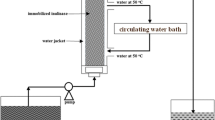
Abstract
A detailed study on the newer methods of coupling urease to diverse matrices have been investigated towards optimizing the efficiency of the immobilized enzyme action and cost effectiveness for the industrial applications. With some of the matrices like collagen, DEAE-cellulose and nylon, even the crude extract of jack bean meal, a major and inexpensive form of urease has been found to be extremely efficient and has indicated that factors other than covalent bonding contribute to the enzyme immobilization. Increased flow rate of the substrate has also resulted in increased reaction kinetics. Apparently, the rapid removal of the product, ammonium ion is largely responsible for this effect. Based on these informations, a large scale column with nylon net has been tested for its efficiency in handling urea effluent from fertilizer plants.
Similar content being viewed by others
References
Sunmer, J.B.: Methods in enzymology, Vol. II, p 378, New York: Academic Press, 1955
Khoklova, T.D.;Yamuna, M.M.;Nikitin Yu.S.;Kurganov, N.I.: Micromethod for the determination of urea in blood plasma and serum by using urease immobilized on silochrome. Zh. Anal. khim. 43 (1980) 1875–1877
Murachi, T.: Use of immobilized enzyme reactors in automated clinical analysis. In: Chibata, Fukui, Wingard (Eds): Enzyme Engineering, New York: Plenum Press, 1982, 369
Stanislaw, G.;Robert, K.;Ewa, K.;Izabela W.;Adam, H.: Urea sensors based on PVC membrane pH electrode. Talanta 41 (1994) 1201–1205
Krajewska, N.;Leszko, M.;Zaborska, W.: Urease immobilized on chitosan membrane: Preparation and properties. J. Chem. Technol. Biotechnol. 48 (1990) 337–350
Srinivasa Rao, M.;Chellapandian, M.;Krishnan, M.R.V.: Immobilization of urease on gelatin-poly (HEMA) copolymer: Preparation and characterization. Bioprocess Engineering 13 (1995) 211–214
Sundaram, P.V.: Cost analysis and viability of immobilized enzymes in routine analysis. In: Chibata, Fukui, Wingard (Eds) Enzyme Engineering, New York, Plenum Press, 1982, 405–407
Sundaram, P.V.;Apps, D.K.: Preparation and properties of nylon tube supported nicotinamide adenine dinucleotide kinase. Biochem J. 161 (1977) 441–443
Martins, M.B.F.;Cruz, M.E.M.;Cabral, J.M.S.;Kennedy, J.F.: Urease immobilization on an alkaline derivative of titanium (IV)-porous silica: Kinetics and operational stability. J. Chem. Technol. Biotechnol. 39 (1987) 201–213
Sundaram, P.V.;Jgloi, M.P.;Wassemam, R.;Hinsch, W.;Knoke, K.J.: Immobilized enzyme nylon tube reactors for routine determination of urea and citrulline in serum. Clin. Chem. 24 (1978) 234–239
Chaney, A.L.;Marbach, E.P.: Modified reagents for the determination of urea and ammonia. Clin. Chem. 8 (1962) 130–132
Lowry, O.H.;Rosenbrough, N.J.;Farr, A.L.;Randall, R.J.: Protein measurement with the Folin-Phenol reagent. J. Biol. Chem. 193 (1951) 265–275
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
George, S., Chellapandian, M., Sivasankar, B. et al. Flow rate dependent kinetics of urease immobilized onto diverse matrices. Bioprocess Engineering 15, 311–315 (1996). https://doi.org/10.1007/BF02426440
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02426440