Summary
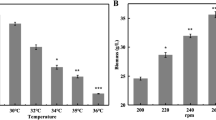
Phosphate, ammonium and magnesium salts interfered with rapamycin production byStreptomyces hygroscopicus at concentrations optimal for growth. These observations point to the existence of phosphorus, magnesium and nitrogen-negative regulation mechanisms for rapamycin biosynthesis. On the other hand, Fe2+ stimulated rapamycin production at concentrations greater than that required for growth.
Similar content being viewed by others
References
Brana, A.F. and A.L. Demain. 1988. Nitrogen control of antibiotic biosynthesis in actinomycetes. In: Nitrogen Source Control of Microbial Processes (Sanchez-Esquivel, S., ed.), pp. 99–119. CRC Press, Boca Raton, Florida.
Dreyfuss, M., E. Harri, H. Hofmann, H. Kobel, W. Pache and H. Tscherter. 1976. Cyclosporine A and C, new metabolites fromTrichoderma polysporum (Link et Pers.) Rifai. Appl. Microbiol. 3: 125–133.
Kojima, I., Y.R. Cheng, V. Mohan and A.L. Demain. 1995. Carbon source nutrition of rapamycin biosynthesis inStreptomyces hygroscopicus. J. Ind. Microbiol. (in press).
Martin, J.F. 1988. Molecular mechanisms for the control by phosphate of the biosynthesis of antibiotics and other secondary metabolites. In: Regulation of Secondary Metabolism in Actinomycetes (Shapiro, S., ed.), pp. 213–237. CRC Press, Boca Raton, Florida.
Morris, R.E. 1992. Rapamycins: antifungal, antitumor, antiproliferative, and immunosuppressive macrolides. Transpl. Rev. 16: 39–87.
Omura, S. and Y. Tanaka 1984. Biochemistry, regulation and genetics of macrolide production. In: Macrolide Antibiotics: Chemistry, Biology, and Practice (Omura, S., ed.), pp. 199–229, Academic Press, New York.
Paiva, N.L., A.L. Demain and M.F. Roberts. 1991. Incorporation of acetate, propionate, and methionine into rapamycin byStreptomyces hygroscopicus. J. Nat. Prods. 54: 167–177.
Paiva, N.L., A.L. Demain and M.F. Roberts. 1993. The immediate precursor of the nitrogen-containing ring of rapamycin is free pipecolic acid. Enzyme Microb. Technol. 15: 581–585.
Paiva, N.L., M.F. Roberts and A.L. Demain. 1993. The cyclohexane moiety of rapamycin is derived from shikimic acid inStreptomyces hygroscopicus. J. Ind. Microbiol. 12: 423–428.
Sehgal, S.N., K. Molnar-Kimber, T.D. Ocain and B.M. Weichman. 1994. Rapamycin: a novel immunosuppressive macrolide. Med. Res. Rev. 14: 1–22.
Shapiro, S. 1988. Nitrogen assimilation in actinomycetes and the influence of nitrogen nutrition on actinomycete secondary metabolism. In: Regulation of Secondary Metabolism in Actinomycetes (Shapiro, S., ed.), pp. 135–221, CRC Press, Boca Raton, Florida.
Tanaka, H., H. Kuroda, H. Marusawa, T. Hatanaka, T. Kino, T. Goto, M. Hashimoto and T. Taga. 1987. Structure of FK506: a novel immunosuppressant isolated fromStreptomyces. J. Amer. Chem. Soc. 109: 5031–5033.
Tkacz, J.S., R.A. Giacobbe and R.L. Monaghan. 1993. Improvement in the titer of echinocandin-type antibiotics: a magnesiumlimited medium supporting the biphasic production of pneumocandins Ao and Bo. J. Ind. Microbiol. 11: 95–103.
Vezina, C., A. Kudelski and S.N. Sehgal. 1975. Rapamycin (AY-22, 989), a new antifungal antibiotic: I. Taxonomy of the producting streptomycete and isolation of the active principle. J. Antibiot. 28: 721–726.
Weinberg, E.D. 1988. Roles of micronutrients in secondary metabolism of actinomycetes. In: Regulation of Secondary Metabolism in Actinomycetes (Shapiro, S., ed.), pp. 239–261, CRC Press, Boca Raton, Florida.
Weinberg, E.D. 1962. Trace metal control of specific biosynthetic processes. Persp. Biol. Med. 5: 432–445.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cheng, Y.R., Hauck, L. & Demain, A.L. Phosphate, ammonium, magnesium and iron nutrition ofStreptomyces hygroscopicus with respect to rapamycin biosynthesis. Journal of Industrial Microbiology 14, 424–427 (1995). https://doi.org/10.1007/BF01569962
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01569962