Abstract
Background
Alterations in intercellular and cell-extracellular matrix connections contribute to tumour development. This study investigates the expression of specific cell adhesion molecules (CAMs) in salivary gland tumors (SGTs).
Methods
Formalin–fixed, paraffin– embedded tissue specimens of different types of 34 benign and 31 malignant SGTs and normal salivary glands were studied using Envision/HRP immunohistochemical technique for Desmoglein-2 (Dsg-2), beta4-integrin, CD44s and ICAM-1. Intensity of staining was evaluated in a semi-quantitative manner. Results were analyzed using Kendall’s τ and Spearman’s ρ as correlation criteria.
Results
Dsg-2 in intercellular space, beta4-integrin in cell-basal membrane, and CD44s in both types of contacts were strongly expressed in normal acinar and ductal cells, whereas ICAM-1 was expressed only at the endothelium and sparse stromal cells and monocytes. Strong correlation was found between Dsg-2 expression in adenomas and controls and between adenocarcinomas and controls. In adenomas, a distinct cytoplasmic presence of Dsg-2 was observed in addition to the usual membranous expression, with decreased expression in comparison with normal tissue. In malignant SGTs, Dsg-2 expression was absent. In most SGTs, beta4-integrin was expressed also with a distinct pattern, involving the cytoplasm and the unpolarised membrane, while CD44 was found only on the membrane. Strong correlation between beta4-integrin expression in adenomas and controls was noted, while CD44 expression was found to be correlated significantly between adenocarcinomas and controls (p < 0.001). Regarding ICAM-1, its expression was found increased in adenomas, with non-specific distribution in malignant SGTs and strong correlation between the histological subtypes and controls (p < 0.001).
Conclusion
The different expression profile of CAMs in SGTs could possibly suggest a role on their pathogenesis, representing a model of how neoplastic cells can take advantage of normal tissue architecture and cell-extracellular matrix interactions.
Similar content being viewed by others
Background
The development and organization of multicellular organs, such as salivary glands, requires adhesion structures for intercellular communication, anchorage to the extracellular matrix (ECM), and triggering of intracellular pathways. This cell adhesion is mediated by subfamilies of transmembrane glycoprotein receptors named Cell Adhesion Molecules (CAMs) who mediate cell–cell and cell–matrix interaction and play important role in tissue morphogenesis, integrity and maintenance, as well as in immune responses [1,2,3,4,5]. Alteration of CAMs expression is associated with pathogenesis and progression of benign and malignant solid neoplasms of various tissues [6,7,8,9,10]. Desmoglein-2 (Dsg-2) is the main desmoglein expressed in desmosomal junctions (desmosomes) of mono/bilayer epithelia [11,12,13] (like salivary gland epithelium). Although the role of desmosomes in carcinogenesis is still unknown, previous studies have shown that Dsg-2 can act either as a tumor suppressor or an oncogene. Dsg-2 expression was upregulated in skin squamous cell carcinoma and head and neck cancer. In contrast, Dsg-2 expression was downregulated in pancreatic cancer, melanoma cells, and diffuse-type gastric cancer [14].
ICAM-1 is a cell-surface glycoprotein member of the immunoglobulin (Ig) superfamily. ICAM-1 is expressed on the surface of most cells and its expression is correlated with chronic inflammatory conditions like atherosclerosis, diabetes and cerebral malaria [15]. Previous studies have emphasised its role in elucidating cancer prognosis and progression by measuring the concentration of its soluble form as, for example, in advanced non-small cell lung cancer [16].
Moreover, beta4-integrin is also implicated in normal tissue epithelial-stromal architecture, neoplasm formation and immune responses by affecting cell cycle. Integrins are transmembrane receptors that facilitate cell-extracellular matrix adhesion; they form a critical connection between the extracellular matrix and the cell interior. Beta4-integrin electively binds with the alpha-6 subunit and then to laminin. In non-small cell lung cancer, tyrosine kinase receptor has been found to interact with integrin beta-4/alpha-6 ligand to facilitate tumor invasion, creating a microenvironment that promotes this type of cancer [17].
Pleomorphic adenomas (PAs) are the most common benign tumors of the salivary glands, which mainly arise from the major salivary glands, including the parotid and submandibular glands. Although most PAs are benign, recurrence may occur as well as malignant transformation. Previous studies have shown that CD44 cells can act as tumor inducing cells promoting tumor transformation [18].
So far, only a few studies have investigated through immunohistochemistry the expression and location of different CAMs in normal SGs and in specific types of benign or malignant tumours with contradictory results [4, 19,20,21,22,23,24,25,26,27,28,29,30]. The aim of this study was to determine a possibly altered expression profile (in comparison with normal tissue) of specific CAMs, including Dsg-2 (for the first time), beta4-integrin, CD44s and ICAM-1 in both benign and malignant SGTs, which could potentially affect intercellular and cell–matrix interactions, hence contributing to carcinogenesis.
Methods
Specimen
Tissue specimens were retrieved from 34 selected cases of adenomas, including pleomorphic adenoma (n = 30), myoepithelioma (n = 2) and oncocytoma (n = 2), and 31 cases of adenocarcinomas including acinic cell carcinoma (n = 7), epithelial-myoepithelial carcinoma (n = 3), adenoid cystic carcinoma (n = 3), polymorphous low-grade adenocarcinoma (n = 2), salivary ductal carcinoma (n = 3), mucoepidermoid (n = 3), squamous cell carcinoma (n = 5), adenocarcinoma not otherwise specified (NOS, n = 1), oncocytic carcinoma (n = 1), myoepithelial carcinoma (n = 1), and lymphoepithelial carcinoma (n = 2). Specimens were routinely fixed and embedded in paraffin and were retrieved from the archives of the Department of Histopathology of Red Cross Hospital in Athens, and Department of Oral Medicine/Pathology School of Dentistry, Aristotle University of Thessaloniki, Greece between 1999 and 2003. Diagnosis and classification were based on the criteria of the World Health Organization for salivary gland tumours [31]. In addition, 20 normal parotid and submandibular gland tissue adjacent to benign tumours and 7 normal minor labial salivary glands were also studied as controls.
Immunohistochemistry and intensity of staining
The antigen retrieval and the Envision-HRP automated immunohistochemical technique (Dako Cytomation A/S, Glostrup, Denmark), using anti-CAMs antibodies (Santa Cruz, Biotechnology Inc, California, USA), were performed according to manufacturer instructions. Immunoreactivity was semi-quantitatively assessed and positivity was considered if greater than 5% of tumour cells were stained. Positivity was graded according to the percentage of tumour cells stained as I: 5–25%, II: 26–50%, III: 51–100%. The staining intensity was assessed as follows: negative (-), weak to moderate (+), moderate to strong (++) and strong (+++). Staining pattern for each neoplastic cell subpopulation was also characterized as cell membrane and/or cytoplasmic, whereas nuclear staining was not considered as positive. Sections were examined by three of the authors (CB, AE, DA) independently. Sections were re-examined when there were differences, and discussion was occasionally necessary to establish uniformity. Patients had given informed consent, and the whole study was performed according to the declaration of Helsinki II.
Statistics
Rank correlation tests were performed for the type of tissue (taking in pairs: control–adenoma, control–adenocarcinoma, adenoma–adenocarcinoma) and the observed intensity of staining. Furthermore, a similar analysis was performed for the type of tissue and the observed positivity. Non-parametric tests, Kendall’s τ and Spearman’s ρ were used for statistical analysis. Results were processed using SPSS ver. 25 (IBM, USA).
Results
CAMs in normal salivary glands and benign SGTs
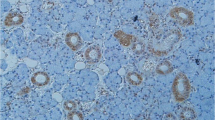
Immunohistochemical results for all examined CAMs were summarized in Table 1 and for benign SGTs in Table 2. In all cases, Dsg-2 was expressed at epithelial cell to cell contacts of acinar and ductal cells but was absent from their luminal-apical surface as well as from basal surface that participates in cell-basement membrane connections. Τhe staining of this receptor was more intense mainly in the ducts than the acini and on basal cells rather than their apical pole (Fig. 1a) while Dsg-2 was not expressed in stromal cells. Regarding beta4-integrin, it was intensely (+++) expressed mainly on the basolateral surface of acinar (serous and mucous), myoepithelial cells, intercalated ductal cells and basal cells of excretory ducts (Fig. 1b). Endothelial cells were also positive for beta4-integrin expression, whereas the rest stromal cell subpopulations were negative. CD44s receptor was strongly expressed on the surface of acinar cells, mainly serous, in all cases. Staining in ductal epithelium was moderate, involving the basolateral surfaces of basal cells, and focally the intercellular contacts of luminal cells (Fig. 1c). Stromal lymphocytes were also positively stained for CD44s. ICAM-1 was absent from the normal glandular epithelium but was expressed in the endothelial and stromal cells including a few inflammatory cells (lymphocytes and macrophages) (Fig. 1d).
Cell adhesion molecules in normal salivary gland tissue. a Desmoglein-2 (Dsg-2): Linear membranous staining of acinar and ductal cells. b Beta4-integrin: Strong immunoreactivity of basolateral portion of serous cell membrane and basal cells of interlobular ducts. c HCAM (CD44s): Strong membranous staining of acinar cells (mainly serous), decreased expression in basolateral surfaces of ductal basal cells and partially in stromal non-epithelial cell subpopulations. d ICAM-1: Negative staining of epithelial cells. Membranous staining of non-epithelial stromal subpopulations such as endothelium, lymphocytes and fibroblasts
Pleomorphic adenoma
Dsg-2 was found in all cases showing mainly a cytoplasmic pattern in more than 50% of neoplastic cells, excluding solitary plasmacytoid cells in myxoid stroma (Fig. 2a). Beta4-integrin showed a diffuse, strong positivity at both cell-membrane and cytoplasm in more than 50% of neoplastic cells in all neoplastic structures (Fig. 2b). In all cases, CD44 was expressed along the membrane of intercellular and cell–matrix contacts of most neoplastic cells, especially in cells of solid structures, cells showing stromal differentiation and non-luminal cells of duct-like structures (Fig. 2c). Lymphocytes, endothelial, and neoplastic cells, located mainly in myxoid stroma were positive for ICAM-1. Moreover, in some of the cases, a continuous positive linear staining on the luminal surface of luminal cells in neoplastic duct-like structures was observed (Fig. 2d).
Cell adhesion molecules in Pleomorphic Adenoma. a Dsg-2: Membranous or diffuse cytoplasmic staining at the intercellular contacts of neoplastic cells of all architecture types except luminal surfaces of luminal cells and basal surfaces of non-luminal cells. No positivity in stromal cells (Envision/HRP × 100). b Beta4-integrin: Strong membranous positivity at cell-basal membrane contacts, intercellular connections of the great majority of cells and stromal cells (Envision/HRP x200). c HCAM (CD44s): Strong membranous expression in the majority of neoplastic cells of all structures especially in stromal cells, cells of solid islands, non-luminal cells and intercellular contacts between luminal and non-luminal cells (Envision/HRP x200). d ICAM-1: Fibroblastoid stromal cells mainly in myxoid areas, linear positivity at the luminal surface of luminal ductal cells and moderate expression in some cells of solid islands (Envision/HRP x200). Strong immunoreactivity of basolateral portion of serous cell membrane and basal cells of interlobular ducts
Myoepithelioma and oncocytoma
In both myoepithelioma and oncocytoma most of neoplastic cells showed a moderate cytoplasmic staining for Dsg-2 whereas beta4-integrin was strongly expressed among the cell membrane and cytoplasm of neoplastic cells. However, the number of positive cells for CD44s in myoepithelioma was less than in oncocytoma (10–25% and > 50%). However, the intensity of staining for oncocytoma was weak. Finally, ICAM-1 was absent in oncocytoma, whereas the two cases of myoepitheliomas showed a heterogeneous positivity.
CAMs in malignant SGTs (adenocarcinomas)
The presence of beta4-integrin, Dsg-2, ICAM-1 and HCAM (CD44s) in malignant tumors was presented in Table 3, emphasizing the localization and intensity of staining in each tumor type. In all cases, beta4-integrin had a moderate to strong expression in unpolarized cell membrane, different to its normal distribution on the basolateral surfaces of cells.
Regarding Dsg-2, the intensity of staining was strong (+++) in 100% of control samples, while in adenomas, 100% of specimens were stained moderately (++). In malignant cases, 77% of cases (24/31) had moderate intensity of staining, while in 10% (3/31) there was weak staining. In 13% of all malignancies, staining was negative (-). Very strong correlation was found in Dsg-2 staining expression between controls and adenomas (Kendall’s τ-c = 0.987, p < 0.001; Spearman’s ρ = 1, p < 0.001; Table 4) and between controls and malignancies (Kendall’s τ-c = 0.995, p < 0.001; Spearman’s ρ = 0.950, p < 0.001; Table 4). Furthermore, in malignant tumors, results of Dsg-2 revealed severe decrease or loss of membrane expression, which was related to the type of malignancy. A cytoplasmic and even membranous expression was noticed in most neoplastic cells in acinic cell carcinoma, epithelial-myoepithelial carcinoma, adenoid cystic carcinoma, polymorphous low-grade adenocarcinoma, salivary ductal carcinoma, squamous cell carcinoma, whereas was focally presented or absent in adenocarcinoma NOS, oncocytic carcinoma, myoepithelial, and lymphoepithelial carcinomas.
Regarding beta4-integrin, intensity of staining was highly correlated between adenomas and controls (Kendall’s τ-c = 0.871, p < 0.001; Spearman’s ρ = 0.877, p < 0.001; Table 4), while malignancies were similar with controls (Table 4). More specifically, while strong (+++) intensity of staining had been observed in 100% of controls, only 12% (4/34) of benign adenomas had had the same feature. In malignancies, on the other hand, 81% (30/34) were strongly (+++) stained.
Regarding CD44, intensity of staining was not significantly correlated between controls and benign adenomas (Table 4). However, controls and adenocarcinomas were significantly correlated (Kendall’s τ-c = 0.578, p < 0.001; Spearman’s ρ = 0.579, p < 0.001; Table 4). All controls and 94% (32/34) of benign adenomas have been moderately stained (++) for CD44, while in malignant cases 21 out of 31 cases (68%) have been strongly (+++) stained for the same antibody. In seven out of 31 cases (23%) moderate (++) staining was noticed, while three out of 31 (10%) had presented weak intensity.
Regarding ICAM-1, strong correlation was noted between controls, adenomas and adenocarcinomas (p < 0.001).
Discussion
Desmosomes, hemidesmosomes and other intercellular and cell–matrix connections are playing an important role in tissue homeostasis and tissue architecture through cell to cell interaction [1]. To the best of our knowledge, the expression of desmosomal component Dsg-2 in salivary gland tissues was only generally referred in a study of Schäfer et al. [11]. In our study we identified the exact pattern of Dsg-2 expression in glandular epithelium as a membrane intercellular connector of all acinar and ductal cells, mainly at the basal pole of acinar and myoepithelial cells and apical pole of luminal cells of excretory ducts.
Limited and conflicting information of different CAMs in the development of pleomorphic adenoma were reported in the literature [27, 28, 32,33,34,35,36,37,38,39,40,41,42]. Furthermore, to our knowledge, Dsg-2, the main desmosomal cadherin in salivary glandular epithelium has not been investigated in depth so far [43]. According to our results, Dsg-2, showed decreased and alternative, mainly cytoplasmic, expression in most of the neoplastic cells excluding the solitary plasmacytoid cells in myxoid stroma. These findings were similar to the abnormal, decreased expression that was reported in gastric and breast adenocarcinomas [44, 45], and suggest that the loss of intercellular desmosome contacts could possibly be an important event during salivary gland neoplastic development. In salivary gland pleomorphic adenoma, this cell to cell detachment may be partially associated with the increased stromal component secretions, which extend the intercellular space breaking the intercellular junctions.
Few papers have evaluated the presence of CAMs subspecies in normal salivary glands. Loducca et al. [35] have described the normal topography of hemidesmosomal beta4-integrin in normal salivary gland tissues. Our study was in agreement with previous data, suggesting that beta4-integrin was strongly expressed at the basolateral membranous portions of serous and mucous acinic cells indicating the existence of laminin not only in basement membrane, but also in intercellular space function as a cell–cell bridge. Loducca et al. [35] also found that beta4-integrin distributed in well-developed clusters on luminal cell pole of intercalated ducts, in a delicate bipolar manner in the striated ducts, and limited to a few intercellular contacts and cell-membrane junctions of excretory ducts. However, we observed strong expression of beta4-integrin at the basolateral surface of intercalated ducts and the overall basal cell membrane of striated and excretory ducts. This can possibly suggest that beta4-integrin and laminin are involved in cell-basement membrane and possibly intercellular connections of ductal epithelium.
There are previous studies, like that of Franchi et al. [29, 41], that had investigated the expression of integrin in pleomorphic adenoma (especially integrin a-subunits) and its participation in cell–cell contacts of tubular and ductal structures, cell–matrix connections and isolated cells in stroma. Furthermore, Lourenço et al. [28] found that integrin a-subunits were intensely expressed on neoplastic luminal cells. Beta-1 subunit was also found to be widely expressed on luminal cells of neoplastic ducts and neoplastic modified myoepithelial cells were positive for a subunit and beta-1 subunit expression at their intercellular contacts. Sunardi-Widyaputra and Van Damme [46] have investigated the expression of beta-1 and beta4-integrins in pleomorphic adenomas. Beta 4-integrin was found again strongly positive on the luminal cells of duct-like structures and their basement membrane. In our study, we found strong immunoreactivity for beta4-integrin in cells of all neoplastic structures in accordance with the above studies. This finding may be associated with the abundant cellular secretion of laminin (the main beta4-integrin ligand) and depositions in stroma [22, 47,48,49]. Normally, beta4-integrin participates in junctions between cells and laminin of basement membrane via α6β4 heterodimer in hemidesmosomes. In pleomorphic adenomas the pattern of beta4-integrin expression is supporting its role in intercellular contacts and could also be associated with cell movement and migration by binding the stromal laminin, a theory that had been originally proposed by Mercurio and Rabinovitz [50].
Instead of the limited expression of HCAM (CD44s) in myoepithelial and rarely in ductal cells that observed by Xing et al. [26], many other studies have detected a more intense expression for HCAM (CD44s) and its isoforms in salivary gland tissues [27, 36, 37, 51]. Similarly, we found strong membranous immunoreactivity in acinar cells (mainly serous), which decreased in intercalated and striated ductal cells. This pattern of expression may be associated with the different hyaluronate or other matrix molecules expression, which act as ligands for HCAM (CD44s) in salivary gland tissues. Interestingly, lymphocytes and macrophages showed positive membranous staining due to the existence of abundant hyaluronate in stroma [26]. These findings suggested a dual role for CD44s, mediating in intercellular and cell–matrix interactions. In pleomorphic adenoma, the expression of CD44s was investigated by Franchi et al. [37] and Xing et al. [26]. Similarly to their findings, we observed a strong immunoreactivity of this receptor in neoplastic cells of every structure (tubular, ductal, solid, isolated cells in myxoid stroma), associated with the excessive presence of hyaluronate, osteopontin and other stromal components, which are the main HCAM (CD44s) ligands [2, 52].
In a study of Perschbacher et al. [27] a diffuse cytoplasmic pattern of staining for ICAM-1, in ductal and sporadically in serous acinar cells had been observed. On the other hand, our results, similarly to Kapsogeorgou et al. [39] and Flipo et al. [40] indicated that in normal salivary gland tissues, ICAM-1 is almost absent from both acinar and ductal epithelium, being present in endothelium, fibroblasts around the ducts and a small number of lymphocytes and monocytes. ICAM-1 had only been examined in few cases of pleomorphic adenoma with insufficient results [27]. According to our results, ICAM-1 showed a linear pattern of immunoreactivity at the luminal-apical pole of luminal cells in neoplastic duct-like structures resembling salivary ducts in sialadenitis. This finding together with ICAM-1 immunoreactivity for endothelium and lymphocytes, may indicate an activation of immunologic mechanisms by neoplastic epithelium to restrict neoplastic transformation and tumour formation. On the other hand, the presence of ICAM-1 in some neoplastic cells located in the stroma, may indicate its participation in tumour cell spreading. The pattern of ICAM-1 expression is also associated with the stromal depositions, as ICAM-1 positive stromal cells were mostly observed in myxoid rather than chondromyxoid stroma.
According to our results, the pattern of the expression of the above described CAMs represents their participation in normal salivary gland architecture and supports a distinct profile of acinar and ductal epithelial subpopulations based on their luminal or non-luminal stratification. The pattern of the examined cell adhesion molecules in pleomorphic adenomas, consisted of qualitative and quantitative alterations of Dsg-2, the unpolarised staining of laminin-receptor beta4-integrin, and the abundant presence of hyaluronate receptor HCAM (CD44s), supporting the idea that this neoplasm resembles a defect form of salivary gland embryogenesis. At this point we would like to emphasize the limitations of our study which are the limited number of other subtypes of benign salivary gland tumors as most of our specimen were pleomorphic adenoma and the use of immunohistochemistry only rather than RT-PCR. It may be hypothesised that unknown genetic or epigenetic factors may evaluate an uncontrolled overproduction of stromal components and abnormal, failed, and incomplete formation of cellular structures that consisted of modified but not entirely differentiated ductal and myoepithelial neoplastic cells, bearing CAMs with altered function. In this phase, intercellular connections are disrupted (decrease or loss of Dsg-2), other receptors help in cell motivation and migration (beta4-integrin, HCAM-CD44s and perhaps ICAM-1), connective tissue is produced and organised, and cell-stroma interactions are formed (HCAM-CD44s) in order to assist tissue growing and differentiation. Finally, the activation of immune system (with participation of ICAM-1) is trying to control and to restrict the neoplastic transformation and cell spreading, in contrast to salivary gland malignancies, in which ICAM seems to help neoplastic cell migration and invasion rather than control them [53].
References
Freemont AJ. Adhesion molecules. J Clin Pathol Mol Pathol. 1998;51:175–84.
Sneath RJ, Mangham DC. The normal structure and function of CD44 and its role in neoplasia. Mol Pathol. 1998;51:191–200.
Mishra MN, Chandavarkar V, Sharma R, Bhargava D. Structure, function and role of CD44 in neoplasia. J Oral Maxillofac Pathol. 2019;23:267–72.
Xu H, Tian Y, Yuan X, Wu H, Liu Q, Pestell RG, et al. The role of CD44 in epithelial-mesenchymal transition and cancer development. Onco Targets Ther. 2015;8:3783–92.
Jang BI, Li Y, Graham DY, Cen P. The role of CD44 in the pathogenesis, diagnosis, and therapy of gastric cancer. Gut Liver. 2011;5:397–405.
Albelda SM. Role of integrins and other cell adhesion molecules in tumour progression and metastasis. Lab Invest. 1993;68:4–17.
Wang Y, Su J, Fu D, Wang Y, Chen Y, Chen R, et al. The role of YB1 in renal cell carcinoma cell adhesion. Int J Med Sci. 2018;15:1304–11.
Piprek RP, Kolasa M, Podkowa D, Kloc M, Kubiak JZ. Cell adhesion molecules expression pattern indicates that somatic cells arbitrate gonadal sex of differentiating bipotential fetal mouse gonad. Mech Dev. 2017;147:17–27.
Ono E, Uede T. Implication of soluble forms of cell adhesion molecules in infectious disease and tumor: insights from transgenic animal models. Int J Mol Sci. 2018;19:239.
Hellquist H, Paiva-Correia A, Vander Poorten V, Quer M, Hernandez-Prera JC, Andreasen S, et al. Analysis of the clinical relevance of histological classification of benign epithelial salivary gland tumours. Adv Ther. 2019;36:1950–74.
Schäfer S, Koch PJ, Franke WW. Identification of the ubiquitous human desmoglein, Dsg2, and the expression catalogue of the desmoglein subfamily of desmosomal cadherins. Exp Cell Res. 1994;211:391–9.
Overmiller AM, Pierluissi JA, Wermuth PJ, Sauma S, Martinez-Outschoorn U, Tuluc M, et al. Desmoglein 2 modulates extracellular vesicle release from squamous cell carcinoma keratinocytes. FASEB J. 2017;31:3412–24.
Peitsch WK, Doerflinger Y, Fischer-Colbrie R, Huck V, Bauer AT, Utikal J, et al. Desmoglein 2 depletion leads to increased migration and upregulation of the chemoattractant secretoneurin in melanoma cells. PLoS ONE. 2014;9:e89491.
Lee S-H, Kim J-M, Lee DG, Lee J, Park JG, Han TS, et al. Loss of desmoglein-2 promotes gallbladder carcinoma progression and resistance to EGFR-targeted therapy through Src kinase activation. Cell Death Differ. 2020. https://doi.org/10.1038/s41418-020-00628-4.
Hsueh PY, Ju Y, Vega A, Edman MC, MacKay JA, Hamm-Alvarez SF. A multivalent ICAM-1 binding nanoparticle which inhibits ICAM-1 and LFA-1 interaction represents a new tool for the investigation of autoimmune-mediated dry eye. Int J Mol Sci. 2020;21:2758.
Gkogkou P, Peponi E, Ntaskagiannis D, Demou A, Ioakeim E, Evangelos B, et al. ICAM-1 expression in patients with advanced non-small cell lung cancer treated with radiotherapy. J Balkan U Oncol. 2020;25:1779–83.
Mohanty A, Nam A, Pozhitkov A, Yang L, Srivastava S, Nathan A, et al. A Non-genetic Mechanism Involving the Integrin β4/Paxillin Axis Contributes to Chemoresistance in Lung Cancer. iScience. 2020;23:101679.
Shen S, Lu H, Liu L, Wang Y, Zhang C, Yang W, et al. Role of CD44 in tumor-initiating cells of salivary gland pleomorphic adenoma: more than a surface biomarker. Oral Dis. 2020;26:547–57.
Harjunpää H, Llort Asens M, Guenther C, Fagerholm SC. Cell adhesion molecules and their roles and regulation in the immune and tumor microenvironment. Front Immunol. 2019;10:1078.
Dan T, Hewitt SM, Ohri N, Ly D, Soule BP, Smith SL, et al. CD44 is prognostic for overall survival in the NCI randomized trial on breast conservation with 25 year follow-up. Breast Cancer Res Treat. 2014;143:11–8.
Yan Y, Zuo X, Wei D. Concise review: emerging role of cd44 in cancer stem cells: a promising biomarker and therapeutic target. Stem Cells Transl Med. 2015;4:1033–43.
Lourenço SV, Lima DMC. Pleomorphic adenoma and adenoid cystic carcinoma: in vitro study of the impact of TGFβ1 on the expression of integrins and cytoskeleton markers of cell differentiation. Int J Exp Pathol. 2007;88:191–8.
Baker OJ, Schulz DJ, Camden JM, Liao Z, Peterson TS, Seye CI, et al. Rat parotid gland cell differentiation in three-dimensional culture. Tissue Eng Part C Methods. 2010;16:1135–44.
Schwartz LE, Begum S, Westra WH, Bishop JA. GATA3 immunohistochemical expression in salivary gland neoplasms. Head Neck Pathol. 2013;7:311–5.
Campolo-González A, Ramírez-Skinner H, Vargas-Díaz A, León-Ramírez A, Goñi- Espildora I, Solár-Gonzalez A. Perfil epidemiológico de neoplasias epiteliales de glándulas salivales. Rev Med Chil. 2018;146:1159–66.
Xing R, Regezi JA, Shuster S, Stern R. Hyaluronan and CD44 expression in minor salivary gland tumours. Oral Dis. 1998;4:241–7.
Perschbacher K, Jackson-Boeters L, Daley T. The adhesion molecules NCAM, HCAM, PECAM,-1 and ICAM-1 in normal salivary gland tissues and salivary gland malignancies. J Oral Pathol Med. 2004;33:230–6.
Lourenço SV, Kapas S, Leite K, Araújo VC. Expression patterns of integrins on pleomorphic adenoma and adenoid cystic carcinoma: study of specimens and in vitro investigation of the effects of extracellular matrix on the expression of these adhesion molecules. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;98:205.
Franchi A, Santoro R, Paglierani M, Bondi R. Comparison of integrin a chain expression in benign and malignant salivary gland tumors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1997;83:588–95.
Economopoulou P, Hanby A, Odell EW. Expression of E-cadherin, cellular differentiation and polarity in epithelial salivary neoplasms. Oral Oncol. 2000;36:515–8.
El-Naggar AK, Chan JKC, Takata T, Grandis JR, Slootweg PJ. The fourth edition of the head and neck World Health Organization blue book: editors’ perspectives. Hum Pathol. 2017;66:10-2.
Lee PS, Solitare-Sabbath M, Redondo TC, Ongcapin EH. Molecular evidence that the stromal and epithelial cells in pleomorphic adenomas of salivary gland arise from the same origin: clonal analysis using human androgen receptor gene (HUMARA) assay. Hum Pathol. 2000;31:498–503.
Zarbo RJ. Salivary gland neoplasia: a review for the practicing pathologist. Mod Pathol. 2002;15:298–323.
Ellis GL, Auclair PL. Tumours of the salivary glands. Armed Forces Institute of Pathology: Atlas of tumor pathology. Washington DC; 1996.
Loducca SVL, Mantesso A, Kapas S, Williams DM, Araújo NS, Araujo VC. Salivary gland tumours: immunoexpression of integrins beta 1, beta 3 and beta 4. Oral Pathol Med. 2003;32:305–9.
Tsunawaki S, Nakamura S, Ohyama Y, Sasaki M, Ikebe-Hiroki A, Hiraki A, et al. Possible function of salivary gland epithelial cells as non-professional antigen-presenting cells in the development of Sjögren’s syndrome. J Rheumatol. 2002;29:1884–96.
Franchi A, Moroni M, Paglierani M, Santucci M. Expression of CD44 standard and variant isoforms in parotid gland and parotid gland tumors. J Oral Pathol Med. 2001;30:564–8.
Fronseca I, Nunes MJF, Soares J. Expression of CD44 isoforms in normal salivary gland tissue: an immunohistochemical and ultrastructural study. Histochem Cell Biol. 2000;114:483–8.
Kapsogeorgou EK, Dimitriou ID, Abu-Helu RF, Moutsopoulos HM, Manoussakis MN. Activation of epithelial and myoepithelial cells in the salivary glands of patients with Sjögren’s syndrome: high expression of intercellular adhesion molecule-1 (ICAM-1) in biopsy specimens and cultured cells. Clin Exp Immunol. 2001;124:126–33.
Flipo RM, Cardon T, Copin MC, Vandecandelaere M, Duquesnoy B, Janin A. ICAM-1, E-selectin, and TNF α expression in labial salivary glands of patients with rheumatoid vasculitis. Ann Rheum Dis. 1997;56:41–4.
Franchi A, Santoro R, Paglierani M, Bondi R. Immunolocalization of α2, α5, and α6 integrin subunits in salivary tissue and adenomas of the parotid gland. J Oral Pathol Med. 1994;23:457–60.
Binmadi N, Elsissi A, Elsissi N. Expression of cell adhesion molecule CD44 in mucoepidermoid carcinoma and its association with the tumor behavior. Head Face Med. 2016;12:8.
Han C-P, Yu Y-H, Wang A-G, Tian Y, Zhang H-T, Zheng Z-M, et al. Desmoglein-2 overexpression predicts poor prognosis in hepatocellular carcinoma patients. Eur Rev Med Pharmacol Sci. 2018;22:5481–9.
Biedermann K, Vogelsang H, Becker I, Plaschke S, Siewert JR, Höfler H, et al. Desmoglein 2 is expressed abnormally rather than mutated in familial and sporadic gastric cancer. J Pathol. 2005;207:199–206.
Davis TL, Cress AE, Dalkin BL, Nagle RB. Unique expression pattern of the α6β4 integrin and laminin-5 in human prostate carcinoma. Prostate. 2001;46:240–8.
Sunardi-Widyaputra S, Van Damme B. Distribution of VLA family of integrins in normal salivary gland and in pleomorphic adenoma. Pathol Res Pr. 1994;190:600–8.
Epivatianos A, Poulopoulos A, Kayavis I, Papapanayotou P. Immunohistochemical study of basement membrane associated substances in pleomorphic adenoma. Balk J Stom. 2000;4:12–5.
Raitz R, Martins MD, Araújo VC. A study of the extracellular matrix in salivary gland tumors. J Oral Pathol Med. 2003;32:290–6.
Saku T, Cheng J, Okabe H, Koyama Z. Immunolocalization of basement membrane molecules in the stroma of salivary gland pleomorphic adenoma. J Oral Pathol Med. 1990;19:208–14.
Mercurio AM, Rabinovitz I. Towards a mechanistic understanding of tumor invasion -lessons from the alpha6beta4 integrin. Semin Cancer Biol. 2001;11:129–41.
Rito M, Fonseca I. Salivary gland neoplasms: does morphological diversity reflect tumor heterogeneity? Pathobiology. 2018;85:85–95.
Caselitz J, Schmitt P, Seifert G, Wustrow J, Schuppan D. Basal membrane associated substances in human salivary glands and salivary gland tumors. Pathol Res Pr. 1988;183:386–94.
Xue F, Zhang Y, Liu F, Jing J, Ma M. Expression of IgSF in salivary adenoid cystic carcinoma and its relationship with invasion and metatstasis. J Oral Pathol Med. 2005;34:295–7.
Acknowledgements
The authors thank the technical assistants M. Kefala and D.O. Pantartzi for their excellent collaboration.
Funding
Dr D. Andreadis was supported by a grant from the Hellenic State Scholarship Foundation.
Author information
Authors and Affiliations
Contributions
AD conception of work. PA acquisition of data. EA design of work. NA acquisition of data. PD data analysis. CK acquisition of data. BC acquisition of data. DK statistical analysis. AT table formatting, data and manuscript revision. TP revision of the manuscript. AD design of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors have consented for publication of this article.
Competing interests
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Andreadis, D., Poulopoulos, A., Epivatianos, A. et al. Cell adhesion molecules’ altered profile in benign and malignant salivary gland tumors. The paradigm of beta4-integrin, desmoglein-2, ICAM-1 and CD44s. J of Biol Res-Thessaloniki 27, 18 (2020). https://doi.org/10.1186/s40709-020-00130-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40709-020-00130-5