Abstract
Background
Like other viral infections, severe acute respiratory syndrome coronavirus-2 infection could affect different human body systems, including host immune responses. Three years after its pandemic, we learn more about this novel coronavirus. As we expected, different co-infections with various organisms, such as viruses, bacteria, and even fungi, have been reported. However, concurrent infection with two severe acute respiratory syndrome coronavirus-2 strains and cytomegalovirus is extremely unusual. We have only a rudimentary understanding of such co-infections and their long-term consequences for patients with cancer.
Case presentation
An 18-year-old young Iranian adult with acute lymphoblastic leukemia presented with abdominal pain, diarrhea, nausea, and vomiting following a recent history of severe acute respiratory syndrome coronavirus-2 infection. The patient never experienced respiratory symptoms, and the chest imaging study was normal on admission. His primary laboratory investigation revealed prerenal azotemia and severe abnormal liver function tests (blood urea nitrogen 32 mg/dL, creatinine 1.75 mg/dL, prothrombin time 66 s, partial thromboplastin time 44.5 s, international normalized ratio 5.14, total bilirubin 2.9 mg/dL, and direct bilirubin 2.59 mg/dL). Cytomegalovirus disease was diagnosed by polymerase chain reaction in his blood and stool samples. The patient’s gastrointestinal signs and symptoms improved shortly after receiving intravenous ganciclovir treatment. His gastrointestinal symptoms continued intermittently for weeks despite maintenance valganciclovir prescription, necessitating frequent hospitalizations. The patient was complicated by the recurrence of gastrointestinal symptoms during the sixth hospitalization, even though he had no respiratory symptoms, and the nasopharyngeal test revealed severe acute respiratory syndrome coronavirus-2 Wuhan strain for the first time. Remdesivir and valganciclovir were administrated due to persistent enteritis and evidence of intestinal tissue invasion by severe acute respiratory syndrome coronavirus 2 and cytomegalovirus on multiple intestinal biopsies, which led to partial clinical responses. Cytomegalovirus and severe acute respiratory syndrome coronavirus-2 fecal shedding continued for more than 6 months despite repeated antiviral therapy, and the Wuhan and Alpha strains were also detected in his nasopharyngeal samples through repeated sampling (confirmed by four nasopharyngeal sampling and multiple stool specimens and several intestinal biopsies). Finally, during the Delta-variant (B.1.617.2) outbreak in Iran, the patient was admitted again with febrile neutropenia and decreased level of consciousness, necessitating respiratory support and mechanical ventilation. During the Delta-variant peak, the patient’s nasopharyngeal sample once more tested positive for severe acute respiratory syndrome coronavirus 2. The patient died a few days later from cardiopulmonary arrest.
Conclusion
The coronavirus disease 2019 pandemic has encountered patients with cancer with critical diagnostic and treatment challenges. Patients who are immunocompromised may co-infect with multiple severe acute respiratory syndrome coronavirus-2 strains and cytomegalovirus, and even with timely diagnosis and treatment, the prognosis may be poor.
Similar content being viewed by others
Introduction
Over time, since the onset of the coronavirus disease 2019 (COVID-19) pandemic, our knowledge regarding pathogenesis, different clinical aspects, diagnosis, treatment, and the various severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) complications has improved. Gastrointestinal (GI) involvement is now one of the well-known presentations of SARS-CoV-2 infection [1,2,3,4,5]. In addition to the common GI symptoms such as nausea, vomiting, diarrhea, and abdominal pain, other GI complications have been described, such as acute hepatitis [6], hemorrhagic colitis [7], appendicitis [8,9,10], pancreatitis [11,12,13], and dysentery [14]. GI involvement could be associated with prolonged viral shedding, and fecal specimens may remain positive for SARS-CoV-2 even 2 weeks after the nasopharynx clearance, especially in those with diarrhea [15, 16]. GI symptoms could be continued for up to 3 weeks [16, 17], while infected patients may have asymptomatic fecal shedding for a more extended period, even more than 8 weeks [15,16,17,18]. The clinical significance of prolonged fecal excretion of the virus is not well understood; however, affected patients may be implicated in viral transmission [18,19,20]. There are few reports on SARS-CoV-2 and cytomegalovirus (CMV) co-infection in the literature [21,22,23,24]. Our current knowledge need to improve due to the significant complication of such co-infections [23]. The odds of fatal COVID-19 outcomes are higher among immunocompromised patients, even those receiving primary vaccination [25]. Although, co-infection of SARS-CoV-2 with more common seasonal viral infections such influenza viruses, respiratory syncytial virus, or adenoviruses has been thoroughly investigated [26], more research should be done on less frequent viral co-infections. This report aims to represent a young adult male with acute lymphoblastic leukemia (ALL) who developed CMV enteritis concomitant with SARS-CoV-2 Alpha variant (B.1.1.7) and original wild-type (Wuhan strain) SARS-CoV-2 co-infection. We also discuss possible mechanisms of CMV reactivation during SARS-CoV-2 infection.
Case presentation
Clinical history
On 27 December 2020, an 18-year-old young Iranian adult with a history of ALL who had been undergoing maintenance chemotherapy for more than 18 months and was in remission was admitted to our hospital owing to abdominal pain, diarrhea, nausea, and vomiting. He has a history of contracting SARS-CoV-2 infection 1 month before admission and subsequently repeated hospitalization. The patient never developed respiratory symptoms and did not fulfill the criteria for remdesivir treatment on first-time detection of SARS-CoV-2 via reverse transcription polymerase chain reaction (RT–PCR) test. The patient chemotherapy regimen consists of monthly vincristine (1.5 mg/m2), prednisolone (40 mg/m2) 5 days per month, weekly methotrexate (10 mg/m2), and 6-mercaptopurine (50 mg) every night. He also received trimethoprim/sulfamethoxazole 400/80 mg twice per day/three times per week for Pneumocystis jirovecii prophylaxis and voriconazole (VCZ) 200 mg twice per day concurrent with his chemotherapy. He has a history of invasive pulmonary aspergillosis and pan-sinusitis (8 and 4 weeks before admission, respectively). He was hospitalized with severe GI signs and symptoms in another academic hospital (leading to intensive care unit admission) without clinical improvement, leading to self-discharge. On admission, he was febrile (tympanic temperature 38.8 °C). He suffered from nausea/vomiting, abdominal pain, diarrhea, oral aphthous lesions, skin rashes, excessive drooling, low appetite, weight loss, and decreased urine output. The results of his primary lab testing are summarized in Table 1.
On the basis of presenting signs and symptoms, severe abnormal liver function tests, and prerenal azotemia, we considered several differential diagnoses, including sepsis, drug–drug interactions or toxicities [methotrexate (MTX), vincristine (VNC), VCZ], SARS-CoV-2-induced gastroenteritis, biliary obstruction, portal venous thrombosis secondary to SARS-CoV-2 infection or VNC-induced veno-occlusive disease (VOD), and, finally, transfusion-associated graft-versus-host disease (TA-GVHD), given the history of the repeated transfusion (especially non-irradiated blood transfusion). Accordingly, he was worked up for drug–drug interactions. No significant interaction was found except VCZ/VNC. VCZ was withheld and VCZ serum levels were checked for three consecutive days, and all were within therapeutic or subtherapeutic ranges, 0.329 µg/mL, 0.241 µg/mL, and 0.526 µg/mL, respectively. MTX serum level also was checked, which was not in the toxic range (< 0.1 µmol/L), CYP2C19A polymorphism was also checked, which was compatible with homozygote CYP2C19*17 alleles (ultrarapid metabolizer phenotype). Serum galactomannan was negative on two occasions [0.3 and 0.2 optical density index (ODI)]. Given the prolonged prothrombin time (PT), partial thromboplastin time (PTT), and international normalized ratio (INR), a mixing test was requested. The mixing test corrected PT, PTT, and INR values in about 2 h. Factor assay was also performed, which showed decreased factor II and X levels, with acceptable factor V levels. On abdominopelvic ultrasonographic (US) study, the liver was normal in size with diffusely increased parenchymal echogenicity compatible with fatty liver change grade 2. Color Doppler sonography (CDS) of mesenteric and portal veins was done to investigate chemotherapy-induced VOD, which was normal.
When leukemia was diagnosed, the patient’s primary investigations revealed that he was CMV seropositive. Accordingly, a CMV blood PCR test was performed to check for CMV reactivation. The high viral load [98,500 copies/mL; cycle threshold (CT) 29] was suggestive of a significant CMV viremia. Stool was assessed for CMV by PCR because of hepatic impairment (abnormal liver function tests) and GI signs and symptoms. A fecal sample showed extremely high viral shedding (295,480,000 copies/mL; CT: 25). So, the patient was investigated for the possibility of disseminated CMV disease, including CMV encephalitis, despite having no central nervous system (CNS) signs or symptoms due to concurrent CMV viremia and CMV enteritis. CMV was not detected by PCR in CSF and urine specimens. We also investigated SARS-CoV-2 nasopharyngeal and stool PCR.
SARS-CoV-2 nasopharyngeal and stool RT–PCR also were positive (CT 31 and 24, respectively) for the Alpha strain. B.1.1.7 (Alpha), B.1.351 (Beta), and P1 (Gamma) variant’s single-nucleotide deletions were investigated for SARS-CoV-2 by a multiplex PCR using type-specific probes described by Vogels et al. (Additional file 1, Molecular Analysis for SARS-CoV-2 Variants Identification) [27].
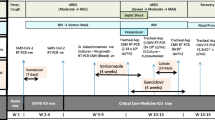
A summary of viral marker test results is presented in Table 1. The patient’s clinical course timeline and key clinical events during repeated hospitalization are shown in Fig. 1.
The results of his virologic investigation are summarized in Table 2. Figure 2 also illustrates the patient’s SARS-CoV-2 status compared with the national data of transmission peaks.
The patient’s SARS-CoV-2 PCR status compared with the national data of peaks of transmission in Iran. Data extracted from the WHO website last updated on 30 August 2021 (available at https://covid19.who.int/region/emro/country/ir) and Iran Ministry of Health and Medical Education website (available at https://behdasht.gov.ir/). The Alpha variant (B.1.1.7) was first sequenced in September 2020 in the UK and subsequently spread globally (https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/959438/Technical_Briefing_VOC_SH_NJL2_SH2.pdf)
The patient was treated with intravenous ganciclovir (5 mg/kg/dose Q12 hours as a loading dose) on 29 December 2020 once blood and stool CMV PCR confirmed CMV reactivation. Intragastrointestinal CMV replication was suppressed significantly after 1 week of ganciclovir treatment (stool PCR 20,800 copies/mL with CT 31.5). Blood viral load also decreased gradually up to the low level of detection (97,850, 1200, and 560 copies/mL) on repeated testing. Stool CMV PCR was negative (nondetectable) on follow-up testing in the sixth and seventh weeks of treatment. The patient’s treatment course was continued with oral valganciclovir 450 mg Q12 hours for 4 weeks. He had, however, experienced intermittent GI symptoms for several weeks following that, leading to recurrent hospitalizations. He received a short course of intravenous rehydration therapy (3–7 days) and supplementary electrolytes for correcting hypokalemia and hyponatremia in each admission. On 21 April 2021, a diagnostic rectosigmoidoscopy and colonoscopy were carried out, and several biopsies from the right colon, terminal ileum, and rectum were taken. No macroscopic or microscopic abnormality was detected during the operation and immunohistopathologic evaluation. It should be noted that diagnostic rectosigmoidoscopy and colonoscopy were postponed several times due to severe coagulopathy, profound neutropenia, and thrombocytopenia, in addition to the patient’s self-discharge events from the hospital.
CMV was detected by PCR in all tissue samples. SARS-CoV-2 Alpha variant (B.1.1.7) and original wild-type SARS-CoV-2 were also found in all three specimens. No specific CMV-cytopathic changes were reported on immunohistopathologic examination (Fig. 3a–d). The SARS-CoV-2 nasopharyngeal test was also positive for the Wuhan strain (CT 28). On 8 May 2021, the patient was treated with intravenous remdesivir (200 mg as a loading dose and 100 mg daily for 4 days) despite having no respiratory signs or symptoms. All GI manifestations resolved a few days after antiviral treatment, and he was encouraged to undergo close follow-up monitoring.
a Low-power view of the terminal ileum with normal villi without ileitis [hematoxylin and eosin (H&E) ×40]; b higher-power view of the same picture in Fig. 1. No inflammation or endothelial abnormality is seen (H&E ×200); c low-power view of colon mucosa biopsy shows normal-looking gland and surface epithelium. No colitis is present. A benign-looking lymphoid nodule is seen (H&E ×40); d higher-power view of the colon mucosa. No colitis is present. No abnormality of endothelium is seen, which is not indicative of CMV infection. Only scattered plasma cells and lymphocytes are present
Outcome and follow-up
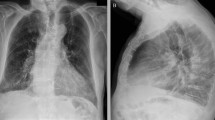
Finally, the patient was admitted again on 27 July, with febrile neutropenia and decreased level of consciousness necessitating respiratory support and mechanical ventilation. The patient’s nasopharyngeal specimen tested positive again, and blood and stool CMV PCR also were positive (30,500 and 19,700 copies/mL, respectively). The patient’s chest X-rays revealed bilateral multifocal parenchymal involvement (Fig. 4). The patient died after cardiopulmonary arrest on 27 July 2021.
We also perform immunological workups during the two periods of patient admissions, as shown in Fig. 5 (including the last two admissions where the second was 1 day before the patient died). The patient experienced severe neutropenia in both periods. Analysis of the serum cytokines revealed a similar pattern on two occasions; however, a substantial increase in IL-6 was found during the last admission (2593.5 versus 38 pg/mL) 1 day before the patient died. At this time, IL-17A and IL-17F decreased significantly to the nondetectable values. Analysis of the last lymphocyte immunophenotyping showed that the frequency of CD3+ CD4+ T cells was high (92%), whereas the levels of CD3− CD16+ CD56+ lymphocytes (natural killer cells) as well as B lymphocytes (CD3− CD19+ CD20+) were low (2% and 6%, respectively).
a, b The graphs depict variations in the white blood cell counts and immunological factors in the last two periods of patient hospitalization (period 1 and period 2, respectively). Period 2 represents the day before the patient died. a White blood cell counts during the last 3 days of each hospitalization period; b the patient’s cytokine assay during period 1 and period 2
Discussion
Here, we discuss a case with ALL that also had co-infection with CMV and two SARS-CoV-2 strains. The patient’s last bone marrow aspiration confirmed that he was in remission. The patient did not have other convincing reasons for the CMV reactivation, including the disease relapse, protocol change, anticancer agent dose intensification, or even nonadherence with chemotherapy treatment. On the other hand, CMV reactivation signs and symptoms (hepatic and gastrointestinal involvement) begin to develop immediately after COVID-19 infection. The patient was treated with ganciclovir due to CMV viremia and CMV enteritis even though he did not match the requirements for remdesivir treatment in the first detection of the SARS-CoV-2 RT-PCR test because he never developed respiratory symptoms. A nasopharyngeal test for the SARS-CoV-2 Wuhan strain showed up positive on the fifth admission. The patient was treated with remdesivir and valganciclovir for persistent enteritis and evidence of SARS-CoV-2 and CMV invasion of the intestinal tissue.
SARS-CoV-2 PCR re-positivity and re-infection
Aside from the facts concerning the patient’s clinical condition during repeated admissions, there are some important points regarding interpreting the patient’s repeated positive viral markers.
A positive PCR test within 3 months of past COVID-19 infection in an asymptomatic individual with a CT value higher than 35 is usually considered a PCR re-positivity, and there is no need for respiratory isolation [28, 29]. However, all CT values were lower than 35 in our case during more than 6 months of follow-up (Table 2), which could be explained by the patient’s immunosuppressed status [30]. Accordingly, we considered respiratory precautions and isolation in his ambulatory visits and admissions. On the basis of available guidelines, those experiencing atypical clinical courses and having a history of exposure during outbreaks are suspected of re-infection [28]. So, according to prolonged PCR re-positivity with low CT values and also due to his immunosuppressed status, he was suspected of contracting a new mutant SARS-CoV-2 strain, and he was investigated for other variants of concern (VOCs), including B.1.351 (Beta) and P1 (Gamma).
Interestingly, the SARS-CoV-2 Wuhan strain was detected in the patient’s pharyngeal sample. The re-infection with SARS-CoV-2 Wuhan strain (CT value 28) is particularly intriguing, given that the patient’s pharyngeal specimen was positive about 16 days earlier with Alpha strain (CT value 21). It is unusual and unreasonable to document a new SARS-CoV-2 strain while the patient has recently had a positive pharyngeal test with another SARS-CoV-2 strain. It is worth noting that, unlike some bacterial strains, such as Haemophilus influenza, which can have pharyngeal colonization for months, documentation of viral infection (such as the flu) in the upper respiratory tract is evidence of acute infection. The simultaneous presence of Wuhan and Alpha SARS-CoV-2 strains (all CT values lower than 35; Table 2), in addition to CMV in the patient’s intestinal tissue samples, is also a thought-provoking finding in our patient.
SARS-CoV-2 GI involvement
Our patient suffers from prolonged SARS-CoV-2 shedding confirmed by consecutive positive PCR results from his nasopharyngeal and stool specimens for about 6 months. Although prolonged viral shedding is a well-known phenomenon in immunocompromised hosts after COVID-19 infection, it has never been reported for such a long time [31, 32]. It should be noticed that the prolonged SARS-CoV-2 shedding in cancer patients is associated with poor prognosis and increased cancer-associated death and comorbidities [33], as we described in this report.
GI, hepatic, hypercoagulability state, and prerenal azotemia successfully improved within a few days after treatment. Similar clinical presentation of COVID-19 was our main challenge; however, as we discussed here, CMV reactivation is the most probable cause of multiorgan involvement in this case. The possible CMV reactivation mechanisms are also briefly discussed here on the basis of current data regarding CMV reactivation in critically ill patients.
Angiotensin-converting enzyme 2 (ACE2) is highly expressed in the lung cells, esophagus epithelial cells, and absorptive enterocytes of the ileum and colon [1]. As a result, GI involvement could occur during SARS-CoV-2 infection and worsen the patient’s outcome [15]. Diarrhea, nausea/vomiting, and abdominal pain are the most common signs and symptoms [16]. GI involvement may be the sole manifestation of the SARS-CoV-2 infection in about 20% of affected individuals [15]. The most common reported lab findings are elevated liver enzymes (mostly aspartate transaminase), elevated alkaline phosphatase, bilirubin, and abnormal PT values [34]. The median time for GI symptom presentation is about 4 days after infection, and may last for up to 2 weeks [34], although researchers found a shorter course in those with milder COVID phenotypes [15]. Nonspecific, moderate microvesicular steatosis, mild mixed lobular, focal/portal necrosis, and portal lymphocyte infiltration suggestive of hepatic vascular involvement could be seen on liver histology [35]. Fecal SARS-CoV-2 shedding may occur in about half of cases [34], which usually lasts longer than nasopharyngeal viral shedding (about 2 weeks or longer) [36]. GI manifestations rarely continued for more than 3 weeks [16]. Altogether, based on the natural history of SARS-CoV-2 infection, intermittent fecal viral shedding, PCR CT values higher than 30 in fecal specimens (in all tested samples), and the too late presentation, the patient’s signs and symptoms could not be attributed to COVID-19.
Possible CMV reactivation mechanisms in the context of SARS-CoV-2 infection
Understanding the likely mechanism of CMV reactivation in SARS-CoV-2 infection is also a debatable issue with considerable importance.
CMV is a genetically diverse large virus with more than 200 open reading frames producing effector proteins, about one-quarter of them committed to CMV replication. It may potentially alter innate and adaptive host immune responses [37,38,39,40]. There is very little information regarding CMV reactivation mechanisms in the literature. Immediate early (IE) CMV gene, which is expressed by TNF-α stimulation, sepsis-induced immunosuppression, decreased IFN-γ T-cell responses, natural killer (NK) dysfunction, and impaired dendritic cell function all could be attributed to CMV reactivation after sepsis from a bacterial origin [41,42,43,44,45,46]. The mechanisms of CMV reactivation during viral infections are much less known. However, elevated TNF-α and cyclic-AMP resulting from pro-inflammatory prostaglandins and stress catecholamine stimulation may play a role [47]. Finally, the role of T-regulatory/T-helper 17 (Treg/Th17) imbalance has been described previously in the pathogenesis of cytomegalovirus reactivation [48].
On the basis of previous discussions, reactivation of cytomegalovirus may occur through previously known mechanisms in the context of COVID-19 infection. Gastrointestinal endothelial cells could be affected indirectly by lung infection due to immune regulation of the “gut–lung axis” in the absence of detectable SARS-CoV-2 in the stool of patients with COVID-19. Following SARS-CoV-2 lung infection, CD4+ T cells infiltrate the infected site and express CCR9 upon stimulation. After that, lung-derived CCR9+ CD4+ T cells migrate by the systemic circulation to the small intestine where epithelial cells express CCL25. Interaction between CCR9 and CCL25 is essential for recruiting effector T cells to the intestine [49]. Upon infiltration of CCR9+ CD4+ T cells, a disturbance occurs in the intestine flora, affecting the balance between Th17 and regulatory T cells, leading to the increased polarization of Th17 and, consequently, more IL-17A production. IL-17A recruits neutrophils into the intestine, which can cause intestinal immune damage [50]. As discussed earlier, Treg/Th17 balance is essential in the pathogenesis of cytomegalovirus infection. During CMV reactivation/replication, decreased Treg/Th17 ratio could be observed with decreased Treg-related factors and increased Th17-related factors (Fig. 6), while in latent infection Treg/Th17 ratio usually increased secondary to robust Treg responses [48]. Thus, mediators of host immune responses could be affected due to the SARS-CoV-2 leading to the CMV reactivation. However, the hypothesis will require additional investigation through thoughtful in vitro experiments.
A few considerations should be considered regarding the justification of the negative cytopathic effect in the histopathologic examination. First, sample biopsies may be taken from the unaffected areas. Second, the likelihood of identifying a cytopathic effect in a critically symptomatic patient with severe diarrhea may be minimal due to the significant mucosal shedding of the injured endothelium. Finally, previous effective antiviral treatment may playing an important role. Despite the absence of the expected cytopathic effect, the extremely high fecal viral load shows extensive GI tract involvement when pathological investigation results are inconclusive. Besides, high-level CMV fecal shedding during CMV viremia without invasive intestinal disease may occur in immunocompromised patients as described previously [51, 52].
Cytokine and biomarker investigation during CMV and SARS-CoV-2 co-infection in critically ill patient with cancer
We observed that, in contrast to the reduced leukocyte count, the levels of IL-6, which is essential for Th17 differentiation [53], and IL-22 produced by Th22 and Th17 [54] increased significantly in the final stages; however, IL-17A and IL-17F secreted by Th17 had the lowest levels. Regarding T-cell exhaustion in viral infections such as CMV and SARS-CoV-2 [55, 56], it seems that, besides leukopenia, higher IL-6 levels may also affect T-cell exhaustion and secretion of IL-17 by Th17 cells. So, the Treg/Th17 balance is disturbed at the terminal stages of the CMV/SARS-CoV-2 co-infection. To confirm bone marrow (BM) remission during the last admission, the patient underwent a trephine bone marrow aspiration/biopsy once more.
CMV co-infection may be fatal in high-risk patients such as transplant recipients [23]. Prolonged intermittent GI symptoms in leukemic individuals during the COVID-19 pandemic may mimic more severe and prevalent GI illnesses such as GVHD or irritable bowel syndrome (IBS), especially in the setting of CMV co-infection.
Conclusion
Patients with cancer have encountered critical diagnostic and treatment challenges as a result of the COVID-19 pandemic. Human immune responses may be affected by SARS-CoV-2/CMV co-infection. Such reports may help us with current virus–virus interactions during the COVID-19 pandemic. However, further research is needed to confirm CMV reactivation/replication mechanisms in the context of SARS-CoV-2 infection.
Availability of data and materials
All data generated or analyzed during this study are included in this article.
Abbreviations
- SARS-CoV-2:
-
Severe acute respiratory syndrome coronavirus-2
- CMV:
-
Cytomegalovirus
- ALL:
-
Acute lymphoblastic leukemia
- PCR:
-
Polymerase chain reaction
- GI:
-
Gastrointestinal
- COVID-19:
-
Coronavirus disease 2019
- MTX:
-
Methotrexate
- VNC:
-
Vincristine
- VCZ:
-
Voriconazole
- aGVHD:
-
Acute graft-versus-host disease
- VOD:
-
Veno-occlusive disease
- ODI:
-
Optical density index
- PT:
-
Prothrombin time
- PTT:
-
Partial thromboplastin time
- INR:
-
International normalized ratio
- CSF:
-
Cerebrospinal fluid
- CT:
-
Cycle threshold
- US:
-
Ultrasonographic
- CDS:
-
Color Doppler sonography
- ACE2:
-
Angiotensin-converting enzyme 2
- NK:
-
Natural killer
- Treg:
-
T-regulatory
- Th17:
-
T-helper 17
References
Xiao F, et al. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology. 2020;158(6):1831–3.
Pan L, et al. Clinical characteristics of COVID-19 patients with digestive symptoms in Hubei, China: a descriptive, cross-sectional, multicenter study. Am J Gastroenterol. 2020;115:766.
Lamers MM, et al. SARS-CoV-2 productively infects human gut enterocytes. Science. 2020;369(6499):50–4.
Gupta A, et al. Extrapulmonary manifestations of COVID-19. Nat Med. 2020;26(7):1017–32.
Wong SH, Lui RN, Sung JJ. Covid-19 and the digestive system. J Gastroenterol Hepatol. 2020;35(5):744–8.
Wander P, Epstein M, Bernstein D. COVID-19 presenting as acute hepatitis. Am J Gastroenterol. 2020;115:941.
Carvalho A, et al. SARS-CoV-2 gastrointestinal infection causing hemorrhagic colitis: implications for detection and transmission of COVID-19 disease. Am J Gastroenterol. 2020;115:942.
Prichard C, et al. Case series of acute appendicitis association with SARS-CoV-2 infection. BMC Infect Dis. 2021;21(1):1–4.
Meyer JS, et al. Acute appendicitis in four children with SARS-CoV-2 infection. J Pediatr Surg Case Rep. 2021;64: 101734.
Ahmad S, et al. SARS-CoV-2 isolation from an appendix. J Surg Case Rep. 2020;2020(8): rjaa245.
Kandasamy S. An unusual presentation of COVID-19: acute pancreatitis. Ann Hepatobiliary Pancreat Surg. 2020;24(4):539.
Al Mazrouei SS, Saeed GA, Al Helali AA. COVID-19-associated acute pancreatitis: a rare cause of acute abdomen. Radiol Case Rep. 2020;15(9):1601–3.
Aloysius MM, et al. COVID-19 presenting as acute pancreatitis. Pancreatology. 2020;20(5):1026–7.
Tariverdi M, et al. Dysentery as the only presentation of COVID-19 in a child: a case report. J Med Case Rep. 2021;15(1):1–3.
Han C, et al. Digestive symptoms in COVID-19 patients with mild disease severity: clinical presentation, stool viral RNA testing, and outcomes. Am J Gastroenterol. 2020;115:923.
Cheung KS, et al. Gastrointestinal manifestations of SARS-CoV-2 infection and virus load in fecal samples from a Hong Kong cohort: systematic review and meta-analysis. Gastroenterology. 2020;159(1):81–95.
Gupta S, et al. Persistent viral shedding of SARS-CoV-2 in faeces—a rapid review. Colorectal Dis. 2020;22(6):611–20.
Zapor M. Persistent detection and infectious potential of SARS-CoV-2 virus in clinical specimens from COVID-19 patients. Viruses. 2020;12(12):1384.
Gu J, Han B, Wang J. COVID-19: gastrointestinal manifestations and potential fecal–oral transmission. Gastroenterology. 2020;158(6):1518–9.
Tian Y, et al. Gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51(9):843–51.
Gozzi-Silva SC, et al. SARS-CoV-2 infection and CMV dissemination in transplant recipients as a treatment for Chagas cardiomyopathy: a case report. Trop Med Infect Dis. 2021;6(1):22.
Amaral PH, et al. COVID-19 and cytomegalovirus co-infection: a challenging case of a critically ill patient with gastrointestinal symptoms. Eur J Case Rep Intern Med. 2020;7(10):001911.
Molaei H, et al. Iranian kidney transplant recipients with COVID-19 infection: clinical outcomes and cytomegalovirus coinfection. Transpl Infect Dis. 2021;23(1): e13455.
Zhu X, et al. Co-infection with respiratory pathogens among COVID-2019 cases. Virus Res. 2020;285: 198005.
Yek C, et al. Risk factors for severe COVID-19 outcomes among persons aged ≥ 18 years who completed a primary COVID-19 vaccination series—465 health care facilities, United States, December 2020–October 2021. Morb Mortal Wkly Rep. 2022;71(1):19.
Swets MC, et al. SARS-CoV-2 co-infection with influenza viruses, respiratory syncytial virus, or adenoviruses. Lancet. 2022;399(10334):1463–4.
Vogels C, Fauver J, Grubaugh N. Multiplexed RT-qPCR to screen for SARS-COV-2 B. 1.1. 7. B. 1: p. 1.
Yahav D, et al. Definitions for coronavirus disease 2019 reinfection, relapse and PCR re-positivity. Clin Microbiol Infect. 2021;27(3):315–8.
Kim J-M, et al. Viral shedding among re-positive severe acute respiratory syndrome coronavirus-2 positive individuals in Republic of Korea. Viruses. 2021;13(10):2089.
Singanayagam A, et al. Duration of infectiousness and correlation with RT-PCR cycle threshold values in cases of COVID-19, England, January to May 2020. Eurosurveillance. 2020;25(32):2001483.
Nakajima Y, et al. Prolonged viral shedding of SARS-CoV-2 in an immunocompromised patient. J Infect Chemother. 2021;27(2):387–9.
Rogado J, et al. Prolonged SARS-CoV-2 viral shedding in patients with solid tumors and associated factors. Eur J Cancer. 2021;148:58.
GoubetAG, et al. Prolonged SARS-CoV-2 RNA virus shedding and lymphopenia are hallmarks of COVID-19 in cancer patients with poor prognosis. medRxiv, 2021.
Mao R, et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5(7):667–78.
Fan Z, Chen L, Li J. Clinical features of COVID-19-related liver damage. medRxiv. 2020;27. Epub ahead of print
Elshazli RM, et al. Gastroenterology manifestations and COVID-19 outcomes: a meta-analysis of 25,252 cohorts among the first and second waves. J Med Virol. 2021;93:2740.
Murphy E, Shenk TE. Human cytomegalovirus genome. Human cytomegalovirus, 2008: p. 1–19.
Murphy E, et al. Coding potential of laboratory and clinical strains of human cytomegalovirus. Proc Natl Acad Sci USA. 2003;100(25):14976–81.
Dunn W, et al. Functional profiling of a human cytomegalovirus genome. Proc Natl Acad Sci USA. 2003;100(24):14223–8.
Miller-Kittrell M, Sparer TE. Feeling manipulated: cytomegalovirus immune manipulation. Virol J. 2009;6(1):1–20.
Papazian L, et al. Cytomegalovirus reactivation in ICU patients. Intensive Care Med. 2016;42(1):28–37.
Döcke W, et al. Cytomegalovirus reactivation and tumour necrosis factor. Lancet. 1994;343(8892):268–9.
Hummel M, Abecassis MM. A model for reactivation of CMV from latency. J Clin Virol. 2002;25:123–36.
von Müller L, et al. Cellular immunity and active human cytomegalovirus infection in patients with septic shock. J Infect Dis. 2007;196(9):1288–95.
Chiche L, et al. Interferon-γ production by natural killer cells and cytomegalovirus in critically ill patients. Crit Care Med. 2012;40(12):3162–9.
Avdic S, McSharry BP, Slobedman B. Modulation of dendritic cell functions by viral IL-10 encoded by human cytomegalovirus. Front Microbiol. 2014;5:337.
Ahmed A. Immunopathology of CMV co-infection: review. MOJ Immunol. 2014;1(3):00017.
Tan Y, et al. Role of Treg/Th17 balance in the pathogenesis of cytomegalovirus infection. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2012;28(6):649–51.
Mavigner M, et al. Altered CD4+ T cell homing to the gut impairs mucosal immune reconstitution in treated HIV-infected individuals. J Clin Investig. 2012;122(1):62–9.
Ye Q, et al. The mechanism and treatment of gastrointestinal symptoms in patients with COVID-19. Am J Physiol Gastrointest Liver Physiol. 2020;319(2):G245–52.
Ganzenmueller T, et al. Detection of cytomegalovirus (CMV) by real-time PCR in fecal samples for the non-invasive diagnosis of CMV intestinal disease. J Clin Virol. 2014;61(4):517–22.
Michel D, et al. Intestinal cytomegalovirus disease in immunocompromised patients may be ruled out by search for cytomegalovirus DNA in stool samples. J Clin Microbiol. 1995;33(11):3064–7.
Zhou L, et al. IL-6 programs TH-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat Immunol. 2007;8(9):967–74.
Perez LG, et al. TGF-β signaling in Th17 cells promotes IL-22 production and colitis-associated colon cancer. Nat Commun. 2020;11(1):1–14.
Chen Z, Wherry EJ. T cell responses in patients with COVID-19. Nat Rev Immunol. 2020;20(9):529–36.
Kato T, et al. Correlation of serum IL-6 level with exhaustion of cytomegalovirus-specific T cells after hematopoietic stem cell transplantation. Blood. 2011;118(21):360.
Acknowledgements
Our thanks go to Professor Alborzi clinical microbiology research center staff for their technical support and assistance.
Funding
The authors have no support or funding to report.
Author information
Authors and Affiliations
Contributions
The study concept and design: AA; acquisition of data: AA, MZ, MRB, MS, SBH; analysis and interpretation of data: AA, MZ, MK; drafting of the manuscript: AA, MK; virological studies: MZ, MJ, NHM; final approval of the manuscript and agreement to be accountable for all aspects of the paper: AA, MS, MRB, SBH, MZ, MJ, MK, and NHM. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Research Ethics Committee of Shiraz University of Medical Sciences. The study was in line with the ethical principles and the national norms and standards for conducting Medical Research in Iran with the approval ID IR.SUMS.REC.1400.144 on 23 May 2021.
Consent for publication
Written informed consent was obtained from the patient’s next of kin for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Molecular Analysis for SARS-CoV-2 Variants Identification.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Amanati, A., Shahriari, M., Bordbar, M.R. et al. Severe acute respiratory syndrome coronavirus-2 Alpha variant (B.1.1.7), original wild-type severe acute respiratory syndrome coronavirus 2, and cytomegalovirus co-infection in a young adult with acute lymphoblastic leukemia, case report, and review of the possible cytomegalovirus reactivation mechanisms. J Med Case Reports 17, 66 (2023). https://doi.org/10.1186/s13256-022-03750-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13256-022-03750-8