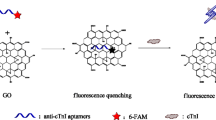
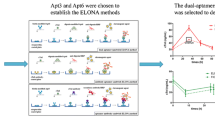
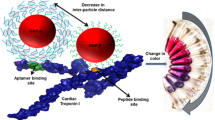
Abstract
The development of a pragmatic biosensor for diagnosing acute myocardial infarction (AMI) is of paramount significance. However, the detection of cardiac Troponin I (cTnI) presents formidable challenges owing to the intricate composition and scant concentration of cTnI in clinical samples. Therefore, we propose an innovative approach employing an aptamer biosensor predicated on MXene/multi-wall carbon nanotubes (MWCNTs) to discern cTnI. By integrating the MXene/MWCNTs nanomaterial, our aptasensor manifests remarkable amplification effects, thereby enabling an extensive detection range spanning from 10 to 106 pg/mL, while attaining an unparalleled detection sensitivity with an exceptionally low limit of 6.21 pg/mL. Furthermore, our aptasensor exhibits favourable selectivity, reproducibility, and stability. Moreover, our aptasensor showcases a robust clinical correlation equation of y = 1.202x + 0.0147, accompanied by an R2 value of 0.9797. A notable p-value discrepancy is observed between the AMI and healthy control groups. Remarkably, the area under the curve (AUC) achieved amounts to 0.981, with a sensitivity of 0.938 and specificity of 0.923. These outstanding attributes, coupled with its exceptional clinical performance, position our aptasensor as the optimal choice for the practical monitoring of acute myocardial infarction (AMI) disease.






Similar content being viewed by others
References
Chen J, Li Y, Jiang Y, Mao L, Lai M, Jiang L, Liu H, Nie Z (2021) TiO2/MXene-assisted LDI-MS for urine metabolic pofiling in urinary disease. Adv Funct Mater 31:1–9. https://doi.org/10.1002/adfm.202106743
Dong H, Chen H, Jiang J, Zhang H, Cai C, Shen Q (2018) Highly sensitive electrochemical detection of tumor exosomes based on aptamer recognition-induced multi-DNA release and cyclic enzymatic amplification. Anal Chem 90:4507–4513. https://doi.org/10.1021/acs.analchem.7b04863
Driscoll N, Erickson B, Murphy BB, Richardson AG, Robbins G, Apollo NV, Mentzelopoulos G, Mathis T, Hantanasirisakul K, Bagga P, Gullbrand SE, Sergison M, Reddy R, Wolf JA, Chen HI, Lucas TH, Dillingham TR, Davis KA, Gogotsi Y, Medaglia JD, Vitale F (2021) MXene-infused bioelectronic interfaces for multiscale electrophysiology and stimulation. Sci Transl Med 13:1–15. https://doi.org/10.1126/scitranslmed.abf8629
Eom W, Shin H, Ambade RB, Lee SH, Lee KH, Kang DJ, Han TH (2020) Large-scale wet-spinning of highly electroconductive MXene fibers. Nat Commun 11:2. https://doi.org/10.1038/s41467-020-16671-1
Guo Y, Zhang D, Yang Y, Wang Y, Bai Z, Chu PK, Luo Y (2021) MXene-encapsulated hollow Fe3O4nanochains embedded in N-doped carbon nanofibers with dual electronic pathways as flexible anodes for high-performance Li-ion batteries. Nanoscale 13:4624–4633. https://doi.org/10.1039/d0nr09228b
Han Z, Tang Z, Jiang K, Huang Q, Meng J, Nie D, Zhao Z (2020) Dual-target electrochemical aptasensor based on co-reduced molybdenum disulfide and Au NPs (rMoS2-Au) for multiplex detection of mycotoxins. Biosens Bioelectron 150:111894. https://doi.org/10.1016/j.bios.2019.111894
Liang Y, Wu C, Figueroa-Miranda G, Offenhäusser A, Mayer D (2019) Amplification of aptamer sensor signals by four orders of magnitude via interdigitated organic electrochemical transistors. Biosens Bioelectron 144:111668. https://doi.org/10.1016/j.bios.2019.111668
Lin X, Li C, Meng X, Yu W, Duan N, Wang Z, Wu S (2022) CRISPR-Cas12a-mediated luminescence resonance energy transfer aptasensing platform for deoxynivalenol using gold nanoparticle-decorated Ti3C2Tx MXene as the enhanced quencher. J Hazard Mater 433:128750. https://doi.org/10.1016/j.jhazmat.2022.128750
Liu Q, Aroonyadet N, Song Y, Wang X, Cao X, Liu Y, Cong S, Wu F, Thompson ME, Zhou C (2016) Highly sensitive and quick detection of acute myocardial infarction biomarkers using In2O3 nanoribbon biosensors fabricated using shadow masks. ACS Nano 10:10117–10125. https://doi.org/10.1021/acsnano.6b05171
Ma P, Fang D, Liu Y, Shang Y, Shi Y, Yang HY (2021a) MXene-based materials for electrochemical sodium-ion storage. Adv Sci 8:202003185. https://doi.org/10.1002/advs.202003185
Ma Y, Wang Y, Liu Y, Shi L, Yang D (2021b) Multi-carbon dots and aptamer based signal amplification ratiometric fluorescence probe for protein tyrosine kinase 7 detection. J Nanobiotechnol 19:1–11. https://doi.org/10.1186/s12951-021-00787-7
Mokhtari Z, Khajehsharifi H, Hashemnia S, Solati Z, Azimpanah R, Shahrokhian S (2020) Evaluation of molecular imprinted polymerized methylene blue/aptamer as a novel hybrid receptor for Cardiac Troponin I (cTnI) detection at glassy carbon electrodes modified with new biosynthesized ZnONPs. Sensors Actuators B Chem 320:128316. https://doi.org/10.1016/j.snb.2020.128316
Saraf N, Woods ER, Peppler M, Seal S (2018) Highly selective aptamer based organic electrochemical biosensor with pico-level detection. Biosens Bioelectron 117:40–46. https://doi.org/10.1016/j.bios.2018.05.031
Sharma A, Han CH, Jang J (2016) Rapid electrical immunoassay of the cardiac biomarker troponin I through dielectrophoretic concentration using imbedded electrodes. Biosens Bioelectron 82:78–84. https://doi.org/10.1016/j.bios.2016.03.056
Sinha A, Tai TY, Li KH, Gopinathan P, Chung YD, Sarangadharan I, Ma HP, Huang PC, Shiesh SC, Wang YL, Lee GB (2019) An integrated microfluidic system with field-effect-transistor sensor arrays for detecting multiple cardiovascular biomarkers from clinical samples. Biosens Bioelectron 129:155–163. https://doi.org/10.1016/j.bios.2019.01.001
Sun D, Luo Z, Lu J, Zhang S, Che T, Chen Z, Zhang L (2019) Electrochemical dual-aptamer-based biosensor for nonenzymatic detection of cardiac troponin I by nanohybrid electrocatalysts labeling combined with DNA nanotetrahedron structure. Biosens Bioelectron 134:49–56. https://doi.org/10.1016/j.bios.2019.03.049
Wang H, Li H, Huang Y, Xiong M, Wang F, Li C (2019) A label-free electrochemical biosensor for highly sensitive detection of gliotoxin based on DNA nanostructure/MXene nanocomplexes. Biosens Bioelectron 142:111531
Wang Z, Wang F, Hermawan A, Asakura Y, Hasegawa T, Kumagai H, Kato H, Kakihana M, Zhu J, Yin S (2021) SnO-SnO2 modified two-dimensional MXene Ti3C2Tx for acetone gas sensor working at room temperature. J Mater Sci Technol 73:128–138. https://doi.org/10.1016/j.jmst.2020.07.040
Yang Y, Lei D, Huang S, Yang Q, Song B, Guo Y, Shen A, Yuan Z, Li S, Qing FL, Ye X, You Z, Zhao Q (2019) Elastic 3D-printed hybrid polymeric scaffold improves cardiac remodeling after myocardial infarction. Adv Healthc Mater 8:1–15. https://doi.org/10.1002/adhm.201900065
Zhang T, Ma N, Ali A, Wei Q, Wu D, Ren X (2018) Electrochemical ultrasensitive detection of cardiac troponin I using covalent organic frameworks for signal amplification. Biosens Bioelectron 119:176–181. https://doi.org/10.1016/j.bios.2018.08.020
Zhang Z, Li Y, Liang C, Yu G, Zhao J, Luo S, Huang Y, Su C, Xing G (2020) In situ growth of MAPbBr 3 nanocrystals on few-layer MXene nanosheets with efficient energy transfer. Small 16:1. https://doi.org/10.1002/smll.201905896
Zhu YC, Jiang XZ, Bai QK, Deng SH, Zhang Y, Zhang ZP, Jiang Q (2019) Evaluating the efficacy of atorvastatin on patients with carotid plaque by an innovative ultrasonography. J Stroke Cerebrovasc Dis 28:830–837. https://doi.org/10.1016/j.jstrokecerebrovasdis.2018.11.027
Zhu YC, Zhang Y, Shan J, Deng SH, Shi XR, Jiang Q (2021) Added value of superb microvascular imaging and virtual touch imaging quantification in assisting thyroid cancer classification. Ultrasound Med Biol 47:3364–3371. https://doi.org/10.1016/j.ultrasmedbio.2021.07.017
Zouggari Y, Ait-Oufella H, Bonnin P, Simon T, Sage AP, Guérin C, Vilar J, Caligiuri G, Tsiantoulas D, Laurans L, Dumeau E, Kotti S, Bruneval P, Charo IF, Binder CJ, Danchin N, Tedgui A, Tedder TF, Silvestre JS, Mallat Z (2013) B lymphocytes trigger monocyte mobilization and impair heart function after acute myocardial infarction. Nat Med 19:1273–1280. https://doi.org/10.1038/nm.3284
Acknowledgements
This project is funded by the Z202120 (Wuxi Health Commission) and M202140 (Wuxi Health Commission).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Informed consent statement
The Ethics Committee of Jiangyin People's Hospital approved this project (No:2021-039).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jinsong, T., Jin, H., Weizhang, L. et al. Integrating MXene/MWCNTs into aptasensor capable of ultrasensitive quantification of cTnI towards the diagnosis of acute myocardial infarction. Chem. Pap. 78, 3205–3212 (2024). https://doi.org/10.1007/s11696-024-03304-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-024-03304-w