell B, Gutteridge JMC (eds) (1999) Free radicals biology and medicine. Science Publications, Oxford
Harada M (1995) Minamata disease: methylmercury poisoning in Japan caused by environmental pollution. Crit Rev Toxicol 25:1–24. doi:10.3109/10408449509089885
Heinecke JW (1998) Oxidants and antioxidants in the pathogenesis of atherosclerosis: implications for the oxidized low density lipoprotein hypothesis. Atherosclerosis 141:1–15. doi:10.1016/S0021-9150(98)00173-7
Hibbs JB, Westenfelder C Jr, Taintor R, Vavrin Z, Kablitz C, Baranowski RL, Ward JH, Menlove RL, McMurry MP, Kushner JP (1992) Evidence for cytokine-inducible nitric oxide synthesis from l-arginine in patients receiving interleukin-2 therapy. J Clin Invest 89:867–877. doi:10.1172/JCI115666
Houston MC (2007) The role of mercury and cadmium heavy metals in vascular disease, hypertension, coronary heart disease, and myocardial infarction. Altern Ther Health Med 13:128–133
Huang YL, Cheng SL, Lin TH (1996) Lipid peroxidation in rats administrated with mercuric chloride. Biol Trace Elem Res 52:193–206. doi:10.1007/BF02789461
Hunter D, Russell DL (1954) Focal cerebral and cerebellar atrophy in a human subject due to organic mercury compounds. J Neurol Neurosurg Psychiatry 17:235–241. doi:10.1136/jnnp.17.4.235
Hussain S, Atkinson A, Thompson SJ, Khan AT (1999) Accumulation of mercury and its effect on antioxidant enzymes in brain, liver, and kidneys of mice. J Environ Sci Health B 34:645–660. doi:10.1080/03601239909373219
Jina X, Loka E, Bondya G, Caldwella D, Muellera R, Kapala K, Cheryl A, Taylora M, Kubowc S, Mehtaa R, Chan HM (2007) Modulating effects of dietary fats on methylmercury toxicity and distribution in rats. Toxicology 230:22–44. doi:10.1016/j.tox.2006.10.023
Kelm M, Preik-Steinhoff H, Preik M, Strauer BE (1999) Serum nitrite sensitively reflects endothelial NO formation in human forearm vasculature: evidence for biochemical assessment of the endothelial l-arginine–NO pathway. Cardiovasc Res 41:765–772. doi:10.1016/S0008-6363(98)00259-4
Kim SH, Sharma RP (2004) Mercury-induced apoptosis and necrosis in murine macrophages: role of calcium-induced reactive oxygen species and p38 mitogen-activated protein kinase signaling. Toxicol Appl Pharmacol 196:47–57. doi:10.1016/j.taap.2003.11.020
Kishimoto T, Oguri T, Tada M (1995) Effect of methylmercury (CH3HgCl) injury on nitric oxide synthase (NOS) activity in cultured human umbilical vascular endothelial cells. Toxicology 103:1–7. doi:10.1016/0300-483X(95)99370-R
Kleinbongard P, Dejam A, Lauer T, Rassaf T, Schindler A, Picker O, Scheeren T, Godecke A, Schrader J, Schulz R, Heusch G, Schaub GA, Bryan NS, Feelisch M, Kelm M (2003) Plasma nitrite reflects constitutive nitric oxide synthase activity in mammals. Free Radic Biol Med 35:790–796. doi:10.1016/S0891-5849(03)00406-4
Kono Y, Fridovich I (1982) Superoxide radical inhibits catalase. J Biol Chem 257:5751–5754
Lauer T, Preik M, Rassaf T, Strauer BE, Deussen A, Feelisch M, Kelm M (2001) Plasma nitrite rather than nitrate reflects regional endothelial nitric oxide synthase activity but lacks intrinsic vasodilator action. Proc Natl Acad Sci USA 98:12814–12819. doi:10.1073/pnas.221381098
Lauer T, Kleinbongard P, Kelm M (2002) Indexes of NO bioavailability in human blood. News Physiol Sci 17:251–255
Li Y, Yan XP, Chen C, Xia YL, Jiang Y (2007) Human serum albumin–mercurial species interactions. J Proteome Res 6:2277–2286. doi:10.1021/pr0700403
Lund BO, Miler DM, Woods JS (1991) Mercury-induced H2O2 formation and lipid peroxidation in vitro in rat kidney mitochondria. Biochem Pharmacol 42:181–187. doi:10.1016/0006-2952(91)90408-W
Mahboob M, Shireen KF, Atkinson A, Khan AT (2001) Lipid peroxidation and antioxidant enzyme activity in different organs of mice exposed level of mercury. J Environ Sci Health B 36:687–697. doi:10.1081/PFC-100106195
Meister A (1983) Selective modification of glutathione metabolism. Science 220:472–477. doi:10.1126/science.6836290
Miller DM, Woods JS (1993) Urinary porphyrins as biological indicators of oxidative stress in the kidney. Interaction of mercury and cephaloridine. Biochem Pharmacol 46:2235–2241. doi:10.1016/0006-2952(93)90614-3
NRC (National Research Council) (2000) Toxicological effects of methylmercury. National Academy Press, Washington, DC
Osterud B, Bjorklid E (2003) Role of monocytes in atherogenesis. Physiol Rev 83:1069–1112
Park EJ, Park K (2007) Induction of reactive oxygen species and apoptosis in BEAS-2B cells by mercuric chloride. Toxicol In Vitro 21:789–794. doi:10.1016/j.tiv.2007.01.019
Passos CJS, Sampaio DS, Lemire M, Fillion M, Guimarães JRD, Lucotte M, Mergler D (2008) Daily mercury intake in fish-eating populations in the Brazilian Amazon. Exp Sci Environ Epidemiol 18:76–87. doi:10.1038/sj.jes.7500599
Perottoni J, Lobato LP, Silveira A, Rocha JB, Emanuelli T (2004) Effects of mercury and selenite on delta-aminolevulinate dehydratase activity and on selected oxidative stress parameters in rats. Environ Res 95:166–173. doi:10.1016/j.envres.2003.08.007
Rabenstein DL, Fairhurst MT (1975) Nuclear magnetic resonance studies of the solution chemistry of metal complexes XI. Binding of methylmercury by sulfhydryl-containing amino acids and by glutathione. J Am Chem Soc 97:2086–2092. doi:10.1021/ja00841a015
Refsvik T (1978) Excretion of methyl mercury in rat bile: the effect of diethylmaleate, cyclohexene oxide and acrylamide. Acta Pharmacol Toxicol (Cph) 42:135–141
Reus IS, Bando I, Andre’s D, Cascales M (2003) Relationship between expression of HSP70 and metallothionein and oxidative stress during mercury chloride induced acute liver injury in rats. J Biochem Mol Toxicol 17:161–168. doi:10.1002/jbt.10074
Rosselli M, Imthurn B, Keller PJ, Jackson EK, Dubey RK (1995) Circulating nitric oxide (nitrite/nitrate) levels in postmenopausal women substituted with 17 beta-estradiol and norethisterone acetate. A two-year follow-up study. Hypertension 25:848–853
Roulet M, Lucotte M, Farella N, Serique G, Coelho H, Passos CJS, da Silva EJ, de Andrade PS, Mergler D, Guimarães JRD, Amorim M (1999) Effects of recent human colonization on the presence of mercury in Amazonian ecosystems. Water Air Soil Pollut 112:297–313
Salonen JT, Seppänen K, Nyyssönen K, Korpela H, Kauhanen J, Kantola M, Tuomilehto J, Esterbauer H, Tatzber F, Salonen R (1995) Intake of mercury from fish, lipid peroxidation, and the risk of myocardial infarction and coronary, cardiovascular, and any death in eastern Finish men. Circulation 91:645–655
Sarafian TA (1999) Methylmercury-induced generation of free radical: biological implications. Met Ions Biol Syst 36:415–444
Shaw CA, Taylor EL, Megson IL, Rossi AG (2005) Nitric oxide and the resolution of inflammation: implications for atherosclerosis. Mem Inst Oswaldo Cruz 100:67–71. doi:10.1590/S0074-02762005000900012
Shimizu N, Kobayashi K, Hayashi K (1984) The reaction of superoxide radical with catalase. J Biol Chem 259:4414–4418
Sirois JE, Atchison WD (2000) Methylmercury affects multiple subtypes of calcium channels in rat cerebellar granule cells. Toxicol Appl Pharmacol 167:1–11. doi:10.1006/taap.2000.8967
Stern AH (2005) A review of the studies of the cardiovascular health effects of methylmercury with consideration of their suitability for risk assessment. Environ Res 98:133–142. doi:10.1016/j.envres.2004.07.016
Stringari J, Meotti FC, Souza DO, Santos ARS, Farina M (2006) Postnatal methylmercury exposure induces hyperlocomotor activity and cerebellar oxidative stress in mice: dependence on the neurodevelopmental period. Neurochem Res 31:563–569. doi:10.1007/s11064-006-9051-9
Stringari J, Nunes AKC, Franco JL, Bohrer D, Garcia SC, Dafre AL, Milatovic D, Souza DO, Rocha JBT, Aschner M, Farina M (2008) Prenatal methylmercury exposure hampers glutathione antioxidant system ontogenesis and causes long-lasting oxidative stress in the mouse brain. Toxicol Appl Pharmacol 227:147–154. doi:10.1016/j.taap.2007.10.010
Su L, Wang M, Yin ST, Wang HL, Chen L, Sun LG, Ruan DY (2008) The interaction of selenium and mercury in the accumulations and oxidative stress of rat tissues. Ecotoxicol Environ Saf 70:483–489. doi:10.1016/j.ecoenv.2007.05.018
Thompson SA, White CC, Krejsa CM, Eaton DL, Kavanagh TJ (2000) Modulation of glutathione and glutamate-l-cysteine ligase by methylmercury during mouse development. Toxicol Sci 57:141–146. doi:10.1093/toxsci/57.1.141
Uchino M, Tanaka Y, Ando Y, Yonehara T, Hara A, Mishima I, Okajima T, Ando M (1995) Neurological features of chronic Minamata disease (organic mercury poisoning) and incidence of complications with aging. J Environ Sci Health B 30:699–715. doi:10.1080/03601239509372961
Virtanen JK, Rissanen TH, Voutilainen S, Tuomainen TP (2007) Mercury is a risk factor for cardiovascular diseases. J Nutr Biochem 18:75–85. doi:10.1016/j.jnutbio.2006.05.001
Wakita Y (1987) Hypertension induced by methyl mercury in rats. Toxicol Appl Pharmacol 89:144–147. doi:10.1016/0041-008X(87)90185-2
Wolf MB, Baynes JW (2007) Cadmium and mercury cause an oxidative stress-induced endothelial dysfunction. Biometals 20:73–81. doi:10.1007/s10534-006-9016-0
Yoshizawa K, Rimm EB, Morris JS, Spate VL, Hsieh CC, Spiegelman D, Stampfer MJ, Willett WC (2002) Mercury and the risk of coronary heart disease in men. N Engl J Med 347:1755–1760. doi:10.1056/NEJMoa021437
Zahir F, Rizwi SJ, Haq SK, Khan RH (2005) Low dose mercury toxicity and human health. Environ Toxicol Pharmacol 20:351–360. doi:10.1016/j.etap.2005.03.007
Acknowledgements
The authors would like to thank the financial support of the São Paulo State Foundation for Scientific Research (FAPESP, Brazil) and the Brazilian National Council for Scientific and Technologic Development (CNPq).
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grotto, D., de Castro, M.M., Barcelos, G.R.M. et al. Low level and sub-chronic exposure to methylmercury induces hypertension in rats: nitric oxide depletion and oxidative damage as possible mechanisms. Arch Toxicol 83, 653–662 (2009). https://doi.org/10.1007/s00204-009-0437-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-009-0437-8