Summary
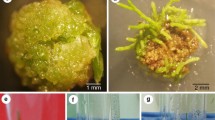
Gene transfer and plant regeneration systems have been developed for European larch (Larix decidua Mill.) in our laboratory. Aseptically germinated young seedlings were hypocotyl wound-inoculated withAgrobacterium rhizogenes strains 11325 containing a wild-type Ri (root-inducing) plasmid. Swollen stems appeared at infected wounds followed by either abundant hairy roots or adventitious shoot buds that developed within 3 to 4 wk after inoculation. No symptoms were seen on wounded but uninoculated seedlings. We demonstrated agrobacteria attached to larch cells by examination of scanning electron micrographs. Subsequently, calli derived from symptomatic tissues exhibited phytohormone autotrophic growth. Adventitious buds were elongated and rooted in vitro before being transferred to the greenhouse where the transformed whole plants grew normally. Transformants tested positive for opine production and transformation was further confirmed by Southern blot analysis with larch genomic DNAs isolated from both proliferated calli and needle tissue of transgenic plants.
Similar content being viewed by others
References
Ahuja, M. R. Gene transfer in forest trees. In: Hanover, J.; Kiethley, D., eds. Genetic manipulation of woody plants. New York: Plenum Press; 1988:25–41.
Anderson, A. R.; Moore, L. W. Host specificity in the genusAgrobacterium. Phytopathology 69:320–323; 1979.
Bekkaoui, F.; Detla, R. S. S.; Pilon, M., et al. The effects of promoter on transient expression in conifer cell lines. Theor. Appl. Genet. 79:353–359; 1990.
Birnboim, M. C.; Doly, J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 7:1513–1523; 1979.
Brown, C. L.; Lawrence, R. H. Culture of pine callus on a defined medium. For. Sci. 14:62–64; 1968.
Chiang, V. L.; Mroz, G. D.; Shaler, S. M., et al. Pulp production of a 16-year-old larch stand. Tappi J. 71:179–180; 1988.
Chilton, M.-D.; Farrand, S. K.; Bendich, A. J., et al.Agrobacterium tumefaciens DNA and PS8 bacteriophage DNA not detected in crown gall tumors. Proc. Natl. Acad. Sci. USA 71:3672–3676; 1974.
Dandekar, A. M.; Gupta, P. K.; Durzan, D. J., et al. Transformation and foreign gene expression in micropropagated Douglas-firPseudotsuga menziesii. Bio/Technology 5:587–590; 1987.
Davis, J. M.; Keathley, D. E. Detection and analysis of T-DNA in crown gall tumors and kanamycin-resistant callus ofRobinia pseudoacacia. Can. J. For. Res. 19:1118–1123; 1989.
De Cleene, M.; De Ley, J. The host range of crown gall. Bot. Rev. 42:389–446; 1976.
Diner, A. M.; Strickler, A.; Karnosky, D. F. Initiation, elongation and remultiplication ofLarix decidua micropropagules. N. Z. J. For. Sci. 16:306–318; 1986.
Diner, A. M.; Karnosky, D. F. Differential responses of two conifers toin vitro inoculation withAgrobacterium rhizogenes. Eur. J. For. Pathol. 17:211–216; 1987.
Ellis, D. D.; Wayner, W. R.; Roberts, D. R., et al. The effect of antibiotics on elongation and callus and bud formation from embryonic tissue ofPicea glauca. Can. J. For. Res. 19:1343–1346; 1989.
Einspahr, D. W.; Wyckoff, G. W.; Fiscus, M. H. Larch: A fast-growing fiber source for the Lake States and Northeast. J. For. 82:104–106; 1984.
Feinberg, A. P.; Vogelstein, B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal. Biochem. 132:6–13; 1983.
Fillatti, J. J.; Sellmer, J.; McCown, B., et al.Agrobacterium-mediated transformation and regeneration ofPopulus. Mol. Gen. Genet. 206:192–199; 1987.
Gresshoff, P. M.; Doy, C. H. Development and differentiation of haploidLycopersicon esculentum (Tomato). Plants 107:161–170; 1972.
Hauptmann, R. M.; Widholm, J. M.; Paxton, J. D. Benomyl: a broad spectrum fungicide for use in plant cell and protoplast culture. Plant Cell Rep. 4:129–132; 1985.
Hood, E. E.; Clapham, D. H.; Ekberg, I., et al. T-DNA presence and opine production in tumors ofPicea abies (L.) Karst induced byAgrobacterium tumefaciens A281. Plant Mol. Biol. 14:111–117; 1990.
Huang, Y.; Shin, D. I.; Karnosky, D. F. Evidence forAgrobacterium-mediated genetic transformation inLarix decidua. In: Ahuja, M. R., ed. Biotechnology of woody plant. New York: Plenum Press; 1991:In press.
Huang, Y.; Stokke, D. D.; Barnes, W. M., et al. Virulence ofAgrobacterium onLarix decidua and their cellular interactions as depicted by scanning electron microscopy. Plant Cell Reports. Submitted.
Karnosky, D. F.; Diner, A. M. A cotyledon culture system for cloningLarix decidua andPinus banksiana. In: TAPPI research and Development Conference. Technical Association of Pulp and Paper Industry, Atlanta, Georgia. 1984:13–15.
Karnosky, D. F.; Mulcahey, A. Explant orientation and media manipulation affectLarix decidua adventitious bud quality and quantity. In Vitro Cell. Dev. Biol. 24:51A; 1988.
Klimaszewska, K. Recovery of somatic embryos and plantlets from protoplast cultures ofLarix X eurolepis. Plant Cell Rep. 8:440–444; 1989.
Laliberte, S.; Lalonde, M. Sustained caulogenesis in callus cultures ofLarix X eurolepis initiated from short-shoot buds of a 12-year-old tree. Am. J. Bot. 75:767–777; 1988.
Langor, D. W.; Raske, A. G. The Eastern larch beetle: another threat to our forestsColeoptera: Scolytidae). For. Chron. 65:276–279; 1989.
Lanier, G. N. Larch beetleDendroctonus simplex. In: Forest insect and disease problems for New York State, 1980–1981. Committee on Insects and Diseases. New York Section, Syracuse, New York; 1981:3.
Lippincott, J. A.; Lippincott, B. B. Tumor-initiating ability and nutrition in the genusAgrobacterium. J. Gen. Microbiol. 59:57–75; 1969.
Litvay, J. D.; Johnson, M. A.; Verma, D., et al. Conifer suspension culture medium development using analytical data from developing seeds. Inst. Paper Chem. Technical Paper Series, Institute of Paper Chemistry, Appleton, WI. no. 115:1–71; 1981.
Loopstra, C. A.; Stomp, A.-M.; Sederoff, R. R.Agrobacterium-mediated DNA transfer in sugar pine. Plant Mol. Biol. 15:1–9; 1990.
Mackay, J.; Seguin, A.; Lalonde, M. Genetic transformation of ninein vitro clones ofAlnus andBetula byAgrobacterium tumefciens. Plant Cell Rep. 7:229–232; 1988.
Maniatis, T.; Fritsch, E. F.; Sambrook, J. Molecular cloning: a laboratory manual. New York: Cold Spring Harbor Laboratory; 1982:76–389.
Matthysse, A. G.; Holmes, K. V.; Gurlitz, R. H. G. Elaboration of cellulose fibrils byAgrobacterium tumefaciens during attachment to carrot cell. J. Bacteriol. 145:583–585; 1981.
McGranahan, G. H.; Leslie, C. A.; Uratsu, S. L., et al.Agrobacterium-mediated transformation of walnut somatic embryos and regeneration of transgenic plants. Bio/Technology 6:800–804; 1988.
Mehra-Palta, A.; Smeltzer, R. H.; Mott, R. L. Hormonal control of induced organogenesis: Experiments with excised plant parts of loblolly pine. Tappi J. 61:37–40; 1978.
Moore, L. W.; Anderson, A.; Kado, C. I. Gram-negative bacterium:Agrobacterium. In: Schaad, N. W., ed. Laboratory guide for identification of plant pathogenic bacteria. St. Paul, MN: American Phytopathological Society; 1980:22–23.
Mroz, G. D.; Reed, D. D.; Liechty, H. O. Volume production of a 16-year-old European larch stand. North. J. Appl. For. 5:160–161; 1988.
Nagmani, R.; Bonga, J. M. Embryogenesis in subcultured callus ofLarix decidua. Can. J. For. Res. 15:1088–1091; 1985.
Noda, T.; Tanaka, N.; Mano, Y., et al. Regeneration of horseradish hairy roots incited byAgrobacterium rhizogenes infection. Plant Cell Rep. 6:283–286; 1987.
Ooms, G.; Hooykaas, P. J. J.; Moolenaar, G., et al. Crown gall plant tumors of abnormal morphology induced byAgrobacterium tumefaciens carrying mutant octopine Ti plasmids: Analysis of T-DNA functions. Gene 14:33–50; 1981.
Otten, L.; DeGreve, H.; Hernalsteens, J. P., et al. Mandelian transmission of genes introduced into plants by the Ti plasmids ofAgrobacterium tumefaciens. Mol. Gen. Genet. 183:209–213; 1981.
Otten, L. A. B. M.; Schilperoort, R. A. A rapid micro-scale method for the detection of lysopine and nopaline dehydrogenase activities. Biochem. Biophys. Acta 527:497–500; 1978.
Petit, A.; David, C.; Dahl, G. A., et al. Further extension of the opine concept: plasmids inAgrobacterium tumefaciens cooperate for opine degradation. Mol. Gen. Genet. 190:204–214; 1983.
Shahin, E. A. Totipotency of tomato protoplasts. Theor. Appl. Genet. 69:235–241; 1985.
Stachel, S. E.; Messens, E.; Van Montagu, M., et al. Identification of the sign molecules produced by wounded plant cells that activate T-DNA transfer inAgrobacterium tumefaciens. Nature 318:624–629; 1985.
Tepfer, M.; Casse-Delbart, F.Agrobacterium rhizogenes as a vector for transforming higher plants. Microbiol. Sci. 4:24–28; 1987.
Von Aderkas, P.; Bonga, J. M. Formation of haploid embryoids ofLarix decidua: Early embryogenesis. Am. J. bot. 75:619–628; 1988.
White, F. F.; Ghidossi, G.; Gordon, M. P., et al. Tumor induction byAgrobacterium rhizogenes involves the transfer of plasmid DNA to the plant genome. Proc. Natl. Acad. Sci. USA 79:3193–3197; 1982.
White, F. F.; Nester, E. W. Relationship of plamids responsible for hairy root and crown gall tumorigenicity. J. Bacteriol. 144:710–720; 1980.
Wullems, G. J.; Molendijk, L.; Schilperoort, R. Differential expression of crown gall tumor markers in transformants obtained afterin vitro Agrobacterium tumefaciens-induced transformation of cell wall regenerating protoplasts derived fromNicotiana tabacum. Proc. Natl. Acad. Sci. USA 78:4344–4348; 1981.
Yang, F.; Simpson, R. B. Revertant seedlings from crown gall tumors retain a portion of the bacterial Ti plasmid DNA sequences. Proc. Natl. Acad. Sci. USA 78:4151–4155; 1981.
Zimmer, E. A.; Rivin, C. J.; Walbot, V. A DNA isolation procedure suitable for most higher plant species. Plant. Mol. Biol. Newsletter. 2:93–96; 1981.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Huang, Y., Diner, A.M. & Karnosky, D.F. Agrobacterium rhizogenes-mediated genetic transformation and regeneration of a conifer:Larix decidua . In Vitro Cell Dev Biol - Plant 27, 201–207 (1991). https://doi.org/10.1007/BF02632217
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02632217