Abstract
Introduction
A longitudinal study was established to investigate bone compositional information in spinal cord injury (SCI) rat model.
Methods
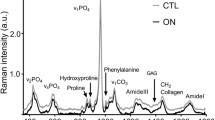
Raman spectroscopy was applied to detect the distal femur and humeri of SCI, sham-operated (SO), and age-matched control (CON) male Sprague-Dawley (SD) rats at first, second, third, and fifth weeks after surgery. One-way ANOVA and Tukey’s HSD post hoc multiple comparison tests were used to analyze the longitudinal data of mineral to matrix ratio and carbonate substitution.
Results
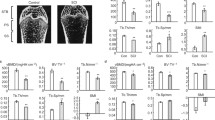
Relative mineral decrease was found in SCI group by more than 20% in femur and approximately 12% in humeri compared with CON group. No significant changes in carbonate substitution were observed.
Conclusions
Severe bone loss in the early stage of SCI was confirmed by a continuous decrease of the mineral to collagen matrix ratio. The decrease in the humeri suggested hormone level variations might participate in the etiology of SCI-induced osteoporosis.



Similar content being viewed by others
References
Kiratli BJ, Smith AE, Nauenberg T et al (2000) Bone mineral and geometric changes through the femur with immobilization due to spinal cord injury. J Rehabil Res Dev 37:225–233
Frey-Rindova P, de Bruin ED, Stussi E et al (2000) Bone mineral density in upper and lower extremities during 12 months after spinal cord injury measured by peripheral quantitative computed tomography. Spinal Cord 38:26–32
Liu CC, Theodorou DJ, Theodorou SJ et al (2000) Quantitative computed tomography in the evaluation of spinal osteoporosis following spinal cord injury. Osteoporos Int 11:889–896
Lazo MG, Shirazi P, Sam M et al (2001) Osteoporosis and risk of fracture in men with spinal cord injury. Spinal Cord 39:208–214
Jiang SD, Jiang LS, Dai LY (2006) Spinal cord injury causes more damage to bone mass, bone structure, biomechanical properties and bone metabolism than sciatic neurectomy in young rats. Osteoporos Int 17:1552–1561
Jiang SD, Shen C, Jiang LS et al (2007) Differences of bone mass and bone structure in osteopenic rat models caused by spinal cord injury and ovariectomy. Osteoporos Int 18:743–750
Bieringsorensen F, Bohr H, Schaadt O (1988) Bone-mineral content of the lumbar spine and lower-extremities years after spinal-cord lesion. Paraplegia 26:293–301
Siris ES, Brenneman SK, Miller PD et al (2004) Predictive value of low BMD for 1-year fracture outcomes is similar for postmenopausal women ages 50–64 and 65 and older: results from the National Osteoporosis Risk Assessment (NORA). J Bone Miner Res 19:1215–1220
Poetzschner R, Abendroth K (2001) Relationship of risk factors, clinical and X-ray findings of osteoporosis with BMD results of different QCT and pQCT measurements. J Bone Miner Res 16(suppl):518–518
Carter DR, Hayes WC (1977) Compressive behavior of bone as a 2-phase porous structure. J Bone Joint Surg Am 59:954–962
Majumdar S, Kothari M, Augat P et al (1998) High-resolution magnetic resonance imaging: three-dimensional trabecular bone architecture and biomechanical properties. Bone 22:445–454
Lee C, Almagor O, Dunlop DD et al (2007) Self-reported fractures and associated factors in women with systemic lupus erythematosus. J Rheumatol 34:2018–2023
Bolotin HH (2007) DXA in vivo BMD methodology: an erroneous and misleading research and clinical gauge of bone mineral status, bone fragility, and bone remodelling. Bone 41:138–154
Rubin MA, Rubin J, Jasiuk I (2004) SEM and TEM study of the hierarchical structure of C57BL/6J and C3H/HeJ mice trabecular bone. Bone 35:11–20
Rubin MA, Jasiuk I, Taylor J et al (2003) TEM analysis of the nanostructure of normal and osteoporotic human trabecular bone. Bone 33:270–282
Barbosa CJ, Vaillancourt FH, Eltis LD et al (2002) The power distribution advantage of fiber-optic coupled ultraviolet resonance Raman spectroscopy for bioanalytical and biomedical applications. J Raman Spectrosc 33:503–510
Vo-Dinh T, Yan F, Wabuyele MB (2006) Surface-enhanced Raman scattering for biomedical diagnostics and molecular imaging. In: Kneipp K, Moskovits M, Kneipp H (eds) Surface-enhanced Raman scattering: physics and applications. Springer, Berlin, pp 409–426
Walton AG, Deveney MJ, Koenig JL (1970) Raman spectroscopy of calcified tissue. Calcif Tissue Res 6:162–167
Draper ERC, Morris MD, Camacho NP et al (2005) Novel assessment of bone using time-resolved transcutaneous Raman spectroscopy. J Bone Miner Res 20:1968–1972
Penel G, Delfosse C, Descamps M et al (2005) Composition of bone and apatitic biomaterials as revealed by intravital Raman microspectroscopy. Bone 36:893–901
Penel G, Leroy N, Van Landuyt P et al (1999) Raman microspectrometry studies of brushite cement: in vivo evolution in a sheep model. Bone 25(suppl):81–84
Weiner S (2006) Transient precursor strategy in mineral formation of bone. Bone 39:431–433
Carden A, Rajachar RM, Morris MD et al (2003) Ultrastructural changes accompanying the mechanical deformation of bone tissue: a Raman imaging study. Calcif Tissue Int 72:166–175
McCreadie BR, Morris MD, Chen TC et al (2006) Bone tissue compositional differences in women with and without osteoporotic fracture. Bone 39:1190–1195
Gadeleta SJ, Boskey AL, Paschalis E et al (2000) A physical, chemical, and mechanical study of lumbar vertebrae from normal, ovariectomized, and nandrolone decanoate-treated cynomolgus monkeys (Macaca fascicularis). Bone 27:541–550
Faibish D, Gomes A, Boivin G et al (2005) Infrared imaging of calcified tissue in bone biopsies from adults with osteomalacia. Bone 36:6–12
Ou-Yang H, Paschalis EP, Mayo WE et al (2001) Infrared microscopic imaging of bone: spatial distribution of CO 2-3 . J Bone Miner Res 16:893–900
Bohic S, Rey C, Legrand A et al (2000) Characterization of the trabecular rat bone mineral: effect of ovariectomy and bisphosphonate treatment. Bone 26:341–348
Paschalis EP, Betts F, DiCarlo E et al (1997) FTIR microspectroscopic analysis of human iliac crest biopsies from untreated osteoporotic bone. Calcif Tissue Int 61:487–492
Jiang SD, Dai LY, Jiang LS (2006) Osteoporosis after spinal cord injury. Osteoporos Int 17:180–192
Maimoun L, Couret I, Micallef JP et al (2002) Use of bone biochemical markers with dual-energy X-ray absorptiometry for early determination of bone loss in persons with spinal cord injury. Metabolism 51:958–963
Roberts D, Lee W, Cuneo RC et al (1998) Longitudinal study of bone turnover after acute spinal cord injury. J Clin Endocrinol Metab 83:415–422
Dauty M, Verbe BP, Maugars Y et al (2000) Supralesional and sublesional bone mineral density in spinal cord-injured patients. Bone 27:305–309
Jiang SD, Jiang LS, Dai LY (2006) Mechanisms of osteoporosis in spinal cord injury. Clin Endocrinol 65:555–565
Iida-Klein A, Hughes C, Lu SS et al (2006) Effects of cyclic versus daily hPTH(1–34) regimens on bone strength in association with BMD, biochemical markers, and bone structure in mice. J Bone Miner Res 21:274–282
Akkus O, Adar F, Schaffler MB (2004) Age-related changes in physicochemical properties of mineral crystals are related to impaired mechanical function of cortical bone. Bone 34:443–453
Huang RY, Miller LM, Carlson CS et al (2003) In situ chemistry of osteoporosis revealed by synchrotron infrared microspectroscopy. Bone 33:514–521
Acknowledgements
This work is financially supported by National Natural Science Foundation of China (No. 20705025, No. 30772058).
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Shen, J., Fan, L., Yang, J. et al. A longitudinal Raman microspectroscopic study of osteoporosis induced by spinal cord injury. Osteoporos Int 21, 81–87 (2010). https://doi.org/10.1007/s00198-009-0949-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-009-0949-3