Abstract
Key message
Bark stripping wounds by red deer (Cervus elavus L.) were assessed on 9026 Norway spruce trees. Wound variables (length, width, area, relative width, height above ground, and angle) were analysed using generalized additive models with spatial soap film smoothers. Wounds located at the uphill side of trees were larger in summer than winter, and wound size depended on the diameter at breast height (DBH) and was spatially clustered.
Context
In Austria, red deer (Cervus elaphus L.) is the main species causing bark stripping wounds. In winter, they often gnaw at the bark because of food scarcity; in summer, large pieces of bark are detached to help digestion, water, and nutrient uptake or as social behaviour.
Aims
The aim of this study was to analyse wound size (length, width, area, relative width (i.e., width divided by stem circumference)) and wound position (height above ground, angle (i.e., deviation between wound azimuth from slope line)) for winter and summer bark stripping wounds by red deer depending on stand attributes and to describe the spatial patterns of wound size within stands.
Methods
A total of 3832 wounds on 9026 trees in nine experimental stands of Norway spruce (Picea abies (L.) Karst.) located at 47° 19’ N and 14° 46’ E at an elevation of 1009–1622 m were analysed. A linear regression model was fit for wound length over wound width for each season. For all wound variables (wound length, width, area, relative width, position, height above ground, and angle) generalized additive models (GAM) with soap film smoothers, which predict spatial patterns, were fitted.
Results
Of all wounds, 79.5 % were inflicted in winter and 20.5 % in summer. Wound length (31.9 cm ± 31.2 SD), width (11.7 cm ± 6.0 SD), area (446.5 cm2± 558.1 SD), and relative wound width (0.177 cm ± 0.098 SD) were modelled depending on summer or winter bark peeling, DBH, and tree coordinates. For wound height above ground (119.4 cm± 26.8 SD) and angle (− 1.9 ± 97.3 SD), no meaningful GAM could be calculated. Seasonal differences between wound length and area were more pronounced than for wound width; differences in height above ground were minimal, but significant. Analyses further showed that wounds were mainly located at the uphill side of the trees.
Conclusion
The spatial clustering of wound sizes might reduce the efficiency of thinning to remove heavily damaged trees in bark-peeled stands and might increase the number of sample points required to assess deer impact in forest inventories. Also, the uphill location of damages is an important information in inventories.
Similar content being viewed by others
1 Introduction
1.1 Bark stripping damage on Norway spruce (Picea abies L. Karst.)
Many European Cervidaes are known to strip bark. Bark stripping damage is reported for red deer (Cervus elavus L.), moose (Alces alces L.) (Vasiliauskas 1998; Arhipova et al. 2015), sika deer (Cervus nippon L.) (Welch et al., 1988), and fallow deer (Dama dama L.) (Gill, 1992). Roe deer (Capreolus capreolus L.) and reindeer (Rangifer tarandus L.) do not strip bark in winter, but forage small trees and grass or lichens, respectively (Gill, 1992).
The red deer is the most widespread bark stripping cervid in Europe, resident in all countries, absent only in northern Fenno-Scandia (Lovari, et al., 2019). Similarly, the moose is common, but is restricted to the northern part of Europe (Hundertmark, 2016). Sika deer and follow deer have been introduced from Japan and Turkey, respectively (European Mammal Assessment Team, 2007), and the sika deer is now resident in Great Britain, Czech Republic, and other Central European countries. Populations of all Cervidaes, native or introduced, are stable or increasing in Europe (European Mammal Assessment Team, 2007; Harris, 2015). In particular, populations of red deer (Côté et al., 2004; Burneviča et al., 2016; Candaele et al., 2021) and sika deer (Vacek et al., 2020) have been reported to increase rapidly in many areas, causing severe damage to forest stands (Čermák & Strejček, 2007; Vacek et al., 2020), with sika deer causing more severe damage than the native red deer in some areas (Vacek et al., 2020).
Moose is the largest of the Cervidaes with a shoulder height of 150–220 cm; shoulder height for red deer varies between 84 and 110 cm and is considerably smaller than that of moose. The other cervids only grow up to 100 cm (sika deer: 65–95 cm; fallow deer: 80–100 cm) (Flamm et al., 1992). Accordingly, the wound height, width, and length inflicted by different cervid species also differ significantly with wound heights above ground and sizes being higher and larger for the larger animals (Gill, 1992; Månsson and Jarnemo, 2013; Arhipova et al., 2015; Vacek et al., 2020). Bark stripping wounds by red deer were found at a height above ground of 97–110 cm (Arhipova et al., 2015), corresponding to red deer shoulder height. In scientific literature, wound direction was reported to be arbitrary (Welch et al., 1988) however, in steep terrain wounds, seem to be mostly found on the uphill side of the stem, but wound position in steep terrain has not been documented before.
Preferred species are Norway spruce (Picea abies (L.) Karst.) (Čermák et al., 2004; Vospernik, 2006; Cukor et al., 2019a, b, Vacek et al., 2020; Candaele et al., 2021), lodge pole pine (Pinus contorta Douglas ex Loudon) (Arhipova et al., 2015), Douglas fir (Pseudotsuga menziesii), (Candele et al., 2021), European ash (Fraxinus excelsior L.), (Vasiliauskas & Stenlid, 1998; Vospernik, 2006; Candaele et al., 2021), linden (Tilia spp.) (Fehér et al., 2016), maple (Acer spp.) (Féher et al., 2016), sweet chestnut (Castanea sativa Mill.) (Vospernik, 2006), and Sorbus spp. (Vospernik, 2006), whereas thick-barked species are often avoided (Gill, 1992; Vospernik, 2006; Candaele et al., 2021). Open wounds exposed 4–4355 cm2 of sapwood (Čermák & Strejček, 2007; Arhipova et al., 2015), and repeated assessment or dendrochronological analysis showed that the same tree was debarked several times (Gill, 1992; Arhipova et al., 2015; White, 2019; Nagaike, 2020), with an average of 1.8–2 wounds per tree (Arhipova et al., 2015). Wounds were inflicted in stands aged 8–10 to 40–60 years (Čermák et al., 2004; Fehér et al., 2016; Cukor et al., 2019a, b; Vacek et al., 2020; Candaele et al., 2021) depending on site productivity and deer abundance (Candaele et al., 2021), with the age of vulnerability corresponding to mean diameters of 6–24 cm for thin-barked species (Vasiliauskas & Stenlid, 1998; Arhipova et al., 2015; Vacek et al., 2020) and a considerably shorter period of sensitivity and smaller diameters for thick-barked species (Gill, 1992; Vospernik, 2006; Candaele et al., 2021).Wound size increases with diameter at breast height (DBH) (Arhipova et al., 2015; Vacek et al., 2020), and wounds inflicted in summer are reported to be larger than winter wounds (Månsson & Jarnemo, 2013; Candaele et al., 2021).
Food scarcity in winter is thought to be the main reason for winter bark stripping (Gill, 1992) The phloem of trees provides a nutrient-rich food that lacks toxins and feeding deterrents, and bark stripping often occurs in late winter and early spring, when the phloem is more nutrient rich (White, 2019). Reasons for summer bark peeling are less clear. Summer bark peeling has a beneficial effect for the digestion in the rumen (Månsson & Jarnemo, 2013) and has also been related to water scarcity (König, 1968). The two types of wounds can be easily distinguished, since winter bark stripping wounds show teeth marks. In winter, the bark is firmly attached to the stem, whereas in summer, the bark is more loosely attached, and thus, large stripes of bark can be removed from the trees (Gill, 1992; Candaele et al., 2021).
Bark stripping greatly damages saplings and trees by destroying water conductivity and increasing fungal infection (Nagaike, 2020; Vacek et al., 2020). Wounds with more than 65% of stem circumference resulted in increased tree mortality (Nagaike, 2020), and recent studies have also evidenced a significantly decreased productivity and increased sensitivity of stripped trees to drought and heatwaves (Cukor et al., 2019a, b; Vacek et al., 2020). Another important consequence of bark stripping is fungal infection and subsequent wood decay (Vasiliauskas, 1998; Vasiliauskas & Stenlid, 1998; Čermák et al., 2004; Čermák & Strejček, 2007; Butin, 2011; Vasaitis et al., 2012; Rönnberg et al., 2013; Arhipova et al., 2015; Burneviča et al., 2016).
Infection spreads vertically along the tree (Arhipova et al., 2015; Burneviča et al., 2016) and is linearly correlated with wound length, whereas the spread of wound decay beyond the wound margin is limited (Vasiliauskas & Stenlid, 1998; Mäkinen et al., 2007; Arhipova et al., 2015). Decay in the Norway spruce was reported to range from 2.7–62.5 cm·year−1, finally reaching an extent of 1.5–6 m (Čermák et al., 2004; Čermák & Strejček, 2007; Rönnberg et al., 2013; Vacek, et al., 2020) depending on tree age and the age of the wounds (Čermák & Strejček, 2007). Further, decay was reported to increase with site fertility (Vasaitis et al., 2012; Burneviča et al., 2016). Even though wound sizes for different tree species vary a little, pathogenic consequences vary between species, since fungi colonizing wounds are tree species specific (Mäkinen et al., 2007; Metzler et al., 2012) and have different spread rates (Vasiliauskas, 1998; Mäkinen et al., 2007; Metzler et al., 2012).
For example, Mäkinen et al. (2007) isolated different fungi from the Norway spruce (Picea abies (L.) Karst.) and Scots pine (Pinus sylvestris L.) and Metzler et al. (2012) from the Norway spruce and silver fir (Abies alba Mill.). In the Scots pine, a lower proportion of trees were decayed than in the Norway spruce (Mäkinen et al., 2007). Similarly, infection rate for the Norway spruce with pathogens was 4.6 times higher than for silver fir (Metzler et al., 2012). Fungal infection and limited water uptake further increase stand sensitivity to wind and snow damage (Vasiliauskas, 1998; Čermák et al., 2004; Čermák & Strejček, 2007; Burneviča et al., 2016; Snepsts et al., 2020). Damaged stands had a 1.68 times higher probability for wind damage (Snepsts et al., 2020), and pulling experiments evidenced a lower resistance of bark-stripped trees to pulling force (Krisans et al., 2020). However, contrary to expectations, most trees in the pulling experiment were uprooted, and only one bark-stripped tree broke at the wound (Krisans et al., 2020).
In Austria, bark stripping by red deer prevails, whereas wounds inflicted by other cervids are rare (Völk, 1997). The tree species most affected by bark stripping are the Norway spruce, European ash (Fraxinus excelsior L.), sweet chestnut (Castanea sativa Mill.), and Sorbus spp. (Vospernik, 2006). Of these tree species, bark stripping damage of the Norway spruce is considered most important, because of its high economic importance for Austrian forestry, since it constitutes 60.4% of the standing timber volume (BFW, 2021). While the Austrian National Forest Inventory (BFW, 2021) records the presence/absence of bark stripping wounds, wound size, position, and the spatial distribution of damage within stands is not assessed. In particular, within stand spatial patterns have been little investigated in detail before, although bark stripping damage has been reported to be clustered (Gill, 1992).
Further, bark stripping is more common in areas with higher agriculture (Månsson & Jarnemo, 2013; Sun et al., 2020) and increases with the distance to forest roads (Kiffner et al., 2008). Moreover, a substantial increase in damage is found around diversionary feeding stations, constructed to protect forestry and natural habitats (Putman & Staines, 2004).
Deer animals and bark stripping wounds are a frequent source of debate between wildlife managers and forest managers (Reimoser & Gossow, 1996). On the one hand, bark stripping causes important economic damage and seriously devaluates timber (Čermák et al., 2004; Čermák & Strejček, 2007; Vacek et al., 2020; Candaele et al., 2021), and forest managers have a high interest in controlling red deer populations (Kiffner et al., 2008; Candele et al., 2021). On the other hand, deer animals are a part of the forest ecosystem and are highly valued for hunting and aesthetic reasons (Ehrhart et al., 2022). An objective assessment of bark stripping damage is the basis to settle these differences. For damage assessment, between-stand and within-stand variation of bark stripping wounds is important. Wound size is an important factor for the economic evaluation since the decay extent is closely linked to it (Arhipova et al., 2015; Vacek et al., 2020).
1.2 Hypotheses and research questions analysed in this paper
The aim of this study was to analyse wound size, position, and its spatial patterns. Specifically, the following hypotheses were analysed:
-
Wound size is larger in summer than in winter.
-
Winter bark stripping is more common than summer bark stripping.
-
Wounds are larger on larger trees.
-
Wounds are located at the uphill side of the trees.
-
Wound height is animal specific and is therefore at a constant height above ground.
-
Wound sizes are larger on trees close to winter feedings and forest roads.
-
Wound sizes are spatially clustered.
2 Methods
2.1 Study area
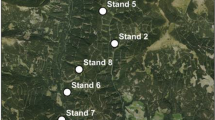
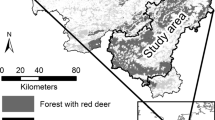
The study was conducted in Gaal/Austria (Fig 1). Precise stand coordinates are given in Table 1. The study area is an area of a high red deer population density; the average number of hunted animals is 1.21 deer per 100 ha of forestland (Reimoser & Reimoser, 2019). The measurements were collected in nine stands, located on moderately steep slopes (40 to 50%) at an elevation of 1009–1622 m (Table 1). Stands were selected based on a visually estimated damage rate of the stands (three stands with a low, medium, and high bark-peeling rate, respectively). However, the following precise assessment of the stands rather showed a continuum of bark stripping rates than three distinct classes. Trees grow on Cambisols on silicate bedrock (quarz, granite, and feldspar). Soils are classified as moist or fresh (Kilian et al., 1994; FAO, 2015). The study area is classified as a warm, summer continental climate acording to Köppen (Wikipedia, 2022). At the nearest climate station in Seckau (Lat = 47.2°, Lon = 15.1°, 863 m above sea level), an annual mean temperature of 6.6 °C and a mean annual precipitation of 817 mm were recorded from 1981 to 2010. The coldest month is January with a mean temperature of − 3.4 °C, and the warmest month is July with a mean temperature of 16.6 °C. In Seckau, there are 141 frost days and 92 days with snow cover (> 1 cm) of which 24 days have a snow cover of more than 20 cm (ZAMG, 2021). Stands are almost totally composed of Norway spruce with admixtures of European larch (Larix decidua Mill.), and broad-leaved trees represent less than 5% of the total basal area, except in stand 8 with a larch proportion of 15%.
2.2 Data collection and design
In a first step, each stand was scanned with a laser scanner (“FARO Focus3D X330”—Faro Technologies Inc., Lake Mary, FL, USA). The scans were done in multiscan mode with the resolution parameter set to 1/4, resulting in a resolution of r = 6.136 mm/10 m, and the quality parameter set to 4×, resulting in a measuring time of 8 μs per scan point. Scans were co-registered, and tree coordinates were automatically derived from the point clouds using an algorithm (Ritter et al., 2017; Gollob et al., 2019; Gollob et al., 2020). From the laser scans, detailed digital elevation models (DTM) were obtained for each of the nine stands.
Subsequent field assessment encompassed verification of calculated coordinates and diameter at breast height (DBH) and wound characteristics were measured on each tree. In total, 9026 trees with 3832 bark stripping wounds were assessed. For each tree, DBH and tree species were noted, and all wounds were searched for and recorded. Wounds were classified as winter or summer bark stripping damage, and for each damage, the following measurements were taken (Fig. 2), and the following calculations were done:
-
Wound length defined as the maximum vertical extent
-
Wound width defined as the maximal horizontal extent
-
Wound height defined as the distance of the centre of the wound to the ground
-
Wound azimuth was measured in degrees
-
Wound area was calculated as wound length times wound width (Eq. 1)
-
Wound angle was calculated as the difference between the azimuth and the local slope line derived from a 1-m resolution digital elevation model (DTM) from the laser scan (Eq. 2)
-
Relative wound width as the quotient of the absolute wound width and the circumference of the tree (Eqs. 3and 4)
with Area Calculated area of the bark stripping wound (cm2)
Length Length of the bark stripping wound (cm)
Width Width of the bark stripping wound (cm)
Angle Calculated difference between the wound azimuth and the local slope line (°)
Az Azimuth of the bark stripping wound (°) (Fig. 2)
SL Local slope line (°) calculated from the laser scan data
CF Circumference (cm) of the tree
DBH Diameter at breast height (cm) of the tree
π Mathematical constant
Rel. width Relative width of the wound (-)
2.3 Statistical analysis and modelling of wound sizes with general additive models
Generalized additive models (Hastie & Tibshirani, 1990; Wood, 2017) are flexible models where the dependent variable is modelled by smooth functions of the independent variables. For fitting the models, the r-package mgcv (Wood, 2020) and, for model diagnostics, the r-package mgcViz (Fasiolo et al., 2021) were used. The mgcv-package allows for the inclusion of two-dimensional smoothers, parametric terms, the specification of different distribution families as well as the specification of different link functions.
The generalized additive models (GAM) equation in general is given in Eq. 5:
with β0 Intercept
βi Coefficient for the ith parametric term
xi ith paramtric term
s(yi) Smooth function for the jth variable (yj)
In this research, the gamma distribution and the log link function were used to relate the damage length, width, and area and relative wound width (wound width divided by stem circumference) to the predictive variables. Categorial variables such as summer or winter bark peeling and tree species were included as parametric terms. In addition, the nine stands were included as a random effect, but the random effect was not significant.
All variables, except the tree coordinates, were modeled usin g thin plate regression splines, which are the default smooth function in the mgcv-package with 10 knots. If the default number of knots was appropriate, it was verified with the mgcViz-package (function check), which computes a k-index for each smooth term. A k-index of less than one indicates that the number of knots is too low. The mgcViz-package provides also an automatic model diagnostic plot for each model. This plot contains 4 sub-plots: (i) a quantile–quantile (Q-Q) plot, where the theoretical quantiles are calculated from Monte Carlo simulation and plotted against the observed quantiles, (ii) the histogram of the residuals, (iii) the plot of residuals versus the linear predictor, and (iv) the plot of response versus fitted values (Augustin et al., 2012).
Numerous covariates and covariate combinations were tested for each dependent variable, and the resulting models were ranked according to the adjusted coefficient of determination (adj. r2) and the Akaike information criterion (AIC) (Akaike, 1973). For each dependent variable, the five highest ranked models were considered in detail, and biological plausibility was checked. The highest ranked model for wound height and angle (using also the gamma distribution and the log link function) did not explain more then 3.0% and 10.7% of the total variation, respectively, and therefore, modelling of these characteristics was not pursued. For the other variables (wound length, width, area, and relative width), final model choice was a model formulation that was consistent across all four size variables because the differences in AIC (Akaike, 1973) and adjusted r2 between the five best ranked models were minimal.
For the spatial component, we used the soap film smoother (Wood et al., 2008), which is a finite-area smoother using boundary polygons. In our case, spatial wound size distribution was smoothed within each of the nine stands using the stand boundary line as constraint for the smooth.
Finally, the following model was chosen for the four wound size variables (Eq. 6):
with Length Length of the bark stripping wound (cm)
Width Width of the bark stripping wound (cm)
Area Calculated area of the wound (cm2)
Rel. width Relative width of the wound (-)
β0 Intercept
β1 Coefficient for the parametric term w
w Parametric term; w = 1 for winter damage; w = 0 for summer damage
s(xLambert, yLambert, bs = ‘so’) Soap film smooth (bs = ‘so’) for the spatial distribution of wounds using the Lambert coordinates of the trees with wounds
s(DBH) Smoother for DBH (cm). The number of knots (k) was set to k = 50 in response to the results of the r-function check
The coefficients (β0 and β1) and the smooth functions for the coordinates and the DBH were modeled for length, width, area and relative width separately.
3 Results
3.1 Descriptive statistics and t test
In total, 3832 wounds were measured with an average length of 31.9 cm and an average width of 11.7 cm, resulting in an average wound area of 446.5 cm2; the wounds have an average relative width of 0.177. The average height of the wound centre is 119.4 cm above ground, and wounds were located on the uphill side of the trees with very little deviation from the uphill slope line as indicated by the angle (Table 2). Between stands, variations in length, width, height, area, and angle were minor. In particular, between stand differences in wound height and angle were very small (Table 2).
The difference between the azimuth of damages and the local slope line was tested with a paired Student’s t test. The difference between the two azimuths was not significant (t value = − 0.982; p value 0.326), confirming that damages were located on the uphill side of the trees. Figure 3 shows the histogram of observed angles, peaking at angle = 0.
Winter damage accounted for 79.5% and summer bark stripping damage accounted for 20.5% of all damages observed. Differences in wound height above ground between summer and winter (winter: 119.0 cm; summer: 119.8 cm) were minimal but significant because of the large sample size. Similarly, differences in wound width, length, and area between winter and summer bark peeling damage were significant. Wound width (winter: 11.5 cm; summer: 14.5 cm) differed less than wound length (winter: 26.7 cm; summer: 53.0 cm) or wound area (winter: 360.4 cm2, summer: 850.3 cm2). The distribution of wound height above ground, wound width, length, and area by season is shown in the boxplots in Fig. 4, and the relationship between wound length and wound width for both seasons are shown in Fig. 5. For the same wound width, summer bark peeling damage has a longer wound length as is shown by the different regression lines (Fig. 5), explaining 29% and 30% of the variation in wound length.
3.2 Modelling with GAM
A detailed analysis revealed that wound length, width, area, and relative wound width depended on season, Lambert coordinates, and the DBH of the trees; model statistics are given in Table 3. The Q–Q plots created by gam.check are given in Fig. 6.
Residual diagnostic plots for the final models for wound length (cm), width (cm), area (cm2), and relative width (width divided by stem circumference) created by the R-function “gam.check” of the package mgcViz (Augustin et al., 2012)
Wound length, width, and area all initially increased with DBH; they had a maximum at a DBH of approximately 35 cm, and then gradually decreased for larger trees. Relative wound width decreased with DBH. Wounds inflicted on trees in summer were considerably larger than wounds formed in winter. In Fig. 7, this information is plotted for stand 1. This relationship was constant across stands; therefore, plots for the other stands are not shown.
Predicted wound length (cm), width (cm), area (cm2) and relative wound width, the 95% confidence interval over diameter at breast height (DBH (cm)), and stem circumference (CF (cm)) for the trees of stand No. 1 for winter and summer bark stripping. Tree coordinates are fixed at the median values of the stand. 1/8: threshold for no damage; 1/3: threshold for severe damage defined by Cukor et al. (2019); 65%: threshold for increased mortality defined by Nagaike (2020)
Wound length, width, area, and relative wound width varied spatially (Figs. 8, 9, 10, and 11). Spatial patterns for all four variables were similar, but were more pronounced for wound length and area than for wound width and relative wound width. Spatial patterns could be related to the presence of supplementary feedings in two of the nine stands (stand 2: Feeding in the Southeast; stand 3: Feeding in the Northeast). These 2 stands showed a decreasing gradient in damage size with distance to the supplementary feedings. The other four stands also showed considerable spatial variation of damage, but without any clear explanation for it.
4 Discussion
4.1 Wound position and wound size
In practical forestry, it is common knowledge that in steep terrain, the bark peeling wounds are located on the uphill side of the stem. This side is more accessible for red deer because the local terrain is flatter than on the downhill side, and the animals are able to reach higher parts of the tree with thinner bark, which they prefer. This behaviour was almost invariable in our study: in contrast, on flat terrain, no dependence of wound occurrence on direction was observed (Welch et al., 1988). It remains unclear, at which slope these two contrasting patterns switch. This interesting fact, which is related to animal behaviour, has also important consequences for the monitoring of bark stripping damages. If the wounds are exclusively located at one side, this has to be accounted for by the sampling design.
Wound height above ground is specific for different cervid species, and in studies where multiple bark stripping cervids are present, it is used to attribute damage to a specific cervid species (Månsson & Jarnemo, 2013). The certain identification of the causing animal species from wound height above ground is, however, difficult (Månsson & Jarnemo, 2013, Fehér et al., 2016). Wound height observed in this study (Table 2) is closely related to reported heights above ground inflicted by red deer from previous studies (Welch et al., 1988; Arhipova et al., 2015; Burneviča et al., 2016).
Studies on bark stripping of the Norway spruce by red deer report a damage width between 6 cm (Welch et al., 1988) and 10 cm (Burneviča et al., 2016) and a damage length between 12 cm (Welch et al., 1988) and 33 cm (Burneviča et al., 2016) resulting in a wounded area between 200 cm2 (Månsson & Jarnemo, 2013) and 589 cm2 (Burneviča et al., 2016). In our study, the wound width observed is within the range reported in the literature but rather on the upper side of it (Table 2). Moreover, these wound sizes inflicted by red deer on Norway spruce were also similar to those found on other tree species (Welch et al., 1988; Vasiliauskas & Stenlid 1998; Arhipova et al., 2015), so that wound size is rather animal specific than a tree species characteristic. Similarly, Welch et al. (1988) also found no differences in wound size between tree species.
In our study, wound length and wound width were measured in the field, whereas wound area was calculated as rectangular area (wound width × wound length). Wound area was calculated in the same way by Vasiliauskas & Stenlid (1998). In contrast, Arhipova et al. (2015) measured wound size directly in the field by drawing the wound area on a transparent paper and measuring the area in the laboratory with a digital planimeter. This procedure gives more precise area measurements, but was too cost-intensive in this study for the large sample size. Possibly, wound area could be determined from the laser scans, but respective classification algorithms are not yet developed. The wound area calculated in this study, however, overestimates actual wound size.
Studies reporting relative wound size report means of approximately 30% (Cukor et al., 2019a, b; Vacek et al., 2020). Our model shows that percentages of more than 1/3 are mostly observed for smaller trees. A percentage of 65% defined as threshold for increased tree mortality by Nagaike (2020) is seldom observed in our study and mostly occurs on very small trees. Thus, we do not expect a considerably higher mortality due to bark peeling in our study area, but we expect an increased vulnerability to drought and wind. An increased vulnerability to drought was reported by Cukor et al. (2019a, b) and Vacek et al. (2020) and is probably due to the wounding of the cambium, and we speculate that this is more closely linked to damage width which differs little between summer and winter wounds. An increased vulnerability to wind was reported by Snepsts et al. (2020) and Krisans et al. (2020) and is very probable in our study area since seven stands are located in areas which are highly prone to wind damage.
Wound size clearly differed between winter and summer bark stripping damage. This is also reported in other studies (Månsson & Jarnemo, 2013; Candaele et al., 2021). In our study, the presence of teeth marks was used to distinguish between winter and summer bark stripping. In winter, the bark is difficult to detach and is gnawed of by red deer leaving teeth marks on the stems. With the rehydration of stems in spring, the bark becomes loosely attached to the tree and can be easily peeled off by red deer resulting in larger wounds. This differentiation between the two types of wounds was in some cases ambiguous in our study, in particular for old wounds, but many wounds could be unambiguously classified. Moreover, assessment of damage type might differ between different field teams; this was however not verified in this study. Further, identification of winter and summer wounds would be more certain, if wounds were assessed yearly, as was done in the study of Candaele et al. (2021).
Interestingly, season of wounding is reported in few wound size studies, possibly because in central Europe winter bark peeling is prevalent (Völk 1997; Månsson and Jarnemo 2013). The percentage of summer bark stripping is 20.5% in our study, which is comparable with other studies (Welch et al., 1988; Månsson and Jarnemo, 2013; Candaele et al., 2021), but percentages may vary regionally. For example, Månsson and Jarnemo (2013) reported a summer bark peeling percentage of 26–28% in Southern Sweden and 1–6% in Northern Sweden.
Wounds in general (bark stripping, logging, rockfall, etc.) are entry gates for fungal colonisation (Vasiliauskas, 1998; Vasiliauskas & Stenlid, 1998). Wound infection is linearly related to wound length (Vasiliauskas & Stenlid, 1998; Arhipova et al., 2015), and the decay colon increases with the age of wounds (Čermák & Strejček, 2007). Thus, decay extent in our study will be larger for wounds inflicted in summer. Fungi isolated from the wounds are tree species specific (Vasiliauskas & Stenlid, 1998; Metzler et al., 2012; Arhipova et al., 2015). On the Norway spruce, Stereum sanguinolentum (Alb. & Schwein.) Fr. is the most common basidiomycete on infected wounds (Butin, 2011). This fungus is a fast-spreading species (Čermák & Strejček, 2007), and, therefore, the Norway spruce is more affected than other tree species (Mäkinen et al., 2007; Metzler et al., 2012). Increment losses for individual trees in case of fungal infection are small for harvesting damage (Mäkinen et al., 2007), but higher increment losses are reported for bark stripping damage (Cukor et al., 2019a, b). Nevertheless, the wood devaluation through decay causes much larger economic losses. For instance, Čermák & Strejček (2007) found that the amount of decayed wood accounted for 22–70% (mean 42 %) of the merchantable stem volume.
4.2 Modelling with generalized additive models
Preferences of red deer for particular stands depend on habitat structure, silvicultural systems, and ungulate dynamics. Red deer require not only food but also hiding cover and protection from climate (Reimoser & Gossow, 1996). As a result, large between-stand variations in the presence of deer and bark stripping damage have been reported in numerous studies (Gill, 1992; Vospernik, 2006; Kiffner et al., 2008; Månsson & Jarnemo, 2013). Also, within stands, there are clear dependencies of the occurrence of bark stripping dependent on a number of tree-specific variables (DBH, local density, local slope) (Hahn C, Vospernik S, Gollob C, Ritter T: Bark stripping damage by red deer (Cervus elaphus L.): assessing the spatial distribution on the stand level using generalized additive models, submitted). There are considerably less independent variables explaining differences in wound length, width, area, and relative wound width. As discussed above, the season of wounding is a decisive factor. In addition, only DBH and the spatial distribution are explanatory variables for size variables.
For presence/absence of bark peeling of trees, DBH is an important factor, and the presence of wounds often decreases with DBH because small trees have a more digestible bark (Gill, 1992; Kiffner et al., 2008). Similarly, studies on wound size report a smaller DBH of wounded trees than for healthy trees (Cukor et al., 2019a, b; Vacek et al., 2020).
In contrast, wound size itself initially increases with DBH. Obviously, larger wounds can only be inflicted on large-diameter trees. The decrease in older trees (DBH ~ 35 cm) might be due to wound healing and the removal of the most heavily damaged trees during the thinning of the stands. Probably the latter factor is more important since the Norway spruce has a notoriously slow wound healing rate (Vasaitis et al., 2012).
Both presence and absence of bark stripping (Hahn C, Vospernik S, Gollob C, Ritter T: Bark stripping damage by red deer (Cervus elaphus L.): assessing the spatial distribution on the stand level using generalized additive models, submitted) and wound size are spatially aggregated. In our study, these patterns can mainly be related to the vicinity of disturbances and forage (feeding sites) and local preferences for hiding cover. Red deer are sensitive animals and avoid disturbances and are usually not found close to forest roads (Kiffner et al., 2008) (Hahn C, Vospernik S, Gollob C, Ritter T: Bark stripping damage by red deer (Cervus elaphus L.): assessing the spatial distribution on the stand level using generalized additive models, submitted) or human activities (Kiffner et al., 2008). The spatial patterns revealed in this study for wound size, however, do not show a clear dependence on the proximity to forest roads.
Furthermore, the location of forest stands close to winter feedings has been reported as an important factor for bark peeling damage (Putman and Staines, 2004; Kiffner et al. 2008; Sun et al. 2020) (Hahn C, Vospernik S, Gollob C, Ritter T: Bark stripping damage by red deer (Cervus elaphus L.): assessing the spatial distribution on the stand level using generalized additive models, submitted). The animals aggregate close to the feeding stations at high densities, causing severe damage in the vicinity. Usually, these effects are reported for the presence/absence of bark peeling damage. In our study, a similar effect has been observed for wound size. So not only do the occurrence of wounds increase in the vicinity of feeding stations but also the wound size. This may be due to repeated debarking of the same tree, which has been reported in other studies (Gill, 1992; Arhipova et al., 2015; White, 2019; Nagaike, 2020). In the field assessement, it might not be possible to distinguish this multiple debarking damages, and they may thus be recorded as larger wound size. Because of the high damage rates close to supplementary feedings, it has been much debated in forestry practice and in the scientific literature if supplementary feedings are an appropriate measure to reduce environmental damage (e.g. see Putman & Staines (2004)).
Local preferences of red deer seem to be an important factor in our study, and patterns of wound size clearly differ spatially, with average wound size differences of several centimetres. Such aggregated patterns have been reported for the occurrence of damage (Gill, 1992) and are also found for wound size. It remains however unclear what the causal factors are. Factors that might explain spatial patterns could be hunting regime or population and foraging properties or winter snow cover. The first two factors are however rather homogenous in the investigating stands; for the latter factor, the within-stand variation is not known on a local scale for the studied stands. The multitude of possible influencing factors and their impact has been reviewed by Gill (1992) and more recently by Gerhardt et al. (2013).
5 Conclusion
This study has important implications for silviculture of bark stripping damaged stands and the assessment of bark stripping in forest inventories.
-
The high rates of bark stripping in stands and the large wound sizes found in this study in conjunction with results from other studies that report that the spread of rot is linearly related to wound length suggest high economic losses in the investigated stands and in similar stands in Austria. For the pole stands already stripped, heavy thinning interventions and a short rotation of stands might help to reduce economic losses by reducing damage rates by selectively removing the most damaged trees. Thinning might however not be an efficient tool to reduce damage rates, when damages are clustered, which is shown in this study. Further, it remains unclear what thinning rate to select. Conventional thinning in Austria removes 10–20% of the standing volume, and thinning rates of more than 30% in Norway spruce would result in stand growth reductions. Detailed knowledge of the spatial distribution of damage and damage sizes forms the basis for detailed growth and yield scenario analysis to manage bark-stripped stands; also, information on growth reductions in damaged trees would need to be included.
-
To avoid game damage in young stands, reducing the cover of forest stands through increased spacing and admixtures of deciduous species might be an important strategy. An important and probably contributing within-stand factor that is hard to assess is the distribution of within-stand snow cover, which might cause spatial aggregation of red deer in winter.
-
The high economic losses through bark stripping and the following infection with wood destroying fungi cause conflicts between forest managers and hunters. An effective and objective damage assessment can therefore provide important information to settle disagreements. Because of the high costs of forest inventories, the design should also be efficient for the detection of bark stripping damages. Our study showed that the damages are not only clustered between stands but also within stands, which needs to be accounted for in forest inventory designs. The knowledge that wounds are mainly found on the uphill side of the stems also needs to be incorporated in forest inventories.
References
Akaike H (1973) Information theory as an extension of the maximum likelihood principle. In: Petrov BN, Csaksi F (eds) Second international symposium on information theory. Akademiai Kiado, Budapest, pp 267–281
Arhipova N, Jansons A, Zaļuma A, Gaitnieks T, Vasaitis R (2015) Bark stripping of Pinus contorta caused by moose and deer: wounding patterns, discoloration of wood and associated fungi. Can J Forest Res 45(10):1434–1438. https://doi.org/10.1139/cjfr-2015-0119
Augustin NH, Sauleaub EA, Wood SN (2012) On quantile qantile plots for generalized linear models. Comput Stat Data Analysis 56(8):2404–3409. https://doi.org/10.1016/j.csda.2012.01.026
BFW. (2021) Zwischenauswertung der ÖWI 2016/18 – Bund. https://www.bfw.gv.at/wp-content/uploads/Bundesergebnisse_OEWI_16_18.pdf [26.08.2021]
Burneviča N, Jansons Ā, Zaļuma A, Kļaviņa D, Jansons J, Gaitnieks T (2016) Fungi inhabiting bark stripping wounds made by large game on stems of Picea abies (L.) Karst. in Latvia. Baltic Forestry 22(1):2–7
Butin H (2011) Krankheiten der Wald- und Parkbäume: Diagnose-Biologie-Bekämpfung, 4. Auflage edn. Eugen Ulmer, Stuttgart (Hohenheim)
Candaele R, Lejeune P, Licoppe A, Malengreaux C, Brostaux Y, Morelle K, Latte N (2021) Mitigation of bark stripping on spruce: the need for red deer population control. Eur J Forest Res 140(1):227–240. https://doi-1org-10006d0fj1a6e.pisces.boku.ac.at/10.1007/s10342-020-01326-z
Čermák P, Jankovský L, Glogar J (2004) Progress of spreading Stereum sanguinolentum (Alb. et Schw.: Fr.) Fr. wound rot and its impact on the stability of spruce stands. J Forest Sci 50(8):360–365
Čermák P, Strejček M (2007) Stem decay by Stereum sanguinolentum after red deer damage in the Českomoravská vrchovina Highland. J Forest Sci 53(12):567–572
Côté SD, Rooney TP, Tremblay JP, Dussault C, Waller DM (2004) Ecological impacts of deer overabundance. Annu. Rev. Ecol. Evol. Syst. 35(1):113–147. https://doi.org/10.1146/annurev.ecolsys.35.021103.105725
Cukor J, Vacek Z, Linda R, Sharma RP (2019a) Afforested farmland vs. forestland: effects of bark stripping by Cervus elaphus and climate on production potential and structure of Picea abies forests. PLoS ONE 14(8):e80221082
Cukor J, Vacek Z, Linda R, Vacek S, Marada P, Šimůnek V, Havránek F (2019b) Effects of bark stripping on timber production and structure of Norway spruce forests in relation to climatic factors. Forests 10:320. https://doi.org/10.3390/f10040320
Ehrhart S, Stühlinger M, Schraml U (2022) The relationship of stakeholders' social identities and wildlife value orientations with attitudes toward red deer management. Human Dimension Wildlife 27(1):69–83. https://doi.org/10.1080/10871209.2021.1885767
European Mammal Assessment Team. (2007). The IUCN red list of threatened species: e.T42188A10656222. (03.08.2021).
FAO (2015). World reference base for soil resources 2014: international soil classification system for naming soils and creating legends for soil maps. Update 2015. Rome.
Fasiolo M, Nedellec R, Goude Y, Capezza C, & Wood SN (2021) mgcViz: visualisations for generalized additive models. Version 0.1.8. Website: https://cran.r-project.org/web/packages/mgcViz/mgcViz.pdf (26.08.2021).
Fehér Á, Szemethy L, Katona K (2016) Selective debarking by ungulates in temperate deciduous forests: preference towards tree species and stem girth. Eur J of Forest Res. 135(6):1131–1143. https://doi.org/10.1007/s10342-016-1000-9
Flamm J, Flasch RF, Hager E, Kutzer E, Leitner WC, Nussbaumer J, Sternath M (1992) Der neue Jagdprüfungsbehelf, 12. Auflage edn. Österreichischer Jagd- und Fischereiverlag, Wien
Gerhardt P, Arnold JM, Hackländer K, Hochbichler E (2013) Determinants of deer impact in European forests - a systematic literature analysis. Forest Ecol Manag 310:173–186. https://doi.org/10.1016/j.foreco.2013.08.030
Gill RM (1992) A review of damage by mammals: 1. Deer. Forestry 65(2):145–169. https://doi.org/10.1093/forestry/65.2.145
Gollob C, Ritter T, Nothdurft A (2020) Forest inventory with long range and high-speed personal laser scanning (PLS) and simultaneous localization and mapping (SLAM) technology. Remote Sensing 12(9):1509. https://doi.org/10.3390/rs12091509
Gollob C, Ritter T, Wassermann C, Nothdurft A (2019) Influence of scanner position and plot size on the accuracy of tree detection and diameter estimation using terrestrial laser scanning on forest inventory plots. Remote Sensing 11(13):1602. https://doi.org/10.3390/rs11131602
Harris RB (2015) Cervus nippon. The IUCN Red List of Threatened Species. https://doi.org/10.2305/IUCN.UK.2015-2.RLTS.T41788A22155877.en (03.08.2021).
Hastie TJ, Tibshirani RJ (1990) Generalized additive models. Chapman & Hall / CRC, Boca Raton, New York, Washington, D.C.
Hundertmark K (2016) Alces alces. The IUCN Red List of Threatened Species: e.T56003281A22157381. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T56003281A22157381.en (03.08.2021).
Kiffner C, Rößiger E, Trisl O, Schulz R, Rühe F (2008) Probability of recent bark stripping damage by red deer (Cervus elaphus) on Norway spruce (Picea abies) in a low mountain range in Germany - a preliminary analysis. Silva Fennica 42(1):125–134. https://doi.org/10.14214/sf.269
Kilian W, Müller F, Starlinger F (1994) Die forstlichen Wuchsgebiete Österreichs: Eine Naturraumgliederung nach waldökologischen Grundlagen. Forstliche Bundesversuchsanstalt, Wien
König E (1968) Der Einfluss des jahresperiodischen Verlaufs des Wasser- und Zuckergehaltes einiger Baumarten auf das Schälen durch Rotwild (Cervus elaphus L.). Doctoral thesis. University Freiburg, Freiburg
Krisans O, Saleniece R, Rust S, Elferts D, Kapostins R, Jansons Ā, Matisons R (2020) Effects of bark-stripping on mechanical stability of Norway spruce. Forests 11(3):357. https://doi.org/10.3390/f11030357
Lovari S, Lorenzini R, Masseti M, Pereladova O, Carden RF, Brook SM, & Mattioli (2019) The IUCN red list of threatened species 2018: e.T55997072A142404453. https://doi.org/10.2305/IUCN.UK.2018-2.RLTS.T55997072A142404453.en. (03.08.2021).
Mäkinen H, Hallaksela M, Isomäki A (2007) Increment and decay in Norway spruce and Scots pine after artificial logging damage. Can J Forest Res 73(11):2130–2141. https://doi.org/10.1139/X07-087
Månsson J, Jarnemo A (2013) Bark-stripping on Norway spruce by red deer in Sweden: level of damage and relation to tree characteristics. Scand J Forest Res 28(2):117–125. https://doi.org/10.1080/02827581.2012.701323
Metzler B, Hecht U, Nill M, Brüchert F, Fink S, Kohnle U (2012) Comparing Norway spruce and silver fir regarding impact of bark wounds. Forest Ecol Manag 274:99–107. https://doi.org/10.1016/j.foreco.2012.02.016
Nagaike T (2020) Effects of heavy, repeated bark stripping by Cervus neppon on survival of Abies vietchii in a subalpine coniferous forest in central Japan. J Forestry Res 31(4):1139–1135. https://doi.org/10.1007/s11676-019-00940-x
Putman RJ, Staines BW (2004) Supplementory winter feeding of wild red deer Cervus elaphus in Europe and North America: justifications, feeding practice and effectiveness. Mammal Rev 34(4):258–306. https://doi.org/10.1111/j.1365-2907.2004.00044.x
Reimoser F, Gossow H (1996) Impact of ungulates on forest vegetation and its dependence on the silvicultural system. Forest Ecol Manag 88(1-2):107–119. https://doi.org/10.1016/S0378-1127(96)03816-9
Reimoser F, & Reimoser S (2019). Das Rotwild (Cervus elaphus) in Österreich. 25. Österreichische Jägertagung:5-12.
Ritter T, Schwarz M, Tockner A, Leisch F, Nothdurft A (2017) Automatic mapping of forest stands based on three-dimensional point clouds derived from terrestrial laser-scanning. Forests 8(8):265. https://doi.org/10.3390/f8080265
Rönnberg J, Berglund M, Johansson U, Clear M (2013) Incidence of Heterobasidion spp. following different thinning regimes in Norway spruce in southern Sweden. Forest Ecol Manag 289:409–415. https://doi.org/10.1016/j.foreco.2012.10.013
Snepsts G, Kitenberga M, Elferts D, Donis J, Jansons A (2020) Stem damage modifies the impact of wind on Norway spruces. Forests 11(4):463. https://doi.org/10.3390/f11040463
Sun Y, Yu Y, Guo J, Zhang M (2020) The winter habitat selection of red deer (Cervus elaphus) based on a multi-scale model. Animals 10(2454). https://doi.org/10.3390/ani10122454
Vacek Z, Cukor J, Linda R, Vacek S, Šimůnek V, Brichta J, Prokůpková A (2020) Bark stripping, the crucial factor affecting stem rot development and timber production of Norway spruce forests in Central Europe. Forest Ecol Manag 474:118360. https://doi.org/10.1016/j.foreco.2020.118360
Vasaitis R, Lygis V, Vailiauskaite I, Valiliauskas A (2012) Wound occlusion and decay in Picea abies. Eur J Forest Res 131(4):1211–1216. https://doi.org/10.1007/s10342-011-0592-3
Vasiliauskas R (1998) Patterns of wounding and decay in stems of Quercus robur due to bark peeling. Scand J Forest Res 13(1-4):437–441. https://doi.org/10.1080/02827589809383004
Vasiliauskas R, Stenlid J (1998) Discoloration following bark stripping wounds on Frasinus excelsior. Eur J Forest Pathology 28(6):383–390. https://doi.org/10.1111/j.1439-0329.1998.tb01192.x
Völk F (1997) Schälschäden und Rotwildmanagement in Abhängigkeit von Jagdgesetz und Waldbau in Österreich. Doctoral thesis, Universität für Bodenkultur.
Vospernik S (2006) Probability of bark stripping damage by red deer (Cervus elaphus) in Austria. Silva Fennica 40(4):589–601. https://doi.org/10.14214/sf.316
Welch D, Staines BW, Scott D, Catt DC (1988) Bark stripping damage by red deer in a Sitka spruce forest in western Scotland: II. Wound size and position. Forestry:245–254. https://doi.org/10.1093/forestry/61.3.245
White T (2019) The cauce of bark stripping of young plantation trees. Ann Forest Sci 76(4):105. https://doi.org/10.1007/s13595-019-0888-x
Wikipedia. (2022). Köppen climate classification. https://en.wikipedia.org/wiki/K%C3%B6ppen_climate_classification (10.01.2022)
Wood SN (2017) Generalized additive models: an introduction with R, 2nd edn. CRC Press, Boca Raton, London, New York. https://doi.org/10.1201/9781315370279
Wood SN (2020). mgcv: mixed GAM computation vehicle with automatic smoothness estimation. Version 1.8-33. Website: https://cran.r-project.org/web/packages/mgcv/mgcv.pdf (30.09.2020).
Wood SN, Bravington MV, Hedley SL (2008) Soap film smoothing. J Royal Stat Soc B Stat Method 70(5):931–955. https://doi.org/10.1111/j.1467-9868.2008.00665.x
ZAMG. (2021). Climate station "Sekau". downloaded 20.12.2021 https://www.zamg.ac.at/cms/de/klima/messnetze
Acknowledgements
We are thankful to the forest company Wasserberg/Stift Heiligenkreuz, and in particular to P. Cœlestin Klemens Nebel OCist. for the opportunity to make the measurements for our study on their sites and for their support. We thank Ralf Krassnitzer, Franz Gollob, and Philipp Waltl for the careful fieldwork.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Author information
Authors and Affiliations
Contributions
Conceptualization was done by CH and SV. Methodology was done by CH and SV. Formal analysis was done by CH, SV. Data curation was done by CH and SV. Writing—original draft was done by CH and SV. Writing—review & editing was done by CH and SV. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The authors declare that they follow the rules of good scientific practice.
Consent for publication
All authors gave their informed consent to this publication and its content.
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Marco Ferretti
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Hahn, C., Vospernik, S. Position, size, and spatial patterns of bark stripping wounds inflicted by red deer (Cervus elavus L.) on Norway spruce using generalized additive models in Austria. Annals of Forest Science 79, 13 (2022). https://doi.org/10.1186/s13595-022-01134-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13595-022-01134-y