Abstract
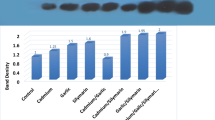
The aim of this study was to evaluate the effects of chronic exposure to cadmium (Cd) on the renal cytochrome P450-dependent monooxygenase system. For this purpose, male Wistar rats were intoxicated with Cd administered in drinking water at a concentration of 5 or 50 mg Cd/l for 6, 12 and 24 weeks. Concentrations of cytochrome P450 and cytochrome b5 as well as activities of NADPH-cytochrome P450 reductase and NADH-cytochrome b5 reductase were determined in the kidney microsomal fraction. Protein content of CYP1A1, CYP2E1 and CYP3A1 cytochrome P450 isoforms was evaluated as well. In the rats exposed to 5 mg Cd/l, the concentration of cytochrome P450 decreased (by 41%) after 24 weeks of the experiment. The activity of NADPH-cytochrome P450 reductase decreased (by 24%) after 6 and 12 weeks, whereas after 24 weeks it remained unchanged, compared with the control group. Moreover, a decrease in the concentration of cytochrome b5 (by 25, 15 and 26% at 6, 12 and 24 weeks, respectively) and the activity of its NADH reductase (by 26 and 31% at 6 and 24 weeks, respectively) was noted in these animals. At the exposure to 50 mg Cd/l, the concentrations of cytochrome P450 and cytochrome b5 and the activities of their corresponding reductases were decreased at each time-point. Western blot analysis revealed that all isoforms of cytochrome P450 studied were affected by Cd and the effect was dependent on the level and the duration of exposure. The results of this study indicate that chronic exposure to Cd in a dose- and time-dependent manner affects the kidney cytochrome P450-dependent monooxygenase system by decreasing the concentrations of cytochrome P450 and cytochrome b5 and inhibiting the activities of their corresponding reductases. The effect of Cd on the cytochrome P450 content is associated with its ability to stimulate or inhibit of various P450 isoforms. A very important finding of this study is that Cd affects the kidney cytochrome P450-dependent monooxygenase system at relatively low exposure and low kidney Cd accumulation (2.40±0.15 μg/g). As the experimental model used reflects human exposure to Cd, we conclude that Cd can affect the kidney cytochrome P450-dependent monooxygenase system in environmentally exposed humans. Previously we have reported disorders in the system in the liver of rats at the same levels of exposure as in this study. Thus, we hypothesize that the metabolism and detoxification of many substances, including xenobiotics, may be seriously affected in Cd-exposed subjects.





Similar content being viewed by others
References
Alexidis AN, Rekka EA, Kourounakis PN (1994) Influence of mercury and cadmium intoxication on hepatic microsomal CYP2E and CYP3A subfamilies. Res Commun Mol Pathol Pharmacol 85:67–72
Baker JR, Satarug S, Reilly PE, Edwards RJ, Ariyoshi N, Kamataki T, Moore MR, Williams DJ (2001) Relationships between non-occupational cadmium exposure and expression of nine cytochrome P450 forms in human liver and kidney cortex samples. Biochem Pharmacol 62:713–721
Bompart G, Orfila C, Manuel Y (1991) Cisplatin nephrotoxicity in cadmium-pretreated rats. Enzymatic, functional and morphological studies. Nephron 58:68–74
Brzóska MM, Kamiński M., Supernak-Bobko D, Zwierz K, Moniuszko-Jakoniuk J (2003) Changes in the structure and function of the kidney of rats chronically exposed to cadmium. I. Biochemical and histopathological studies. Arch Toxicol 77:344–352
Dallner G (1974) Isolation of rough and smooth microsomes—general. Methods Enzymol 32:191–215
Degawa M, Arai H, Miura S, Hashimoto Y (1993) Preferential inhibitions of hepatic P450IA2 expression and induction by lead nitrate in the rat. Carcinogenesis 14:1091–1094
Degawa M, Arai H, Kubota M, Hashimoto Y (1994) Ionic lead, a unique metal ion as an inhibitor for cytochrome P450IA2 (CYP1A2) expression in the rat liver. Biochem Biophys Res Commun 200:1086–1092
Degawa M, Arai H, Kubota M, Hashimoto Y (1995) Ionic lead, but not other ionic metals (Ni2+, Co2+ i Cd2+), suppresses 2-methoxy-4-aminoazobenzene-mediated cytochrome P450IA2 (CYP1A2) induction in rat liver. Biol Pharm Bull 18:1215–1218
Ding HP, Liu YG (1992) A comparative study of the effects of cadmium on microsomal mixed-function oxidase activities between kidney cortex and liver. J Tongji Med Univ 12:85–89
Estabrook RW, Werringloer J (1978) The measurement of difference spectra: application to the cytochromes of microsomes. Methods Enzymol 52:212–220
Guengerich FP (1997) Comparisons of catalytic selectivity of cytochrome P450 subfamily enzymes from different species. Chem Biol Interact 106:161–182
Guengerich FP (2003) Cytochrome P450 oxidations in the generation of reactive electrophiles: epoxidation and related reactions. Arch Biochem Biophys 409:59–71
Hodges TK, Leonard RT (1974) Purification of a plasma membrane-bound adenosine triphosphatase from plant roots. Methods Enzymol 32:392–406
Iszard MB, Liu J, Klaassen CD (1995a) Effect of several metallothionein inducers on oxidative stress defense mechanisms in rats. Toxicology 104:25–33
Iszard MB, Liu J, Dalton T, Andrews GK, Palmiter RD, Klaassen CD (1995b) Characterization of metallothionein-I-transgenic mice. Toxicol Appl Pharmacol 133:305–312
Kido T, Nogawa K, Ohmichi M, Honda R, Tsuritani I, Ishizaki M, Yamada Y (1992) Significance of urinary cadmium concentration in Japanese population environmentally exposed to cadmium. Arch Environ Health 47:196–202
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lauwerys R, Bernard AM, Roels HA, Buchet JP (1994) Cadmium: exposure markers as predictors of nephrotoxic effects. Clin Chem 40:1391–1394
Lowry OH, Rosebrough NJ, Farr AL, Randal RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Novak RF, Woodcroft KJ (2000) The alcohol-inducible form of cytochrome P450 (CYP 2E1): role in toxicology and regulation of expression. Arch Pharm Res 23:267–282
Nordberg GF, Jin T, Nordberg M (1994) Subcellular targets of cadmium nephrotoxicity: cadmium binding to renal membrane proteins in animals with or without protective metallothionein synthesis. Environ Health Perspect 102[Suppl 3]:191–194
Omura T, Sato R (1964) The carbon monoxide-binding pigment of liver microsomes. I. Evidence for its hemoprotein nature. J Biol Chem 239:2370–2378
Plewka A, Brzóska MM, Moniuszko-Jakoniuk J, Plewka D, Nowaczyk G, Wiaderkiewicz A, Kamiński M (1999) Effect of cadmium on liver cytochrome P450-dependent monooxygenase system. Pol J Environ Stud 8:154–157
Schnell RC, Yuhas EM, Pence DH, Means JR, Roberts SA, Yau ET, Miya S, Mennear JH (1978) Effect of acute and chronic cadmium treatment on hepatic drug metabolism in male rats. Arch Toxicol 40:269–277
Schnell RC, Means JR, Roberts SA, Pence DH (1979) Studies on cadmium-induced inhibition of hepatic microsomal drug biotransformation in the rat. Environ Health Perspect 28:273–277
Templeton D, Cherian G (1991) Toxicological significance of metallothionein. Methods Enzymol 205:11–24
UengY-F, Liu C, Lai C-F, Meng L-M, Hung Y-Y, Ueng T-H (1996) Effects of cadmium and environmental pollution on metallothionein and cytochrome P450 in tilapia. Bull Environ Contam Toxicol 57:125–131
Virgo BB, Virgo NS (1984) Neonatal exposure to cadmium alters the response of the hepatic monooxygenases to phenobarbital and to cadmium in adult male rats. J Pharmacol Exp Ther 231:700–704
Acknowledgements
The present study was supported by Grant No. 4PO5D 012 19 from the Committee for Scientific Research (KBN, Poland).
Author information
Authors and Affiliations
Corresponding author
Additional information
Part of this work was presented at the EUROTOX 2001, Istanbul, Turkey, 2001
Rights and permissions
About this article
Cite this article
Plewka, A., Plewka, D., Nowaczyk, G. et al. Effects of chronic exposure to cadmium on renal cytochrome P450-dependent monooxygenase system in rats. Arch Toxicol 78, 194–200 (2004). https://doi.org/10.1007/s00204-003-0529-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-003-0529-9