Abstract
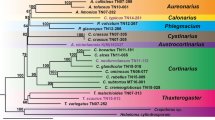
The development and application of molecular methods in oats has been relatively slow compared with other crops. Results from the previous analyses have left many questions concerning species evolutionary relationships unanswered, especially regarding the origins of the B and D genomes, which are only known to be present in polyploid oat species. To investigate the species and genome relationships in genus Avena, among 13 diploid (A and C genomes), we used the second intron of the nuclear gene FLORICAULA/LEAFY (FL int2) in seven tetraploid (AB and AC genomes), and five hexaploid (ACD genome) species. The Avena FL int2 is rather long, and high levels of variation in length and sequence composition were found. Evidence for more than one copy of the FL int2 sequence was obtained for both the A and C genome groups, and the degree of divergence of the A genome copies was greater than that observed within the C genome sequences. Phylogenetic analysis of the FL int2 sequences resulted in topologies that contained four major groups; these groups reemphasize the major genomic divergence between the A and C genomes, and the close relationship among the A, B, and D genomes. However, the D genome in hexaploids more likely originated from a C genome diploid rather than the generally believed A genome, and the C genome diploid A. clauda may have played an important role in the origination of both the C and D genome in polyploids.



Similar content being viewed by others
References
Ahearn KP, Johnson HA, Weigel D, Wagner DR (2001) NFL1, a Nicotiana tabacum LFY-like gene, controls meristem initiation and floral structure. Plant Cell Physiol 42:1130–1139
Akaike H (1974) A new look at the statistical model identification. IEEE T Automat Contr 19:716–723
Alicchio R, Aranci L, Conte L (1995) Restriction fragment length polymorphism based phylogenetic analysis of Avena L. Genome 38:1279–1284
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Archambault A, Bruneau A (2001) How useful is the LEAFY gene for the phylogeny reconstruction in the Caesalpinioideae? Am J Bot 88(Suppl):97
Archambault A, Bruneau A (2004) Phylogenetic utility of the LEAFY/FLORICAULA gene in the caesalpinioideae (Leguminosae): gene duplication and a novel insertion. Syst Bot 29:609–626
Badaeva E, Loskutov I, Shelukhina O, Pukhalsky V (2005) Cytogenetic analysis of diploid Avena L. species containing the as genome. Russ J Genet 41:1428–1433
Bailey CD, Doyle JJ (1999) Potential phylogenetic utility of the low-copy nuclear gene pistillata in dicotyledonous plants: comparison to nrDNA ITS and trnL intron in Sphaerocardamum and other Brassicaceae. Mol Phylogenet Evol 13:20–30
Baker WJ, Hedderson TA, Dransfield J (2000) Molecular phylogenetics of subfamily Calamoideae (Palmae) based on nrDNA ITS and cpDNA rps16 intron sequence data. Mol Phylogenet Evol 14:195–217
Baldwin BG, Preston RE, Wessa BL, Wetherwax M (2001) A biosystematic and phylogenetic assessment of sympatric taxa in Blepharizonia (Compositae-Madiinae). Syst Bot 26:184–194
Baum BR (1977) Oats: wild and cultivated, a monograph of the genus Avena L. (Poaceae). Minister of supply and services Canada. Agriculture Canada, Ottawa, Ontario
Baum BR, Rajhathy T, Sampson DR (1973) An important new diploid Avena species discovered on the Canary Islands. Can J Bot 51:4759–4762
Blázquez MA, Soowal LN, Lee I, Weigel D (1997) LEAFY expression and flower initiation in Arabidopsis. Development 124:3835–3844
Bomblies K, Doebley JF (2005) Molecular evolution of FLORICAULA/LEAFY orthologs in the Andropogoneae (Poaceae). Mol Biol Evol 22:1082–1094
Chen Q, Armstrong K (1994) Genomic in situ hybridization in Avena sativa. Genome 37:607–612
Cheng DW, Armstrong KC, Drouin G, McElroy A, Fedak G, Molnar SD (2003) Isolation and identification of Triticeae chromosome 1 receptor-like kinase genes (Lrk10) from diploid, tetraploid, and hexaploid species of the genus Avena. Genome 46:119–127
Coen ES, Romero JM, Doyle S, Elliott R, Murphy G, Carpenter R (1990) Floricaula: a homeotic gene required for flower development in antirrhinum majus. Cell 63:1311–1322
Craig IL, Murray BE, Rajhathy T (1974) A. canariensis: mor-phological and electrophoretic polymorphism and relationship to the A. magna-A. murphyi complex and A. sterilis. Can J Genet Cytol 16:677–689
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Drossou A, Katsiokis A, Leggett JM, Loukas M, Tsakas S (2004) Genome and species relationships in genus Avena based on RAPD and AFLP molecular markers. Theor Appl Genet 109:48–54
Fominaya A, Vega C, Ferrer E (1988a) Giemsa C-banded karyotypes of Avena species. Genome 30:627–632
Fominaya A, Vega C, Ferrer E (1988b) C-banding and nucleolar activity of tetraploid Avena species. Genome 30:633–638
Fominaya A, Hueros G, Loarce Y, Ferrer E (1995) Chromosomal distribution of a repeated DNA sequence from C-genome heterochromatin and the identification of a new ribosomal DNA locus in the Avena genus. Genome 38:548–557
Frohlich MW, Meyerowitz EM (1997) The search for flower homeotic gene homologs in basal Angiosperms and Gnetales: a potential new source of data on the evolutionary origin of flowers. Int J Plant Sci 158:S131–S142
Frohlich MW, Parker DS (2000) The mostly male theory of flower evolutionary origins: from genes to fossils. Syst Bot 25:155–170
Fu YB, Williams DJ (2008) AFLP variation in 25 Avena species. Theor Appl Genet 117:333–342
Galtier N, Gouy M, Gautier C (1996) SEAVIEW and PHYLO_WIN: two graphic tools for sequence alignment and molecular phylogeny. Comput Appl Biosci 12:543–548
Ge S, Sang T, Lu BR, Hong DY (1999) Phylogeny of rice genomes with emphasis on origins of allotetraploid species. Proc Natl Acad Sci USA 96:14400–14405
Grob GBJ, Gravendeel B, Eurlings MCM (2004) Potential phylogenetic utility of the nuclear FLORICAULA/LEAFY second intron: comparison with three chloroplast DNA regions in Amorphophallus (Araceae). Mol Phylogenet Evol 30:13–23
Hillis DM (1987) Molecular versus morphological approaches to systematics. Annu Rev Ecol Syst 18:23–42
Hofer J, Turner L, Hellens R, Ambrose M, Matthews P, Michael A, Ellis N (1997) UNIFOLIATA regulates leaf and flower morphogenesis in pea. Curr Biol 7:581–587
Hoot SB, Taylor WC (2001) The utility of nuclear ITS, a LEAFY homolog intron, and chloroplast atpB-rbcL spacer region data in phylogenetic analyses and species delimitation in Isoetes. Am Fern J 91:166–177
Howarth DG, Baum DA (2005) Gencalogical wbidence of homoploid hybrid speciation in an adaptive radiation of Scaevola (Goodenraceae) in the Hawarran islands. Evolution 59:948–961
Irigoyen ML, Loarce Y, Linares C, Ferrer E, Leggett M, Fominaya A (2001) Discrimination of the closely related A and B genomes in AABB tetraploid species of Avena. Theor Appl Genet 103:1160–1166
Irigoyen ML, Ferrer E, Loarce Y (2006) Cloning and characterization of resistance gene analogs from Avena species. Genome 49:54–63
Jellen EN, Gill B (1996) C-banding variation in the Moroccan oat species Avena agadiriana (2n = 4x = 28). Theor Appl Genet 92:726–732
Jellen EN, Phillips RL, Rines HW (1993) C-banded karyotypes and polymorphisms in hexaploid oat accessions (Avena. spp.) using Wright’s stain. Genome 36:1129–1137
Jellen EN, Philips RL, Rines HW (1994a) Chromosomal localization and polymorphisms of ribosomal DNA in oat (Avena spp.). Genome 37:23–32
Jellen EN, Gill BS, Cox TS (1994b) Genomic in situ hybridization differentiates between A/D- and C-genome chromatin and detects intergenomic translocations in polyploid oat species (genus Avena). Genome 37:613–618
Jiang J, Gill BS (1994) Different species-specific chromosome translocations in Triticum timopheevii and T. turgidum support the diphyletic origin of polyploid wheats. Chromosome Res 2:59–64
Jin H, Domier LL, Kolb FL, Brown CM (1999) Identification of quantitative loci for tolerance to barley yellow dwarf virus in oat. Phytopathology 88:410–415
Katsiotis A, Schmidt T, Heslop-Harrison JS (1996) Chromosomal and genomic organization of Ty1-copia-like retrotransposon sequences in the genus Avena. Genome 39:410–417
Katsiotis A, Loukas M, Heslop-Harrison JS (1997) The close relationship between the A and B genomes in Avena L. (Poaceae) determined by molecular cytogenetic analysis of total genomic, tandemly and dispersed repetitive DNA sequences. Ann Bot 79:103–109
Kelly AJ, Bonnlander MB, Meeks-Wagner DR (1995) NFL, the tobacco homolog of FLORICAULA and LEAFY, is transcriptionally expressed in both vegetative and floral meristems. Plant Cell 7:225–234
Kianian SF, Egli MA, Phillips RL, Rines HW, Somers DA, Gengenbach BG, Wesenberg DM, Stuthman DD, Fulcher RG (1999) Association of a major groat oil content QTL and an acetyl-COA carboxylase gene in oat. Theor Appl Genet 98:884–894
Kihara H, Nishiyama I (1932) The genetics and cytology of certain cereals III. Different compatibility in reciprocal crosses of Avena, with reference to tetraploid hybrids between hexaploid and diploid species. Jpn J Bot 6:245–305
Kimura M (1980) A simple method for estimating evolutionary rate of base substitution through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Ladizinsky G (1973) Genetic control of bivalent pairing in the Avena strigosa polyploid complex. Chromosoma 42:105–110
Ladizinsky G (1974) Genome relationships in the diploid oats. Chromosoma 47:109–117
Ladizinsky G (1988) Biological species and wild genetic resources in Avena. In: Mattsson B, Lyhagen R, Svalof AB (eds) Proceedings of the 3rd international oat conference, Lund, Sweden, 4–8 July, 1988
Ladizinsky G (1998) A new species of Avena from Sicily, possibly the tetraploid progenitor of hexaploid oats. Genet Resour Crop Evol 45:263–269
Ladizinsky G (1999) Cytogenetic relationships between Avena insularis (2n = 28) and both A. strigosa (2n = 14) and A. murphyi (2n = 28). Genet Resour Crop Evol 46:501–504
Ladizinsky G, Fainstein R (1977) Intergression between the cultivated hexaploid oat A. sativa and the tetraploid wild A. magna and A. murphyi. Can J Genet Cytol 19:59–60
Ladizinsky G, Zohary D (1968) Genetic relationships between the diploids and tetraploids in the series Eubarbatae of Avena. Can J Genet Cytol 10:68–81
Ladizinsky G, Zohary D (1971) Notes on species delimitation, species relationships and polyploidy in Avena. Euphytica 20:380–395
Leggett JM (1984a) Morphology and metaphase chromosome pairing in three Avena hybrids. Can J Genet Cytol 26:641–645
Leggett JM (1984b) Cytoplasmic substitutions involving six Avena species. Can J Genet Cytol 26:698–700
Leggett JM (1987) Interspecific hybrids involving the recently described taxon Avena atlantica. Genome 29:361–364
Leggett JM (1989) Interspecific diploid hybrids in Avena. Genome 32:346–348
Leggett JM (1996) Using and conserving Avena genetic resources. In: Scoles GJ, Rossnagel BG (eds) Barley chromosome coordinators’ workshop at the V international oat conference & VII international barley genetics symposium, Saskatoon, Saskatchewan, Canada, 31 July 1996. University of Saskatchewan, pp 128–132
Leggett JM (1998) Chromosome and genomic relationships between the diploid species Avena strigosa, A. eriantha and the tetraploid A. maroccana. Heredity 80:361–363
Leggett JM, Markhand GS (1995) The genomic structure of Avena revealed by GISH. In: Brandham PE, Bennett MD (eds) Kew chromosome conference IV. HMSO, UK, pp 133–139
Leggett JM, Thomas H (1995) Oat evolution and cytogenetics. In: Welch RW (ed) The oat crop: production and utilization. Chapman and Hall, London, UK
Li CD, Rossnagel BG, Scoles GJ (2000a) The development of oat microsatellite markers and their use in identifying relationships among Avena species and oat cultivars. Theor Appl Genet 101:1259–1268
Li CD, Rossnagel BG, Scoles GJ (2000b) Tracing the phylogeny of the hexaploid oat Avena sativa with satellite DNAs. Crop Sci 40:1755–1763
Li WT, Peng YY, Wei YM, Baum BR, Zheng YL (2009) Relationship among Avena species as revealed by consensus chloroplast simple sequence repeat (ccSSR) markers. Genet Resour Crop Evol 56:465–480
Linares C, Vega C, Ferrer E, Fominaya A (1992) Identification of C-banded chromosomes in meiosis and the analysis of nucleolar activity in Avena byzantina C. Koch cv 'Kanota'. Theor Appl Genet 83:650–654
Linares C, Gónzalez J, Ferrer E, Fominaya A (1996) The use of double FISH to physically map the positions of 5S rDNA genes in relation to the chromosomal location of 18S–5.8S–26S rDNA and a C genome specific DNA sequence in the genus Avena. Genome 39:535–542
Linares C, Ferrer E, Fominaya A (1998) Discrimination of the closely related A and D genomes of the hexaploid oat Avena sativa L. Proc Natl Acad Sci USA 95:12450–12455
Linares C, Irigoyen ML, Fominaya A (2000) Identification of C-genome chromosomes involved in intergenomic translocations in Avena sativa L., using cloned repetitive DNA sequences. Theor Appl Genet 100:353–360
Liston A, Robinson WA, Oliphant JM, Alvarez-Buylla ER (1996) Length variation in the nuclear ribosomal DNA internal transcribed spacer region of non-flowering seed plants. Syst Bot 21:109–120
Liu Q, Ge S, Tang H, Zhang X, Zhu G, Lu BR (2006) Phylogenetic relationships in Elymus (Poaceae: Triticeae) based on the nuclear ribosomal internal transcribed spacer and chloroplast trnL-F sequences. New Phytol 170:411–420
Loskutov IG (2001) Interspecific crosses in the genus Avena L. Rus J Genet 37:467–475
Loskutov IG (2008) On evolutionary pathways of Avena species. Genet Resour Crop Evol 55:211–220
Loskutov IG, Perchuk IN (2000) Evaluation of interspecific diversity in Avena genus by RAPD analysis. Oat Newsletter 46 (http://wheat.pw.usda.gov/ggpages/oatnewsletter/v46/)
Marshall HG, Myers WM (1961) A cytogenetic study of certain interspecific Avena hybrids and the inheritance of resistance in diploid and tetraploid varieties to races of crown rust. Crop Sci 1:29–34
Martin AP, Burg TM (2002) Perils of paralogy: using HSP70 genes for inferring organismal phylogenies. Syst Biol 51:570–587
Mason-Gamer RJ (2001) Origin of North American Elymus (Poaceae: Triticeae) allotetraploids based on granule-bound starch synthase gene sequences. Syst Bot 26:757–768
Mason-Gamer RJ, Weil CF, Kellogg EA (1998) Granule-bound starch synthase: structure, function, and phylogenetic utility. Mol Biol Evol 15:1658–1673
Murai K, Tsunewaki K (1987) Chloroplast genome evolution in the genus Avena. Genetics 116:613–621
Nicholas KB, Nicholas HB Jr, Deerfield DW II (1997) GeneDoc: a tool for editing and annotating multiple sequence alignments. Embnew News 4:1–4
Nikoloudakis N, Katsiotis A (2008) The origin of the C-genome and cytoplasm of Avena polyploids. Theor Appl Genet 117:273–281
Nikoloudakis N, Skaracis G, Katsiotis A (2008) Evolutionary insights inferred by molecular analysis of the ITS1–5.8S-ITS2 and IGS Avena sp. sequences. Mol Phylogenet Evol 46:102–115
Nishiyama I (1984) Interspecific cross-incompatibility system in the genus Avena. Bot Mag 97:219–231
Nishiyama I, Yabuno T (1975) Meiotic chromosome pairing in two interspecific hybrids and a criticism of the evolutionary relationship of diploid Avena. Jpn J Genet 50:443–451
Nocelli E, Giovannini T, Bioni M, Alicchio R (1999) RFLP- and RAPD-based genetic relationships of seven diploid species of Avena with the A genome. Genome 42:950–959
O’Donoughue LS, Kianian SF, Rayapati PJ, Penner GA, Sorrells ME, Tanksley SD et al (1995) A molecular linkage map of cultivated oat. Genome 38:368–380
Oh S-H, Potter D (2003) Phylogenetic utility of the second intron of LEAFY in Neillia Stephanandra (Rosaceae) and implications for the origin of Stephanandra. Mol Phylogenet Evol 29:203–215
Oh S-H, Potter D (2005) Molecular phylogenetic systematics and biogeography of tribe Neillieae (Rosaceae) using DNA sequences of cpDNA, rDNA and LEAFY. Am J Bot 92:179–192
Oinuma T (1952) Karyomorphology of cereals. Biol J Okayama Univ 1:12–71
Peng YY, Wei YM, Baum BR, Zheng YL (2008) Molecular diversity of 5S rDNA gene and genomic relationships in genus Avena (Poaceae: Aveneae). Genome 51:137–154
Posada D (2008) jModelTest: phylogenetic model averaging. Mol Biol Evol 25:1253–1256
Potter D, Luby JJ, Harrison RE (2000) Phylogenetic relationships among species of Fragaria (Rosaceae) inferred from non-coding nuclear and chloroplast DNA sequences. Syst Bot 25:337–348
Rajhathy T (1966) Evidence and an hypothesis for the origin of the C genome of hexaploid Avena. Can J Genet Cytol 8:774–779
Rajhathy T (1991) The chromosomes of Avena. In: Gupta PK, Tsuchiya T (eds) Chromosome engineering in plants: genetics, breeding, evolution. Elsevier Science Publishers, The Netherlands, pp 447–465
Rajhathy T, Baum BR (1972) Avena damascena: a new diploid oat species. Can J Genet Cytol 14:645–654
Rajhathy T, Morrison JW (1959) Chromosome morphology in the genus Avena. Can J Bot 37:372–377
Rajhathy T, Morrison JW (1960) Genome homology in the genus Avena. Can J Genet Cytol 2:278–285
Rajhathy T, Thomas H (1974) Cytogenetics of oats (Avena L.). Misc Publ Genet Soc Can, Ottawa
Rines HW, Gengenbach BG, Boylan KL, Storey KK (1988) Mitochondrial DNA diversity in oat cultivars and species. Crop Sci 28:171–176
Roalson EH, Columbus JT, Friar EA (2001) Phylogenetic relationships in Cariceae (Cyperaceae) based on ITS (nrDNA) and trnT-L (cpDNA) region sequences: assessment of subgeneric and sectional relationships in Carex with emphasis on section Acrocystis. Syst Bot 26:318–341
Rodionov AV, Tyupa NB, Kim ES, Machs EM, Loskutov IG (2005) Genomic configuration of the autotetraploid oat species Avena macrostachya inferred from comparative analysis of ITS1 and ITS2 sequences: on the oat karyotype evolution during the early events of the Avena species divergence. Russ J Genet 41:518–528
Ronald PS, Penner GA, Brown PD, Brule-Babel A (1997) Identification of RAPD markers for percent hull in oat. Genome 40:873–878
Sadasivaiah RS, Rajhathy T (1968) Genome relationships in tetraploid Avena. Can J Genet Cytol 10:655–669
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstruction phylogenetic trees. Mol Biol Evol 4:406–425
Sánchez de la Hoz P, Fominaya A (1989) Studies of isozymes in oat species. Theor Appl Genet 77:735–741
Schlüter PM, Gudrun K, Stuessy TF, Paulus HF (2007) A screen of low-copy nuclear genes rebeals the LFY gene as phylogenetically informative in closely related species of orchids (Ophrys). Taxon 56:493–504
Schultz EA, Haughn GW (1991) LEAFY, a homeotic gene that regulates inflorescence development in Arabidopsis. Plant Cell 3:771–781
Shelukhina OYU, Badaeva ED, Loskutov IG, Pukhal’sky VA (2007) A comparative cytogenetic study of the tetraploid oat species with the A and C genomes: A. insularis, A. maroccana and A. murphyi. Russ J Genet 43:613–626
Shimodaira H, Hasegawa M (1999) Multiple comparisons of log-likelihoods with applications to phylogenetic inference. Mol Biol Evol 16:1114–1116
Shitsukawa N, Takagishi A, Ikari C, Takumi S, Murai K (2006) WFL, a wheat FLORICAULA/LEAFY ortholog, is associated with spikelet formation as lateral branch of the inflorescence meristem. Genes Genet Syst 81:13–20
Small RL, Ryburn JA, Cronn RC, Seelanan T, Wendel JF (1998) The tortoise and the hare: choosing between noncoding plastome and nuclear Adh sequences for phylogeny reconstruction in a recently diverged plant group. Am J Bot 85:1301–1315
Soltis DE, Soltis PS (1998) Choosing an approach and an appropriate gene for phylogenetic analysis. In: Soltis DE, Soltis PS, Doyle JJ (eds) Molecular systematics of plants II: DNA sequencing. Kluwer Academic Publishers, Boston, pp 1–42
Soltis DE, Soltis PS, Tate JA (2003) Advances in the study of polyploidy since plant speciation. New Phytol 161:173–191
Southerton SG, Strauss SH, Oliver MR, Hercourt RL, Decroocq V, Zhu X, Llewellyn DJ, Dennis ES (1998) Eucalyptus has a functional equivalent of the Arabidopsis floral meristem identity gene LEAFY. Plant Mol Biol 37:897–910
Swofford DL (1998) PAUP*. Phylogenetic analysis using parsimony (*and other methods). Version 4.0b10. Sinauer Associates, Sunderland, Mass
Tank DC, Sang T (2001) Phylogenetic utility of the glycerol-3-phosphate acyltransferase gene: evolution and implications in Paeonia (Paeoniaceae). Mol Phylogenet Evol 19:421–429
Theissen G (2000) Plant breedings: FLO-like meristem identity genes: from basic science to crop plant design. In: Progress in botany, Springer, Berlin, 61:167–183
Thomas H (1992) Cytogenetics of Avena. In: Marshall HG, Sorrells ME (eds) Oat science and technology, monograph 33, agronomy series. ASA and CSSA, Madison, Wisconsin, pp 473–508
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTALW: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Vision TJ, Brown DG, Tanksley SD (2000) The origins of genomic duplications in Arabidopsis. Science 290:2114–2117
Wada M, Cao Q, Nobuhiro K, Soejima J, Masuda T (2002) Apple has two orthologs of FLORICAULA/LEAFY involved in flowering. Plant Mol Biol 49:567–577
Weigel D (1995) The genetics of flower development: from floral induction to ovule morphogenesis. Annu Rev Genet 29:19–39
Wendel JF (2000) Genome evolution in polyploids. Plant Mol Biol 42:225–249
Whitcher IN, Wen J (2001) Phylogeny and biogeography of Corylus (Betulaceae): inferences from ITS sequences. Syst Bot 26:283–298
Wilson CA (2003) Phylogenetic relationships in Iris series Californicae based on ITS sequences of nuclear ribosomal DNA. Syst Bot 28:39–46
Yang Q, Hanson L, Bennett MD, Leitch IJ (1999) Genome structure and evolution in the allohexaploid weed Avena fatua L. (Poaceae). Genome 42:512–518
Acknowledgments
We thank Agriculture and Agri-Food Canada (AAFC), the National Small Grains Collection, ARS, USDA, and Dr. Eric N. Jellen, Brigham Young University, Provo, Utah, USA, for kindly providing the Avena material. We are very grateful to Mrs Charlene Wight, Eastern Cereal and Oilseed Research Centre, for her most valuable help and comments on the manuscript. We also wish to thank two anonymous reviewers for helpful comments that improved the manuscript. This work was supported by the Earmarked fund for Modern Agro-industry Technology Research System (NYCYTX-14). Yuan-Ying Peng was supported by the Excellent Doctoral Dissertation Fund of Sichuan Agricultural University (YBPY0802).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by P. Heslop-Harrison.
Y.-Y. Peng and Y.-M. Wei contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Peng, YY., Wei, YM., Baum, B.R. et al. Phylogenetic inferences in Avena based on analysis of FL intron2 sequences. Theor Appl Genet 121, 985–1000 (2010). https://doi.org/10.1007/s00122-010-1367-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-010-1367-9