Abstract
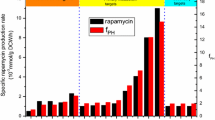
A chemically defined medium was developed which satisfies the nutritional needs of theStreptomyces hygroscopicus strain producing the immunosuppressant rapamycin. More than a doubling of rapamycin biosynthesis was observed on addition of 57 mmol/L of exogenous shikimate. The previously observed inhibition of the synthesis by phenylalanine was not reversed by shikimate.
Similar content being viewed by others
References
Byrne K., Shafee A., Nielsen J.B., Arison B., Monaghan R.L., Kaplan L.: The biosynthesis and enzymology of an immunosuppressant, immunomycin, produced byStreptomyces hygroscopicus var.ascomyceticus.Dev. Ind. Microbiol.32, 29–45 (1993).
Cheng Y.R., Hauck L., Demain A.L.: Phosphate, ammonium, magnesium and iron nutrition ofStreptomyces hygroscopicus with respect to rapamycin biosynthesis.J. Ind. Microbiol.14, 424–427 (1995a).
Cheng Y.R., Fang A., Demain A.L.: Effect of amino acids on rapamycin biosynthesis byStreptomyces hygroscopicus.Appl. Microbiol. Biotechnol.43, 1096–1098 (1995b).
Findlay J.A., Radics L.: On the chemistry and high field nuclear magnetic resonance spectroscopy of rapamycin.Can. J. Chem.58, 579–590 (1980).
Floss H.G., Cho H., Casati R., Reynolds K.A., Kennedy E., Moore B.S., Belae J.M., Mocek V.M., Poralla K.: Diversions of the shikimate pathway—The biosynthesis of cyclohexane-carboxylic acid, pp. 77–88, in:Secondary-Metabolite Biosynthesis and Metabolism (R.J. Petroski and S.P. McCormick, Eds). Plenum, New York 1992.
Hatanaka H., Kino T., Miyata S., Imunara N., Kuroda A., Goto T., Tanaka H., Okuhara M.: FR-900520 and FR-900523, novel immunosuppressants isolated fromStreptomyces. 2. Fermentation, isolation and physico-chemical and biological characteristics.J. Antibiot.41, 1592–1601 (1987).
Kino T., Hatanaka H., Hashimoto M., Nishiyama M., Goto T., Okuhara M., Kohsaka M., Aoki H., Imanaka H.: FK-506, a novel immunosuppressant isolated from aStreptomyces. 1. Fermentation, isolation, and physico-chemical and biological characteristics.J. Antibiot.40, 1249–1255 (1987).
Kojima I., Cheng Y.R., Mohan V., Demain A.L.: Carbon source nutrition of rapamycin biosynthesis inStreptomyces hygroscopicus.J. Ind. Microbiol.14, 436–439 (1995).
Morris R.E.: Rapamycins: antifungal, antitumor, antiproliferative, and immunosuppressive macrolides.Transpl. Rev.16, 39–87 (1992).
Paiva N.L., Demain A.L., Roberts M.F.: Incorporation of acetate, propionate, and methionine into rapamycin byStreptomyces hygroscopicus.J. Nat. Prod.54, 167–177 (1991).
Paiva N.L., Roberts M.F., Demain A.L.: The cyclohexane moiety of rapamycin derived from shikimic acid inStreptomyces hygroscopicus.J. Ind. Microbiol.12, 423–428 (1993).
Sehgal S.N., Molnar-Kimber K., Ocain T.D., Weichman B.M.: Rapamycin: a novel immunosuppressive macrolide.Med. Res. Rev.14, 1–22 (1994).
Tanaka H., Kuroda A., Marusawa H., Hatanaka H., Kino T., Goto M., Hashimoto M., Taga S.T.: Structure of FK506: a novel immunosuppressant isolated fromStreptomyces.J. Amer. Chem. Soc.109, 5031–5033 (1987).
Vezina C., Kudelski A., Sehgal S.N.: Rapamycin (AY-22 989) a new antifungal antibiotic. 1. Taxonomy of the producing streptomycete and isolation of the active principle.J. Antibiot.28, 721–726 (1975).
Vining L.C.: Antibiotic biosynthesis.Biotech. Adv.3, 171–194 (1985).
Wallace K.K., Reynolds K.A., Koch K., McArthur H.A.I., Brown M.S., Wax R.G., Moore B.S.: Biosynthetic studies of ascomycin (FK520): formation of the (1R, 3R, 4R)-3,4-dihydroxycyclohexane carboxylic acid-derived moiety.J. Amer. Chem. Soc.116, 11600–11601 (1994).
Werner R.G.: Secondary metabolites with antibiotic activity from the primary metabolism of aromatic amino acids.Prog. Clin. Biochem. Med.1, 47–115 (1984).
Author information
Authors and Affiliations
Additional information
Dedicated to Dr. J. Spížek on the occasion of his 60th birthday
Rights and permissions
About this article
Cite this article
Fang, A., Demain, A.L. Exogenous shikimic acid stimulates rapamycin biosynthesis inStreptomyces hygroscopicus . Folia Microbiol 40, 607–610 (1995). https://doi.org/10.1007/BF02818516
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02818516