Abstract
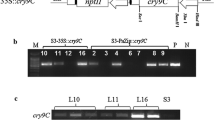
Co-expression of multiple genes encoding different kinds of insect resistant proteins has been developed to confer a broader spectrum of pest control. Tetraploid Isatis indigotica Fort was transformed with a plasmid, p3300BP, containing Bacillus thuringiensis Cry1Ac gene (Bt) and Pinellia ternata agglutinin gene (Pta) and the selectable marker herbicide resistance gene (Bar) driven by the CaMV35S promoter via Agrobacterium tumefaciens-mediated transformation. The integration and expression of introduced genes in regenerated transgenic plants were confirmed by PCR and Western blot assays. Insect bioassay test demonstrated transgenic lines had significant inhibition to diamondback moths (Plutella xylostella L.) and peach potato aphids (Myzus persicae Sulzer) simultaneously. Our study reported here would be a great motivation for field culture of tetraploid I. indigotica, also providing an efficient molecular breeding strategy to provide insect tolerant plants.




Similar content being viewed by others
References
Wu XY, Liu YH, Sheng WY et al (1997) Chemical constituents of Isatis indigotica. Planta Med 63:55–57
Chen M, Chen XF, Gu ZL (2002) Progress in pharmacological effects and clinical application of Isatis indigotica Fort. Chin Wild Plant Res 2:3–6
Qiao CZ, Wu MS, Dai FB et al (1989) Studies on polyploid breeding of Isatis indigotica Fort. Acta Bot Sin 31:678–683
Tang JD, Shelton AM, Rie J et al (1996) Toxicity of Bacillus thuringiensis spore and crystal protein to resistant diamondback moth (Plutella xylostella). Appl Environ Microbiol 62:564–569
Margaritopoulos JT, Kasprowicz L, Malloch GL et al (2009) Tracking the global dispersal of a cosmopolitan insect pest, the peach potato aphid. BMC Ecol 9:13
Ferry N, Edwards MG, Gatehouse J et al (2006) Transgenic plants for insect pest control: a forward looking scientific perspective. Transgenic Res 15:13–19
Jabeen R, Khan MS, Zafar Y et al (2010) Codon optimization of cry1Ab gene for hyper expression in plant organelles. Mol Biol Rep 37:1011–1017
Kumar S, Chandra A, Pandey KC (2008) Bacillus thuringiensis (Bt) transgenic crop: an environment friendly insect-pest management strategy. J Environ Biol 29:641–653
Bukhari DA, Shakoori AR (2009) Cloning and expression of Bacillus thuringiensis cry11 crystal protein gene in Escherichia coli. Mol Biol Rep 36:1661–1670
Deka S, Barthakur S (2010) Overview on current status of biotechnological interventions on yellow stem borer Scirpophaga incertulas (Lepidoptera: Crambidae) resistance in rice. Biotechnol Adv 28:70–81
Yao JH, Zhao XY, Liao ZH et al (2003) Cloning and molecular characterization of a novel lectin gene from Pinellia ternate. Cell Res 13:301–308
Yao JH, Pang YZ, Qi HX et al (2003) Transgenic tobacco expressing Pinellia ternata agglutinin confers enhanced resistance to aphids. Transgenic Res 12:715–722
Sambrook J, Russell D, Maniatis T (2002) Molecular cloning a laboratory manual. Cold Springer Harbor Laboratory Press, Cold Springer Harbor
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Sadeghi A, Smagghe G, Broeders S et al (2008) Ectopically expressed leaf and bulb lectins from garlic (Allium sativum L.) protect transgenic tobacco plants against cotton leaf worm (Spodoptera littoralis). Transgenic Res 17:9–18
Hilder VA, Powell KS, Gatehouse AMR et al (1995) Expression of snowdrop lectin in transgenic tobacco plants results in added protection against aphids. Transgenic Res 4:18–25
James C (2002) Global status of commercialisation of transgenic crops: 2002. ISAAA briefs No. 27, Ithaca, NY. http://www.isaaa.org
Ralph B (2007) Plastid biotechnology: prospects for herbicide and insect resistance, metabolic engineering and molecular farming. Curr Opin Biotechnol 18:100–106
Hu Q, Tao SS (2005) The latest progress of study and development on Pinellia ternata. J Southwest Univ Sci Technol 20:63–68
Bano-Maqbool S, Christou P (1999) Multiple traits of agronomic importance in transgenic indica rice plants: analysis of transgene integration patterns, expression levels and stability. Mol Breed 5:471–480
Tang K, Sun X, Wu A et al (2001) Transgenic rice plants expressing the ferredoxin-like protein (AP1) from sweet pepper show enhanced resistance to Xanthomonas oryzae pv. Oryzae. Plant Sci 160:1031–1038
Cao J, Anthony MS, Elizabeth DE (2008) Sequential transformation to pyramid two Bt genes in vegetable Indian mustard (Brassica juncea L.) and its potential for control of diamondback moth larvae. Plant Cell Rep 27:479–487
Jung SC, Kim YG (2007) Potentiating effect of Bacillus thuringiensis spp. kurstaki on pathogenicity of entomopathogenic bacterium Xenorhabdus nematophila K1 against diamondback moth (Lepidoptera: Plutellidae). J Econ Entomol 100:246–250
Rang C, Lacey LA, Frutos R (2000) The crystal proteins from Bacillus thuringiensis spp. thompsoni display a synergistic activity against the codling moth, Cydia pomonella. Curr Microbiol 40:200–204
Li M, Wu G, Liu C et al (2009) Expression and activity of a probable toxin from Photorhabdus luminescens. Mol Biol Rep 36:785–790
Roh JY, Choi JY, Li MS et al (2007) Bacillus thuringiensis as a specific, safe, and effective tool for insect pest control. J Microbiol Biotechnol 17:547–559
Acknowledgments
This research was financially supported by National Natural Science Foundation of China (30900786); Modernization of Traditional Chinese Medicine Foundation (08DZ1971502) and Domestic Science and Technology Cooperation Projects (10495801400, 10395820200), Shanghai Science and Technology Committee.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Xiao, Y., Wang, K., Ding, R. et al. Transgenic tetraploid Isatis indigotica expressing Bt Cry1Ac and Pinellia ternata agglutinin showed enhanced resistance to moths and aphids. Mol Biol Rep 39, 485–491 (2012). https://doi.org/10.1007/s11033-011-0762-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-011-0762-x