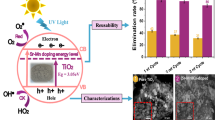
Abstract
Highly effective Z-scheme La–ZnO/GCN nanocomposite (LZG) were synthesized via hydrothermal and ultrasonication methods. The prepared samples were further analyzed through varies techniques like X-ray diffraction (XRD), high-resolution scanning electron microscopy (HRSEM), X-ray photoelectron spectroscopy (XPS) and UV–visible spectroscopy. XRD confirms the non-detection of secondary phase formation and decrementing pattern of crystallite size confirm La ions presence in host lattice. Presence of La–ZnO nanorods on nanosheets of GCN are well observed from the HRSEM analysis. Enhancement in pollutant degradation was accredited due to higher charge transfer property observed from EIS (Electrochemical impedance spectroscopy). First-order Langmuir–Hinshelwood relation reveals about the higher rate of reaction (0.01796 × 10–2 min−1), around 84% of TC pollutant degradation by 10-10LZG nanocomposite within the time span of 80 min. The current research supports a novel design of nanocomposite with an electron trapper for hindering charge recombination process and enhancing the degradation of pharmaceutical pollutants.
Graphical abstract











Similar content being viewed by others
Data availability
The datasets used or analyzed during the current study are included in this published article.
Code availability
Not applicable.
References
M.B. Gawande, A. Goswami, F.X. Felpin, T. Asefa, X. Huang, R. Silva, X. Zou, R. Zboril, R.S. Varma, Cu and Cu-based nanoparticles: synthesis and applications in catalysis. Chem. Rev. 116, 3722–3811 (2016). https://doi.org/10.1021/acs.chemrev.5b00482
N.R. Khalid, U. Mazia, M.B. Tahir, N.A. Niaz, M.A. Javid, Photocatalytic degradation of RhB from an aqueous solution using Ag3PO4/N-TiO2 heterostructure. J. Mol. Liq. 313, 113522 (2020). https://doi.org/10.1016/j.molliq.2020.113522
Y.M. Hunge, A.A. Yadav, S. Kang, H. Kim, Photocatalytic degradation of tetracycline antibiotics using hydrothermally synthesized two-dimensional molybdenum disulfide/titanium dioxide composites. J. Colloid Interface Sci. 606, 454–463 (2022). https://doi.org/10.1016/j.jcis.2021.07.151
F.E. Titchou, H. Zazou, H. Afanga, J. El Gaayda, R.A. Akbour, M. Hamdani, Removal of persistent organic pollutants (POPs) from water and wastewater by adsorption and electrocoagulation process. Groundw. Sustain. Dev. 13, 100575 (2021). https://doi.org/10.1016/j.gsd.2021.100575
I.M.F. Cardoso, R.M.F. Cardoso, J.C.G. Esteves de Silva, Advanced oxidation processes coupled with nanomaterials for water advanced oxidation processes coupled with nanomaterials for water treatment. Nanomaterials 11(8), 2045 (2021). https://doi.org/10.3390/nano11082045
M. Qiu, B. Hu, Z. Chen, H. Yang, L. Zhuang, X. Wang, Challenges of organic pollutant photocatalysis by biochar-based catalysts. Biochar. 3, 117–123 (2021). https://doi.org/10.1007/s42773-021-00098-y
R.R. Chandrapal, S. Bharathkumar, G. Bakiyaraj, V. Ganesh, J. Archana, M. Navaneethan, Hydrothermally synthesized strontium-modified ZnO hierarchical nanostructured photocatalyst for second-generation fluoroquinolone degradation. Appl. Nanosci. 12, 1869–1884 (2022). https://doi.org/10.1007/s13204-022-02414-9
S. Yu, H. Pang, S. Huang, H. Tang, S. Wang, M. Qiu, Z. Chen et al., Recent advances in metal-organic framework membranes for water treatment: A review. Sci. Total Environ. 800, 149662 (2021). https://doi.org/10.1016/j.scitotenv.2021.149662
L. Liang, F. Xi, W. Tan, X. Meng, B. Hu, X. Wang, Review of organic and inorganic pollutants removal by biochar and biochar-based composites. Biochar 3, 255–281 (2021). https://doi.org/10.1007/s42773-021-00101-6
F. Liu, Y. Lou, F. Xia, B. Hu, Immobilizing nZVI particles on MBenes to enhance the removal of U (VI) and Cr (VI) by adsorption-reduction synergistic effect. J. Chem. Eng. 454, 140318 (2023). https://doi.org/10.1016/j.cej.2022.140318
M.S. Chavali, M.P. Nikolova, Metal oxide nanoparticles and their applications in nanotechnology. SN Appl. Sci. 1, 607 (2019). https://doi.org/10.1007/s42452-019-0592-3
M. Tahir, S. Tasleem, B. Tahir, Recent development in band engineering of binary semiconductor materials for solar driven photocatalytic hydrogen production. Int. J. Hydrogen Energy. 45, 15985–16038 (2020). https://doi.org/10.1016/j.ijhydene.2020.04.071
C. Klingshirn, ZnO: material, physics and applications. ChemPhysChem 8, 782–803 (2007). https://doi.org/10.1002/cphc.200700002
M. Samadi, M. Zirak, A. Naseri, E. Khorashadizade, A.Z. Moshfegh, Recent progress on doped ZnO nanostructures for visible-light photocatalysis. Thin Solid Films 605, 2–19 (2016). https://doi.org/10.1016/j.tsf.2015.12.064
T. Jia, W. Wang, F. Long, Z. Fu, H. Wang, Q. Zhang, Fabrication, characterization and photocatalytic activity of La-doped ZnO nanowires. J. Alloys Compd. 484, 410–415 (2009). https://doi.org/10.1016/j.jallcom.2009.04.153
P. Pascariu, M. Homocianu, C. Cojocaru, P. Samoila, A. Airinei, M. Suchea, Preparation of La doped ZnO ceramic nanostructures by electrospinning–calcination method: Effect of La3+ doping on optical and photocatalytic properties. Appl. Surf. Sci. 476, 16–27 (2019). https://doi.org/10.1016/j.apsusc.2019.01.077
R. Bomila, S. Srinivasan, S. Gunasekaran, A. Manikandan, Enhanced photocatalytic degradation of methylene blue dye, opto-magnetic and antibacterial behaviour of pure and La-doped ZnO nanoparticles. J. Supercond. Nov. Magn. 31, 855–864 (2018). https://doi.org/10.1007/s10948-017-4261-8
N.S. Alharbi, B. Hu, T. Hayat, S.O. Rabah, A. Alsaedi, L. Zhuang, X. Wang, Efficient elimination of environmental pollutants through sorption-reduction and photocatalytic degradation using nanomaterials. Front. Chem. Sci. Eng. 14, 1124–1135 (2020). https://doi.org/10.1007/s11705-020-1923-z
J. Wen, J. Xie, X. Chen, X. Li, A review on g-C3N4-based photocatalysts. Appl. Surf. Sci. 391, 72–123 (2017). https://doi.org/10.1016/j.apsusc.2016.07.030
J. Fu, J. Yu, C. Jiang, B. Cheng, g-C3N4 -Based heterostructured photocatalysts. Adv. Energy Mater. 8(3), 1701503 (2018). https://doi.org/10.1002/aenm.201701503
M. Zhou, Z. Hou, X. Chen, The effects of hydrogenation on graphitic C3N4 nanosheets for enhanced photocatalytic activity. Part. Part. Syst. Charact. 35, 1700038 (2018). https://doi.org/10.1002/ppsc.201700038
W. Yu, D. Xu, T. Peng, Enhanced photocatalytic activity of g-C3N4 for selective CO2 reduction to CH3OH via facile coupling of ZnO: A direct Z- scheme mechanism. J. Mater. Chem. A 3, 19936–19947 (2015). https://doi.org/10.1039/C5TA05503B
G. Liu, M. Liao, Z. Zhang, H. Wang, D. Chen, Y. Feng, Enhanced photodegradation performance of Rhodamine B with g-C3N4 modified by carbon nanotubes. Sep. Purif. Technol. 244, 116618 (2020). https://doi.org/10.1016/j.seppur.2020.116618
P.V. Korake, R.S. Dhabbe, A.N. Kadam, Y.B. Gaikwad, K.M. Garadkar, Highly active lanthanum doped ZnO nanorods for photodegradation of metasystox. J. Photochem. Photobiol. B: Biol. 130, 11–19 (2014). https://doi.org/10.1016/j.jphotobiol.2013.10.012
M.U. Yousaf, E. Pervaiz, S. Minallah, M.J. Afzal, L. Honghong, M. Yang, Tin oxide quantum dots decorated graphitic carbon nitride for enhanced removal of organic components from water: Green process. Results Phys. 14, 102455 (2019). https://doi.org/10.1016/j.rinp.2019.102455
V.P. Dinesh, P. Biji, A. Ashok, S.K. Dhara, M. Kamruddin, A.K. Tyagi, B. Raj, Plasmon -mediated highly enhanced photocatalytic degradation of industrial textile effluent dyes using hybrid ZnO@Ag core-shell nanorods. RSC Adv. 103, 58930–58940 (2014). https://doi.org/10.1039/C4RA09405K
S.Y. Pung, W.P. Lee, A. Aziz, Kinetic study of organic dye degradation using ZnO particles with different morphologies as a photocatalyst. Int. J. Inorg. Chem. (2012). https://doi.org/10.1155/2012/608183
Y. Sheng, Y. Jiang, X. Lan, C. Wang, S. Li, X. Liu, H. Zhong, Mechanism and growth of flexible ZnO nanostructure arrays in a facile controlled way. J. Nanomater. (2011). https://doi.org/10.1155/2011/473629
R. Hao, S. Luo, F. Wang, X. Pan, J. Yao, J. Wu, H. Fang, W. Li, Enhancement of fluorescence and anti-tumor effect of ZnO QDs by La doping. Front. Chem. 10, 1042038 (2022). https://doi.org/10.3389/fchem.2022.1042038
M. Kumar, K. Negi, A. Umar, M.S. Chauhan, Photocatalytic and fluorescent chemical sensing applications of La-doped ZnO nanoparticles. Chem. Pap. 75, 1555–1566 (2021). https://doi.org/10.1007/s11696-020-01388-8
Y. Al-Hadeethi, A. Umar, K. Singh, A.A. Ibrahim, S. Al-Heniti, B.M. Raffah, Ytterbium-doped ZnO flowers based phenyl hydrazine chemical sensor. J. Nanosci. Nanotechnol. 19, 4199–4204 (2019). https://doi.org/10.1166/jnn.2019.16283
G. Nagaraju, S.A. Prashanth, M. Shastri, K.V. Yathish, C. Anupama, D. Rangappa, Electrochemical heavy metal detection, photocatalytic, photoluminescence, biodiesel production and antibacterial activities of Ag–ZnO nanomaterial. Mater. Res. Bull. 94, 54–63 (2017). https://doi.org/10.1016/j.materresbull.2017.05.043
L.V. Bettina, M. Döblinger, J. Sehnert, L. Seyfarth, J. Senker, O. Oeckler, W. Schnick, Unmasking melon by a complementary approach employing electron diffraction, solid-state NMR spectroscopy, and theoretical calculations—structural characterization of a carbon nitride polymer. Chem. Eur. J. 13, 4969–4980 (2007). https://doi.org/10.1002/chem.200601759
D.R. Paul, S. Gautam, P. Panchal, S.P. Nehra, P. Choudhary, A. Sharma, ZnO-modified g-C3N4: a potential photocatalyst for environmental application. ACS Omega 5, 3828–3838 (2020). https://doi.org/10.1021/acsomega.9b02688
A.K. Zak, M.E. Abrishami, W.H.A. Majid, R. Yousefi, S.M. Hosseini, Effects of annealing temperature on some structural and optical properties of ZnO nanoparticles prepared by a modified sol–gel combustion method. Ceram. Int. 37, 393–398 (2011). https://doi.org/10.1016/j.ceramint.2010.08.017
I. Khan, K. Saeed, I. Khan, Nanoparticles : Properties, applications and toxicities. Arab. J. Chem. 12, 908–931 (2019). https://doi.org/10.1016/j.arabjc.2017.05.011
S.J. Gilliland, J.A. Sans, J.F. Sánchez-Royo, G. Almonacid, B. Garcìa, A. Segura, G. Tobias, E. Canadell, Role of p-d and s-d interactions in the electronic structure and band gap of Zn1 − x MxO (M=Cr, Mn, Fe Co, Ni, and Cu): Photoelectron and optical spectroscopy and first-principles band structure calculations. Phys. Rev. B 86, 155203 (2012). https://doi.org/10.1103/PhysRevB.86.155203
M.K. Debanath, S. Karmakar, Study of blueshift of optical band gap in zinc oxide (ZnO) nanoparticles prepared by low-temperature wet chemical method. Mater. Lett. 111, 116–119 (2013). https://doi.org/10.1016/j.matlet.2013.08.069
Q. Li, X. Ma, H. Liu, Z. Chen, H. Chen, S. Chu, Self-organized growth of two-dimensional GaTe nanosheet on ZnO nanowires for heterojunctional water splitting applications. ACS Appl. Mater. Interfaces 22, 18836–18844 (2017). https://doi.org/10.1021/acsami.7b04199
H.D. Zhang, M. Yu, J.C. Zhang, C.H. Sheng, X. Yan, W.P. Han, Y.C. Liu, S. Chen, G.Z. Shen, Y.Z. Long, Fabrication and photoelectric properties of La-doped p-type ZnO nanofibers and crossed p–n homojunctions by electrospinning. Nanoscale 23, 10513–10518 (2015). https://doi.org/10.1039/c5nr02191j
W. Lu, D. Zhu, Synthesis and characterization of La- and Ce-codoped polycrystal ZnO prepared by hydrothermal method for 1,2-propanediol. Appl. Phys. A 125, 68 (2019). https://doi.org/10.1007/s00339-019-2380-8
C.K. Swamy, A. Hezam, A.M. Ramesh, D.H. Ramakrishnegowda, D.K. Purushothama, J. Krishnegowda, K.S. Rangappa, S. Shivanna, Microwave hydrothermal synthesis of copper induced ZnO/gC3N4 heterostructure with efficient photocatalytic degradation through S-scheme mechanism. J. Photochem. Photobiol. A Chem. 418, 113394 (2021). https://doi.org/10.1016/j.jphotochem.2021.113394
G. Xu, F. You, X. Li, La-doped ZnO / g-C3N4 heterojunction for efficient degradation of organic contamination under visible light irradiation. J. Inorg. Organomet. Polym. Mater. 31, 375–383 (2021). https://doi.org/10.1007/s10904-020-01779-0
S. Pareek, J.K. Quamara, Dielectric and optical properties of graphitic carbon nitride – titanium dioxide nanocomposite with enhanced charge seperation. J. Mater. Sci. 53, 604–612 (2018). https://doi.org/10.1007/s10853-017-1506-7
H. Qin, Y. Zuo, J. Jin, W. Wang, Y. Xu, L. Cui, H. Dang, ZnO nanorod arrays grown on g-C3N4 micro-sheets for enhanced visible light photocatalytic H2 evolution. RSC Adv. 9, 24483–24488 (2019). https://doi.org/10.1039/c9ra03426a
Z. Lan, G. Zhang, X. Wang, A facile synthesis of Br-modified g-C3N4 semiconductors for photoredox water splitting. Appl. Catal. B Environ. 192, 116–125 (2016). https://doi.org/10.1016/j.apcatb.2016.03.062
L. Wei, J. Tian, Q. Wang, Y. Liu, Y. Yu, C. Yang, A study on the mechanism and kinetic of nitrate reduction by the nZVI–g-C3N4/TiO2 composite under the simulated sunlight. J. Mater. Sci. Mater. Electron. 32, 15864–15881 (2021). https://doi.org/10.1007/s10854-021-06139-3
J.T. Schneider, D.S. Firak, R.R. Ribeiro, P.P. Zamora, Use of scavenger agents in heterogeneous photocatalysis: truths, half-truths, and misinterpretations. Phys. Chem. Chem. Phys. 22, 15723–15733 (2020). https://doi.org/10.1039/D0CP02411B
J. Qiu, M. Guo, Y. Feng, X. Wang, Electrochemical deposition of branched hierarchical ZnO nanowire arrays and its photoelectrochemical properties. Electrochim. Acta. 56, 5776–5782 (2011). https://doi.org/10.1016/j.electacta.2011.04.059
D.T. Dimitrov, M.M. Milanova, R.P. Kralshevska, Lanthanide oxide doped titania photocatalysts for degradation of organic pollutants under UV and visible light illumination. Bulg. Chem. Commun. 43(4), 489–501 (2011)
Q.T.H. Ta, G. Namgung, J.S. Noh, Facile synthesis of porous metal-doped ZnO/g-C3N4 composites for highly efficient photocatalysts. J. Photochem. Photobiol. A: Chem. 368, 110–119 (2019). https://doi.org/10.1016/j.jphotochem.2018.09.049
M. Rostami, Construction of La-doped TiO2@La-doped ZnO–B- doped reduced graphene oxide ternary nanocomposites for improved visible light photocatalytic activity. RSC Adv. 7, 43424–43431 (2017). https://doi.org/10.1039/C7RA06767D
M.A. Qamar, M. Javed, S. Shahid, M. Sher, Fabrication of g-C3N4/transition metal (Fe Co, Ni, Mn and Cr)-doped ZnO ternary composites: Excellent visible light active photocatalysts for the degradation of organic pollutants from wastewater. Mater. Res. Bull. 147, 111630 (2022). https://doi.org/10.1016/j.materresbull.2021.111630
M.A. Qamar, S. Shahid, M. Javed, S. Iqbal, M. Sher, A. Bahadur, M.M. Al-Anazy, A. Laref, D. Li, Designing of highly active g-C3N4/Ni–ZnO photocatalyst nanocomposite for the disinfection and degradation of the organic dye under sunlight radiations. Colloids Surf. A Physicochem. Eng. Asp. 614, 126176 (2021). https://doi.org/10.1016/j.colsurfa.2021.126176
C. Blanco, M. Granda, A. Patricia, N.G. Asenjo, R. Santamarı, R. Mene, Correct use of the Langmuir–Hinshelwood equation for proving the absence of a synergy effect in the photocatalytic degradation of phenol on a suspended mixture of titania and activated carbon. Carbon 55, 62–69 (2013). https://doi.org/10.1016/j.carbon.2012.12.010
F.A. Aisien, N.A. Amenaghawon, E.F. Ekpenisi, Photocatalytic decolourisation of industrial wastewater from a soft drink company. J. Eng. Appl. Sci. 9, 11–16 (2013)
Z. Ren, F. Chen, K. Wen, J. Lu, Enhanced photocatalytic activity for tetracyclines degradation with Ag modified g-C3N4 composite under visible light. J. Photochem. Photobiol. A Chem. 389, 112217 (2020). https://doi.org/10.1016/j.jphotochem.2019.112217
V. Jayaraman, D. Sarkar, R. Rajendran, B. Palanivel, C. Ayappan, M. Chellamuthu, A. Mani, Synergistic effect of band edge potentials on BiFeO3/V2O5 composite: enhanced photo catalytic activity. J. Environ. Manage. 247, 104–114 (2019). https://doi.org/10.1016/j.jenvman.2019.06.041
L. Lu, Z. Lv, Y. Si, M. Liu, S. Zhang, Recent progress on band and surface engineering of graphitic carbon nitride for artificial photosynthesis. Appl. Surf. Sci. 462, 693–712 (2018). https://doi.org/10.1016/j.apsusc.2018.08.131
S. Prabhu, M. Pudukudy, S. Harish, M. Navaneethan, S. Sohila, K. Murugesan, R. Ramesh, Facile construction of djembe-like ZnO and its composite with g-C3N4 as a visible-light-driven heterojunction photocatalyst for the degradation of organic dyes. Mater. Sci. Semicond. Process. 106, 104754 (2020). https://doi.org/10.1016/j.mssp.2019.104754
Acknowledgments
Authors thankfully acknowledge the SRM Institute of science and technology (SRM IST), Department of Physics and Nanotechnology for the constant support to carry out this research work. Authors also gratefully acknowledge the Nanotechnology Research Center (NRC) for characterization facility.
Funding
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Author information
Authors and Affiliations
Contributions
RRC: conceptualization; investigation; methodology; software; writing-original draft. JR: methodology; data curation; software; GB: conceptualization; supervision; validation; visualization; writing-review & editing. SB: conceptualization; methodology; data curation; software; validation. VG: methodology; validation; data curation. JA: validation; data curation. MN: data curation; validation; resources. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that there is no conflict of interest in this research manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chandrapal, R.R., Raveena, J., Bakiyaraj, G. et al. Enhancing the photocatalytic performance of g-C3N4 (GCN) via La–ZnO nanocomposite (Z-scheme mechanism) against toxic pharmaceutical pollutant. Journal of Materials Research 38, 3585–3601 (2023). https://doi.org/10.1557/s43578-023-01087-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/s43578-023-01087-6