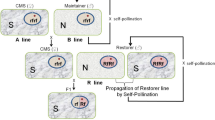
Abstract
This chapter retraces the history of using somatic hybridization to modify the cytoplasmic male sterility (CMS)-inducing Ogura radish cytoplasm and its application in hybrid seed production for Brassica crops. First, we present hybrid seed production and the issues behind the introduction of the Ogura radish cytoplasm into Brassica crops. We then detail the procedures of the protoplast fusion experiments used to produce cybrids. Recombination between radish and Brassica mitochondrial genomes ultimately led to the creation of the Ogu-INRA system. Not only have cybrids contributed to hybrid seed production but also molecular analysis of cybrids helped identify the mitochondrial gene causing CMS, and contributed basic knowledge on plant mitochondrial gene expression. Furthermore, restoration of male fertility is useful for some crops, and the Ogu-INRA system has also been used to identify restorer genes. The Ogu-INRA system is now widely used in agriculture.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Akagi H, Taguchi T, Fujimura T (1995) Stable inheritance and expression of the CMS traits introduced by asymmetric protoplast fusion. Theor Appl Genet 91:563–567. doi:10.1007/BF00223280
Ashutosh KP, Dinesh Kumar V, Sharma PC, Prakash S, Bhat SR (2008) A novel orf108 co-transcribed with the atpA gene is associated with cytoplasmic male sterility in Brassica juncea carrying Moricandia arvensis cytoplasm. Plant Cell Physiol 49:284–289. doi:10.1093/pcp/pcm182
Bannerot H, Boulidard L, Cauderon Y, Tempé J (1974) Transfer of cytoplasmic male sterility from Raphanus sativus to Brassica oleracea. In: Eucarpia Meeting Cruciferae. pp 52–54
Bassene J, Berti L, Carcouet E, Dhuique-Mayer C, Fanciullino AL, Bouffin J, Ollitrault P, Froelicher Y (2008) Influence of mitochondria origin on fruit quality in a citrus cybrid. J Agric Food Chem 56:8635–8640
Beal WJ (1880) Indian corn. Rept Michigan State Board Agric 279–289.
Bellaoui M, Pelletier G, Budar F (1997) The steady-state level of mRNA from the Ogura cytoplasmic male sterility locus in Brassica cybrids is determined post-transcriptionally by its 3ʹ region. EMBO J 16:5057–5068. doi:10.1093/emboj/16.16.5057
Bellaoui M, Martin-Canadell A, Pelletier G, Budar F (1998) Low-copy-number molecules are produced by recombination, actively maintained and can be amplified in the mitochondrial genome of Brassicaceae: relationship to reversion of the male sterile phenotype in some cybrids. Mol Gen Genet 257:177–185
Bellaoui M, Grelon M, Pelletier G, Budar F (1999) The restorer Rfo gene acts post-translationally on the stability of the ORF138 Ogura CMS-associated protein in reproductive tissues of rapeseed cybrids. Plant Mol Biol 40:893–902
Belliard G, Vedel F, Pelletier G (1979) Mitochondrial recombination in cytoplasmic hybrids of Nicotiana tabacum by protoplast fusion. Nature 281:401–403. doi:10.1038/281401a0
Beversdorf WD, Weiss-Lerman J, Erickson LR, Souza-Machado V (1980) Transfer of cytoplasmically inherited triazine resistance from birds-rape to cultivated oil seed rape (B. campestris and B. napus). Can J Genet Cytol 22:167–172
Binder S, Brennicke A (2003) Gene expression in plant mitochondria: transcriptional and post-transcriptional control. Philos Trans R Soc Lond, B, Biol Sci 358:181–8; discussion 188–9. doi:10.1098/rstb.2002.1179
Bonhomme S, Budar F, Férault M, Pelletier G (1991) A 2.5 kb NcoI fragment of Ogura radish mitochondrial DNA is correlated with cytoplasmic male-sterility in Brassica cybrids. Curr Genet 19:121–127. doi:10.1007/BF00326293
Bonhomme S, Budar F, Lancelin D, Small I, Defrance MC, Pelletier G (1992) Sequence and transcript analysis of the Nco2.5 Ogura-specific fragment correlated with cytoplasmic male sterility in Brassica cybrids. Mol Gen Genet 235:340–348
Braun CJ, Brown GG, Levings CSI (1992) Cytoplasmic male sterility. In: Hermann R (ed) Cell organelles. Plant Gene Research. Springer, New-York, pp 219–245
Brown GG, Formanová N, Jin H, Wargachuk R, Dendy C, Patil P, Laforest M, Zhang J, Cheung WY, Landry BS (2003) The radish Rfo restorer gene of Ogura cytoplasmic male sterility encodes a protein with multiple pentatricopeptide repeats. Plant J 35:262–272
Budar F, Pelletier G (2001) Male sterility in plants: occurrence, determinism, significance and use. C R Acad Sci III Sci Vie 324:543–550
Budar F, Touzet P, De Paepe R (2003) The nucleo-mitochondrial conflict in cytoplasmic male sterilities revisited. Genetica 117:3–16
Cardi T, Earle ED (1997) Production of new CMS Brassica oleracea by transfer of “Anand” cytoplasm from B. rapa through protoplast fusion. Theor Appl Genet 94:204–212
Chatterjee G, Sikdar SR, Das S, Sen SK (1988) Intergeneric somatic hybrid production through protoplast fusion between Brassica juncea and. Diplotaxis muralis Theor Appl Genet 76:915–922
Chetrit P, Mathieu C, Vedel F, Pelletier G, Primard C (1985) Mitochondrial DNA polymorphism induced by protoplast fusion in Cruciferae. Theor Appl Genet 69:361–366
Darwin C (1876) The effects of cross and self fertilisation in the vegetable kingdom. John Murray, London
Delourme R, Eber F (1992) Linkage between an isozyme marker and a restorer gene in radish cytoplasmic male sterility of rapeseed (Brassica napus L.). Theor Appl Genet 85:222–228. doi: 10.1007/BF00222863
Delourme R, Eber F, Renard M (1994) Transfer of radish cytoplasmic male sterility from Brassica napus to B. juncea and B. rapa. Eucarpia Cruciferae Newsletter 79.
Delourme R, Foisset N, Horvais R, Barret P, Champagne G, Cheung WY, Landry BS, Renard M (1998) Characterisation of the radish introgression carrying the Rfo restorer gene for the Ogu-INRA cytoplasmic male sterility in rapeseed (Brassica napus L.). Theor Appl Genet 97:129–134
Desloire S, Gherbi H, Laloui W, Marhadour S, Clouet V, Cattolico L, Falentin C, Giancola S, Renard M, Budar F, Small I, Caboche M, Delourme R, Bendahmane A (2003) Identification of the fertility restoration locus, Rfo, in radish, as a member of the pentatricopeptide-repeat protein family. EMBO Rep 4:588–594. doi:10.1038/sj.embor.embor848
Ducos E, Touzet P, Boutry M (2001) The male sterile G cytoplasm of wild beet displays modified mitochondrial respiratory complexes. Plant J 26:171–180
Duroc Y, Gaillard C, Hiard S, Defrance MC, Pelletier G, Budar F (2005) Biochemical and functional characterization of ORF138, a mitochondrial protein responsible for Ogura cytoplasmic male sterility in Brassiceae. Biochimie 87:1089–1100. doi:10.1016/j.biochi.2005.05.009
Duroc Y, Hiard S, Vrielynck N, Ragu S, Budar F (2009) The Ogura sterility-inducing protein forms a large complex without interfering with the oxidative phosphorylation components in rapeseed mitochondria. Plant Mol Biol 70:123–137. doi:10.1007/s11103-009-9461-6
Falk KC, Rakow G, Downey RK, Spurr DT (1994) Performance of inter-cultivar summer turnip rape hybrids in Saskatchewan. Can J Plant Sci 74:441–445
Fu D (1981) Production and research on rapeseed in the Peoples Republic in China. Eucarpia Cruciferae Newslett 6:6–7
Fu D, Yang GS (1995) Rapeseed heterosis breeding in China. In: 9th International Rapeseed Congress. Cambridge, pp 119–121
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exptl Cell Research 50:151–158
Giancola S, Rao Y, Chaillou S, Hiard S, Martin-Canadell A, Pelletier G, Budar F (2007) Cytoplasmic suppression of Ogura cytoplasmic male sterility in European natural populations of. Raphanus raphanistrum Theor Appl Genet 114:1333–1343. doi:10.1007/s00122-007-0520-6
González-Melendi P, Uyttewaal M, Morcillo CN, Hernández Mora JR, Fajardo S, Budar F, Lucas MM (2008) A light and electron microscopy analysis of the events leading to male sterility in Ogu-INRA CMS of rapeseed (Brassica napus). J Exp Bot 59:827–838. doi:10.1093/jxb/erm365
Gray MW, Lang BF, Cedergren R, Golding GB, Lemieux C, Sankoff D, Turmel M, Brossard N, Delage E, Littlejohn TG, Plante I, Rioux P, Saint-Louis D, Zhu Y, Burger G (1998) Genome structure and gene content in protist mitochondrial DNAs. Nucleic Acids Res 26:865–878
Grelon M, Budar F, Bonhomme S, Pelletier G (1994) Ogura cytoplasmic male-sterility (CMS)-associated orf138 is translated into a mitochondrial membrane polypeptide in male-sterile Brassica cybrids. Mol Gen Genet 243:540–547
Handa H, Bonnard G, Grienenberger JM (1996) The rapeseed mitochondrial gene encoding a homologue of the bacterial protein Ccl1 is divided into two independently transcribed reading frames. Mol Gen Genet 252:292–302
Hanson MR (1991) Plant mitochondrial mutations and male sterility. Annu Rev Genet 25:461–486. doi:10.1146/annurev.ge.25.120191.002333
Hanson MR, Bentolila S (2004) Interactions of mitochondrial and nuclear genes that affect male gametophyte development. Plant Cell 16(Suppl):154–169. doi:10.1105/tpc.015966
Heath DW, Earle ED, Dickson MH (1994) Introgressing cold-tolerant Ogura cytoplasm from rapeseed into pak choi and Chinese cabbage. HortScience 29:202–203
Horlow C, Defrance MC, Pollien JM, Goujaud J, Delon R, Pelletier G (1993) Transfer of cytoplasmic male sterility by spontaneous androgenesis in tobacco (Nicotiana tabacum L.). Euphytica 66:45–53
Iwabuchi M, Koizuka N, Fujimoto H, Sakai T (1999) Identification and expression of the Kosena radish (Raphanus sativus cv. Kosena) homologue of the ogura radish CMS-associated gene, orf138. Plant Mol Biol 39:183–188
Jarl CI, Bornman CH (1988) Correction of chlorophyll‐defective, male‐sterile winter oilseed rape (Brassica napus) through organelle exchange: phenotypic evaluation of progeny. Hereditas 108:97–102
Jarl CI, Grinsven MQJM, Mark F (1989) Correction of chlorophyll-defective male-sterile winter oilseed rape (Brassica napus) through organelle exchange: molecular analysis of the cytoplasm of parental lines and corrected progeny. Theor Appl Genet 77:135–141. doi:10.1007/BF00292328
Jones HA, Clarke AE (1943) Inheritance of male sterility in the onion and the production of hybrid seed. Proc Amer Soc Hort Sci 43:189–194
Kao HM, Keller WA, Gleddie S, Brown GG (1992) Synthesis of Brassica oleracea/Brassica napus somatic hybrid plants with novel organeile DNA compositions. Theor Appl Genet 83:313–320
Kirti PB, Banga SS, Prakash S, Chopra VL (1995) Transfer of Ogu cytoplasmic male sterility to Brassica juncea and improvement of the male sterile line through somatic cell fusion. Theor Appl Genet 91:517–521
Koizuka N, Imai R, Iwabuchi M, Sakai T, Imamura J (2000) Genetic analysis of fertility restoration and accumulation of ORF125 mitochondrial protein in the Kosena radish (Raphanus sativus cv. Kosena) and a Brassica napus restorer line. Theor Appl Genet 100:949–955
Koizuka N, Imai R, Fujimoto H, Hayakawa T, Kimura Y, Kohno-Murase J, Sakai T, Kawasaki S, Imamura J (2003) Genetic characterization of a pentatricopeptide repeat protein gene, orf687, that restores fertility in the cytoplasmic male-sterile Kosena radish. Plant J 34:407–415
Krishnasamy S, Makaroff C (1993) Characterization of the radish mitochondrial orfB locus: possible relationship with male sterility in Ogura radish. Curr Genet 24:156–163
Krishnasamy S, Makaroff CA (1994) Organ-specific reduction in the abundance of a mitochondrial protein accompanies fertility restoration in cytoplasmic male-sterile radish. Plant Mol Biol 26:935–946
Kubo T, Kitazaki K, Matsunaga M, Itchoda N, Estiati A, Mikami T (2011) Male sterility-inducing mitochondrial genomes: how do they differ? Crit Rev Plant Sci 30:378–400
Landgren M, Zetterstrand M, Sundberg E, Glimelius K (1996) Alloplasmic male-sterile Brassica lines containing B. tournefortii mitochondria express an ORF 3ʹ of the atp6 gene and a 32 kDa protein. Plant Mol Biol 32:879–890
Lefort-Buson M, Dattée Y (1982) Genetic study of some agronomic characters in winter oilseed rape (Brassica napus L.) I Heterosis II genetic parameters. Agronomie 2:315–322
L’Homme Y, Brown GG (1993) Organizational differences between cytoplasmic male sterile and male fertile Brassica mitochondrial genomes are confined to a single transposed locus. Nucleic Acids Res 21:1903–1909
L'Homme Y, Stahl RJ, Li X-Q, Hameed A, Brown GG (1997) Brassica nap cytoplasmic male sterility is associated with expression of a mtDNA region containing a chimeric gene similar to the polCMS-associated orf224gene. Curr Genet 31:321–335
Liu JH, Dixelius C, Eriksson I, Glimelius K (1995) Brassica napus(+) B. tournefortii, a somatic hybrid containing traits of agronomic importance for rapeseed breeding. Plant Sci 109:75–86
Luo D, Xu H, Liu Z, Guo J, Li H, Chen L, Fang C, Zhang Q, Bai M, Yao N, Wu H, Wu H, Ji C, Zheng H, Chen Y, Ye S, Li X, Zhao X, Li R, Liu YG (2013) A detrimental mitochondrial-nuclear interaction causes cytoplasmic male sterility in rice. Nat Genet 45:573–577. doi:10.1038/ng.2570
Maltais B, Bouchard CJ (1978) Une moutarde des oiseaux (B. rapa) résistante à l’atrazine. Phytoprotection 59:117–119
Menassa R, El-Rouby N, Brown GG (1997) An open reading frame for a protein involved in cytochrome c biogenesis is split into two parts in Brassica mitochondria. Curr Genet 31:70–79
Menczel L, Morgan A, Brown S, Maliga P (1987) Fusion-mediated combination of Ogura-type cytoplasmic male sterility with Brassica napus plastids using X-irradiated CMS protoplasts. Plant Cell Rep 6:98–101
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Ogura H (1968) Studies on the new male sterility in Japanese radish, with special references to utilization of this sterility towards the practical raising of hybrid seeds. Mem Fac Agric Kagoshima Univ 6:39–78
Okhawa Y, Shiga T (1981) Possibility of hybrid seed production by use of cytoplasmic male sterility in Brassica campestris, Chinese cabbage. In: 1st Symp. Asian Veg. Res. Taiwan, pp 301–311
Pelletier G, Primard C, Vedel F, Chetrit P, Remy R, Rousselle P, Renard M (1983) Intergeneric cytoplasmic hybridization in Cruciferae by protoplast fusion. Mol Gen Genet 191:244–250
Pelletier G, Primard C, Vedel F, Chétrit P (1986) Genetic improvement of cytoplasmic traits through cytoplasmic hybridization in Crucifera. In: Horn W, Jensen CJ, Oldenbach W, Scheider O (eds) Genetic manipulation in plant breeding. Walter de Gruyter & Co, Berlin, pp 653–661
Pelletier G, Primard C, Férault M, Vedel F, Chétrit P, Renard M, Delourme R (1988) Use of protoplasts in plant breeding: cytoplasmic aspects. Plant Cell Tiss Organ Cult 12:173–180
Pradhan AK, Sodhi YS, Mukhopadhyay A, Pental D (1993) Heterosis breeding in Indian mustard (Brassica juncea L. Czern & Coss): analysis of component characters contributing to heterosis for yield. Euphytica 69:219–229. doi:10.1007/BF00022368
Prakash S, Kirti PB, Bhat SR, Gaikwad K, Kumar VD, Chopra VL (1998) A Moricandia arvensis-based cytoplasmic male sterility and fertility restoration system in. Brassica juncea Theor Appl Genet 97:488–492
Primard-Brisset C, Poupard JP, Horvais R, Eber F, Pelletier G, Renard M, Delourme R (2005) A new recombined double low restorer line for the Ogu-INRA cms in rapeseed (Brassica napus L.). Theor Appl Genet 111:736–746. doi:10.1007/s00122-005-2059-8
Sabar M, Gagliardi D, Balk J, Leaver CJ (2003) ORFB is a subunit of F1F(O)-ATP synthase: insight into the basis of cytoplasmic male sterility in sunflower. EMBO Rep 4:381–386. doi:10.1038/sj.embor.embor800
Sakai T, Imamura J (1990) Intergeneric transfer of cytoplasmic male sterility between Raphanus sativus (cms line) and Brassica napus through cytoplast-protoplast fusion. Theor Appl Genet 80:421–427
Sakai T, Imamura J (1992) Alteration of mitochondrial genomes containing atpA genes in the sexual progeny of cybrids between Raphanus sativus cms line and Brassica napus cv. Westar. Theor Appl Genet 84:923–929
Schull GH (1952) Beginnings of the heterosis concept. In: Gowen JW (ed) Heterosis. Iowa State College Press, Iowa, pp 14–48
Uyttewaal M, Arnal N, Quadrado M, Martin-Canadell A, Vrielynck N, Hiard S, Gherbi H, Bendahmane A, Budar F, Mireau H (2008) Characterization of Raphanus sativus Pentatricopeptide repeat proteins encoded by the fertility restorer locus for Ogura cytoplasmic male sterility. Plant Cell 20:3331–3345. doi:10.1105/tpc.107.057208
Vedel F, Mathieu C, Lebacq P, Ambard-Bretteville F, Rémy R, Pelletier G, Renard M, Rousselle P (1982) Comparative macromolecular analysis of the cytoplasms of normal and cytoplasmic male sterile. Brassica napus Theor Appl Genet 62:255–262. doi:10.1007/BF00276248
Walters TW, Earle ED (1993) Organellar segregation, rearrangement and recombination in protoplast fusion-derived Brassica oleracea calli. Theor Appl Genet 85:761–769. doi:10.1007/BF00225017
Walters TW, Mutschler MA, Earle ED (1992) Protoplast fusion-derived Ogura male sterile cauliflower with cold tolerance. Plant Cell Rep 10:624–628
Wen L, Ruesch KL, Ortega VM, Kamps TL, Gabay-Laughnan S, Chase CD (2003) A nuclear restorer-of-fertility mutation disrupts accumulation of mitochondrial ATP synthase subunit alpha in developing pollen of S male-sterile maize. Genetics 165:771–779
Yamagishi H, Terachi T (1994a) Molecular and biological studies on male -sterile cytoplasm in the Cruciferae. I. The origin and distribution of Ogura male-sterile cytoplasm in Japanese wild radishes (Raphanus sativus L.) revealed by PCR-aided assay of their mitochondrial DNAs. Theor Appl Genet 87:996–1000
Yamagishi H, Terachi T (1994b) Molecular and biological studies on male sterile cytoplasm in Cruciferae. II. The origin of Ogura male sterile cytoplasm inferred from the segregation pattern of male sterility in the F1 progeny of wild and cultivated radishes (Raphanus sativus L.). Euphytica 80:201–206. doi:10.1007/BF00039651
Yamagishi H, Terachi T (1996) Molecular and biological studies on male-sterile cytoplasm in the Cruciferae. III. Distribution of Ogura-type cytoplasm among Japanese wild radishes and Asian radish cultivars. Theor Appl Genet 93:325–332
Yamagishi H, Terachi T (1997) Molecular and biological studies on male-sterile cytoplasm in the Cruciferae. IV. Ogura-type cytoplasm found in the wild radish. Raphanus raphanistrum Plant Breed 116:323–329
Yamagishi H, Terachi T (2001) Intra-and inter-specific variations in the mitochondrial gene orf138 of Ogura-type male-sterile cytoplasm from Raphanus sativus and. Raphanus raphanistrum Theor Appl Genet 103:725–732
Yarrow SA, Burnett LA, Wildeman RP, Kemble RJ (1990) The transfer of “Polima”cytoplasmic male sterility from oilseed rape (Brassica napus) to broccoli (B. oleracea) by protoplast fusion. Plant Cell Rep 9:185–188
Zirckle C (1952) Early ideas on inbreeding and crossbreeding. In: Gowen JW (ed) Heterosis. Iowa State College Press, Iowa, pp 1–13
Acknowledgments
We are very grateful to our colleagues at the INRA Rennes Center for their very impressive work on rapeseed breeding and for our long-running and very fruitful collaboration, particularly with Michel Renard and Régine Delourme. We acknowledge the excellent contributions of all colleagues and students involved in Versailles and Rennes INRA Centers who have worked for over 30 years on the Ogu-INRA CMS in Brassica. Our research on this CMS system has been partly funded by PROMOSOL, CETIOM, and Genoplante. We are also very grateful to Céline Duroc, joint head of the CETIOM technical center, for her help in retrieving data on the use of the Ogu-INRA system in agriculture.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Pelletier, G., Budar, F. (2015). Brassica Ogu-INRA Cytoplasmic Male Sterility: An Example of Successful Plant Somatic Fusion for Hybrid Seed Production. In: Li, XQ., Donnelly, D., Jensen, T. (eds) Somatic Genome Manipulation. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-2389-2_9
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2389-2_9
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-2388-5
Online ISBN: 978-1-4939-2389-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)