Abstract
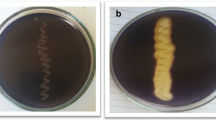
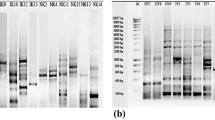
The present study was conducted to isolate α-amylase-producing thermophilic bacteria from DargeÇit hot springs in Turkey. The morphological, biochemical and physiological characterisation, as well as genetic analysis by 16S rRNA sequences indicated that the isolated strain AH1 was a member of Anoxybacillus genus. The strain was aerobe, Gram-positive and spore-forming rod, exhibiting optimum growth temperature and pH of 60°C and 7.0–7.5, respectively. Optimization of growth medium and enzyme assay conditions for extracellular α-amylase production by the novel thermophilic Anoxybacillus sp. AH1 were carried out in many different media containing a variety of carbon and nitrogen sources. Among various carbon and nitrogen sources, peptone (2054.1 U/mL) at 1% and maltose (1862.9 U/mL) at 0.5% increased α-amylase activity, compared to controls. Moreover, a high enzyme production was observed with potato starch at 0.5% and 1% (2668.4 U/mL and 3627 U/mL, respectively), as well as with 1% soluble starch (2051.9 U/mL). The enzyme activity was found to be rather high in the presence of CaCl2 up to 100 mM.
Similar content being viewed by others
Abbreviations
- BM:
-
basal medium
- LB:
-
Luria broth
- NB:
-
nutrient broth
- TLC:
-
thin-layer chromatography
References
Aiyer P.V.D. 2004. Effect of C:N ratio on α-amylase production by Bacillus licheniformis SPT 27. Afr. J. Biotechnol. 3: 519–522.
Altschul S.F., Gish W., Miller W., Myers E.W. & Lipman D.J. 1990. Basic local alignment search tool. J. Mol. Biol. 215. 403–410.
Asgher M., Asad M.J., Rahman S.U. & Legge R.L. 2007. A thermostable α-amylase from a moderately thermophilic Bacillus subtilis strain for starch processing. J. Food. Eng. 79. 950–955.
Belduz A.O., Dulger S. & Demirbag Z. 2003. Anoxybacillus gonensis sp. nov., a moderately thermophilic, xylose-utilizing, endospore-forming bacterium. Int. J. Syst. Evol. Microbiol. 53. 1315–1320.
Bernfeld P. 1955. Amylases, α and β. Methods Enzymol. 1. 149–158
Chai Y.Y., Kahar U.M., Salleh M.M., Illias R.M. & Goh K.M. 2012a. Isolation and characterization of pullulan-degrading Anoxybacillus species isolated from Malaysian hot springs. Environ. Technol. 33. 1231–1238.
Chai YY, Rahman RN, Illias RM, Goh KM. 2012b. Cloning and characterization of two new thermostable and alkalitolerant α-amylases from the Anoxybacillus species that produce high levels of maltose. J. Ind. Microbiol. Biotechnol. 39. 731–741.
Cihan A.C., Ozcan B. & Cokmus C. 2011. Anoxybacillus salavatliensis sp. nov., an α-glucosidase producing, ther-mophilic bacterium isolated from Salavatli, Turkey. J. Basic Microbiol. 51. 136–146.
Cordeiro C.A.M., Martins M.L.L. & Luciano A.B. 2002. Production and properties of α-amylase from thermophilic Bacillus sp. Braz. J. Microbiol. 33. 57–61.
Das K., Doley R. & Mukherjee A.K. 2004. Purification and biochemical characterization of a thermostable, alkaliphilic, extracellular α-amylase from Bacillus subtilis DM-03, a strain isolated from the traditional fermented food of India. Biotechnol. Appl. Biochem. 40. 291–298.
De Clerck E., Rodríguez-Díaz M., Vanhoutte T., Heyrman J., Logan N.A., De Vos P. 2004. Anoxybacillus contaminans sp. nov. and Bacillus gelatini sp. nov., isolated from contaminated gelatin batches. Int. J. Syst. Evol. Microbiol. 54. 941–946.
Deep K., Poddar A. & Das S.K. 2013. Anoxybacillus suryakundensis sp. nov, a moderately thermophilic, alkalitolerant bacterium isolated from hot spring at Jharkhand, India. PLoS One 8: e85493.
Derekova A., Sjøholm C., Mandeva R. & Kambourova M. 2007. Anoxybacillus rupiensis sp. nov., a novel thermophilic bacterium isolated from Rupi basin (Bulgaria). Extremophiles 11: 577–583.
Devi B., Unni B.G., Wann S.B. & Samanta R. 2012. A study on partial purification and characterization of extracellular amylases from Bacillus subtilis. Adv. Appl. Sci. Res. 3. 2739–2744.
Dulger S., Demirbag Z. & Belduz A.O. 2004. Anoxybacillus ayderensis sp. nov. and Anoxybacillus kestanbolensis sp. nov. Int. J. Syst. Evol. Microbiol. 54. 1499–1503.
Dussault H.P. 1955. An improved technique for staining for halophilic bacteria. J. Bacteriol. 70. 484–485.
Felsenstein J. 1993. PHYLIP (Phylogeny Inference Package), version 3.5.1., University of Washington, Seattle, USA.
Goh K.M., Kahar U.M., Chai Y.Y., Chong C.S., Chai K.P., Ranjani V., Illias R.M. & Chan K.G. 2013. Recent discoveries and applications of Anoxybacillus. Appl. Microbiol. Biotechnol. 97. 1475–1488.
Goh K.M., Gan H.M., Chan K.G., Chan G.R., Chan G.F., Shabar S., Chong C.S., Kahar U.M. & Chai K.P. 2014. Analysis of Anoxybacillus genomes from the aspects of a lifestyle adaptations, arophage diversity, and carbohydrate metabolism. PLoS One 9: e90549.
Gordon R.H.W.C. & Pang C.H.N. 1973. The Genus Bacillus. Washington, D.C., USA.
Gul-Güven R., Güven K., Poli A. & Nicolaus B. 2008. Anoxybacillus kamchatkensis subsp. asaccharedens subsp. nov., a thermophilic bacterium isolated from a hot spring in Batman. J. Gen. Appl. Micbiobol. 54. 327–334.
Hamilton L.M., Kelly C.T. & Fogarty W.M. 1999. Production and properties of the raw starch-digesting α-amylase of Bacillus sp. IMD 435. Process Biochem. 35. 27–31.
Hauli I., Sarkar B., Mukherjee T. & Mukhopadhyay S.K. 2013. Isolation and identification of a novel thermo-alkaline, thermostable, SDS and chelator resistant amylase producing Anoxybacillus sp. IB-A from hot spring of Bakreswar, West Bengal (India): first report. Adv. Appl. Sci. Res. 4. 202–212.
Heinen W., Lauwers A.M. & Mulders J.W.M. 1982. Bacillus flavothermus, a newly isolated facultative thermophile. Antonie van Leeuwenhoek 48: 265–272.
Inan K., Belduz A.O. & Canakci S. 2013. Anoxybacillus kaynarcensissp. nov., a moderately thermophilic, xylanase producing bacterium. J. Basic Microbiol. 53. 410–419.
Janecek S., Svensson B. & MacGregor E.A. 2014. α-Amylase: an enzyme specificity found in various families of glycoside hydrolases. Cell. Mol. Life Sci. 71. 1149–1170.
Kanimozhi M., Johny M., Gayathri N. & Subashkumar R. 2014. Optimization and production of α-amylase from halophilic Bacillus species isolated from Mangrove soil sources. J. Appl. Environ. Microbiol. 2. 70–73.
Kevbrin V.K., Zengler K., Lysenko A.M. & Wiegel J. 2005. Anoxybacillus kamchatkensis sp. nov., a novel thermophilic facultative aerobic bacterium with a broad pH optimum from the Geyser valley, Kamchatka. Extremophiles 9: 391–398.
Khan J.A. & Priya R. 2011. A study on partial purification and characterization of extracellular amylases from Bacillus subtilis. Adv. Appl. Sci. Res. 2. 509–519.
Kikani B.A. & Singh S.P. 2012. The stability and thermodynamic parameters of a very thermostable and calcium-independent α-amylase from a newly isolated bacterium, Anoxybacillus beppuensis TSSC-1. Process Biochem. 47. 1791–1798.
Kim O.S., Cho Y.J., Lee K., Yoon S.H., Kim M., Na H., Park S.C., Jeon Y.S., Lee J.H., Yi H., Won S. & Chun J. 2012. Introducing EzTaxon: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int. J. Syst. Evol. Microbiol. 62. 716–721.
Konsoula Z. & Liakopoulou-Kyriakides M. 2007. Co-production of α-amylase and β-galactosidase by Bacillus subtilis in complex organic substrates. Bioresour. Technol. 98. 150–157.
Lanyi B. 1987. Classical and rapid identification methods for medically important bacteria. Methods Microbiol. 19. 1–67.
Lowry O.H., Rosebrough N.J. & Farr A.L. & Randall R.J. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193. 265–275.
Maidak B.L., Cole J.R., Parker C.T.J., Garrity G.M., Larsen N., Li B., Lilburn T.G., Mc Caughey M.J., Olsen G. J., Overbeek R., Pramanik S., Schmidt T.M., Tiedje J.M. & Woese C.R. 1999. A new version of the RDP (Ribosomal Database Project). Nucleic Acids Res. 27. 171–173.
Matpan Bekler F. & Güven K. 2014. Isolation and production of thermostable α-amylase from thermophilic Anoxybacillus sp. KP1 from Diyadin hot spring in Ağri, Turkey. Biologia 69: 419–427.
Nicolaus B., Manca M.C. Lama L., Esposito E. & Gambacorta A. 2001. Lipid modulation by environmental stresses in two models of extremophiles isolated from Antarctica. Polar Biol. 24. 1–8.
Pikuta E., Cleland D. & Tang J. 2003. Aerobic growth of Anoxy-bacillus pushchinoensis K1T: emended descriptions of A. pushchinoensis and the genus Anoxybacillus. Int. J. Syst. Evol. Microbiol. 53. 1561–1562.
Pikuta E., Lysenko A., Chuvilskaya N., Mendrock U., Hippe H., Suzina N., Nikitin D., Osipov G. & Laurinavichius K. 2000. Anoxybacillus pushchinensis gen. nov., sp. nov., a novel anaerobic, alkaliphilic, moderately thermophilic bacterium from manure, and description of Anoxybacillus flavithermus comb. nov. Int. J. Syst. Evol. Microbiol. 50. 2109–2117.
Poli A., Esposito E., Lama L., Orlando P., Nicolaus G., Appolonia F., Gambacorta A. & Nicolaus B. 2006. Anoxybacillus amylolyticus sp. nov., a thermophilic amylase producing bacterium isolated from Mount Rittmann (Antarctica). Syst. Appl. Microbiol. 29. 300–307.
Poli A., Romano I., Cordella P., Orlando P., Nicolaus B. & Ceschi Berrini C. 2009. Anoxybacillus thermarum sp. nov., a novel thermophilic bacterium isolated from thermal mud in Eu-ganean hot springs, Abano Terme, Italy. Extremophiles 13: 867–874.
Prakash B., Vidyasagar M., Madhukumar M.S., Muralikrishna G. & Sreeramulu K. 2009. Production, purification, and characterization of two extremely halotolerant, thermostable, and alkali-stable α-amylases from Chromohalobacter sp. TVSP 101. Process Biochem. 44. 210–215.
Rainey F.A., Fritze D. & Stackebrandt E. 1994. The phylogenetic diversity of thermophilic members of the genus Bacillus as revealed by 16S rRNA analysis. FEMS Microbiol. Lett. 115. 205–212.
Rainey F.A., Ward-Rainey N., Kroppenstedt R.M. & Stackbrandt E. 1996. The genus Nocardiopsis represents a phyloge-netically coherent taxon and a distinct Actinomycete lineage: proposal of Nocardiopsaceae fam. nov. Int. J. Syst. Bacteriol. 46. 1088–1092.
Ranjani V., Janecek S., Chai K.W., Shahir S., Abdul Rahman R.N., Chan K.G. & Goh K.M. 2014. Protein engineering of selected residues from conserved sequence regions of a novel Anoxybacillus α-amylase. Sci. Rep. 4. 5850.
Saitou N. & Nei M. 1987. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4. 406–425.
Saw J.H., Mountain B.W., Feng L., Omelchenko M.W., Hou S., Saito J.A., Stott, M.B., Li D., Zhao G., Wu J., Galperin M.Y., Koonin E.V., Makarova K.S., Wolf Y.I., Rigden D.J., Dun-field P.F., Wang L. & Alam M. 2008. Encapsulated in silica: genome, proteome and physiology of the thermophilic bacterium Anoxybacillus flavithermus WK1. Genome Biol. 9. R161.
Tawil G., Viksø-Nielsen A., Rolland-Sabaté A., Colonna P. & Buléon A. 2012. Hydrolysis of concentrated raw starch: a new very efficient α-amylase from Anoxybacillus flavother-mus. Carbohydr. Polym. 87. 46–52.
Yang S.J., Lee H.S., Park C.S., Kim Y.R., Moon T.W. & Park K.H. 2004. Enzymatic analysis of an amylolytic enzyme from the hyper-thermophilic archaeon Pyrococcus furiosus reveals its novel catalytic properties as both an α-amylase and a cyclodextrin-hydrolyzing enzyme. Appl. Environ. Microbiol. 70. 5988–5995.
Yang X., Zhang F., Ren W., Nie G., Li S., Ming H., Tang S., Zhang L. & Li W. 2012. A study on the best condition for thermostable α-amylase fermented by response surface methodology by strain Anoxybacillus sp. YIM 342. Journal of Yunnan University 4: 490–496.
Yumoto I., Hirota K., Kawahara T., Nodasaka Y., Okuyama H., Matsuyama H., Yokota Y., Nakajima K. & Hoshino T. 2004. Anoxybacillus voinovskiensis sp. nov., a moderately thermophilic bacterium from a hot spring in Kamchatka. Int. J. Syst. Evol. Microbiol. 54. 1239–1242.
Zhang C.M., Huang, X.W., Pan W.Z., Zhang J., Wei K.B., Klenk H.P., Tang S.K., Li W.J. & Zhang, K. 2010. Anoxybacillus tengchongensis sp. nov. and Anoxybacillus eryuanensis sp. nov., facultatively anaerobic, alkalitolerant bacteria from hot springs. Int. J. Syst. Evol. Microbiol. 61. 118–122.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Acer, Ö., Pirinççioğlu, H., Matpan Bekler, F. et al. Anoxybacillus sp. AH1, an α-amylase-producing thermophilic bacterium isolated from Dargeçit hot spring. Biologia 70, 853–862 (2015). https://doi.org/10.1515/biolog-2015-0111
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2015-0111