Abstract
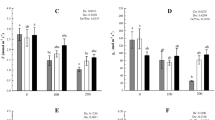
The effects of 24-epibrassinolide (EBR) on chlorophyll fluorescence, leaf surface morphology and cellular ultrastructure of grape seedlings (Vitis vinifera L.) under water stress were investigated. The grape seedlings were subjected to 10 % (w/v) polyethylene glycol (PEG-6000) and treated with 0.05, 0.10 or 0.20 mg L−1 EBR, respectively. EBR application increased chlorophyll contents, the effective photochemical quantum yield of PSII, maximum photochemical efficiency of PSII, maximal fluorescence and non-photochemical quenching coefficient under water stress in each concentration. Compared with water stress control, higher stomatal density and stomatal length were observed in young leaves under EBR treatments, but not in mature leaves. In-depth analysis of the ultrastructure of leaves indicated that water stress induced disappearance of nucleus, chloroplast swelling, fractured mitochondrial cristae and disorder of thylakoid arrangement both in young leaves and mature leaves. However, EBR application counteracted the detrimental effects of water stress on the structure of the photosynthetic apparatus better in young leaves than in mature leaves. Compared to the other treatments, treatment of 0.10 mg L−1 EBR had best ameliorative effect against water stress. These results suggested that exogenous EBR could alleviate water stress-induced inhibition of photosynthesis on grape possibly through increasing chlorophyll content, lessening the stomatal and non-stomatal limitation of photosynthesis performance.





Similar content being viewed by others
Abbreviations
- BRs:
-
Brassinosteroids
- EBR:
-
24-Epibrassinolide
- PEG-6000:
-
Polyethylene glycol-6000
- Chl:
-
Chlorophyll
- PSII:
-
Photosystem II
- F o :
-
Minimal fluorescence
- F v/F m :
-
Maximum photochemical quantum yield of PSII
- ΦPSII:
-
Effective photochemical quantum yield of PSII
- NPQ:
-
Non-photochemical quenching coefficient
- LHC:
-
Light-harvesting complex
- Chr:
-
Chromatin
- CW:
-
Cell wall
- Gt:
-
Grana thylakoid
- M:
-
Mitochondrion
- Nu:
-
Nucleolus
- Pg:
-
Plastoglobule
- SG:
-
Starch grain
References
Adams DB, Adams WW (1996) The role of xanthophylls cycle carotenoids in the protection of photosynthesis. Trends Plant Sci 1:21–26
Ali B, Hayat S, Ahmad A (2007) 28-Homobrassinolide ameliorates the saline stress in chickpea (Cicer arietinum L.). Environ Exp Bot 59:217–223
Ali Q, Athar HR, Ashraf M (2008) Modulation of growth, photosynthetic capacity and water relations in salt stressed wheat plants by exogenously applied 24-epibrassinolide. Plant Growth Regul 56:107–116
Ali S, Farooq MA, Yasmeen T, Hussain S, Arif MS, Abbas F, Bharwana SA, Zhang G (2013) The influence of silicon on barley growth, photosynthesis and ultra-structure under chromium stress. Ecotox Environ Safe 89:66–72
Ali B, Qian P, Jin R, Ali S, Khan M, Aziz R, Tian T, Zhou W (2014) Physiological and ultra-structural changes in brassica napus seedlings induced by cadmium stress. Biol Plantarum 58:131–138
BajguzA HayatS (2009) Effect of brassinosteroids on the plant responses to environmental stresses. Plant Physiol Biochem 47:1–8
Basile A, Sorbo S, Conte B, Cardi M, Esposito S (2013) Ultrastructural changes and heat shock proteins 70 induced by atmospheric pollution are similar to the effects observed under in vitro heavy metals stress in Conocephalum conicum (Marchantiales-Bryophyta). Environ Pollut 182:209–216
Bilger W, Björkman O (1990) Role of the xanthophyll cycle in photoprotection elucidated by measurements of light-induced absorbance changes, fluorescence and photosynthesis in leaves of Hedera canariensis. Photosynth Res 25:173–185
Castle J, Montoya T, Bishop GJ (2003) Selected physiological responses of brassinosteroids: a historical approach. In: Hayat S, Ahmad A (eds) Brassinosteroids: Bioactivity and Crop Productivity. Kluwer Academic Publishers, Dordrecht, pp 45–68
Fariduddin Q, Khanam S, Hasan SA, Ali B, Hayat S, Ahmad A (2009) Effect of 28-homobrassinolide on the drought stress-induced changes in photosynthesis and antioxidant system of Brassica juncea L. Acta Physiol Plant 31:889–897
Gao JF (2006) Experimental guide for Plant Physiology. Higher Education Press, Beijing
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Giles KL, Beardsell MF, Cohen D (1974) Cellular and ultrastructural changes in mesophyll and bundle sheath cells of maize in response to water stress. Plant Physiol 54:208–212
Giles KL, Cohen D, Beardsell MF (1976) Effects of water stress on the ultrastructure of leaf cells of Sorghum bicolor. Plant Physiol 57:11–14
Gomes MMA, Netto AT, Campostrini E, Smith RB, Zullo MAT, Ferraz TM, Siqueira LN, Leal NR, VÁZquez MN (2013) Brassinosteroid analogue affects the senescence in two papaya genotypes submitted to drought stress. Theor Exp Plant Physiol 25:186–195
Grove MD, Spencer GF, Rohwedder WK, Mandava N, Worley JF, Warthen JD, Steffens GL, Flippen-Anderson JL, Cook JC (1979) Brassinolide, a plant growth-promoting steroid isolated from Brassica napus pollen. Nature 281:216–217
Gunning BES, Steer MW (1996) Plant Cell Biology: structure and function. Jones and Bartlett Publishers, Sudbury
Hayat S, Ali B, Hasan SA, Ahmad A (2007) Brassinosteroid enhanced the level of antioxidants under cadmium stress in Brassica juncea. Environ Exp Bot 60:33–41
Hu WH, Yan XH, Xiao YA, Zeng JJ, Qi HJ, Ogweno JO (2013) 24-Epibrassinosteroid alleviate drought-induced inhibition of photosynthesis in Capsicum annuum. Sci Hortic 150:232–237
Khripach V, Zhabinskii V, De Groot A (2000) Twenty years of brassinosteroids: steroidal plant hormones warrant better crops for the XXI century. Ann Bot 86:441–447
Kitajima M, Butler WL (1975) Excitation spectra for photosystem I and photosystem II in chloroplasts and the spectral characteristics of the distributions of quanta between the two photosystems. Biochimicaet Biophysica Acta 408:297–305
Li YH, Liu YJ, Xu XL, Jin M, An LZ, Zhang H (2012) Effect of 24-epibrassinolide on drought stress-induced changes in Chorisporabungeana. BiolPlantarum 56:192–196
Liu ZJ, Guo YK, Bai JG (2010) Exogenous hydrogen peroxide changes antioxidant enzyme activity and protects ultrastructure in leaves of two cucumber ecotypes under osmotic stress. J Plant Growth Regul 29:171–183
Melkozernov AN (2006) Photosynthetic functions of chlorophylls. In: Blankenship RE, Grimm B, Porra RJ, Rüdiger W, Scheer H (eds) Chlorophylls and bacteriochlorophylls: biochemistry, biophysics, functions and applications. Springer, Dordrecht, pp 397–412
Ogweno JO, Song XS, Shi K, Hu WH, Mao WH, Zhou YH, You JQ (2008) Nogués S.Brassinosteroids alleviate heat-induced inhibition of photosynthesis by increasing carboxylation efficiency and enhancing antioxidant systems in Lycopersicon esculentum. J Plant Growth Regul 27:49–57
Pääkkönen E, Vahala J, Pohjola M, Holopainen T, Kärenlampi L (1998) Physiological, stomatal and ultrastructural ozone responses in birch (Betula pendula Roth.) are modified by water stress. Plant Cell Environ 21:671–684
Qi W, Tan H, Zhai H (2006) Photosynthetic characters and fluorescence parameters of different grape stocks under water stress. Chin J Appl Ecol 17:835–838
Qin HY, Ai J, Xu PL, Wang ZX, Zhao Y, Yang YM, Fan ST, Shen YJ (2013) Chlorophyll fluorescence parameters and ultrastructure in amur grape (Vitis amurensis Rupr.) under salt stress. Acta Bot Boreal 33:1159–1164
Sasse J (1999) Physiological actions of brassinosteroids. In: Sakurai A, Yokota T, Clouse SD (eds) Brassinosteroids: steroidal plant hormones. Springer Verla gGmbh, Tokyo, pp 137–161
Sasse JM (2003) Physiological actions of brassinosteroids: an update. J Plant Growth Regul 22:276–288
Sayed OH (2003) Chlorophyll fluorescence as a tool in cereal crop research. Photosynthetica 41:321–330
Shahbaz M, Ashraf M, Athar HR (2008) Does exogenous application of 24-epibrassinolide ameliorate salt induced growth inhibition in wheat (Triticuma estivumL.)? Plant Growth Regul 55:51–61
Swamy KN, Rao SSR (2008) Influence of 28-homobrassinolide on growth, photosynthesis metabolite and essential oil content of geranium (Pelargonium graveolens (L.) Herit). Amer J Plant Physiol 3:173–179
Wu XX, Zhu ZW, Yao XF, Zhang H, Chen JL, Zha DS (2014) Brassinosteroids protect photosynthesis and antioxidant system of eggplant seedlings from high-temperature stress. Acta Physiol Plant 36:251–261
Xi ZM, Sun WJ, Zhang ZW (2007) Effect of exogenous Ca2+ on drought resistance physiological indexes of wine grape cultivar Pinot Noir under water stress. J Northwest A&F Univ 35:137–146
Xu S, Li JL, Zhang XQ, Wei H, Cui LJ (2006) Effects of heat acclimation pretreatment on changes of membrane lipid peroxidation, antioxidant metabolites, and ultrastructure of chloroplasts in two cool-season turfgrass species under heat stress. Environ Exp Bot 56:274–285
Yu JQ, Huang LF, Hu WH, Zhou YH, Mao WH, Ye SF, Nogués S (2004) A role for brassinosteroids in the regulation of photosynthesis in Cucumis sativus. J Exp Bot 55:1135–1143
Yuan GF, Jia CG, Li Z, Sun B, Zhang LP, Liu N, Wang QM (2010) Effect of brassinosteroids on drought resistance and abscisic acid concentration in tomato under water stress. Sci Hortic 126:103–108
Zhang GL, Chen LY, Zhang ST, Zheng H, Liu GH (2009) Effects of high temperature stress on microscopic and ultrastructural characteristics of mesophyll cells in flag leaves of rice. Rice Sci 16:65–71
Zhou SX, Duursma RA, Medlyn BE, Kelly Jeff WG, Prentice IC (2013) How should we model plant responses to drought? An analysis of stomatal and non-stomatal responses to water stress. Agr Forest Meteorol 182–183:204–214
Acknowledgments
This study was supported by the National Technology System for Grape Industry (CARS-30-zp-9), the Natural Science Foundation of Shaanxi Province (2011JM3004). Thanks for the Key Laboratory of Horticultural Plant Biology and Germplasm Innovation in Northwest, Ministry of Agriculture of China. The authors are obliged to Tong Lu, M.S (Texas Christian University), who provided some useful comments on an earlier draft of this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P.K. Nagar.
Rights and permissions
About this article
Cite this article
Wang, Z., Zheng, P., Meng, J. et al. Effect of exogenous 24-epibrassinolide on chlorophyll fluorescence, leaf surface morphology and cellular ultrastructure of grape seedlings (Vitis vinifera L.) under water stress. Acta Physiol Plant 37, 1729 (2015). https://doi.org/10.1007/s11738-014-1729-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-014-1729-z