Abstract
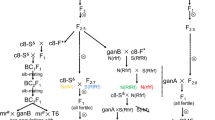
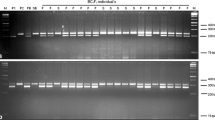
Backcross substitution of Brassica juncea (2n = 36; AABB) nucleus into the cytoplasm of a wild crucifer, Brassica fruticulosa helped in development of a new cytoplasmic male sterility (CMS) system. Male sterility was complete, stable, and expressed as rudimentary anthers containing sterile pollen grains. There was no impact on other floral as well as vegetative characters. All the natural B. juncea genotypes evaluated maintained the sterility. Gene for fertility restoration could be successfully introgressed from cytoplasm donor species. Genomic in situ hybridization studies revealed B. fruticulosa intogression in at least three chromosomes of the recipient species. Genetic studies carried out on F1, F2 and test cross progenies derived from hybridizing cytoplasmic male sterile and fertility restoring genotypes revealed a monogenic dominant control for the fertility restoration. Bulked segregant analysis with 588 SSR and 30 ISSR primers allowed identification of putative primers associated with fertility restoration. Co-segregation analysis of ten such primers with Rff gene revealed that Rf was flanked by two markers, namely cnu_m316 and nia_ m22, which were located 27.1 (LOD = 3.0) and 19.7 cM (LOD = 3.0) respectively around the gene in question. Distinctness of new CMS system from the ogura CMS was also demonstrated. This newly developed CMS-fertility-restorer system has a significant potential for hybrid seed production programs in mustard as an alternative to currently popular ogura CMS system.



Similar content being viewed by others
References
Atri C, Kumar B, Kumar H, Kumar S, Sharma S, Banga SS (2012) Development and characterization of Brassica juncea—fruticulosa introgression lines exhibiting resistance to mustard aphid (Lipaphis erysimi Kalt). BMC Genet. doi:10.1186/1471-2156-13-104
Banga SS, Banga SK (1998) Hybrid cultivar development. Springer/Narosa Publishing House New Delhi, New Delhi
Banga SS, Labana KS (1985) Male sterility in Indian mustard (Brassica juncea (L.) Coss.). Genetics and cytology of MS-4. Can J Genet Cytol 27:487–490
Banga SS, Deol JS, Banga SK (2003) Alloplasmic male sterile Brassica juncea with Enarthrocarpus lyratus cytoplasm and the introgression of gene(s) for fertility restoration from cytoplasm donor species. Theor Appl Genet 106:1390–1395
Bannerot H, Boulidard L, Cauderon Y, Temp J (1974) Transfer of cytoplasmic male sterility from Raphanus sativus to Brassica oleracea. Eucarpia Meet Cruciferae Crop Sect 25:52–54
Bannerot H, Boulidard L, Cauderon Y (1977) Unexpected difficulties met with the radish cytoplasm. Eucarpia Cruciferae Newslett 2:16
Chandra A, Gupta ML, Banga SS, Banga SK (2004) Production of an interspecific hybrid between Brassica fruticulosa and B. rapa. Plant Breed 123:497–498
Deol JS, Shivanna KR, Prakash S, Banga SS (2003) Enarthrocarpus lyratus based cytoplasmic male sterility and fertility restorer system in Brassica rapa. Plant Breed 122:438–440
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Duroc Y, Gaillard C, Hiard S, Tinchant C, Berthomé R, Pelletier G, Budar F (2006) Nuclear expression of a cytoplasmic male sterility gene modifies mitochondrial morphology in yeast and plant cell. Plant Sci 170:755–767
Garg H, Atri C, Sandhu PS, Kaur B, Renton M, Banga SK, Singh H, Singh C, Barbetti MJ, Banga SS (2010) High level of resistance to Sclerotinia sclerotiorum in introgression lines derived from hybridization between wild crucifers and the crop Brassica species B. napus and B. juncea. Field Crop Res 117:51–58
Heyn FW (1976) Transfer of restorer genes from Raphanus to cytoplasmic male sterile Brassica napus. Cruciferae Newslett 1:15–16
Hinata K, Konno N (1979) Studies on a male-sterile strain with a Brassica campestris nucleus and Diplotaxis muralis cytoplasm. The breeding process and some characteristics of the strain. Jpn J Breed 29:305–311
Janeja HS, Banga SK, Bhaskar PB, Banga SS (2003) Alloplasmic male sterile Brassica napus with Enarthrocarpus lyratus cytoplasm. Introgression and molecular mapping of lyratus chromosome segment carrying fertility restoring gene. Genome 46:792–797
Jean M, Brown GG, Landry BS (1997) Genetic mapping of nuclear fertility restorer gene for the ‘polima’ cytoplasmic male sterility in canola (Brassica napus L) using DNA markers. Theor Appl Genet 95:321–328
Kaur H, Banga SS (2015) Discovery and mapping of Brassica juncea Sdt 1 gene associated with determinate plant growth habit. Theor Appl Genet 128:235–245
Kaur G, Banga SK, Gogna KPS, Joshi S, Banga S (2004) Moricandia arvensis cytoplasm based system of cytoplasmic male sterility in Brassica juncea: reappraisal of fertility restoration and agronomic potential. Euphytica 138:271–276
Kim DH, Kang JG, Kim BD (2007) Isolation and characterization of the cytoplasmic male sterility-associated orf456 gene of chilli pepper (Capsicum annuum L.). Plant Mol Biol 63:519–532
Kirti PB, Banga SS, Prakash S, Chopra VL (1995) Transfer of ogu cytoplasmic male sterility to Brassica juncea and improvement of the male sterile through somatic cell fusion. Theor Appl Genet 91:517–521
Kumar S, Atri C, Sangha MK, Banga SS (2011) Screening of wild crucifers for resistance to mustard aphid, Lipaphis erysimi (Kaltenbach) and attempt at introgression of resistance gene (s) from Brassica fruticulosa to Brassica juncea. Euphytica 179:461–470
Kumar P, Vasupalli N, Sirinivasan R, Bhat SR (2012) An evolutionarily conserved mitochondrial orf108 is associated with cytoplasmic male sterility in different alloplasmic lines of Brassica juncea and induces male sterility in transgenic Arabidopsis thaliana. J Exp Bot 63:2921–2932
Labana KS, Banga SK (1989) Transfer of Ogura cytoplasmic male sterility of Brassica napus into genetic background of Brassica juncea. Crop Improv 16:82–83
Lander E, Green P, Abrahamson J, Barlow A, Daly MJ, Lincon SE, Newburg L (1987) MAPMAKER: an interactive computer package for constructing primary genetic linkage maps of experimental and natural populations. Genomics 1:174–181
Maureen R, Hanson L, Stephane Bentolia S (2004) Interactions of mitochondrial and nuclear genes that affect male gametophyte development. Plant Cell 16:154–169
Ogura H (1968) Studies on the new male-sterility in Japanese radish with special reference to the utilization of this sterility towards the practical raising of hybrid seeds. Mem Fac Agric Kagoshima Univ 6:39–78
Pearson OH (1972) Cytoplasmically inherited male-sterility characters and flavour components from the species cross Brassica nigra (L.) Koch -B. oleracea L. J Am Soc Hort Sci 97:398–402
Pelletier G, Primard C, Vedel F, Chétrit P, Rémy R, Rousselle P, Renard M (1983) Intergeneric cytoplasmic hybridization in Cruciferae by protoplast fusion. Mol Gen Genet 191:244–250
Prakash S (2001) Utilization of wild germplasm of Brassica allies in developing cytoplasmic male sterility/fertility restoration systems in Indian mustard—Brassica juncea. In: Liu H, Fu TD (eds) Proceedings of the international symposium rapeseed Science. Science Press, New York, pp 63–68
Prakash S, Bhat SR, Quiro´s CF, Kirti PB, Chopra VL (2009) Brassica and its close allies: cytogenetics and evolution. Plant Breed Rev 31:21–187
Prakash S, Bhat SR, Quiros CF, Kirti PB, Chopra VL (2010) Brassica and its close allies: cytogenetics and Evolution. Plant Breed Res 3:2–187
Schwarzacher T, Heslop-Harrison P (2000) Practical In situ Hybridization. BIOS Scientific Publishers Oxford. p 203
Stadler T, Delph LF (2002) Ancient mitochondrial haplotypes and evidence for intragenic recombination in a gynodioecious plant. Proc Natl Acad Sci USA 99:11730–11735
Yang JH, Liu XY, Yang XD, Zhang MF (2010) Mitochondrially-targeted expression of a cytoplasmic male sterility-associated orf220 gene causes male sterility in Brassica juncea. BMC Plant Biol 10:231. doi:10.1186/1471-2229-10-231
Acknowledgments
The research was supported by Indian Council of Agricultural Research in the form of ICAR National Professor Chair project “Broadening the genetic base of Indian mustard (Brassica juncea) through alien introgressions and germplasm enhancement” awarded to SSB. We are also thankful to anonymous reviewers for their helpful suggestions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Atri, C., Kaur, B., Sharma, S. et al. Substituting nuclear genome of Brassica juncea (L.) Czern & Coss. In cytoplasmic background of Brassica fruticulosa results in cytoplasmic male sterility. Euphytica 209, 31–40 (2016). https://doi.org/10.1007/s10681-015-1628-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-015-1628-4