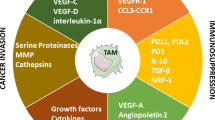
Abstract
Tumor microenvironment (TME) is a complex and constantly evolving entity that consists not only of cancer cells, but also of resident host cells and immune-infiltrating cells, among which macrophages are significant components, due to their diversity of functions through which they can influence the immune response against tumor cells. Macrophages present in tumor environment are termed as tumor-associated macrophages (TAMs). They are strongly plastic cells, and depending on the TME stimuli (i.e., cytokines, chemokines), TAMs polarize to antitumoral (M1-like TAMs) or protumoral (M2-like TAMs) phenotype. Both types of TAMs differ in the surface receptors’ expression, activation of intracellular signaling pathways, and ability of production and various metabolites release. At the early stage of tumor formation, TAMs are M1-like phenotype, and they are able to eliminate tumor cells, i.e., by reactive oxygen species formation or by presentation of cancer antigens to other effector immune cells. However, during tumor progression, TAMs M2-like phenotype is dominating. They mainly contribute to angiogenesis, stromal remodeling, enhancement of tumor cells migration and invasion, and immunosuppression. This wide variety of TAMs’ functions makes them an excellent subject for use in developing antitumor therapies which mainly is based on three strategies: TAMs’ elimination, reprograming, or recruitment inhibition.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- AKT:
-
serine-threonine protein kinase
- Ang-2:
-
angiopoietin-2
- APCs:
-
antigen-presenting cells
- ARG1:
-
arginase-1
- CCL:
-
C-C chemokine ligand
- CTLs:
-
cytotoxic T lymphocytes
- CXCL:
-
C-X-C motif chemokine ligand
- DCs:
-
dendritic cells
- ECM:
-
extracellular matrix
- EGF:
-
epidermal growth factor
- EMT:
-
epithelial–mesenchymal transition
- GM-CSF/CSF-2:
-
granulocyte-macrophage colony-stimulating factor
- HIF:
-
hypoxia-inducible factor
- ICB:
-
immune-checkpoint blockade
- IFN:
-
interferon
- IL:
-
interleukin
- JAK:
-
Janus kinase
- M1:
-
classically activated macrophages
- M2:
-
alternatively activated macrophages
- M-CSF/CSF-1:
-
macrophage colony-stimulating factor
- MHC:
-
major histocompatibility complex
- MIF:
-
migration inhibitory factor
- MMP:
-
metalloproteinase
- NF-κB:
-
nuclear factor kappa B
- NK:
-
natural killer
- PD-1:
-
programmed cell death protein 1
- PDGF:
-
platelet-derived growth factor
- PD-L1:
-
programmed cell death ligand 1
- PI3K:
-
phosphatidylinositol 3-kinase
- PlGF:
-
placenta growth factor
- RNS:
-
reactive nitrogen species
- ROS:
-
reactive oxygen species
- STAT:
-
signal transducer and activator of transcription
- TAMs:
-
tumor-associated macrophages
- TEMs:
-
TIE-2-expressing monocytes
- TGF:
-
transforming growth factor
- TIE:
-
Tek tyrosine kinase receptor
- TLR:
-
Toll-like receptor
- TME:
-
tumor microenvironment
- TNF:
-
tumor necrosis factor
- VEGF:
-
vascular endothelial growth factor
References
Achyut BR, Shankar A, Iskander ASM et al (2015) Bone marrow derived myeloid cells orchestrate antiangiogenic resistance in glioblastoma through coordinated molecular networks. Cancer Lett 369:416–426. https://doi.org/10.1016/j.canlet.2015.09.004
Advani R, Flinn I, Popplewell L et al (2018) CD47 blockade by Hu5F9-G4 and rituximab in non-Hodgkin’s lymphoma. N Engl J Med 379:1711–1721. https://doi.org/10.1056/NEJMoa1807315
Allavena P, Sica A, Solinas G et al (2008) The inflammatory micro-environment in tumor progression: the role of tumor-associated macrophages. Crit Rev Oncol Hematol 66:1–9. https://doi.org/10.1016/j.critrevonc.2007.07.004
Anderson NM, Simon MC (2020) The tumor microenvironment. Curr Biol 30:R921–R925. https://doi.org/10.1016/j.cub.2020.06.081
Anfray C, Ummarino A, Torres Andón F, Allavena P (2019) Current strategies to target tumor-associated-macrophages to improve anti-tumor immune responses. Cell 9. https://doi.org/10.3390/cells9010046
Argyle D, Kitamura T (2018) Targeting macrophage-recruiting chemokines as a novel therapeutic strategy to prevent the progression of solid tumors. Front Immunol 9:2629. https://doi.org/10.3389/fimmu.2018.02629
Arlauckas SP, Garris CS, Kohler RH et al (2017) In vivo imaging reveals a tumor-associated macrophage-mediated resistance pathway in anti-PD-1 therapy. Sci Transl Med 9. https://doi.org/10.1126/scitranslmed.aal3604
Badawi MA, Abouelfadl DM, El-Sharkawy SL et al (2015) Tumor-associated macrophage (TAM) and angiogenesis in human colon carcinoma. Open Access Maced J Med Sci 3:209–214. https://doi.org/10.3889/oamjms.2015.044
Baghban R, Roshangar L, Jahanban-Esfahlan R et al (2020) Tumor microenvironment complexity and therapeutic implications at a glance. Cell Communication and Signaling 18:59. https://doi.org/10.1186/s12964-020-0530-4
Balkwill F (2004) Cancer and the chemokine network. Nat Rev Cancer 4:540–550. https://doi.org/10.1038/nrc1388
Balkwill F, Coussens LM (2004) Cancer: an inflammatory link. Nature 431:405–406. https://doi.org/10.1038/431405a
Balkwill FR, Capasso M, Hagemann T (2012) The tumor microenvironment at a glance. J Cell Sci 125:5591–5596. https://doi.org/10.1242/jcs.116392
Beatty GL, Chiorean EG, Fishman MP et al (2011) CD40 agonists alter tumor stroma and show efficacy against pancreatic carcinoma in mice and humans. Science 331:1612–1616. https://doi.org/10.1126/science.1198443
Belgiovine C, D’Incalci M, Allavena P, Frapolli R (2016) Tumor-associated macrophages and anti-tumor therapies: complex links. Cell Mol Life Sci 73:2411–2424. https://doi.org/10.1007/s00018-016-2166-5
Benoit M, Desnues B, Mege J-L (2008) Macrophage polarization in bacterial infections. J Immunol 181:3733–3739. https://doi.org/10.4049/jimmunol.181.6.3733
Biswas SK, Chittezhath M, Shalova IN, Lim J-Y (2012) Macrophage polarization and plasticity in health and disease. Immunol Res 53:11–24. https://doi.org/10.1007/s12026-012-8291-9
Bocanegra Gondan AI, Ruiz-de-Angulo A, Zabaleta A et al (2018) Effective cancer immunotherapy in mice by polyIC-imiquimod complexes and engineered magnetic nanoparticles. Biomaterials 170:95–115. https://doi.org/10.1016/j.biomaterials.2018.04.003
Boimel PJ, Smirnova T, Zhou ZN et al (2012) Contribution of CXCL12 secretion to invasion of breast cancer cells. Breast Cancer Res 14:R23. https://doi.org/10.1186/bcr3108
Borgoni S, Iannello A, Cutrupi S et al (2018) Depletion of tumor-associated macrophages switches the epigenetic profile of pancreatic cancer infiltrating T cells and restores their anti-tumor phenotype. Onco Targets Ther 7:e1393596. https://doi.org/10.1080/2162402X.2017.1393596
Boussiotis VA, Chatterjee P, Li L (2014) Biochemical signaling of PD-1 on T cells and its functional implications. Cancer J 20:265–271. https://doi.org/10.1097/PPO.0000000000000059
Bowman RL, Klemm F, Akkari L et al (2016) Macrophage ontogeny underlies differences in tumor-specific education in brain malignancies. Cell Rep 17:2445–2459. https://doi.org/10.1016/j.celrep.2016.10.052
Bromberg J, Wang TC (2009) Inflammation and cancer: IL-6 and STAT3 complete the link. Cancer Cell 15:79–80. https://doi.org/10.1016/j.ccr.2009.01.009
Bron S, Henry L, Faes-van’t Hull E et al (2015) TIE-2-expressing monocytes are lymphangiogenic and associate specifically with lymphatics of human breast cancer. Onco Targets Ther 5. https://doi.org/10.1080/2162402X.2015.1073882
Cai X, Yin Y, Li N et al (2012) Re-polarization of tumor-associated macrophages to pro-inflammatory M1 macrophages by microRNA-155. J Mol Cell Biol 4:341–343. https://doi.org/10.1093/jmcb/mjs044
Cai J, Xia L, Li J et al (2019) Tumor-associated macrophages derived TGF-β–induced epithelial to mesenchymal transition in colorectal cancer cells through Smad2,3-4/snail signaling pathway. Cancer Res Treat 51:252–266. https://doi.org/10.4143/crt.2017.613
Calandra T, Roger T (2003) Macrophage migration inhibitory factor: a regulator of innate immunity. Nat Rev Immunol 3:791–800. https://doi.org/10.1038/nri1200
Carmeliet P (2005) VEGF as a key mediator of angiogenesis in cancer. Oncology 69(Suppl 3):4–10. https://doi.org/10.1159/000088478
Carminati L, Pinessi D, Borsotti P et al (2019) Antimetastatic and antiangiogenic activity of trabectedin in cutaneous melanoma. Carcinogenesis 40:303–312. https://doi.org/10.1093/carcin/bgy177
Cassetta L, Pollard JW (2018) Targeting macrophages: therapeutic approaches in cancer. Nat Rev Drug Discov 17:887–904. https://doi.org/10.1038/nrd.2018.169
Chen Q, Zhang XH-F, Massagué J (2011) Macrophage binding to receptor VCAM-1 transmits survival signals in breast cancer cells that invade the lungs. Cancer Cell 20:538–549. https://doi.org/10.1016/j.ccr.2011.08.025
Chen BJ, Chapuy B, Ouyang J et al (2013) PD-L1 expression is characteristic of a subset of aggressive B-cell lymphomas and virus-associated malignancies. Clin Cancer Res 19:3462–3473. https://doi.org/10.1158/1078-0432.CCR-13-0855
Chen Y, Huang Y, Reiberger T et al (2014) Differential effects of sorafenib on liver versus tumor fibrosis mediated by stromal-derived factor 1 alpha/C-X-C receptor type 4 axis and myeloid differentiation antigen-positive myeloid cell infiltration in mice. Hepatology 59:1435–1447. https://doi.org/10.1002/hep.26790
Chen Z, Feng X, Herting CJ et al (2017) Cellular and molecular identity of tumor-associated macrophages in glioblastoma. Cancer Res 77:2266–2278. https://doi.org/10.1158/0008-5472.CAN-16-2310
Chen Y, Song Y, Du W et al (2019) Tumor-associated macrophages: an accomplice in solid tumor progression. J Biomed Sci 26:78. https://doi.org/10.1186/s12929-019-0568-z
Cheng H, Wang Z, Fu L, Xu T (2019) Macrophage polarization in the development and progression of ovarian cancers: an overview. Front Oncol 9:421. https://doi.org/10.3389/fonc.2019.00421
Cho U, Kim B, Kim S et al (2018) Pro-inflammatory M1 macrophage enhances metastatic potential of ovarian cancer cells through NF-κB activation. Mol Carcinog 57:235–242. https://doi.org/10.1002/mc.22750
Coffelt SB, Chen Y-Y, Muthana M et al (2011) Angiopoietin 2 stimulates TIE2-expressing monocytes to suppress T cell activation and to promote regulatory T cell expansion. J Immunol 186:4183–4190. https://doi.org/10.4049/jimmunol.1002802
Colotta F, Peri G, Villa A, Mantovani A (1984) Rapid killing of actinomycin D-treated tumor cells by human mononuclear cells. I. Effectors belong to the monocyte-macrophage lineage. J Immunol 132:936–944
Cortés M, Sanchez-Moral L, de Barrios O et al (2017) Tumor-associated macrophages (TAMs) depend on ZEB1 for their cancer-promoting roles. EMBO J 36:3336–3355. https://doi.org/10.15252/embj.201797345
Curiel TJ, Coukos G, Zou L et al (2004) Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med 10:942–949. https://doi.org/10.1038/nm1093
Da Silva CG, Camps MGM, Li TMWY et al (2019) Co-delivery of immunomodulators in biodegradable nanoparticles improves therapeutic efficacy of cancer vaccines. Biomaterials 220:119417. https://doi.org/10.1016/j.biomaterials.2019.119417
Davies LC, Jenkins SJ, Allen JE, Taylor PR (2013) Tissue-resident macrophages. Nat Immunol 14:986–995. https://doi.org/10.1038/ni.2705
De Palma M, Lewis CE (2013) Macrophage regulation of tumor responses to anticancer therapies. Cancer Cell 23:277–286. https://doi.org/10.1016/j.ccr.2013.02.013
De Palma M, Naldini L (2011) Angiopoietin-2 TIEs up macrophages in tumor angiogenesis. Clin Cancer Res 17:5226–5232. https://doi.org/10.1158/1078-0432.CCR-10-0171
De Palma M, Venneri MA, Galli R et al (2005) Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 8:211–226. https://doi.org/10.1016/j.ccr.2005.08.002
DeNardo DG, Ruffell B (2019) Macrophages as regulators of tumour immunity and immunotherapy. Nat Rev Immunol 19:369–382. https://doi.org/10.1038/s41577-019-0127-6
DeNardo DG, Brennan DJ, Rexhepaj E et al (2011) Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy. Cancer Discov 1:54–67. https://doi.org/10.1158/2159-8274.CD-10-0028
Denisenko TV, Budkevich IN, Zhivotovsky B (2018) Cell death-based treatment of lung adenocarcinoma. Cell Death Dis 9:117. https://doi.org/10.1038/s41419-017-0063-y
Di Caro G, Cortese N, Castino GF et al (2016) Dual prognostic significance of tumour-associated macrophages in human pancreatic adenocarcinoma treated or untreated with chemotherapy. Gut 65:1710–1720. https://doi.org/10.1136/gutjnl-2015-309193
Edin S, Wikberg ML, Dahlin AM et al (2012) The distribution of macrophages with a M1 or M2 phenotype in relation to prognosis and the molecular characteristics of colorectal cancer. PLoS One 7:e47045. https://doi.org/10.1371/journal.pone.0047045
Epelman S, Lavine KJ, Randolph GJ (2014) Origin and functions of tissue macrophages. Immunity 41:21–35. https://doi.org/10.1016/j.immuni.2014.06.013
Erler JT, Bennewith KL, Cox TR et al (2009) Hypoxia-induced lysyl oxidase is a critical mediator of bone marrow cell recruitment to form the premetastatic niche. Cancer Cell 15:35–44. https://doi.org/10.1016/j.ccr.2008.11.012
Erreni M, Mantovani A, Allavena P (2011) Tumor-associated macrophages (TAM) and inflammation in colorectal cancer. Cancer Microenviron 4:141–154. https://doi.org/10.1007/s12307-010-0052-5
Fan Q-M, Jing Y-Y, Yu G-F et al (2014) Tumor-associated macrophages promote cancer stem cell-like properties via transforming growth factor-beta1-induced epithelial-mesenchymal transition in hepatocellular carcinoma. Cancer Lett 352:160–168. https://doi.org/10.1016/j.canlet.2014.05.008
Feng M, Chen JY, Weissman-Tsukamoto R et al (2015) Macrophages eat cancer cells using their own calreticulin as a guide: roles of TLR and Btk. PNAS 112:2145–2150. https://doi.org/10.1073/pnas.1424907112
Feng M, Marjon KD, Zhu F et al (2018) Programmed cell removal by calreticulin in tissue homeostasis and cancer. Nat Commun 9:3194. https://doi.org/10.1038/s41467-018-05211-7
Feng M, Jiang W, Kim BYS et al (2019) Phagocytosis checkpoints as new targets for cancer immunotherapy. Nat Rev Cancer 19:568–586. https://doi.org/10.1038/s41568-019-0183-z
Franklin RA, Liao W, Sarkar A et al (2014) The cellular and molecular origin of tumor-associated macrophages. Science 344:921–925. https://doi.org/10.1126/science.1252510
Gao L, Zhang W, Zhong W-Q et al (2018) Tumor associated macrophages induce epithelial to mesenchymal transition via the EGFR/ERK1/2 pathway in head and neck squamous cell carcinoma. Oncol Rep 40:2558–2572. https://doi.org/10.3892/or.2018.6657
Germano G, Frapolli R, Belgiovine C et al (2013) Role of macrophage targeting in the antitumor activity of trabectedin. Cancer Cell 23:249–262. https://doi.org/10.1016/j.ccr.2013.01.008
Gomez-Roca CA, Italiano A, Le Tourneau C et al (2019) Phase I study of emactuzumab single agent or in combination with paclitaxel in patients with advanced/metastatic solid tumors reveals depletion of immunosuppressive M2-like macrophages. Ann Oncol 30:1381–1392. https://doi.org/10.1093/annonc/mdz163
Gordon S (2003) Alternative activation of macrophages. Nat Rev Immunol 3:23–35. https://doi.org/10.1038/nri978
Gordon SR, Maute RL, Dulken BW et al (2017) PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature 545:495–499. https://doi.org/10.1038/nature22396
Goswami S, Sahai E, Wyckoff JB et al (2005) Macrophages promote the invasion of breast carcinoma cells via a colony-stimulating factor-1/epidermal growth factor paracrine loop. Cancer Res 65:5278–5283. https://doi.org/10.1158/0008-5472.CAN-04-1853
Goswami KK, Ghosh T, Ghosh S et al (2017) Tumor promoting role of anti-tumor macrophages in tumor microenvironment. Cell Immunol 316:1–10. https://doi.org/10.1016/j.cellimm.2017.04.005
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140:883–899. https://doi.org/10.1016/j.cell.2010.01.025
Gu S, Ni T, Wang J et al (2018) CD47 blockade inhibits tumor progression through promoting phagocytosis of tumor cells by M2 polarized macrophages in endometrial cancer. J Immunol Res 2018:6156757. https://doi.org/10.1155/2018/6156757
Guex N, Crespo I, Bron S et al (2015) Angiogenic activity of breast cancer patients’ monocytes reverted by combined use of systems modeling and experimental approaches. PLoS Comput Biol 11:e1004050. https://doi.org/10.1371/journal.pcbi.1004050
Haabeth OAW, Blake TR, McKinlay CJ et al (2019) Local delivery of Ox40l, Cd80, and Cd86 mRNA kindles global anticancer immunity. Cancer Res 79:1624–1634. https://doi.org/10.1158/0008-5472.CAN-18-2867
Hamada I, Kato M, Yamasaki T et al (2002) Clinical effects of tumor-associated macrophages and dendritic cells on renal cell carcinoma. Anticancer Res 22:4281–4284
Hanahan D, Coussens LM (2012) Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21:309–322. https://doi.org/10.1016/j.ccr.2012.02.022
Hapach LA, Mosier JA, Wang W, Reinhart-King CA (2019) Engineered models to parse apart the metastatic cascade. NPJ Precis Oncol 3:1–8. https://doi.org/10.1038/s41698-019-0092-3
Hasham MG, Baxan N, Stuckey DJ et al (2017) Systemic autoimmunity induced by the TLR7/8 agonist Resiquimod causes myocarditis and dilated cardiomyopathy in a new mouse model of autoimmune heart disease. Dis Model Mech 10:259–270. https://doi.org/10.1242/dmm.027409
Hedlund E-M, Hosaka K, Zhong Z et al (2009) Malignant cell-derived PlGF promotes normalization and remodeling of the tumor vasculature. PNAS 106:17505–17510. https://doi.org/10.1073/pnas.0908026106
Honkanen TJ, Tikkanen A, Karihtala P et al (2019) Prognostic and predictive role of tumour-associated macrophages in HER2 positive breast cancer. Sci Rep 9:10961. https://doi.org/10.1038/s41598-019-47375-2
Huang F, Wang D, Yao Y, Wang M (2017) PDGF signaling in cancer progression. Int J Clin Exp Med 10:9918–9929. http://www.ijcem.com/files/ijcem0055300.pdf
Huang L, Xu H, Peng G (2018) TLR-mediated metabolic reprogramming in the tumor microenvironment: potential novel strategies for cancer immunotherapy. Cell Mol Immunol 15:428–437. https://doi.org/10.1038/cmi.2018.4
Huang X, Zhang X, Lu M (2021) Recent trends in the development of toll-like receptor 7/8-targeting therapeutics. Expert Opin Drug Discov:1–12. https://doi.org/10.1080/17460441.2021.1898369
Hughes R, Qian B-Z, Rowan C et al (2015) Perivascular M2 macrophages stimulate tumor relapse after chemotherapy. Cancer Res 75:3479–3491. https://doi.org/10.1158/0008-5472.CAN-14-3587
Ibberson M, Bron S, Guex N et al (2013) TIE-2 and VEGFR kinase activities drive immunosuppressive function of TIE-2–expressing monocytes in human breast tumors. Clin Cancer Res 19:3439–3449. https://doi.org/10.1158/1078-0432.CCR-12-3181
Ireland LV, Mielgo A (2018) Macrophages and fibroblasts, key players in cancer Chemoresistance. Front Cell Dev Biol 6:131. https://doi.org/10.3389/fcell.2018.00131
Ito M, Minamiya Y, Kawai H et al (2006) Tumor-derived TGFbeta-1 induces dendritic cell apoptosis in the sentinel lymph node. J Immunol 176:5637–5643. https://doi.org/10.4049/jimmunol.176.9.5637
Jensen TO, Schmidt H, Møller HJ et al (2009) Macrophage markers in serum and tumor have prognostic impact in American joint committee on cancer stage I/II melanoma. J Clin Oncol 27:3330–3337. https://doi.org/10.1200/JCO.2008.19.9919
Jeon S-H, Chae B-C, Kim H-A et al (2007) Mechanisms underlying TGF-beta1-induced expression of VEGF and Flk-1 in mouse macrophages and their implications for angiogenesis. J Leukoc Biol 81:557–566. https://doi.org/10.1189/jlb.0806517
Jones CV, Ricardo SD (2013) Macrophages and CSF-1. Organogenesis 9:249–260. https://doi.org/10.4161/org.25676
Jones JD, Sinder BP, Paige D et al (2019) Trabectedin reduces skeletal prostate cancer tumor size in association with effects on M2 macrophages and Efferocytosis. Neoplasia 21:172–184. https://doi.org/10.1016/j.neo.2018.11.003
Joyce JA, Pollard JW (2009) Microenvironmental regulation of metastasis. Nat Rev Cancer 9:239–252. https://doi.org/10.1038/nrc2618
Kalbasi A, Ribas A (2020) Tumour-intrinsic resistance to immune checkpoint blockade. Nat Rev Immunol 20:25–39. https://doi.org/10.1038/s41577-019-0218-4
Kaplan RN, Riba RD, Zacharoulis S et al (2005) VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438:820–827. https://doi.org/10.1038/nature04186
Kaplan RN, Psaila B, Lyden D (2006) Bone marrow cells in the “pre-metastatic niche”: within bone and beyond. Cancer Metastasis Rev 25:521–529. https://doi.org/10.1007/s10555-006-9036-9
Karin M, Greten FR (2005) NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol 5:749–759. https://doi.org/10.1038/nri1703
Katsuya Y, Horinouchi H, Asao T et al (2016) Expression of programmed death 1 (PD-1) and its ligand (PD-L1) in thymic epithelial tumors: impact on treatment efficacy and alteration in expression after chemotherapy. Lung Cancer 99:4–10. https://doi.org/10.1016/j.lungcan.2016.05.007
Keshavarz-Fathi M, Rezaei N (2021) Cancer Immunoprevention: current status and future directions. Arch Immunol Ther Exp 69:3. https://doi.org/10.1007/s00005-021-00604-x
Klug F, Prakash H, Huber PE et al (2013) Low-dose irradiation programs macrophage differentiation to an iNOS+/M1 phenotype that orchestrates effective T cell immunotherapy. Cancer Cell 24:589–602. https://doi.org/10.1016/j.ccr.2013.09.014
Koh YW, Park C, Yoon DH et al (2014) CSF-1R expression in tumor-associated macrophages is associated with worse prognosis in classical Hodgkin lymphoma. Am J Clin Pathol 141:573–583. https://doi.org/10.1309/AJCPR92TDDFARISU
Kong L, Zhou Y, Bu H et al (2016) Deletion of interleukin-6 in monocytes/macrophages suppresses the initiation of hepatocellular carcinoma in mice. J Exp Clin Cancer Res 35:131. https://doi.org/10.1186/s13046-016-0412-1
Krenkel O, Tacke F (2017) Liver macrophages in tissue homeostasis and disease. Nat Rev Immunol 17:306–321. https://doi.org/10.1038/nri.2017.11
Kroemer G, Galluzzi L, Kepp O, Zitvogel L (2013) Immunogenic cell death in cancer therapy. Annu Rev Immunol 31:51–72. https://doi.org/10.1146/annurev-immunol-032712-100008
Kryczek I, Zou L, Rodriguez P et al (2006) B7-H4 expression identifies a novel suppressive macrophage population in human ovarian carcinoma. J Exp Med 203:871–881. https://doi.org/10.1084/jem.20050930
Kuklinski LF, Yan S, Li Z et al (2018) VISTA expression on tumor-infiltrating inflammatory cells in primary cutaneous melanoma correlates with poor disease-specific survival. Cancer Immunol Immunother 67:1113–1121. https://doi.org/10.1007/s00262-018-2169-1
Laoui D, Van Overmeire E, De Baetselier P et al (2014) Functional relationship between tumor-associated macrophages and macrophage Colony-stimulating factor as contributors to cancer progression. Front Immunol 5:489. https://doi.org/10.3389/fimmu.2014.00489
Larionova I, Cherdyntseva N, Liu T et al (2019) Interaction of tumor-associated macrophages and cancer chemotherapy. Onco Targets Ther 8:1596004. https://doi.org/10.1080/2162402X.2019.1596004
Leblond MM, Pérès EA, Helaine C et al (2017) M2 macrophages are more resistant than M1 macrophages following radiation therapy in the context of glioblastoma. Oncotarget 8:72597–72612. https://doi.org/10.18632/oncotarget.19994
Leek RD, Lewis CE, Whitehouse R et al (1996) Association of macrophage infiltration with angiogenesis and prognosis in invasive breast carcinoma. Cancer Res 56:4625–4629
Li S, Xu F, Zhang J et al (2017) Tumor-associated macrophages remodeling EMT and predicting survival in colorectal carcinoma. Onco Targets Ther 7. https://doi.org/10.1080/2162402X.2017.1380765
Lin Y, Xu J, Lan H (2019) Tumor-associated macrophages in tumor metastasis: biological roles and clinical therapeutic applications. J Hematol Oncol 12:76. https://doi.org/10.1186/s13045-019-0760-3
Lissbrant IF, Stattin P, Wikstrom P et al (2000) Tumor associated macrophages in human prostate cancer: relation to clinicopathological variables and survival. Int J Oncol 17:445–451. https://doi.org/10.3892/ijo.17.3.445
Liu Y, Cao X (2015) The origin and function of tumor-associated macrophages. Cell Mol Immunol 12:1–4. https://doi.org/10.1038/cmi.2014.83
Liu KX, Joshi S (2020) “Re-educating” tumor associated macrophages as a novel immunotherapy strategy for neuroblastoma. Front Immunol 11. https://doi.org/10.3389/fimmu.2020.01947
Liu C-Y, Xu J-Y, Shi X-Y et al (2013) M2-polarized tumor-associated macrophages promoted epithelial-mesenchymal transition in pancreatic cancer cells, partially through TLR4/IL-10 signaling pathway. Lab Investig 93:844–854. https://doi.org/10.1038/labinvest.2013.69
Lopez-Castejón G, Baroja-Mazo A, Pelegrín P (2011) Novel macrophage polarization model: from gene expression to identification of new anti-inflammatory molecules. Cell Mol Life Sci 68:3095–3107. https://doi.org/10.1007/s00018-010-0609-y
Loyher P-L, Hamon P, Laviron M et al (2018) Macrophages of distinct origins contribute to tumor development in the lung. J Exp Med 215:2536–2553. https://doi.org/10.1084/jem.20180534
Lu X, Mu E, Wei Y et al (2011) VCAM-1 promotes osteolytic expansion of indolent bone micrometastasis of breast cancer by engaging α4β1-positive osteoclast progenitors. Cancer Cell 20:701–714. https://doi.org/10.1016/j.ccr.2011.11.002
Lv J, Chen F-K, Liu C et al (2020) Zoledronic acid inhibits thyroid cancer stemness and metastasis by repressing M2-like tumor-associated macrophages induced Wnt/β-catenin pathway. Life Sci 256:117925. https://doi.org/10.1016/j.lfs.2020.117925
Ma J, Liu L, Che G et al (2010) The M1 form of tumor-associated macrophages in non-small cell lung cancer is positively associated with survival time. BMC Cancer 10:112. https://doi.org/10.1186/1471-2407-10-112
Macciò A, Gramignano G, Cherchi MC et al (2020) Role of M1-polarized tumor-associated macrophages in the prognosis of advanced ovarian cancer patients. Sci Rep 10:6096. https://doi.org/10.1038/s41598-020-63276-1
Maeda A, Digifico E, Andon FT et al (2019) Poly(I:C) stimulation is superior than Imiquimod to induce the antitumoral functional profile of tumor-conditioned macrophages. Eur J Immunol 49:801–811. https://doi.org/10.1002/eji.201847888
Mantovani A, Allavena P (2015) The interaction of anticancer therapies with tumor-associated macrophages. J Exp Med 212:435–445. https://doi.org/10.1084/jem.20150295
Mantovani A, Sica A (2010) Macrophages, innate immunity and cancer: balance, tolerance, and diversity. Curr Opin Immunol 22:231–237. https://doi.org/10.1016/j.coi.2010.01.009
Mantovani A, Sozzani S, Locati M et al (2002) Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 23:549–555. https://doi.org/10.1016/s1471-4906(02)02302-5
Mantovani A, Sica A, Sozzani S et al (2004) The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol 25:677–686. https://doi.org/10.1016/j.it.2004.09.015
Mantovani A, Marchesi F, Malesci A et al (2017) Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol 14:399–416. https://doi.org/10.1038/nrclinonc.2016.217
Martinez FO, Gordon S, Locati M, Mantovani A (2006) Transcriptional profiling of the human monocyte-to-macrophage differentiation and polarization: new molecules and patterns of gene expression. J Immunol 177:7303–7311. https://doi.org/10.4049/jimmunol.177.10.7303
Mass E, Ballesteros I, Farlik M et al (2016) Specification of tissue-resident macrophages during organogenesis. Science 353. https://doi.org/10.1126/science.aaf4238
Matsubara T, Kanto T, Kuroda S et al (2013) TIE2-expressing monocytes as a diagnostic marker for hepatocellular carcinoma correlates with angiogenesis. Hepatology 57:1416–1425. https://doi.org/10.1002/hep.25965
Mazzieri R, Pucci F, Moi D et al (2011) Targeting the ANG2/TIE2 axis inhibits tumor growth and metastasis by impairing angiogenesis and disabling rebounds of proangiogenic myeloid cells. Cancer Cell 19:512–526. https://doi.org/10.1016/j.ccr.2011.02.005
McKinlay CJ, Vargas JR, Blake TR et al (2017) Charge-altering releasable transporters (CARTs) for the delivery and release of mRNA in living animals. Proc Natl Acad Sci U S A 114:E448–E456. https://doi.org/10.1073/pnas.1614193114
Mills CD, Kincaid K, Alt JM et al (2000) M-1/M-2 macrophages and the Th1/Th2 paradigm. J Immunol 164:6166–6173. https://doi.org/10.4049/jimmunol.164.12.6166
Mosser DM, Edwards JP (2008) Exploring the full spectrum of macrophage activation. Nat Rev Immunol 8:958–969. https://doi.org/10.1038/nri2448
Mullins SR, Vasilakos JP, Deschler K et al (2019) Intratumoral immunotherapy with TLR7/8 agonist MEDI9197 modulates the tumor microenvironment leading to enhanced activity when combined with other immunotherapies. J Immunother Cancer 7:244. https://doi.org/10.1186/s40425-019-0724-8
Murray PJ (2017) Macrophage polarization. Annu Rev Physiol 79:541–566. https://doi.org/10.1146/annurev-physiol-022516-034339
Murray PJ, Allen JE, Biswas SK et al (2014) Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41:14–20. https://doi.org/10.1016/j.immuni.2014.06.008
Nadella V, Singh S, Jain A et al (2018) Low dose radiation primed iNOS + M1macrophages modulate angiogenic programming of tumor derived endothelium. Mol Carcinog 57:1664–1671. https://doi.org/10.1002/mc.22879
Nielsen SR, Schmid MC (2017) Macrophages as key drivers of cancer progression and metastasis. Mediat Inflamm 2017:e9624760. https://doi.org/10.1155/2017/9624760
Nishie A, Ono M, Shono T et al (1999) Macrophage infiltration and heme oxygenase-1 expression correlate with angiogenesis in human gliomas. Clin Cancer Res 5:1107–1113
Noman MZ, Van Moer K, Marani V et al (2018) CD47 is a direct target of SNAI1 and ZEB1 and its blockade activates the phagocytosis of breast cancer cells undergoing EMT. Onco Targets Ther 7:e1345415. https://doi.org/10.1080/2162402X.2017.1345415
Noy R, Pollard JW (2014) Tumor-associated macrophages: from mechanisms to therapy. Immunity 41:49–61. https://doi.org/10.1016/j.immuni.2014.06.010
Nywening TM, Belt BA, Cullinan DR et al (2018) Targeting both tumour-associated CXCR2+ neutrophils and CCR2+ macrophages disrupts myeloid recruitment and improves chemotherapeutic responses in pancreatic ductal adenocarcinoma. Gut 67:1112–1123. https://doi.org/10.1136/gutjnl-2017-313738
O’Brien J, Hayder H, Zayed Y, Peng C (2018) Overview of MicroRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol 9. https://doi.org/10.3389/fendo.2018.00402
Pallasch CP, Leskov I, Braun CJ et al (2014) Sensitizing protective tumor microenvironments to antibody-mediated therapy. Cell 156:590–602. https://doi.org/10.1016/j.cell.2013.12.041
Pantano F, Berti P, Guida FM et al (2013) The role of macrophages polarization in predicting prognosis of radically resected gastric cancer patients. J Cell Mol Med 17:1415–1421. https://doi.org/10.1111/jcmm.12109
Patwardhan PP, Surriga O, Beckman MJ et al (2014) Sustained inhibition of receptor tyrosine kinases and macrophage depletion by PLX3397 and rapamycin as a potential new approach for the treatment of MPNSTs. Clin Cancer Res 20:3146–3158. https://doi.org/10.1158/1078-0432.CCR-13-2576
Perry CJ, Muñoz-Rojas AR, Meeth KM et al (2018) Myeloid-targeted immunotherapies act in synergy to induce inflammation and antitumor immunity. J Exp Med 215:877–893. https://doi.org/10.1084/jem.20171435
Pinto AT, Pinto ML, Cardoso AP et al (2016) Ionizing radiation modulates human macrophages towards a pro-inflammatory phenotype preserving their pro-invasive and pro-angiogenic capacities. Sci Rep 6:18765. https://doi.org/10.1038/srep18765
Pollard JW (2004) Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer 4:71–78. https://doi.org/10.1038/nrc1256
Prenen H, Mazzone M (2019) Tumor-associated macrophages: a short compendium. Cell Mol Life Sci 76:1447–1458. https://doi.org/10.1007/s00018-018-2997-3
Qian B-Z, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141:39–51. https://doi.org/10.1016/j.cell.2010.03.014
Qian B, Deng Y, Im JH et al (2009) A distinct macrophage population mediates metastatic breast cancer cell extravasation, establishment and growth. PLoS One 4:e6562. https://doi.org/10.1371/journal.pone.0006562
Qin W, Hu L, Zhang X et al (2019) The diverse function of PD-1/PD-L pathway beyond cancer. Front Immunol 10:2298. https://doi.org/10.3389/fimmu.2019.02298
Quaranta V, Schmid MC (2019) Macrophage-mediated subversion of anti-tumour immunity. Cell 8. https://doi.org/10.3390/cells8070747
Raggi C, Mousa HS, Correnti M et al (2016) Cancer stem cells and tumor-associated macrophages: a roadmap for multitargeting strategies. Oncogene 35:671–682. https://doi.org/10.1038/onc.2015.132
Redente EF, Higgins DM, Dwyer-Nield LD et al (2010) Differential polarization of alveolar macrophages and bone marrow-derived monocytes following chemically and pathogen-induced chronic lung inflammation. J Leukoc Biol 88:159–168. https://doi.org/10.1189/jlb.0609378
Riabov V, Gudima A, Wang N et al (2014) Role of tumor associated macrophages in tumor angiogenesis and lymphangiogenesis. Front Physiol 5. https://doi.org/10.3389/fphys.2014.00075
Ribas A, Wolchok JD (2018) Cancer immunotherapy using checkpoint blockade. Science 359:1350–1355. https://doi.org/10.1126/science.aar4060
Roelofs AJ, Thompson K, Gordon S, Rogers MJ (2006) Molecular mechanisms of action of bisphosphonates: current status. Clin Cancer Res 12:6222s–6230s. https://doi.org/10.1158/1078-0432.CCR-06-0843
Rolny C, Mazzone M, Tugues S et al (2011) HRG inhibits tumor growth and metastasis by inducing macrophage polarization and vessel normalization through downregulation of PlGF. Cancer Cell 19:31–44. https://doi.org/10.1016/j.ccr.2010.11.009
Ruffell B, Coussens LM (2015) Macrophages and therapeutic resistance in cancer. Cancer Cell 27:462–472. https://doi.org/10.1016/j.ccell.2015.02.015
Saha N, Robev D, Himanen JP, Nikolov DB (2019) ADAM proteases: emerging role and targeting of the non-catalytic domains. Cancer Lett 467:50–57. https://doi.org/10.1016/j.canlet.2019.10.003
Sanchez L, Lucia B, Entenberg D et al (2019) The emerging roles of macrophages in cancer metastasis and response to chemotherapy. J Leukoc Biol 106. https://doi.org/10.1002/jlb.mr0218-056rr
Sangaletti S, Di Carlo E, Gariboldi S et al (2008) Macrophage-derived SPARC bridges tumor cell-extracellular matrix interactions toward metastasis. Cancer Res 68:9050–9059. https://doi.org/10.1158/0008-5472.CAN-08-1327
Sceneay J, Smyth MJ, Möller A (2013) The pre-metastatic niche: finding common ground. Cancer Metastasis Rev 32:449–464. https://doi.org/10.1007/s10555-013-9420-1
Schmall A, Al-Tamari HM, Herold S et al (2015) Macrophage and cancer cell cross-talk via CCR2 and CX3CR1 is a fundamental mechanism driving lung cancer. Am J Respir Crit Care Med 191:437–447. https://doi.org/10.1164/rccm.201406-1137OC
Schumacher N, Rose-John S, Schmidt-Arras D (2020) ADAM-mediated Signalling pathways in gastrointestinal cancer formation. Int J Mol Sci 21. https://doi.org/10.3390/ijms21145133
Shapouri-Moghaddam A, Mohammadian S, Vazini H et al (2018) Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol 233:6425–6440. https://doi.org/10.1002/jcp.26429
Shojaei F, Wu X, Malik AK et al (2007) Tumor refractoriness to anti-VEGF treatment is mediated by CD11b+Gr1+ myeloid cells. Nat Biotechnol 25:911–920. https://doi.org/10.1038/nbt1323
Sica A, Mantovani A (2012) Macrophage plasticity and polarization: in vivo veritas. J Clin Invest 122:787–795. https://doi.org/10.1172/JCI59643
Sica A, Schioppa T, Mantovani A, Allavena P (2006) Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur J Cancer 42:717–727. https://doi.org/10.1016/j.ejca.2006.01.003
Sica A, Erreni M, Allavena P, Porta C (2015) Macrophage polarization in pathology. Cell Mol Life Sci 72:4111–4126. https://doi.org/10.1007/s00018-015-1995-y
Singh N, Baby D, Rajguru JP et al (2019) Inflammation and cancer. Ann Afr Med 18:121–126. https://doi.org/10.4103/aam.aam_56_18
Solinas G, Germano G, Mantovani A, Allavena P (2009) Tumor-associated macrophages (TAM) as major players of the cancer-related inflammation. J Leukoc Biol 86:1065–1073. https://doi.org/10.1189/jlb.0609385
Song W, Mazzieri R, Yang T, Gobe GC (2017) Translational significance for tumor metastasis of tumor-associated macrophages and epithelial-mesenchymal transition. Front Immunol 8:1106. https://doi.org/10.3389/fimmu.2017.01106
Song Y, Tang C, Yin C (2018) Combination antitumor immunotherapy with VEGF and PIGF siRNA via systemic delivery of multi-functionalized nanoparticles to tumor-associated macrophages and breast cancer cells. Biomaterials 185:117–132. https://doi.org/10.1016/j.biomaterials.2018.09.017
Su S, Liu Q, Chen J et al (2014) A positive feedback loop between mesenchymal-like cancer cells and macrophages is essential to breast cancer metastasis. Cancer Cell 25:605–620. https://doi.org/10.1016/j.ccr.2014.03.021
Sumitomo R, Hirai T, Fujita M et al (2019) M2 tumor-associated macrophages promote tumor progression in non-small-cell lung cancer. Exp Ther Med 18:4490–4498. https://doi.org/10.3892/etm.2019.8068
Takanami I, Takeuchi K, Kodaira S (1999) Tumor-associated macrophage infiltration in pulmonary adenocarcinoma: association with angiogenesis and poor prognosis. Oncology 57:138–142. https://doi.org/10.1159/000012021
Thauvin C, Widmer J, Mottas I et al (2019) Development of resiquimod-loaded modified PLA-based nanoparticles for cancer immunotherapy: a kinetic study. Eur J Pharm Biopharm 139:253–261. https://doi.org/10.1016/j.ejpb.2019.04.007
Thomas DA, Massagué J (2005) TGF-beta directly targets cytotoxic T cell functions during tumor evasion of immune surveillance. Cancer Cell 8:369–380. https://doi.org/10.1016/j.ccr.2005.10.012
Torisu H, Ono M, Kiryu H et al (2000) Macrophage infiltration correlates with tumor stage and angiogenesis in human malignant melanoma: possible involvement of TNFalpha and IL-1alpha. Int J Cancer 85:182–188
Truffi M, Sorrentino L, Corsi F (2020) Fibroblasts in the tumor microenvironment. Adv Exp Med Biol 1234:15–29. https://doi.org/10.1007/978-3-030-37184-5_2
Tsutsui S, Yasuda K, Suzuki K et al (2005) Macrophage infiltration and its prognostic implications in breast cancer: the relationship with VEGF expression and microvessel density. Oncol Rep 14:425–431
Tymoszuk P, Evens H, Marzola V et al (2014) In situ proliferation contributes to accumulation of tumor-associated macrophages in spontaneous mammary tumors. Eur J Immunol 44:2247–2262. https://doi.org/10.1002/eji.201344304
Uchida J, Hamaguchi Y, Oliver JA et al (2004) The innate mononuclear phagocyte network depletes B lymphocytes through fc receptor-dependent mechanisms during anti-CD20 antibody immunotherapy. J Exp Med 199:1659–1669. https://doi.org/10.1084/jem.20040119
Varol C, Mildner A, Jung S (2015) Macrophages: development and tissue specialization. Annu Rev Immunol 33:643–675. https://doi.org/10.1146/annurev-immunol-032414-112220
Venneri MA, De Palma M, Ponzoni M et al (2007) Identification of proangiogenic TIE2-expressing monocytes (TEMs) in human peripheral blood and cancer. Blood 109:5276–5285. https://doi.org/10.1182/blood-2006-10-053504
Wang R, Zhang J, Chen S et al (2011) Tumor-associated macrophages provide a suitable microenvironment for non-small lung cancer invasion and progression. Lung Cancer 74:188–196. https://doi.org/10.1016/j.lungcan.2011.04.009
Wang J, Li D, Cang H, Guo B (2019) Crosstalk between cancer and immune cells: role of tumor-associated macrophages in the tumor microenvironment. Cancer Med 8:4709–4721. https://doi.org/10.1002/cam4.2327
Weagel EG, Smith CD, Liu P, et al (2015) Macrophage polarization and its role in cancer. https://doi.org/10.4172/2155-9899.1000338
Whiteside T (2008) The tumor microenvironment and its role in promoting tumor growth. Oncogene 27:5904–5912. https://doi.org/10.1038/onc.2008.271
Willingham SB, Volkmer J-P, Gentles AJ et al (2012) The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc Natl Acad Sci U S A 109:6662–6667. https://doi.org/10.1073/pnas.1121623109
Wu H, Xu J-B, He Y-L et al (2012) Tumor-associated macrophages promote angiogenesis and lymphangiogenesis of gastric cancer. J Surg Oncol 106:462–468. https://doi.org/10.1002/jso.23110
Wyckoff J, Wang W, Lin EY et al (2004) A paracrine loop between tumor cells and macrophages is required for tumor cell migration in mammary tumors. Cancer Res 64:7022–7029. https://doi.org/10.1158/0008-5472.CAN-04-1449
Wyckoff JB, Wang Y, Lin EY et al (2007) Direct visualization of macrophage-assisted tumor cell intravasation in mammary tumors. Cancer Res 67:2649–2656. https://doi.org/10.1158/0008-5472.CAN-06-1823
Xiang X, Wang J, Lu D, Xu X (2021) Targeting tumor-associated macrophages to synergize tumor immunotherapy. Signal Transduct Target Ther 6:1–12. https://doi.org/10.1038/s41392-021-00484-9
Xu J, Escamilla J, Mok S et al (2013) CSF1R signaling blockade stanches tumor-infiltrating myeloid cells and improves the efficacy of radiotherapy in prostate cancer. Cancer Res 73:2782–2794. https://doi.org/10.1158/0008-5472.CAN-12-3981
Yan Y, Zhang J, Li J-H et al (2016) High tumor-associated macrophages infiltration is associated with poor prognosis and may contribute to the phenomenon of epithelial–mesenchymal transition in gastric cancer. Onco Targets Ther 9:3975–3983. https://doi.org/10.2147/OTT.S103112
Yang L, Zhang Y (2017) Tumor-associated macrophages: from basic research to clinical application. J Hematol Oncol 10:58. https://doi.org/10.1186/s13045-017-0430-2
Yang L, Zheng L, Chng WJ, Ding JL (2019) Comprehensive analysis of ERK1/2 substrates for potential combination immunotherapies. Trends Pharmacol Sci 40:897–910. https://doi.org/10.1016/j.tips.2019.09.005
Yin M, Shen J, Yu S et al (2019) Tumor-associated macrophages (TAMs): a critical activator in ovarian cancer metastasis. Onco Targets Ther 12:8687–8699. https://doi.org/10.2147/OTT.S216355
Zhang W, Zhu X-D, Sun H-C et al (2010) Depletion of tumor-associated macrophages enhances the effect of sorafenib in metastatic liver cancer models by antimetastatic and antiangiogenic effects. Clin Cancer Res 16:3420–3430. https://doi.org/10.1158/1078-0432.CCR-09-2904
Zhang Q, Liu L, Gong C et al (2012) Prognostic significance of tumor-associated macrophages in solid tumor: a meta-analysis of the literature. PLoS One 7:e50946. https://doi.org/10.1371/journal.pone.0050946
Zhang M, He Y, Sun X et al (2014) A high M1/M2 ratio of tumor-associated macrophages is associated with extended survival in ovarian cancer patients. J Ovarian Res 7:19. https://doi.org/10.1186/1757-2215-7-19
Zhang X, Chen Y, Hao L et al (2016) Macrophages induce resistance to 5-fluorouracil chemotherapy in colorectal cancer through the release of putrescine. Cancer Lett 381:305–313. https://doi.org/10.1016/j.canlet.2016.08.004
Zhang JQ, Zeng S, Vitiello GA et al (2018) Macrophages and CD8+ T cells mediate the antitumor efficacy of combined CD40 ligation and Imatinib therapy in gastrointestinal stromal tumors. Cancer Immunol Res 6:434–447. https://doi.org/10.1158/2326-6066.CIR-17-0345
Zhang F, Parayath NN, Ene CI et al (2019) Genetic programming of macrophages to perform anti-tumor functions using targeted mRNA nanocarriers. Nat Commun 10:3974. https://doi.org/10.1038/s41467-019-11911-5
Zhao J, Zhang Z, Xue Y et al (2018) Anti-tumor macrophages activated by ferumoxytol combined or surface-functionalized with the TLR3 agonist poly (I : C) promote melanoma regression. Theranostics 8:6307–6321. https://doi.org/10.7150/thno.29746
Zheng P, Luo Q, Wang W et al (2018) Tumor-associated macrophages-derived exosomes promote the migration of gastric cancer cells by transfer of functional apolipoprotein E. Cell Death Dis 9:434. https://doi.org/10.1038/s41419-018-0465-5
Zhou D-Y, Qin J, Huang J et al (2017) Zoledronic acid inhibits infiltration of tumor-associated macrophages and angiogenesis following transcatheter arterial chemoembolization in rat hepatocellular carcinoma models. Oncol Lett 14:4078–4084. https://doi.org/10.3892/ol.2017.6717
Zhou J, Tang Z, Gao S et al (2020) Tumor-associated macrophages: recent insights and therapies. Front Oncol 10:188. https://doi.org/10.3389/fonc.2020.00188
Zhu X-D, Zhang J-B, Zhuang P-Y et al (2008) High expression of macrophage colony-stimulating factor in peritumoral liver tissue is associated with poor survival after curative resection of hepatocellular carcinoma. J Clin Oncol 26:2707–2716. https://doi.org/10.1200/JCO.2007.15.6521
Zhu Y, Knolhoff BL, Meyer MA et al (2014) CSF1/CSF1R blockade reprograms tumor-infiltrating macrophages and improves response to T-cell checkpoint immunotherapy in pancreatic cancer models. Cancer Res 74:5057–5069. https://doi.org/10.1158/0008-5472.CAN-13-3723
Zhu Y, Herndon JM, Sojka DK et al (2017) Tissue resident macrophages in pancreatic ductal adenocarcinoma originate from embryonic hematopoiesis and promote tumor progression. Immunity 47:323–338.e6. https://doi.org/10.1016/j.immuni.2017.07.014
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Szulc-Kielbik, I., Kielbik, M. (2022). Tumor-Associated Macrophages: Reasons to Be Cheerful, Reasons to Be Fearful. In: Klink, M., Szulc-Kielbik, I. (eds) Interaction of Immune and Cancer Cells. Experientia Supplementum, vol 113. Springer, Cham. https://doi.org/10.1007/978-3-030-91311-3_4
Download citation
DOI: https://doi.org/10.1007/978-3-030-91311-3_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-91310-6
Online ISBN: 978-3-030-91311-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)