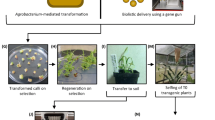
Abstract
Sorghum is a versatile, warm-weather crop that acts as a staple cereal for over 500 million people throughout the world. In addition to human consumption, sorghum is utilized across various markets including for feed, biofuels and biomaterials industries. Sorghum’s nutrient and water use efficiency, as well as its ability to grow in shallow clay soils, make it a viable alternative to other cereal crops that will inevitably be impacted by rapid climate change. With the advancement of genome editing technologies, such as CRISPR/Cas9, biotechnological tools can be integrated into breeding programmes to further improve key phenotypic traits. Current sorghum genome editing techniques have resulted in various trait improvements, such as grain quality. This chapter provides an overview of established and novel methods in sorghum genome editing and transformation, including applications and future directions for sorghum improvement.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Adem M, Beyene D, Feyissa T (2017) Recent achievements obtained by chloroplast transformation. Plant Methods 13(1):30. https://doi.org/10.1186/s13007-017-0179-1
Ahmad N, Michoux F, Lössl AG, Nixon PJ (2016) Challenges and perspectives in commercializing plastid transformation technology. J Exp Bot 67(21):5945–5960. https://doi.org/10.1093/jxb/erw360
Apel W, Bock R (2009) Enhancement of carotenoid biosynthesis in transplastomic tomatoes by induced lycopene-to-provitamin A conversion. Plant Physiol 151(1):59–66. https://doi.org/10.1104/pp.109.140533
Belide S, Vanhercke T, Petrie JR, Singh SP (2017) Robust genetic transformation of sorghum (L.) using differentiating embryogenic callus induced from immature embryos. Plant Methods 13(1):109. https://doi.org/10.1186/s13007-017-0260-9
Bellis ES, Kelly EA, Lorts CM, Gao H, DeLeo VL, Rouhan G, Budden A, Bhaskara GB, Hu Z, Muscarella R, Timko MP, Nebie B, Runo SM, Chilcoat ND, Juenger TE, Morris GP, dePamphilis CW, Lasky JR (2020) Genomics of sorghum local adaptation to a parasitic plant. Proc Natl Acad Sci U S A 117(8):4243–4251. https://doi.org/10.1073/pnas.1908707117
Bock R (2007) Plastid biotechnology: prospects for herbicide and insect resistance, metabolic engineering and molecular farming. Curr Opin Biotechnol 18(2):100–106. https://doi.org/10.1016/j.copbio.2006.12.001
Bock R (2015) Engineering plastid genomes: methods, tools, and applications in basic research and biotechnology. Annu Rev Plant Biol 66(1):211–241. https://doi.org/10.1146/annurev-arplant-050213-040212
Bock R, Warzecha H (2010) Solar-powered factories for new vaccines and antibiotics. Trends Biotechnol 28(5):246–252. https://doi.org/10.1016/j.tibtech.2010.01.006
Boesch P, Weber-Lotfi F, Ibrahim N, Tarasenko V, Cosset A, Paulus F, Lightowlers RN, Dietrich A (2011) DNA repair in organelles: pathways, organization, regulation, relevance in disease and aging. Biochim Biophys Acta 1813(1):186–200. https://doi.org/10.1016/j.bbamcr.2010.10.002
Bohmert-Tatarev K, McAvoy S, Daughtry S, Peoples OP, Snell KD (2011) High levels of bioplastic are produced in fertile transplastomic tobacco plants engineered with a synthetic operon for the production of polyhydroxybutyrate. Plant Physiol 155(4):1690–1708. https://doi.org/10.1104/pp.110.169581
Borrell AK, Hammer GL (2000) Nitrogen dynamics and the physiological basis of stay- green in sorghum. Crop Sci 40(5):1295
Borrell AK, Mullet JE, George-Jaeggli B, van Oosterom EJ, Hammer GL, Klein PE, Jordan DR (2014) Drought adaptation of stay-green sorghum is associated with canopy development, leaf anatomy, root growth, and water uptake. J Exp Bot 65(21):6251–6263. https://doi.org/10.1093/jxb/eru232
Boyles RE, Brenton ZW, Kresovich S (2019) Genetic and genomic resources of sorghum to connect genotype with phenotype in contrasting environments. Plant J 97(1):19–39. https://doi.org/10.1111/tpj.14113
Burow GB, Franks CD, Xin Z (2008) Genetic and physiological analysis of an irradiated bloomless mutant (epicuticular wax mutant) of sorghum. Crop Sci 48(1):41–48. https://doi.org/10.2135/cropsci2007.02.0119
Burow G, Xin ZG, Hayes C, Burke J (2014) Characterization of a multiseeded (msd1) mutant of sorghum for increasing grain yield. Crop Sci 54(5):2030–2037. https://doi.org/10.2135/cropsci2013.08.0566
Cai T, Daly B, Butler L (1987) Callus induction and plant regeneration from shoot portions of mature embryos of high tannin sorghums. Plant Cell Tiss Org 9(3):245–252. https://doi.org/10.1007/BF00040810
Casas AM, Kononowicz AK, Zehr UB, Tomes DT, Axtell JD, Butler LG, Bressan RA, Hasegawa PM (1993) Transgenic sorghum plants via microprojectile bombardment. Proc Natl Acad Sci U S A 90(23):11212–11216. https://doi.org/10.1073/pnas.90.23.11212
Casto AL, Mattison AJ, Olson SN, Thakran M, Rooney WL, Mullet JE (2019) Maturity2, a novel regulator of flowering time in Sorghum bicolor, increases expression of SbPRR37 and SbCO in long days delaying flowering. PLoS One 14(4):e0212154. https://doi.org/10.1371/journal.pone.0212154
Char SN (2019) Development and utilization of genome editing tools in maize, sorghum and rice. ProQuest Dissertations Publishing. http://search.proquest.com/docview/2267899640/
Che P, Anand A, Wu E, Sander JD, Simon MK, Zhu W, Sigmund AL, Zastrow-Hayes G, Miller M, Liu D, Lawit SJ, Zhao ZY, Albertsen MC, Jones TJ (2018) Developing a flexible, high-efficiency Agrobacterium-mediated sorghum transformation system with broad application. Plant Biotechnol J 16(7):1388–1395. https://doi.org/10.1111/pbi.12879
Chen J, Xin Z, Laza H (2019) Registration of BTx623dw5, a new sorghum dwarf mutant. J Plant Reg 13(2):254–257. https://doi.org/10.3198/jpr2018.09.0058crgs
Cheng L, Li HP, Qu B, Huang T, Tu JX, Fu TD, Liao YC (2010) Chloroplast transformation of rapeseed (Brassica napus) by particle bombardment of cotyledons. Plant Cell Rep 29(4):371–381. https://doi.org/10.1007/s00299-010-0828-6
Cherney JH, Cherney DJR, Akin DE, Axtell JD (1991) Potential of brown-midrib, low-lignin mutants for improving forage quality. In: Sparks DL (ed) Advances in agronomy, vol 46. Academic, pp 157–198. https://doi.org/10.1016/S0065-2113(08)60580-5
Childs KL, Miller FR, Cordonnier-Pratt MM, Pratt LH, Morgan PW, Mullet JE (1997) The sorghum photoperiod sensitivity gene, Ma3, encodes a phytochrome B. Plant Physiol 113(2):611–619. https://doi.org/10.1104/pp.113.2.611
Clayton WD, Renvoize SA (1986) Genera graminum. Grasses of the world. H.M.S.O, London
Corsato Alvarenga I, Ou Z, Thiele S, Alavi S, Gregory Aldrich C (2018) Effects of milling sorghum into fractions on yield, nutrient composition, and their performance in extrusion of dog food. J Cereal Sci 82:121–128. https://doi.org/10.1016/j.jcs.2018.05.013
Craig W, Lenzi P, Scotti N, De Palma M, Saggese P, Carbone V, McGrath Curran N, Magee AM, Medgyesy P, Kavanagh TA, Dix PJ, Grillo S, Cardi T (2008) Transplastomic tobacco plants expressing a fatty acid desaturase gene exhibit altered fatty acid profiles and improved cold tolerance. Transgenic Res 17(5):769–782. https://doi.org/10.1007/s11248-008-9164-9
Daniell H (2007) Transgene containment by maternal inheritance: effective or elusive? Proc Natl Acad Sci U S A 104(17):6879–6880. https://doi.org/10.1073/pnas.0702219104
Daniell H, Singh ND, Mason H, Streatfield SJ (2009) Plant-made vaccine antigens and biopharmaceuticals. Trends Plant Sci 14(12):669–679. https://doi.org/10.1016/j.tplants.2009.09.009
Denyer K, Johnson P, Zeeman S, Smith AM (2001) The control of amylose synthesis. J Plant Physiol 158(4):479–487. https://doi.org/10.1078/0176-1617-00360
Department of Agriculture and Fisheries—DAF (2018a) Sorghum—nutrition, irrigation and harvest issues. https://www.daf.qld.gov.au/business-priorities/plants/field-crops-and-pastures/broadacre-field-crops/sorghum/nutrition,-irrigation-and-harvest
Department of Agriculture and Fisheries—DAF (2018b) Insect pest management in sorghum. https://www.daf.qld.gov.au/business-priorities/agriculture/plants/crops-pastures/broadacre-field-crops/insect-pest-management-specific-crops/insect-pest-management-sorghum
Dillon SL, Shapter FM, Henry RJ, Cordeiro G, Izquierdo L, Lee LS (2007) Domestication to crop improvement: genetic resources for Sorghum and Saccharum (Andropogoneae). Ann Bot 100(5):975–989. https://doi.org/10.1093/aob/mcm192
Do PT, Zhang ZJ (2015) Sorghum transformation: achievements, challenges, and perspectives. In: Azhakanandam K, Silverstone A, Daniell H, Davey MR (eds) Recent advancements in gene expression and enabling technologies in crop plants. Springer, New York, pp 291–312. https://doi.org/10.1007/978-1-4939-2202-4_9
Dobrogojski J, Spychalski M, Luciński R, Borek S (2018) Transgenic plants as a source of polyhydroxyalkanoates. Acta Physiol Plant 40(9):162. https://doi.org/10.1007/s11738-018-2742-4
Dolgin E (2019) The bioengineering of cannabis. Nature 572(7771):S5–S7. https://doi.org/10.1038/d41586-019-02525-4
Dunne A, Maple-Grodem J, Gargano D, Haslam RP, Napier JA, Chua NH, Russell R, Moller SG (2014) Modifying fatty acid profiles through a new cytokinin-based plastid transformation system. Plant J 80(6):1131–1138. https://doi.org/10.1111/tpj.12684
Dykes L (2019) Sorghum phytochemicals and their potential impact on human health. In: Z ZY, DJ (eds) Sorghum. Methods in molecular biology, vol. 1931. Humana Press, New York
Ejeta G, Grenier C (2005) Sorghum and its weedy hybrids. Crop ferality and volunteerisim. CRC Press, Boca Raton, pp 123–135
FAO (1999) Sorghum post-harvest operations. http://www.fao.org/fileadmin/user_upload/inpho/docs/Post_Harvest_Compendium_-_SORGHUM.pdf
Farré JC, Araya A (2001) Gene expression in isolated plant mitochondria: high fidelity of transcription, splicing and editing of a transgene product in electroporated organelles. Nucleic Acids Res 29(12):2484–2491. https://doi.org/10.1093/nar/29.12.2484
Fox TD, Sanford JC, McMullin TW (1988) Plasmids can stably transform yeast mitochondria lacking endogenous mtDNA. Proc Natl Acad Sci U S A 85(19):7288–7292. https://doi.org/10.1073/pnas.85.19.7288
Fromm H, Edelman M, Aviv D, Galun E (1987) The molecular basis for rRNA-dependent spectinomycin resistance in Nicotiana chloroplasts. EMBO J 6(11):3233–3237
Fukuoka M, Ohta K, Watanabe H (2002) Determination of the terminal extent of starch gelatinization in a limited water system by DSC. J Food Eng 53(1):39–42. https://doi.org/10.1016/S0260-8774(01)00137-6
Furlani AMC, Clark RB, Maranville JW, Ross WM (1984) Sorghum genotype differences in phosphorus uptake rate and distribution in plant-parts. J Plant Nutr 7(7):1113–1126. https://doi.org/10.1080/01904168409363269
Gabir S, Khristova P, Yossifov N (1990) Composite boards from guar and sorghum (dura) stalks. Biol Wastes 31:311–314. https://doi.org/10.1016/0269-7483(90)90088-A
Getnet Z, Husen A, Fetene M, Yemata G (2015) Growth, water status, physiological, biochemical and yield response of stay green sorghum (Sorghum Bicolor (L.) Moench) varieties—a field trial under drought-prone area in amhara regional state, Ethiopia. J Agron 14:188–202. https://doi.org/10.3923/ja.2015.188.202
Golds T, Maliga P, Koop H-U (1993) Stable plastid transformation in PEG-treated protoplasts of Nicotiana tabacum. Nat Biotechnol 11(1):95–97. https://doi.org/10.1038/nbt0193-95
Gopalan C, Rama Sastri BV, Balasubramanian SC (1996) Nutritive value of Indian foods. Hyderabad
GRDC (2017) Grownotes: sorghum—plant growth and physiology. https://grdc.com.au/__data/assets/pdf_file/0030/370596/GrowNote-Sorghum-North-04-Physiology.pdf
Griswold A (2008) Genome packaging in prokaryotes: the circular chromosome of E. coli. Nat Educ 1:57
Higgins RH, Thurber CS, Assaranurak I, Brown PJ (2014) Multiparental mapping of plant height and flowering time QTL in partially isogenic sorghum families. G3 (Bethesda) 4(9):1593–1602. https://doi.org/10.1534/g3.114.013318
Hilley J, Truong S, Olson S, Morishige D, Mullet J (2016) Identification of Dw1, a regulator of sorghum stem internode length. PLoS One 11(3):e0151271. https://doi.org/10.1371/journal.pone.0151271
Hilley JL, Weers BD, Truong SK, McCormick RF, Mattison AJ, McKinley BA, Morishige DT, Mullet JE (2017) Sorghum Dw2 encodes a protein kinase regulator of stem internode length. Sci Rep 7(1):4616. https://doi.org/10.1038/s41598-017-04609-5
Hirano K, Kawamura M, Araki-Nakamura S, Fujimoto H, Ohmae-Shinohara K, Yamaguchi M, Fujii A, Sasaki H, Kasuga S, Sazuka T (2017) Sorghum DW1 positively regulates brassinosteroid signaling by inhibiting the nuclear localization of brassinosteroid insensitive 2. Sci Rep 7(1):126. https://doi.org/10.1038/s41598-017-00096-w
Hirel B, Le Gouis J, Ney B, Gallais A (2007) The challenge of improving nitrogen use efficiency in crop plants: towards a more central role for genetic variability and quantitative genetics within integrated approaches. J Exp Bot 58(9):2369–2387. https://doi.org/10.1093/jxb/erm097
Hwang HH, Yu M, Lai EM (2017) Agrobacterium-mediated plant transformation: biology and applications. Arabidopsis Book 15:e0186. https://doi.org/10.1199/tab.0186
Jabeen R, Khan MS, Zafar Y, Anjum T (2010) Codon optimization of cry1Ab gene for hyper expression in plant organelles. Mol Biol Rep 37(2):1011–1017. https://doi.org/10.1007/s11033-009-9802-1
Jackson MA, Anderson DJ, Birch RG (2013) Comparison of Agrobacterium and particle bombardment using whole plasmid or minimal cassette for production of high-expressing, low-copy transgenic plants. Transgenic Res 22(1):143–151. https://doi.org/10.1007/s11248-012-9639-6
Jampala B, Rooney WL, Peterson GC, Bean S, Hays DB (2012) Estimating the relative effects of the endosperm traits of waxy and high protein digestibility on yield in grain sorghum. Field Crops Res 139:57–62. https://doi.org/10.1016/j.fcr.2012.09.021
Jannoo N, Grivet L, Chantret N, Garsmeur O, Glaszmann JC, Arruda P, D’Hont A (2007) Orthologous comparison in a gene-rich region among grasses reveals stability in the sugarcane polyploid genome. Plant J 50(4):574–585. https://doi.org/10.1111/j.1365-313X.2007.03082.x
Jiang W, Zhou H, Bi H, Fromm M, Yang B, Weeks DP (2013) Demonstration of CRISPR/Cas9/sgRNA-mediated targeted gene modification in Arabidopsis, tobacco, sorghum and rice. Nucleic Acids Res 41(20):e188. https://doi.org/10.1093/nar/gkt780
Jiao Y, Burke J, Chopra R, Burow G, Chen J, Wang B, Hayes C, Emendack Y, Ware D, Xin Z (2016) A sorghum mutant resource as an efficient platform for gene discovery in grasses. Plant Cell 28(7):1551–1562. https://doi.org/10.1105/tpc.16.00373
Jin S, Daniell H (2015) The engineered chloroplast genome just got smarter. Trends Plant Sci 20(10):622–640. https://doi.org/10.1016/j.tplants.2015.07.004
Johnston SA, Anziano PQ, Shark K, Sanford JC, Butow RA (1988) Mitochondrial transformation in yeast by bombardment with microprojectiles. Science 240(4858):1538–1541. https://doi.org/10.1126/science.2836954
Kawahigashi H, Oshima M, Nishikawa T, Okuizumi H, Kasuga S, Yonemaru J (2013) A novel waxy allele in sorghum landraces in East Asia. Plant Breeding 132(3):305–310. https://doi.org/10.1111/pbr.12054
Kazama T, Nishimura A, Arimura S-i (2018) Rice organelle genomics: approaches to genetic engineering and breeding. In: Sasaki T, Ashikari M (eds) Rice genomics, genetics and breeding. Springer Singapore, Singapore, pp 53–67. https://doi.org/10.1007/978-981-10-7461-5_4
Kohli A, Twyman RM, Abranches R, Wegel E, Stoger E, Christou P (2003) Transgene integration, organization and interaction in plants. Plant Mol Biol 52(2):247–258. https://doi.org/10.1023/a:1023941407376
Koulintchenko M, Konstantinov Y, Dietrich A (2003) Plant mitochondria actively import DNA via the permeability transition pore complex. EMBO J 22(6):1245–1254. https://doi.org/10.1093/emboj/cdg128
Langbecker CL, Ye GN, Broyles DL, Duggan LL, Xu CW, Hajdukiewicz PT, Armstrong CL, Staub JM (2004) High-frequency transformation of undeveloped plastids in tobacco suspension cells. Plant Physiol 135(1):39–46. https://doi.org/10.1104/pp.103.035410
Larosa V, Remacle C (2013) Transformation of the mitochondrial genome. Int J Dev Biol 57(6–8):659–665. https://doi.org/10.1387/ijdb.130230cr
Lemaire G, Charrier X, Hebert Y (1996) Nitrogen uptake capacities of maize and sorghum crops in different nitrogen and water supply conditions. Agronomie 16(4):231–246. https://doi.org/10.1051/agro:19960403
Li H, Huang Y (2017) Expression of brown-midrib in a spontaneous sorghum mutant is linked to a 5’-UTR deletion in lignin biosynthesis gene SbCAD2. Sci Rep 7(1):11664. https://doi.org/10.1038/s41598-017-10119-1
Li X, Li X, Fridman E, Tesso TT, Yu J (2015) Dissecting repulsion linkage in the dwarfing gene Dw3 region for sorghum plant height provides insights into heterosis. Proc Natl Acad Sci U S A 112(38):11823–11828. https://doi.org/10.1073/pnas.1509229112
Li D, Tang N, Fang Z, Xia Y, Cao M (2016a) Co-transfer of TALENs construct targeted for chloroplast genome and chloroplast transformation vector into rice using particle bombardment. J Nanosci Nanotechnol 16(12):12194–12201. https://doi.org/10.1166/jnn.2016.12949
Li Y, Wang R, Hu Z, Li H, Lu S, Zhang J, Lin Y, Zhou F (2016b) Expression of a codon-optimized dsdA gene in tobacco plastids and rice nucleus confers D-serine tolerance. Front Plant Sci 7(640):640. https://doi.org/10.3389/fpls.2016.00640
Li A, Jia S, Yobi A, Ge Z, Sato SJ, Zhang C, Angelovici R, Clemente TE, Holding DR (2018) Editing of an alpha-kafirin gene family increases, digestibility and protein quality in sorghum. Plant Physiol 177(4):1425–1438. https://doi.org/10.1104/pp.18.00200
Liu G, Godwin ID (2012) Highly efficient sorghum transformation. Plant Cell Rep 31(6):999–1007. https://doi.org/10.1007/s00299-011-1218-4
Liu H, Sun B (2018) Effect of fermentation processing on the flavor of Baijiu. J Agric Food Chem 66(22):5425–5432. https://doi.org/10.1021/acs.jafc.8b00692
Liu G, Campbell BC, Godwin ID (2014) Sorghum genetic transformation by particle bombardment. In: Henry RJ, Furtado A (eds) Cereal genomics: methods and protocols. Humana Press, Totowa, pp 219–234. https://doi.org/10.1007/978-1-62703-715-0_18
Lössl A, Bohmert K, Harloff H, Eibl C, Muhlbauer S, Koop HU (2005) Inducible trans-activation of plastid transgenes: expression of the R. eutropha phb operon in transplastomic tobacco. Plant Cell Physiol 46(9):1462–1471. https://doi.org/10.1093/pcp/pci157
Lu Y, Zhao G, Li Y, Fan J, Ding G, Zhao J, Ni X, Xu Y, Wang W (2013) Identification of two novel waxy alleles and development of their molecular markers in sorghum. Genome 56(5):283–288. https://doi.org/10.1139/gen-2013-0047
McIntyre CL, Drenth J, Gonzalez N, Henzell RG, Jordan DR (2008) Molecular characterization of the waxy locus in sorghum. Genome 51(7):524–533. https://doi.org/10.1139/G08-035
Meyers B, Zaltsman A, Lacroix B, Kozlovsky SV, Krichevsky A (2010) Nuclear and plastid genetic engineering of plants: comparison of opportunities and challenges. Biotechnol Adv 28(6):747–756. https://doi.org/10.1016/j.biotechadv.2010.05.022
Moore N, Serafin L, Jenkins L (2014) Grain sorghum. Summer crop production guide. Department of Primary Industries, Sydney, pp 5–16
Multani DS, Briggs SP, Chamberlin MA, Blakeslee JJ, Murphy AS, Johal GS (2003) Loss of an MDR transporter in compact stalks of maize br2 and sorghum dw3 mutants. Science 302(5642):81–84. https://doi.org/10.1126/science.1086072
Murphy RL, Klein RR, Morishige DT, Brady JA, Rooney WL, Miller FR, Dugas DV, Klein PE, Mullet JE (2011) Coincident light and clock regulation of pseudoresponse regulator protein 37 (PRR37) controls photoperiodic flowering in sorghum. Proc Natl Acad Sci U S A 108(39):16469–16474. https://doi.org/10.1073/pnas.1106212108
Murphy RL, Morishige DT, Brady JA, Rooney WL, Yang S, Klein PE, Mullet JE (2014) Ghd7 (Ma6) represses sorghum flowering in long days: Ghd7 alleles enhance biomass accumulation and grain production. Plant Genome 7(2). https://doi.org/10.3835/plantgenome2013.11.0040
Mutisya J, Sitieney JK, Gichuki ST (2010) Phenotypic and physiological aspects related to drought tolerance in sorghum. Afr Crop Sci J 18(4):175–182
NSW Government Department of Primary Industries—NSW DPI (2016) Primefact: salinity tolerance in irrigated crops. https://www.dpi.nsw.gov.au/__data/assets/pdf_file/0005/523643/Salinity-tolerance-in-irrigated-crops.pdf
O’Connell MR, Oakes BL, Sternberg SH, East-Seletsky A, Kaplan M, Doudna JA (2014) Programmable RNA recognition and cleavage by CRISPR/Cas9. Nature 516(7530):263–266. https://doi.org/10.1038/nature13769
Ou-Lee TM, Turgeon R, Wu R (1986) Expression of a foreign gene linked to either a plant-virus or a Drosophila promoter, after electroporation of protoplasts of rice, wheat, and sorghum. Proc Natl Acad Sci U S A 83(18):6815–6819. https://doi.org/10.1073/pnas.83.18.6815
Paterson AH, Bowers JE, Peterson DG, Estill JC, Chapman BA (2003) Structure and evolution of cereal genomes. Curr Opin Genet Dev 13(6):644–650. https://doi.org/10.1016/j.gde.2003.10.002
Paterson AH, Bowers JE, Chapman BA (2004) Ancient polyploidization predating divergence of the cereals, and its consequences for comparative genomics. Proc Natl Acad Sci U S A 101(26):9903–9908. https://doi.org/10.1073/pnas.0307901101
Paterson AH, Bowers JE, Bruggmann R, Dubchak I, Grimwood J, Gundlach H, Haberer G, Hellsten U, Mitros T, Poliakov A, Schmutz J, Spannagl M, Tang H, Wang X, Wicker T, Bharti AK, Chapman J, Feltus FA, Gowik U, Grigoriev IV, Lyons E, Maher CA, Martis M, Narechania A, Otillar RP, Penning BW, Salamov AA, Wang Y, Zhang L, Carpita NC, Freeling M, Gingle AR, Hash CT, Keller B, Klein P, Kresovich S, McCann MC, Ming R, Peterson DG, Mehboob ur R, Ware D, Westhoff P, Mayer KF, Messing J, Rokhsar DS (2009) The Sorghum bicolor genome and the diversification of grasses. Nature 457(7229):551–556. https://doi.org/10.1038/nature07723
Pedersen JF, Rooney WL (2004) Sorghum. In: Mosher LE, Burson BL, Sollenberger LE (eds) Warm-season (C4) grasses. American Society of Agronomy: Crop Science Society of America: Soil Science Society of America, Madison
Pedersen JF, Bean SR, Graybosch RA, Park SH, Tilley M (2005) Characterization of waxy grain sorghum lines in relation to granule-bound starch synthase. Euphytica 144(1–2):151–156. https://doi.org/10.1007/s10681-005-5298-5
Peng C, Wang H, Xu X, Wang X, Chen X, Wei W, Lai Y, Liu G, Godwin ID, Li J, Zhang L, Xu J (2018) High-throughput detection and screening of plants modified by gene editing using quantitative real-time polymerase chain reaction. Plant J 95(3):557–567. https://doi.org/10.1111/tpj.13961
Purnell MP, Petrasovits LA, Nielsen LK, Brumbley SM (2007) Spatio-temporal characterization of polyhydroxybutyrate accumulation in sugarcane. Plant Biotechnol J 5(1):173–184. https://doi.org/10.1111/j.1467-7652.2006.00230.x
Quinby JR, Karper RE (1954) Inheritance of height in sorghum. Agron J 46(5):211–216. https://doi.org/10.2134/agronj1954.00021962004600050007x
Randolph-Anderson BL, Boynton JE, Gillham NW, Harris EH, Johnson AM, Dorthu MP, Matagne RF (1993) Further characterization of the respiratory deficient dum-1 mutation of Chlamydomonas reinhardtii and its use as a recipient for mitochondrial transformation. Mol Gen Genet 236(2–3):235–244. https://doi.org/10.1007/bf00277118
Reece JB, Meyers N, Urry LA, Cain ML, Wasserman SA, Minorsky PV, Jackson RB, Cooke B, Campbell NA (2015) Campbell biology.
Remacle C, Cardol P, Coosemans N, Gaisne M, Bonnefoy N (2006) High-efficiency biolistic transformation of Chlamydomonas mitochondria can be used to insert mutations in complex I genes. Proc Natl Acad Sci U S A 103(12):4771–4776. https://doi.org/10.1073/pnas.0509501103
Remacle C, Larosa V, Salinas T, Hamel P, Subrahmanian N, Bonnefoy N, Kempken F (2012) Transformation and nucleic acid delivery to mitochondria. In: Bock R, Knoop V (eds) Genomics of chloroplasts and mitochondria. Advances in photosynthesis and respiration. Springer, Netherlands, Dordrecht, pp 443–458. https://doi.org/10.1007/978-94-007-2920-9_19
Řepková J (2010) Potential of chloroplast genome in plant breeding. Czech J Genet Plant Breed 46(3):103–113
Rooney WL, Aydin S, Kuhlman LC (2005) Assessing the relationship between endosperm type and grain yield potential in sorghum (Sorghum bicolor L. Moench). Field Crops Res 91(2):199–205. https://doi.org/10.1016/j.fcr.2004.07.011
Roudsari MF, Salmanian A, Mousavi A, Sohi HH, Jafari M (2009) Regeneration of glyphosate-tolerant Nicotiana tabacum after plastid transformation with a mutated variant of bacterial aroA gene. Iran J Biotechnol 7:247–253
Ruf S, Forner J, Hasse C, Kroop X, Seeger S, Schollbach L, Schadach A, Bock R (2019) High-efficiency generation of fertile transplastomic Arabidopsis plants. Nat Plants 5(3):282–289. https://doi.org/10.1038/s41477-019-0359-2
Ryley MJ, Persley DM, Jordan DR, Henzell RG (2008) Status of sorghum and pearl millet diseases in Australia. In: Leslie JF (ed) Sorghum and millets diseases. Wiley, Hoboken
Sant RRP (2012) Development of a transformation system for sorghum (Sorghum bicolor L.). Thesis, Queensland University of Technology. https://eprints.qut.edu.au/50977/
Saski C, Lee SB, Fjellheim S, Guda C, Jansen RK, Luo H, Tomkins J, Rognli OA, Daniell H, Clarke JL (2007) Complete chloroplast genome sequences of Hordeum vulgare, Sorghum bicolor and Agrostis stolonifera, and comparative analyses with other grass genomes. Theor Appl Genet 115(4):571–590. https://doi.org/10.1007/s00122-007-0567-4
Sattler SE, Singh J, Haas EJ, Guo LN, Sarath G, Pedersen JF (2009) Two distinct waxy alleles impact the granule-bound starch synthase in sorghum. Mol Breed 24(4):349–359. https://doi.org/10.1007/s11032-009-9296-5
Sega GA (1984) A review of the genetic effects of ethyl methanesulfonate. Mutat Res 134(2–3):113–142. https://doi.org/10.1016/0165-1110(84)90007-1
Singh V, Nguyen CT, van Oosterom EJ, Chapman SC, Jordan DR, Hammer GL (2015) Sorghum genotypes differ in high temperature responses for seed set. Field Crops Res 171:32–40. https://doi.org/10.1016/j.fcr.2014.11.003
Small ID, Isaac PG, Leaver CJ (1987) Stoichiometric differences in DNA molecules containing the atpA gene suggest mechanisms for the generation of mitochondrial genome diversity in maize. EMBO J 6(4):865–869
Small I, Suffolk R, Leaver CJ (1989) Evolution of plant mitochondrial genomes via substoichiometric intermediates. Cell 58(1):69–76. https://doi.org/10.1016/0092-8674(89)90403-0
Somleva MN, Snell KD, Beaulieu JJ, Peoples OP, Garrison BR, Patterson NA (2008) Production of polyhydroxybutyrate in switchgrass, a value-added co-product in an important lignocellulosic biomass crop. Plant Biotechnol J 6(7):663–678. https://doi.org/10.1111/j.1467-7652.2008.00350.x
Somleva MN, Peoples OP, Snell KD (2013) PHA bioplastics, biochemicals, and energy from crops. Plant Biotechnol J 11(2):233–252. https://doi.org/10.1111/pbi.12039
Stefaniak TR, Dahlberg JA, Bean BW, Dighe N, Wolfrum EJ, Rooney WL (2012) Variation in biomass composition components among forage, biomass, sorghum-sudangrass, and sweet sorghum types. Crop Sci 52(4):1949–1954. https://doi.org/10.2135/cropsci2011.10.0534
Svab Z, Hajdukiewicz P, Maliga P (1990) Stable transformation of plastids in higher plants. Proc Natl Acad Sci U S A 87(21):8526–8530. https://doi.org/10.1073/pnas.87.21.8526
Swigonová Z, Lai J, Ma J, Ramakrishna W, Llaca V, Bennetzen JL, Messing J (2004) Close split of sorghum and maize genome progenitors. Genome Res 14(10A):1916–1923. https://doi.org/10.1101/gr.2332504
Tadesse Y, Sagi L, Swennen R, Jacobs M (2003) Optimisation of transformation conditions and production of transgenic sorghum (Sorghum bicolor) via microparticle bombardment. Plant Cell Tiss Org 75(1):1–18. https://doi.org/10.1023/A:1024664817800
Taylor JRN, Schober TJ, Bean SR (2006) Novel food and non-food uses for sorghum and millets. J Cereal Sci 44(3):252–271. https://doi.org/10.1016/j.jcs.2006.06.009
Tetreault HM, Scully ED, Gries T, Palmer NA, Funnell-Harris DL, Baird L, Seravalli J, Dien BS, Sarath G, Clemente TE, Sattler SE (2018) Overexpression of the Sorghum bicolor SbCCoAOMT alters cell wall associated hydroxycinnamoyl groups. PLoS One 13(10):e0204153. https://doi.org/10.1371/journal.pone.0204153
United States Department of Agriculture—USDA (2020) World agricultural production. https://downloads.usda.library.cornell.edu/usda-esmis/files/5q47rn72z/dz0117445/qv33sf19j/production.pdf
Upadhyaya HD, Wang YH, Sharma S, Singh S (2012) Association mapping of height and maturity across five environments using the sorghum mini core collection. Genome 55(6):471–479. https://doi.org/10.1139/g2012-034
Van Oosterom EJ, Jayachandran R, Bidinger FR (1996) Diallel analysis of the stay-green trait and its components in sorghum. Crop Sci 36(3):549–555. https://doi.org/10.2135/cropsci1996.0011183X003600030002x
Von Caemmerer S, Furbank RT (2003) The C(4) pathway: an efficient CO(2) pump. Photosynth Res 77(2–3):191–207. https://doi.org/10.1023/A:1025830019591
Wang W, Wang J, Yang C, Li Y, Liu L, Xu J (2007) Pollen-mediated transformation of Sorghum bicolor plants. Biotechnol Appl Biochem 48(Pt 2):79–83. https://doi.org/10.1042/ba20060231
Worland B, Robinson N, Jordan D, Schmidt S, Godwin I (2017) Post-anthesis nitrate uptake is critical to yield and grain protein content in Sorghum bicolor. J Plant Physiol 216:118–124. https://doi.org/10.1016/j.jplph.2017.05.026
Wu E, Lenderts B, Glassman K, Berezowska-Kaniewska M, Christensen H, Asmus T, Zhen S, Chu U, Cho MJ, Zhao ZY (2014) Optimized Agrobacterium-mediated sorghum transformation protocol and molecular data of transgenic sorghum plants. In Vitro Cell Dev Biol Plant 50(1):9–18. https://doi.org/10.1007/s11627-013-9583-z
Xin ZG, Wang ML, Burow G, Burke J (2009) An induced sorghum mutant population suitable for bioenergy research. Bioenergy Res 2(1–2):10–16. https://doi.org/10.1007/s12155-008-9029-3
Xin ZG, Burow G, Burke J (2014) Multi-seed mutant of sorghum for increasing grain yield. In: Google Patents.
Xin ZG, Gitz D, Burow G, Hayes C, Burke JJ (2015) Registration of two allelic erect leaf mutants of sorghum. J Plant Registr 9(2):254–257. https://doi.org/10.3198/jpr2014.09.0060crgs
Yamaguchi M, Fujimoto H, Hirano K, Araki-Nakamura S, Ohmae-Shinohara K, Fujii A, Tsunashima M, Song XJ, Ito Y, Nagae R, Wu J, Mizuno H, Yonemaru J, Matsumoto T, Kitano H, Matsuoka M, Kasuga S, Sazuka T (2016) Sorghum Dw1, an agronomically important gene for lodging resistance, encodes a novel protein involved in cell proliferation. Sci Rep 6(1):28366. https://doi.org/10.1038/srep28366
Yang S, Murphy RL, Morishige DT, Klein PE, Rooney WL, Mullet JE (2014) Sorghum phytochrome B inhibits flowering in long days by activating expression of SbPRR37 and SbGHD7, repressors of SbEHD1, SbCN8 and SbCN12. PLoS One 9(8):e105352. https://doi.org/10.1371/journal.pone.0105352
Yerka MK, Toy JJ, Funnell-Harris DL, Sattler SE, Pedersen JF (2015) Registration of N619 to N640 grain sorghum lines with waxy or wild-type endosperm. J Plant Registr 9(2):249–253. https://doi.org/10.3198/jpr2014.06.0043crgs
Yerka MK, Toy JJ, Funnell-Harris DL, Sattler SE, Pedersen JF (2016) Evaluation of interallelic waxy, heterowaxy, and wild-type grain sorghum hybrids. Crop Sci 56(1):113–121. https://doi.org/10.2135/cropsci2015.03.0151
Zegada-Lizarazu W, Monti A (2012) Are we ready to cultivate sweet sorghum as a bioenergy feedstock? a review on field management practices. Biomass Bioenergy 40:1–12. https://doi.org/10.1016/j.biombioe.2012.01.048
Zhang D, Kong W, Robertson J, Goff VH, Epps E, Kerr A, Mills G, Cromwell J, Lugin Y, Phillips C, Paterson AH (2015) Genetic analysis of inflorescence and plant height components in sorghum (Panicoidae) and comparative genetics with rice (Oryzoidae). BMC Plant Biol 15(1):107. https://doi.org/10.1186/s12870-015-0477-6
Źróbek-Sokolnik A (2012) Temperature stress and responses of plants. In: Ahmad P, Prasad MNV (eds) Environmental adaptations and stress tolerance of plants in the era of climate change. Springer, New York, pp 113–134. https://doi.org/10.1007/978-1-4614-0815-4_5
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Wong, A.C.S., Lam, Y., Hintzsche, J., Restall, J., Godwin, I.D. (2022). Genome Editing Towards Sorghum Improvement. In: Zhao, K., Mishra, R., Joshi, R.K. (eds) Genome Editing Technologies for Crop Improvement. Springer, Singapore. https://doi.org/10.1007/978-981-19-0600-8_14
Download citation
DOI: https://doi.org/10.1007/978-981-19-0600-8_14
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-0599-5
Online ISBN: 978-981-19-0600-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)