Abstract
Aims
Tropical tree and lianas in the understory are limited by soil nutrients despite growing in extremely low light. It is not known if nomadic vines are also limited by nutrients in low light conditions.
Methods
We measured differences in root architecture and mycorrhizal colonization, and leaf nutrients of a nomadic vine, Philodendron fragrantissimum (Araceae), in nitrogen (N) and phosphorus (P) fertilization plots in a lowland tropical moist forest in central Panama to measure potential nutrient limitation.
Results
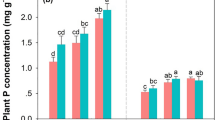
Relative to plants in control plots, leaf P concentration was 54% higher and leaf N concentration was 10% higher for plants in the P- and N-addition treatments, respectively. The N:P of leaves suggested P-limitation in the N-addition treatment and the control but not in the P-addition treatment. Root branching was highest in the P-addition treatment, and P-addition reduced mycorrhizal colonization.
Conclusions
The large effect of P fertilization suggests that, like many tropical plants, P. fragrantissimum has the potential to be P-limited. Although further study is needed, we suggest that nomadic vines be added to the growth forms that respond to nutrient addition in the forest understory and conclude that nutrient-limitation seems like the rule rather than the exception in the light-limited understory.




Similar content being viewed by others
References
Arsenault JL, Pouleur S, Messier C, Guay R (1995) WinRhizo, a root measuring system with a unique overlap correction method. HortScience 30:906
Balcázar-Vargas MP, Peñuela-Mora MC, van Andel TR, Zuidema PA (2012) The quest for a suitable host: size distributions of host trees and secondary hemiepiphytes search strategy. Biotropica 44:19–26
Benner JW, Vitousek PM (2007) Development of a diverse epiphyte community in response to phosphorus fertilization. Ecol Lett 10:628–636
Benzing DH (1990) Vascular epiphytes. General biology and related biota. Cambridge University Press, Cambridge
Bigelow SW (1993) Leaf nutrients in relation to stature and life form in tropical rain forest. J Veg Sci 4:401–408. https://doi.org/10.2307/3235599
Bloom AJ, Chapin FS III, Mooney HA (1985) Resource limitation in plants - an economic analogy. Annu Rev Ecol Evol Syst 16:363–392
Burslem DFRP, Grubb PJ, Turner IM (1996) Responses to simulated drought and elevated nutrient supply among shade-tolerant tree seedlings of lowland tropical forest in Singapore. Biotropica 28:636–648
Chapin FS III (1980) The mineral nutrition of wild plants. Annu Rev Ecol Evol Syst 11:233–260
Chazdon RL, Fetcher N (1984) Light environments of tropical forests. In: Medina E, Mooney HA, Vásquez-Yanes C (eds) Physiol. Ecol. Plants Wet Trop. Dr. W. Junk Publishers, The Hague, pp 553–564
Clark RB, Zeto SK (2000) Mineral acquisition by arbuscular mycorrhizal plants. J Plant Nutr 23:867–902
Coley PD, Bryant JP, Chapin FS III (1985) Resource availability and plant antiherbivore defense. Science 230:895–900
Collins CG, Wright SJ, Wurzburger N (2016) Root and leaf traits reflect distinct resource acquisition strategies in tropical lianas and trees. Oecologia 180:1037–1047
Condit R, Engelbrecht BMJ, Pino D, Perez R, Turner BL (2013) Species distributions in responses to individual soil nutrients and seasonal drought across a community of tropical trees. Proc Natl Acad Sci 110:5064–5068
Coomes DA, Grubb PJ (2000) Impacts of root competition in forests and woodlands: a theoretical framework and review of experiments. Ecol Monogr 70:171–207
Cuevas E, Medina E (1988) Nutrient dynamics within Amazonian forests: fine root growth, nutrient availability, and leaf litter decomposition. Oecologia 76:222–235
D’Angelo E, Crutchfield J, Vandiviere M (2001) Rapid, sensitive, microscale determination of phosphate in water and soil. J Environ Qual 30:2206–2209
Drew MC (1975) Comparison of the effects of a localized supply of phosphate, nitrate, ammonium and potassium on the growth of the seminal root system, and the shoot, in barley. New Phytol 75:479–490
Drew MC, Saker LR (1978) Nutrient supply and the growth of the seminal root system in barley. III. Compensatory increases in growth of lateral roots, and in rates of phosphate uptake in response to a localised supply of phosphate. J Exp Bot 29:435–451
Dubuisson J-Y, Hennequin S, Bary S, Ebihara A, Boucheron-Dubuisson É (2011) Anatomical diversity and regressive evolution in the trichomanoid filmy ferns (Hymenophyllaceae): a phylogenetic approach. C R Biol 334:880–895
Eissenstat DM, Yanai RD (1997) The ecology of root lifespan. Adv Ecol Res 27:1–60
Elser J, Bracken MES, Cleland EE et al (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142
Farley RA, Fitter AH (1999) The response of seven co-occuring woodland herbaceous perennials to localized nutrient-rich patches. J Ecol 87:849–859
Fitter AH (1985) Functional significance of root morphology and root system architecture. In: Fitter AH, Atkinson D, Read DJ, Usher MB (eds) Ecol. Interact. soil. Blackwell Scientific Publications, Oxford, pp 87–106
Fitter AH (1991) Costs and benefits of mycorrhizae: implications for functioning under natural conditions. Experientia 47:350–355
Fitter AH, Williamson L, Linkohr B, Leyser O (2002) Root system architecture determines fitness in an Arabidopsis mutant in competition for immobile phosphate ions but not for nitrate ions. Proc R Soc London B 269:2017–2022
Güsewell S (2004) N:P ratios in terrestrial plants: variation and functional significance. New Phytol 164:243–266
Hättenschwiler S (2002) Liana seedling growth in response to fertilisation in a neotropical forest understorey. Basic Appl Ecol 3:135–143
Hodge A (2004) The plastic plant: root responses to heterogeneous supplies of nutrients. New Phytol 162:9–24
Holdridge LR, Budowski G (1956) Report on an ecological survey of the Republic of Panama. Caribb For 17:92–110
Jackson RB, Manwaring JH, Caldwell MM (1990) Rapid physiological adjustment of roots to localized soil enrichment. Nature 344:58–60
Jones JB, Case BW (1996) Soil testing and plant analysis no. 3. In: Sparks DL (ed) Methods soil anal. Part 3 Chem. Methods. Soil Science Society of America, Madison, pp 389–415
Kaspari M, Garcia MN, Harms KE, Santana M, Wright SJ, Yavitt JB (2008) Multiple nutrients limit litterfall and decomposition in a tropical forest. Ecol Lett 11:35–43
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res 92:486–505
Lambers H, Raven JA, Shaver GR, Smith SE (2008) Plant nutrient-acquisition strategies change with soil age. Trends Ecol Evol 23:95–103
LeBauer DS, Treseder KK (2008) Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 89:371–379
Marschner P (2012) Mineral nutrition of wild plants, 3rd edn. Academic Press, San Diego
Marschner H, Dell B (1994) Nutrient uptake in mycorrhizal symbiosis. Plant Soil 159:89–102
Mayor JR, Wright SJ, Turner BL (2014) Species-specific responses of foliar nutrients to long-term nitrogen and phosphorus additions in a lowland tropical forest. J Ecol 102:36–44. https://doi.org/10.1111/1365-2745.12190
McCormack ML, Dickie IA, Eissenstat DM et al (2015) Redefining fine roots improves understanding of below-ground contributions to terrestrial biosphere processes. New Phytol 207:505–518
McGonigle TP, Miller MH, Evans DG et al (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501
Mirabello MJ, Yavitt JB, Garcia M, Harms KE, Turner BL, Wright SJ (2013) Soil phosphorus responses to chronic nutrient fertilisation and seasonal drought in a humid lowland forest, Panama. Soil Res 51:215–221
Mirmanto E, Proctor J, Green J, Nagy L, Suriantata (1999) Effects of nitrogen and phosphorus fertilization in a lowland evergreen rainforest. Philos Trans R Soc Lond Ser B Biol Sci 354:1825–1829. https://doi.org/10.1098/rstb.1999.0524
Mo Q, Zou B, Li Y et al (2015) Response of plant nutrient stoichiometry to fertilization varied with plant tissues in a tropical forest. Sci Rep 5:1–12
Moffett MW (2000) What’s “up”? A critical look at the basic terms of canopy biology. Biotropica 32:569–596
Nielsen SL, Enríquez S, Duarte CM, Sand-Jensen K (1996) Scaling maximum growth rates across photosynthetic organisms. Funct Ecol 10:167–175
Ostertag R (2001) Effects of nitrogen and phosphorus availability on fine-root dynamics in Hawaiian montane forests. Ecology 82:485–499
Pasquini SC, Wright SJ, Santiago LS (2015) Lianas always outperform tree seedlings regardless of soil nutrients: results from a long-term fertilization experiment. Ecology 96:1866–1876
Putz FE, Holbrook NM (1986) Notes on the natural history of hemiepiphytes. Selbyana 9:61–69
R Development Core Team (2009) R: A language and environment for statistical computing
Robinson D, Rorison IH (1983) A comparison of the responses of Lolium perenne L., Holcus lanatus L., and Deschampsia flexuosa (L.) Trin. To a localized supply of nitrogen. New Phytol 94:263–273
Santiago LS, Wright SJ, Harms KE, Yavitt JB, Korine C, Garcia MN, Turner BL (2012) Tropical tree seedling growth responses to nitrogen, phosphorus and potassium addition. J Ecol 100:309–316
Sayer EJ, Wright SJ, Tanner EVJ et al (2012) Variable responses of lowland tropical forest nutrient status to fertilization and litter manipulation. Ecosystems 15:387–400
Schnitzer SA, Bongers F (2002) The ecology of lianas and their role in forests. Trends Ecol Evol 17:223–230
Schnitzer SA, Kuzee ME, Bongers F (2005) Disentangling above- and below-ground competition between lianas and trees in a tropical forest. J Ecol 93:1115–1125
Sheldrake M, Rosenstock NP, Revillini D, Olsson PA, Wright SJ, Turner BL (2017) A phosphorus threshold for mycoheterotrophic plants in tropical forests. Proc R Soc London B 284:20162093
Soil Staff Survey (1999) Soil taxonomy: a basic system of soil classification for making and interpreting soil surveys
Stewart RH, Stewart JL, Woodring WP (1980) Geologic map of the Panama Canal and vicinity, Republic of Panama. U.S. Geol. Surv. Misc. Investig. Ser. Map I-232
Strong DR, Ray TS (1975) Host tree location behavior of a tropical vine (Monstera gigantea) by skototropism. Science 190:804–806
Tanner EVJ, Kapos V, Franco W (1992) Nitrogen and phosphorus fertilization effects on Venezuelan montane forest trunk growth and litterfall. Ecology 73:78–86. https://doi.org/10.2307/1938722
Tibbet M (2000) Roots, foraging and the exploitation of soil nutrient patches: the role of mycorrhizal symbioses. Funct Ecol 14:397–399
Toledo-Aceves T, Swaine MD (2008) Above- and below-ground competition between the liana Acacia kamerunensis and tree seedlings in contrasting light environments. Plant Ecol 196:233–244
Treseder KK (2004) A meta-analysis of mycorrhizal responses to nitrogen, phosphorus, and atmospheric CO2 in field studies. New Phytol 164:347–355. https://doi.org/10.1111/j.1469-8137.2004.01159.x
Tripler CW, Kaushal SS, Likens GE, Walter MT (2006) Patterns in potassium dynamics in forest ecosystems. Ecol Lett 9:451–466
Turner BL, Yavitt JB, Harms KE, Garcia MN, Romero TE, Wright SJ (2013) Seasonal changes and treatment effects on soil inorganic nutrients following a decade of fertilization in a lowland tropical forest. Soil Sci Soc Am J 77:1357–1369
van Vuuren MMI, Robinson D, Griffiths BS (1996) Nutrient inflow and root proliferation during the exploitation of a temporally and spatially discrete source of nitrogen in soil. Plant Soil 178:185–192
Vitousek PM, Farrington H (1997) Nutrient limitation and soil development: experimental test of a biogeochemical theory. Biogeochemistry 37:63–75
Wanek W, Zotz G (2011) Are vascular epiphytes nitrogen or phosphorus limited? A study of plant 15N fractionation and foliar N:P stoichiometry with the tank bromeliad Vriesea sanguinolenta. New Phytol 192:462–470
Wang L, Mou PP, Jones RH (2006) Nutrient foraging via physiological and morphological plasticity in three plant species. Can J For Res 36:164–173
Williams-Linera G, Lawton RO (1995) The ecology of hemiepiphytes in forest canopies. In: Lowman MD, Nadkarni NM (eds) For. Canopies. Academic Press, New York, New York, pp 255–283
Winkler U, Zotz G (2009) Highly efficient uptake of phosphorus in epiphytic bromeliads. Ann Bot 103:477–484
Woods CL, DeWalt SJ (2013) The conservation value of secondary forests for vascular epiphytes in Central Panama. Biotropica 45:119–127
Wright SJ, Yavitt JB, Wurzburger N, Turner BL, Tanner EVJ, Sayer EJ, Santiago LS, Kaspari M, Hedin LO, Harms KE, Garcia MN, Corre MD (2011) Potassium, phosphorus, or nitrogen limit root allocation, tree growth, or litter production in a lowland tropical forest. Ecology 92:1616–1625
Wright SJ, Turner BL, Yavitt JB, Harms KE, Kaspari M, Tanner EVJ, Bujan J, Griffin EA, Mayor JR, Pasquini SC, Sheldrake M, Garcia MN (2018) Plant responses to fertilization experiments in lowland, species-rich, tropical forests. Ecology 99:1129–1138. https://doi.org/10.1002/ecy.2193
Wurzburger N, Wright SJ (2015) Fine-root responses to fertilization reveal multiple nutrient limitation in a lowland tropical forest. Ecology 96:2137–2146
Yavitt JB, Wright SJ, Wieder RK (2004) Seasonal drought and dry-season irrigation influence leaf-litter nutrients and soil enzymes in a moist, lowland forest in Panama. Austral Ecol 29:177–188
Yavitt JB, Harms KE, Garcia MN, Wright SJ, He F, Mirabello MJ (2009) Spatial heterogeneity of soil chemical properties in a lowland tropical moist forest, Panama. Aust J Soil Res 47:674–687
Yavitt JB, Harms KE, Garcia MN et al (2011) Soil fertility and fine root dynamics in response to 4 years of nutrient (N, P, K) fertilization in a lowland tropical moist forest, Panama. Austral Ecol 36:433–445
Zhu G, Yoh M, Gilliam FS et al (2013) Nutrient limitation in three lowland tropical forests in southern China receiving high nitrogen deposition: insights from the fine root responses to nutrient additions. PLoS One 8:e82661
Zotz G (2013) “Hemiepiphyte”: confusing term and its history. Ann Bot 111:1015–1020
Acknowledgements
The authors thank Mark Wagner for field support and data collection; Christina Wells for the use of her lab to conduct root mycorrhizal analyses as well as use of the WinRhizo program; and the Smithsonian Tropical Research Institute and Barro Colorado Island for logistical support. We thank G. Zotz and an anonymous reviewer for helpful comments and edits on previous versions of our manuscript. Funding for this research was provided by Clemson University and a Wade T. Batson award for field botany to CLW. Financial support for the Gigante Fertilization Project is provided by grants from the Andrew W. Mellon Foundation and the Smithsonian Scholarly Studies program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Hans Lambers.
Rights and permissions
About this article
Cite this article
Woods, C.L., DeWalt, S.J., Cardelús, C.L. et al. Fertilization influences the nutrient acquisition strategy of a nomadic vine in a lowland tropical forest understory. Plant Soil 431, 389–399 (2018). https://doi.org/10.1007/s11104-018-3772-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-018-3772-9