Abstract
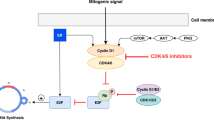
GTI-2040 is a potent antisense to the M2 subunit of the ribonucleotide reductase (RNR), an enzyme involved in the de novo synthesis of nucleoside triphosphates. We hypothesized that combination of GTI-2040 with the cytarabine (Ara-C) could result in an enhanced cytotoxic effect with perturbed intracellular deoxynucleotide/nucleotide (dNTP/NTP) pools including Ara-C triphosphate (Ara-CTP). This study aims to provide a direct experimental support of this hypothesis by monitoring the biochemical modulation effects, intracellular levels of Ara-CTP, dNTPs/NTPs following the combination treatment of Ara-C, and GTI-2040 in K562 human leukemia cells. GTI-2040 was introduced into cells via electroporation. A hybridization–ligation ELISA was used to quantify intracellular GTI-2040 concentrations. Real-time PCR and Western blot methods were used to measure the RNR M2 mRNA and protein levels, respectively. 3-(4,5-Dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt assay was used to measure the cytotoxicity following various drug treatments. A non-radioactive HPLC-UV method was used for measuring the intracellular Ara-CTP, while a LC-MS/MS method was used to quantify intracellular dNTP/NTP pools. GTI-2040 was found to downregulate M2 mRNA and protein levels in a dose-dependent manner and showed significant decrease in dNTP but not NTP pool. When combining GTI-2040 with Ara-C, a synergistic cytotoxicity was observed with no further change in dNTP/NTP pools. Importantly, pretreatment of K562 cells with GTI-2040 was found to increase Ara-CTP level for the first time, and this effect may be due to inhibition of RNR by GTI-2040. This finding provides a laboratory justification for the current phase I/II evaluation of GTI-2040 in combination with Ara-C in patients with acute myeloid leukemia.








Similar content being viewed by others
References
Fontecave M. Ribonucleotide reductases and radical reactions. Cell Mol Life Sci. 1998;54(7):684–95.
Reichard P, Ehrenberg A. Ribonucleotide reductase—a radical enzyme. Science (New York, NY). 1983;221(4610):514–9.
Jordan A, Reichard P. Ribonucleotide reductases. Annu Rev Biochem. 1998;67:71–98.
Kolberg M, Strand KR, Graff P, Andersson KK. Structure, function, and mechanism of ribonucleotide reductases. Biochim Biophys Acta. 2004;1699(1–2):1–34.
Nocentini G. Ribonucleotide reductase inhibitors: new strategies for cancer chemotherapy. Crit Rev Oncol Hematol. 1996;22(2):89–126.
Cory JG. Ribonucleotide reductase as a chemotherapeutic target. Adv Enzyme Regul. 1988;27:437–55.
Lee Y, Vassilakos A, Feng N, Lam V, Xie H, Wang M, et al. GTI-2040, an antisense agent targeting the small subunit component (R2) of human ribonucleotide reductase, shows potent antitumor activity against a variety of tumors. Cancer Res. 2003;63(11):2802–11.
Smith BD, Karp JE. Ribonucleotide reductase: an old target with new potential. Leuk Res. 2003;27(12):1075–6.
Bianchi V, Borella S, Calderazzo F, Ferraro P, Chieco Bianchi L, Reichard P. Inhibition of ribonucleotide reductase by 2′-substituted deoxycytidine analogs: possible application in AIDS treatment. Proc Natl Acad Sci USA. 1994;91(18):8403–7.
Desai AA, Schilsky RL, Young A, Janisch L, Stadler WM, Vogelzang NJ, et al. A phase I study of antisense oligonucleotide GTI-2040 given by continuous intravenous infusion in patients with advanced solid tumors. Ann Oncol. 2005;16(6):958–65.
Estey EH. Therapeutic options for acute myelogenous leukemia. Cancer. 2001;92(5):1059–73.
Ravindranath Y. Recent advances in pediatric acute lymphoblastic and myeloid leukemia. Curr Opin Oncol. 2003;15(1):23–35.
Creasey WA, Papac RJ, Markiw ME, Calabresi P, Welch AD. Biochemical and pharmacological studies with 1-beta-D-arabinofuranosylcytosine in man. Biochem Pharmacol. 1966;15(10):1417–28.
Klisovic RB, Blum W, Wei X, Liu S, Liu Z, Xie Z, et al. Phase I study of GTI-2040, an antisense to ribonucleotide reductase, in combination with high-dose cytarabine in patients with acute myeloid leukemia. Clin Cancer Res. 2008;14(12):3889–95.
Wei X. Pharmacokinetics, pharmacodynamics and metabolism of GTI-2040, a phosphorothioate oligonucleotide targeting R2 subunit of ribonucleotide reductase. Columbus: Ohio State University; 2006. http://rave.ohiolink.edu/etdc/view?acc%5Fnum=osu1143220921.
Wei X, Dai G, Marcucci G, Liu Z, Hoyt D, Blum W, et al. A specific picomolar hybridization-based ELISA assay for the determination of phosphorothioate oligonucleotides in plasma and cellular matrices. Pharm Res. 2006;23(6):1251–64.
Chen P, Liu Z, Liu S, Xie Z, Aimiuwu J, Pang J, et al. A LC-MS/MS method for the analysis of intracellular nucleoside triphosphate levels. Pharm Res. 2009;26(6):1504–15.
Chou TC, Talalay P. Quantitative analysis of dose–effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984;22:27–55.
Avramis VI, Biener R, Krailo M, Finklestein J, Ettinger L, Willoughby M, et al. Biochemical pharmacology of high dose 1-beta-D-arabinofuranosylcytosine in childhood acute leukemia. Cancer Res. 1987;47(24 Pt 1):6786–92.
Braess J, Wegendt C, Jahns-Streubel G, Kern W, Keye S, Unterhalt M, et al. Successful modulation of high-dose cytosine arabinoside metabolism in acute myeloid leukaemia by haematopoietic growth factors: no effect of ribonucleotide reductase inhibitors fludarabine and gemcitabine. Br J Haematol. 2000;109(2):388–95.
Liliemark JO, Plunkett W, Dixon DO. Relationship of 1-beta-D-arabinofuranosylcytosine in plasma to 1-beta-D-arabinofuranosylcytosine 5′-triphosphate levels in leukemic cells during treatment with high-dose 1-beta-D-arabinofuranosylcytosine. Cancer Res. 1985;45(11 Pt 2):5952–7.
Ossenkoppele GJ, Graveland WJ, Sonneveld P, Daenen SM, Biesma DH, Verdonck LF, et al. The value of fludarabine in addition to ARA-C and G-CSF in the treatment of patients with high-risk myelodysplastic syndromes and AML in elderly patients. Blood. 2004;103(8):2908–13.
Ozkaynak MF, Avramis VI, Carcich S, Ortega JA. Pharmacology of cytarabine given as a continuous infusion followed by mitoxantrone with and without amsacrine/etoposide as reinduction chemotherapy for relapsed or refractory pediatric acute myeloid leukemia. Med Pediatr Oncol. 1998;31(6):475–82.
Breithaupt H, Pralle H, Eckhardt T, von Hattingberg M, Schick J, Loffler H. Clinical results and pharmacokinetics of high-dose cytosine arabinoside (HD ARA-C). Cancer. 1982;50(7):1248–57.
Capizzi RL, Poole M, Cooper MR, Richards 2nd F, Stuart JJ, Jackson Jr DV, et al. Treatment of poor risk acute leukemia with sequential high-dose ARA-C and asparaginase. Blood. 1984;63(3):694–700.
Capizzi RL, White JC, Powell BL, Perrino F. Effect of dose on the pharmacokinetic and pharmacodynamic effects of cytarabine. Semin Hematol. 1991;28(3 Suppl 4):54–69.
Estey E, Plunkett W, Dixon D, Keating M, McCredie K, Freireich EJ. Variables predicting response to high dose cytosine arabinoside therapy in patients with refractory acute leukemia. Leukemia. 1987;1(8):580–3.
Qian SX, Li JY, Tian T, Shen YF, Jiang YQ, Lu H, et al. Effect of low-dose cytarabine and aclarubicin in combination with granulocyte colony-stimulating factor priming (CAG regimen) on the outcome of elderly patients with acute myeloid leukemia. Leuk Res. 2007;31(10):1383–8.
Willemze R, Zwaan FE, Colpin G, Keuning JJ. High dose cytosine arabinoside in the management of refractory acute leukaemia. Scand J Haematol. 1982;29(2):141–6.
Yamauchi T, Negoro E, Arai H, Ikegaya S, Takagi K, Takemura H, et al. Combined low-dose cytarabine, melphalan and mitoxantrone for older patients with acute myeloid leukemia or high-risk myelodysplastic syndrome. Anticancer Res. 2007;27(4C):2635–9.
Zhang WG, Wang FX, Chen YX, Cao XM, He AL, Liu J, et al. Combination chemotherapy with low-dose cytarabine, homoharringtonine, and granulocyte colony-stimulating factor priming in patients with relapsed or refractory acute myeloid leukemia. Am J Hematol. 2008;83(3):185–8.
Bauer W, Horvath Z, Hochtl T, Saiko P, Karl D, Fritzer-Szekeres M, et al. Amidox, an inhibitor of ribonucleotide reductase, potentiates the action of Ara-C in HL-60 human promyelocytic leukemia cells. Nucleosides Nucleotides Nucleic Acids. 2004;23(8–9):1541–4.
Fritzer-Szekeres M, Salamon A, Grusch M, Horvath Z, Hochtl T, Steinbrugger R, et al. Trimidox, an inhibitor of ribonucleotide reductase, synergistically enhances the inhibition of colony formation by Ara-C in HL-60 human promyelocytic leukemia cells. Biochem Pharmacol. 2002;64(3):481–5.
Plagemann PG, Behrens M, Abraham D. Metabolism and cytotoxicity of 5-azacytidine in cultured Novikoff rat hepatoma and P388 mouse leukemia cells and their enhancement by preincubation with pyrazofurin. Cancer Res. 1978;38(8):2458–66.
Gandhi V, Estey E, Keating MJ, Chucrallah A, Plunkett W. Chlorodeoxyadenosine and arabinosylcytosine in patients with acute myelogenous leukemia: pharmacokinetic, pharmacodynamic, and molecular interactions. Blood. 1996;87(1):256–64.
Gandhi V, Estey E, Plunkett W. Modulation of arabinosylcytosine metabolism during leukemia therapy. Adv Exp Med Biol. 1994;370:119–24.
Gandhi V, Nowak B, Keating MJ, Plunkett W. Modulation of arabinosylcytosine metabolism by arabinosyl-2-fluoroadenine in lymphocytes from patients with chronic lymphocytic leukemia: implications for combination therapy. Blood. 1989;74(6):2070–5.
Bhalla K, Swerdlow P, Grant S. Effects of thymidine and hydroxyurea on the metabolism and cytotoxicity of 1-B-D arabinofuranosylcytosine in highly resistant human leukemia cells. Blood. 1991;78(11):2937–44.
Colly LP, Richel DJ, Arentsen-Honders MW, Kester MG, ter Riet PM, Willemze R. Increase in Ara-C sensitivity in Ara-C sensitive and -resistant leukemia by stimulation of the salvage and inhibition of the de novo pathway. Ann Hematol. 1992;65(1):26–32.
Iversen N, Birkenes B, Torsdalen K, Djurovic S. Electroporation by nucleofector is the best nonviral transfection technique in human endothelial and smooth muscle cells. Genet Vaccines Therapy. 2005;3(1):2.
Cooper T, Ayres M, Nowak B, Gandhi V. Biochemical modulation of cytarabine triphosphate by clofarabine. Cancer Chemother Pharmacol. 2005;55(4):361–8.
Wang LM, White JC, Capizzi RL. The effect of Ara-C-induced inhibition of DNA synthesis on its cellular pharmacology. Cancer Chemother Pharmacol. 1990;25(6):418–24.
Verschuur AC, Van Gennip AH, Leen R, Voute PA, Brinkman J, Van Kuilenburg AB. Cyclopentenyl cytosine increases the phosphorylation and incorporation into DNA of 1-beta-D-arabinofuranosyl cytosine in a human T-lymphoblastic cell line. Int J Cancer. 2002;98(4):616–23.
Williams JC, Kizaki H, Weber G, Morris HP. Increased CTP synthetase activity in cancer cells. Nature. 1978;271(5640):71–3.
Cros E, Jordheim L, Dumontet C, Galmarini CM. Problems related to resistance to cytarabine in acute myeloid leukemia. Leuk Lymphoma. 2004;45(6):1123–32.
Schilsky RL, Ordway FS. Simultaneous determination of cytosine arabinoside, its nucleotides and metabolites by ion-pair high-performance liquid chromatography. J Chromatogr. 1985;337(1):63–71.
Plunkett W, Hug V, Keating MJ, Chubb S. Quantitation of 1-beta-D-arabinofuranosylcytosine 5′-triphosphate in the leukemic cells from bone marrow and peripheral blood of patients receiving 1-beta-D-arabinofuranosylcytosine therapy. Cancer Res. 1980;40(3):588–91.
Acknowledgment
This work was partially supported by National Cancer Institute/National Institute of Health grant R21 CA133879 (GM/RBK) and grant UO1-CA76576 and Lorus Therapeutics.
Conflict of interest statement
Lorus Therapeutics provided partial support for the development of the Ara-CTP and dNTP/NTP assays.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ping Chen and Josephine Aimiuwu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, P., Aimiuwu, J., Xie, Z. et al. Biochemical Modulation of Aracytidine (Ara-C) Effects by GTI-2040, a Ribonucleotide Reductase Inhibitor, in K562 Human Leukemia Cells. AAPS J 13, 131–140 (2011). https://doi.org/10.1208/s12248-010-9246-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-010-9246-5