Summary
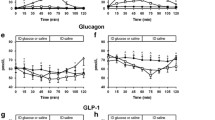
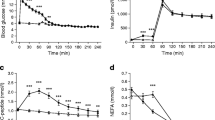
We aimed to assess prandial responses, basal glucose turnover and insulin action (euglycaemic clamp) in a very low-dose neonatal streptozotocin model of Type 2 (noninsulin-dependent) diabetes mellitus. Male Wistar rats were injected at 2 days of age with 45 mg/kg streptozotocin or vehicle (control). At 8 weeks, the groups were subdivided and fed either a high-fat or high-starch diet for 3 weeks. Both the fat diet and streptozotocin treatments had independent hyperglycaemic effects (streptozotocin/fat 9.3±0.3 mmol/l; streptozotocin/starch 7.5±0.3 mmol/l; control/fat 7.4±0.1 mmol/l; all p<0.01 vs control/starch 6.4±0.1 mmol/l). The fat diet effect was associated with both a reduction in basal glucose clearance (p<0.001) and in basal hepatic glucose output (p<0.05). Streptozotocin increased basal hepatic glucose output. Significantly higher prandial glycaemia in the streptozotocin/starch group occurred despite similar insulin levels and appeared to be related to an impaired early insulin response. Whole-body and tissue-specific insulin sensitivity were significantly depressed in fat-fed animals compared to starch-fed animals, however there were no significant effects of streptozotocin treatment. We conclude that fasting hyperglycaemia associated with abnormalities in both glucose production and clearance can exist in the presence of a basal hepatic glucose output which is reduced compared to control animals. Furthermore, dietary-fat-induced insulin resistance is not exacerbated by the relative insulin deficiency and/or mild hyper glycaemia observed when dietary fat and neonatal streptozotocin-treatments are combined.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pascoe WS, Storlien LH (1990) Inducement of basal hyperglycaemia in rats with abnormal β-cell function by fat-feeding: model for study of aetiology and pathogenesis of NIDDM. Diabetes 39: 226–233
Holman RR, Turner RC (1980) The basal plasma glucose: a simple relevant index of maturity-onset diabetes. Clin Endocrinol 14: 279–286
Zimmet P, Whitehouse S, Alford F, Chisholm D (1978) The relationship of insulin response to a glucose stimulus over a wide range of glucose tolerance. Diabetologia 15: 23–27
Halter JB, Graf RJ, Porte D Jr (1979) Potentiation of insulin secretory responses by plasma glucose levels in man: evidence that hyperglycaemia in diabetes compensates for impaired glucose potentiation. J Clin Endocrinol Metab 48: 946–954
Ward WK, Bolgiano DC, McKnight B, Halter JB, Porte D Jr (1984) Diminished β cell secretory capacity in patients with noninsulin-dependent diabetes mellitus. J Clin Invest 74: 1318–1328
Portha B, Picon L, Rosselin G (1979) Chemical diabetes in the adult rat as the spontaneous evolution of neonatal diabetes. Diabetologia 17: 371–377
Kraegen EW, James DE, Jenkins AB, Chisholm DJ (1985) Doseresponse curves for in vivo insulin sensitivity in individual tissues in rats. Am J Physiol 248: E353-E362
Steele R, Watts JS, DeBodo RC, Altszuler N (1959) Measurement of size and turnover rate of body glucose pool by the isotope dilution method. Am J Physiol 187: 15–24
Hansen SA (1975) Thin-layer chromatographic method for identification of oligosaccharides in starch hydrozylates. J Chromatogr 105: 380–390
Morgan CR, Lazarow A (1963) Immunoassay of insulin. Two antibody system: plasma insulin levels of normal, subdiabetic and diabetic rats. Diabetes 12: 115–126
Storlien LH, James DE, Burleigh KM, Chisholm DJ, Kraegen EW (1986) Fat feeding causes widespread in vivo insulin resistance, decreased energy expenditure and obesity in the rat. Am J Physiol 251: E576-E583
Kergoat M, Portha B (1985) In vivo hepatic and peripheral insulin sensitivity in rats with non-insulin-dependent diabetes induced by streptozotocin. Assessment with the insulin-glucose clamp technique. Diabetes 34: 1120–1126
Hother-Nielsen O, Beck-Nielsen H (1990) On the determination of basal glucose production rate in patients with Type 2 (non-insulin-dependent) diabetes mellitus using primed-continuous 3-3H-glucose infusion. Diabetologia 33: 603–610
Heath D (1990) Errors inherent in the primed infusion method for the measurement of the rate of glucose appearance in man when uptake is not forced by glucose or insulin infusion. Clin Sci 79: 201–213
Liljenquist JE, Mueller GL, Cherrington AD, Perry JM, Rabinowitz D (1979) Hyperglycaemia per se (insulin and glucagon withdrawn) can inhibit hepatic glucose production in man. J Clin Endocrinol Metab 48: 171–175
Thorburn AW, Storlien LH, Jenkins AB, Khouri S, Kraegen EW (1989) Fructose-induced in vivo insulin resistance and elevated plasma triglyceride levels in rats. Am J Clin Nutr 49: 1155–1163
Storlien LH, Jenkins AB, Chisholm DJ, Pascoe WS, Khouri S, Kraegen EW (1991) Influence of dietary fat composition on development of insulin resistance in rats: relationship to muscle triglyceride and ω-3 fatty acids in muscle phospholipid. Diabetes 40: 280–289
Bruce DG, Chisholm DJ, Storlien LH, Kraegen EW (1988) The physiological importance of the deficiency in early prandial insulin secretion in non-insulin dependent diabetes. Diabetes 37: 736–744
Kendall DM, Sutherland DER, Najarian JS, Goetz FC, Robert-son RP (1990) Effects of hemipancreatectomy on insulin secretion and glucose tolerance in healthy humans. N Engl J Med 322: 898–903
DeFronzo RA (1988) The triumvirate: β-cell, muscle, liver. A collusion responsible for NIDDM. Diabetes 37: 667–687
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pascoe, W.S., Jenkins, A.B., Kusunoki, M. et al. Insulin action and determinants of glycaemia in a rat model of Type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 35, 208–215 (1992). https://doi.org/10.1007/BF00400919
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00400919