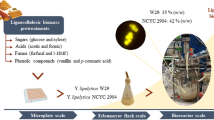
Abstract
Yarrowia lipolytica is an oleaginous yeast species that has attracted attention as a model organism for synthesis of single cell oil. Among over 50 isolates of Y. lipolytica identified, only a few of the strains have been studied extensively. Furthermore, 12 other yeast species were recently assigned to the Yarrowia clade, and most are not well characterized in terms of cell growth and lipid accumulation, especially in industrially relevant conditions. In the present study, we investigated biomass and lipid production by 57 yeast isolates, representing all 13 species in the Yarrowia clade, on a non-detoxified dilute acid-pretreated switchgrass hydrolysate under highly aerobic conditions. The objective was to compare yeast physiology during growth in an abundant, low-cost biomass feedstock and to expand diversity of genetically tractable, oleaginous yeasts available for lipid research. Screening of 45 Y. lipolytica isolates demonstrated considerable variation within the species in terms of lipid accumulation (min = 0.1 g/L; max = 5.1 g/L; mean = 2.3 g/L); three strains (NRRL YB-420, YB-419, and YB-392) were especially promising for cellulosic biomass conversion with average improvements of 43, 57, and 64%, respectively, in final lipid titer as compared to control strain W29. Subsequently, evaluation of strains from 13 distinct species in the Yarrowia clade identified Candida phangngensis PT1-17 as the top lipid producer with a maximum titer of 9.8 g/L lipid, which was over twofold higher than the second-best species in the clade (Candida hollandica NRRL Y-48254). A small set of the most promising strains from the screenings was further characterized to evaluate inhibitor tolerance, lipid production kinetics, and fatty acid distribution. We expect that the results of this study will pave the way for new biotechnological applications involving previously overlooked and under-characterized strains within the Yarrowia clade.





Similar content being viewed by others
References
Almeida JR, Modig T, Petersson A, Hahn-Hägerdal B, Lidén G, Gorwa-Grauslund MF (2007) Increased tolerance and conversion of inhibitors in lignocellulosic hydrolysates by Saccharomyces cerevisiae. J Chem Technol Biotechnol 82(4):340–349
Barth G, Gaillardin C (1996) Yarrowia lipolytica. In: Wolf W (ed) Nonconventional yeasts in biotechnology. Springer, Berlin, pp. 313–388
Beopoulos A, Cescut J, Haddouche R, Uribelarrea J-L, Molina-Jouve C, Nicaud J-M (2009) Yarrowia lipolytica as a model for bio-oil production. Prog Lipid Res 48(6):375–387
Beopoulos A, Nicaud JM, Gaillardin C (2011) An overview of lipid metabolism in yeasts and its impact on biotechnological processes. Appl Microbiol Biotechnol 90(4):1193–1206
Blazeck J, Hill A, Liu L, Knight R, Miller J, Pan A, Otoupal P, Alper HS (2014) Harnessing Yarrowia lipolytica lipogenesis to create a platform for lipid and biofuel production. Nat Commun 5:3131
Chang CF, Chen CC, Lee CF, Liu SM (2013) Identifying and characterizing Yarrowia keelungensis sp. nov., an oil-degrading yeast isolated from the sea surface microlayer. Antonie Van Leeuwenhoek 104(6):1117–1123
Chen D, Beckerich J, Gaillardin C (1997) One-step transformation of the dimorphic yeast Yarrowia lipolytica. Appl Microbiol Biotechnol 48(2):232–235
de Albuquerque TL, da Silva IJ, de Macedo GR, Rocha MVP (2014) Biotechnological production of xylitol from lignocellulosic wastes: a review. Process Biochem 49(11):1779–1789
Dien BS, Slininger PJ, Kurtzman CP, Moser BR, O’Bryan PJ (2016a) Identification of superior lipid producing Lipomyces and Myxozyma yeasts. AIMS Environ Sci 3(1):1–20
Dien BS, Zhu J, Slininger PJ, Kurtzman CP, Moser BR, O’Bryan PJ, Gleisner R, Cotta MA (2016b) Conversion of SPORL pretreated Douglas fir forest residues into microbial lipids with oleaginous yeasts. RSC Adv 6(25):20695–20705
Dujon B, Sherman D, Fischer G, Durrens P, Casaregola S, Lafontaine I, De Montigny J, Marck C, Neuvéglise C, Talla E (2004) Genome evolution in yeasts. Nature 430(6995):35–44
Ellis EM (2002) Microbial aldo-keto reductases. FEMS Microbiol Lett 216(2):123–131
Fickers P, Benetti P-H, Wache Y, Marty A, Mauersberger S, Smit M, Nicaud J-M (2005) Hydrophobic substrate utilisation by the yeast Yarrowia lipolytica, and its potential applications. FEMS Yeast Res 5(6–7):527–543
Gancedo JM (1998) Yeast carbon catabolite repression. Microbiol Mol Biol Rev 62(2):334–361
Groenewald M, Smith MT (2013) The teleomorph state of Candida deformans Langeron & Guerra and description of Yarrowia yakushimensis comb. nov. Antonie Van Leeuwenhoek 103(5):1023–1028
Groenewald M, Boekhout T, Neuvéglise C, Gaillardin C, Van Dijck PW, Wyss M (2014) Yarrowia lipolytica: safety assessment of an oleaginous yeast with a great industrial potential. Crit Rev Microbiol 40(3):187–206
Izard J, Limberger RJ (2003) Rapid screening method for quantitation of bacterial cell lipids from whole cells. J Microbiol Methods 55(2):411–418
Jin M, Slininger PJ, Dien BS, Waghmode S, Moser BR, Orjuela A, da Costa Sousa L, Balan V (2015) Microbial lipid-based lignocellulosic biorefinery: feasibility and challenges. Trends Biotechnol 33(1):43–54
Jönsson LJ, Martín C (2016) Pretreatment of lignocellulose: formation of inhibitory by-products and strategies for minimizing their effects. Bioresour Technol 199:103–112
Kitcha S, Cheirsilp B (2011) Screening of oleaginous yeasts and optimization for lipid production using crude glycerol as a carbon source. Energy Procedia 9:274–282
Knothe G (2005) Dependence of biodiesel fuel properties on the structure of fatty acid alkyl esters. Fuel Process Technol 86(10):1059–1070
Knutsen AK, Robert V, Poot G, Epping W, Figge M, Holst-Jensen A, Skaar I, Smith MT (2007) Polyphasic re-examination of Yarrowia lipolytica strains and the description of three novel Candida species: Candida oslonensis sp. nov., Candida alimentaria sp. nov. and Candida hollandica sp. nov. Int J Syst Evol Microbiol 57(10):2426–2435
Kurtzman CP (2005) New species and a new combination in the Hyphopichia and Yarrowia yeast clades. Antonie Van Leeuwenhoek 88(2):121–130
Kurtzman C (2011) Yarrowia van der Walt & von Arx (1980). In: Kurtzman C, Fell J, Boekhout T (eds) The yeasts, a taxonomic study. Elsevier, London, pp. 927–929
Kurtzman CP, Price NP, Ray KJ, Kuo T-M (2010) Production of sophorolipid biosurfactants by multiple species of the Starmerella (Candida) bombicola yeast clade. FEMS Microbiol Lett 311(2):140–146
Ledesma-Amaro R, Nicaud J-M (2016) Yarrowia lipolytica as a biotechnological chassis to produce usual and unusual fatty acids. Prog Lipid Res 61:40–50
Libkind D, Sommaruga R, Zagarese H, van Broock M (2005) Mycosporines in carotenogenic yeasts. Syst Appl Microbiol 28(8):749–754
Limtong S, Youngmanitchai W, Kawasaki H, Seki T (2008) Candida phangngensis sp. nov., an anamorphic yeast species in the Yarrowia clade, isolated from water in mangrove forests in Phang-Nga Province, Thailand. Int J Syst Evol Microbiol 58(2):515–519
Lindner P (1922) Das Problem der biologischen Fettbildung und Fettgewinnung. Angew Chem 35(19):110–114
Liu L, Alper HS (2014) Draft genome sequence of the oleaginous yeast Yarrowia lipolytica PO1f, a commonly used metabolic engineering host. Genome Announc 2(4):e00652–e00614
Madzak C, Tréton B, Blanchin-Roland S (2000) Strong hybrid promoters and integrative expression/secretion vectors for quasi-constitutive expression of heterologous proteins in the yeast Yarrowia lipolytica. J Mol Microbiol Biotechnol 2(2):207–216
Mäkinen K (1992) Dietary prevention of dental caries by xylitol-clinical effectiveness and safety. J Appl Nutr 44(1):16–28
McNeill J, Barrie F, Buck W, Demoulin V, Greuter W, Hawksworth D, Herendeen P, Knapp S, Marhold K, Prado J (2012) International code of nomenclature for algae, fungi, and plants (Melbourne Code). Regnum Veg 154(1):208
Michely S, Gaillardin C, Nicaud J-M, Neuvéglise C (2013) Comparative physiology of oleaginous species from the Yarrowia clade. PLoS One 8(5):e63356
Nagy E, Niss M, Dlauchy D, Arneborg N, Nielsen DS, Péter G (2013) Yarrowia divulgata fa, sp. nov., a yeast species from animal-related and marine sources. Int J Syst Evol Microbiol 63(12):4818–4823
Nagy E, Dlauchy D, Medeiros AO, Péter G, Rosa CA (2014) Yarrowia porcina sp. nov. and Yarrowia bubula fa sp. nov., two yeast species from meat and river sediment. Antonie Van Leeuwenhoek 105(4):697–707
Nicaud JM (2012) Yarrowia lipolytica. Yeast 29(10):409–418
Nilsson A, Gorwa-Grauslund MF, Hahn-Hägerdal B, Lidén G (2005) Cofactor dependence in furan reduction by Saccharomyces cerevisiae in fermentation of acid-hydrolyzed lignocellulose. Appl Environ Microbiol 71(12):7866–7871
Pan L-X, Yang D-F, Shao L, Li W, Chen G-G, Liang Z-Q (2009) Isolation of the oleaginous yeasts from the soil and studies of their lipid-producing capacities. Food Technol Biotechnol 47(2):215–220
Papanikolaou S, Chevalot I, Komaitis M, Marc I, Aggelis G (2002a) Single cell oil production by Yarrowia lipolytica growing on an industrial derivative of animal fat in batch cultures. Appl Microbiol Biotechnol 58(3):308–312
Papanikolaou S, Muniglia L, Chevalot I, Aggelis G, Marc I (2002b) Yarrowia lipolytica as a potential producer of citric acid from raw glycerol. J Appl Microbiol 92(4):737–744
Perlack RD, Eaton LM, Turhollow Jr AF, Langholtz MH, Brandt CC, Downing ME, Graham RL, Wright LL, Kavkewitz JM, Shamey AM (2011) US billion-ton update: biomass supply for a bioenergy and bioproducts industry. USDoE. Oak Ridge, TN, Oak Ridge National Laboratory
Qiao K, Abidi SHI, Liu H, Zhang H, Chakraborty S, Watson N, Ajikumar PK, Stephanopoulos G (2015) Engineering lipid overproduction in the oleaginous yeast Yarrowia lipolytica. Metab Eng 29:56–65
Rakicka M, Kieroń A, Hapeta P, Neuvéglise C, Lazar Z (2016) Sweet and sour potential of yeast from the Yarrowia clade. Biomass Bioenergy 92:48–54
Ratledge C (1991) Microorganisms for lipids. Acta Biotechnol 11(5):429–438
Ratledge C, Wynn JP (2002) The biochemistry and molecular biology of lipid accumulation in oleaginous microorganisms. Adv Appl Microbiol 51:1–51
Rodriguez GM, Hussain MS, Gambill L, Gao D, Yaguchi A, Blenner M (2016) Engineering xylose utilization in Yarrowia lipolytica by understanding its cryptic xylose pathway. Biotechnol Biofuels 9(1):149
Ryu DD, Kim Y, Kim J (1984) Effect of air supplement on the performance of continuous ethanol fermentation system. Biotechnol Bioeng 26(1):12–16
Ryu S, Hipp J, Trinh CT (2016) Activating and elucidating metabolism of complex sugars in Yarrowia lipolytica. Appl Environ Microbiol 82(4):1334–1345
Sanchez RG, Karhumaa K, Fonseca C, Nogué VS, Almeida JR, Larsson CU, Bengtsson O, Bettiga M, Hahn-Hägerdal B, Gorwa-Grauslund MF (2010) Improved xylose and arabinose utilization by an industrial recombinant Saccharomyces cerevisiae strain using evolutionary engineering. Biotechnol Biofuels 3(1):1
Schneiter R, Daum G (2006) Extraction of yeast lipids. Yeast Protocol 41–45. doi:10.1385/1-59259-958-3:041
Sitepu I, Ignatia L, Franz A, Wong D, Faulina S, Tsui M, Kanti A, Boundy-Mills K (2012) An improved high-throughput Nile red fluorescence assay for estimating intracellular lipids in a variety of yeast species. J Microbiol Methods 91(2):321–328
Sitepu IR, Sestric R, Ignatia L, Levin D, German JB, Gillies LA, Almada LAG, Boundy-Mills KL (2013) Manipulation of culture conditions alters lipid content and fatty acid profiles of a wide variety of known and new oleaginous yeast species. Bioresour Technol 144(0):360–369
Sitepu I, Selby T, Lin T, Zhu S, Boundy-Mills K (2014) Carbon source utilization and inhibitor tolerance of 45 oleaginous yeast species. J Ind Microbiol Biotechnol 41(7):1061–1070
Slininger PJ, Dien BS, Kurtzman CP, Moser BR, Bakota EL, Thompson SR, O’Bryan PJ, Cotta MA, Balan V, Jin M (2016) Comparative lipid production by oleaginous yeasts in hydrolyzates of lignocellulosic biomass and process strategy for high titers. Biotechnol Bioeng 113(8):1676–1690
Tai M, Stephanopoulos G (2013) Engineering the push and pull of lipid biosynthesis in oleaginous yeast Yarrowia lipolytica for biofuel production. Metab Eng 15:1–9
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Thevenieau F, Nicaud J-M, Gaillardin C (2009) Applications of the non-conventional yeast Yarrowia lipolytica. (ed) Yeast biotechnology: diversity and applications. Springer, pp 589–613
Träff K, Jönsson LJ, Hahn-Hägerdal B (2002) Putative xylose and arabinose reductases in Saccharomyces cerevisiae. Yeast 19(14):1233–1241
Tsigie YA, Wang C-Y, Truong C-T, Y-H J (2011) Lipid production from Yarrowia lipolytica Po1g grown in sugarcane bagasse hydrolysate. Bioresour Technol 102(19):9216–9222
Tsigie YA, Wang C-Y, Kasim NS, Diem Q-D, Huynh L-H, Ho Q-P, Truong C-T, Ju Y-H (2012) Oil production from Yarrowia lipolytica Po1g using rice bran hydrolysate. Biomed Res Int 2012
Vogel K, Mitchell R, Casler M, Sarath G (2014) Registration of ‘Liberty’ switchgrass. J Plant Regist 8(3):242–247
Wang J, Li R, Lu D, Ma S, Yan Y, Li W (2009) A quick isolation method for mutants with high lipid yield in oleaginous yeast. World J Microbiol Biotechnol 25(5):921–925
Werpy T, Petersen G, Aden A, Bozell J, Holladay J, White J, Manheim A, Eliot D, Lasure L, Jones S (2004) Top value added chemicals from biomass, vol. 1: results of screening for potential candidates from sugars and synthesis gas. USDoE. Golden, CO, National Renewable Energy Laboratory
Yu X, Zheng Y, Dorgan KM, Chen S (2011) Oil production by oleaginous yeasts using the hydrolysate from pretreatment of wheat straw with dilute sulfuric acid. Bioresour Technol 102(10):6134–6140
Acknowledgements
We would like to acknowledge Novozymes for the kind donation of enzymes and the ARS Culture Collection and Mr. James Swezey for providing many of the yeast strains used in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Disclaimer
The mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. USDA is an equal opportunity provider and employer.
Electronic supplementary material
ESM 1
(PDF 366 kb)
Rights and permissions
About this article
Cite this article
Quarterman, J., Slininger, P.J., Kurtzman, C.P. et al. A survey of yeast from the Yarrowia clade for lipid production in dilute acid pretreated lignocellulosic biomass hydrolysate. Appl Microbiol Biotechnol 101, 3319–3334 (2017). https://doi.org/10.1007/s00253-016-8062-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-8062-y