Abstract
Background
The expiratory time constant (RCEXP), which is defined as the product of airway resistance and lung compliance, enable us to assess the mechanical properties of the respiratory system in mechanically ventilated patients. Although RCEXP could also be applied to spontaneously breathing patients, little is known about RCEXP calculated from the maximal expiratory flow-volume (MEFV) curve. The aim of our study was to determine the reference value for RCEXP, as well as to investigate the association between RCEXP and other respiratory function parameters, including the forced expiratory volume in 1 s (FEV1)/ forced vital capacity (FVC) ratio, maximal mid-expiratory flow rate (MMF), maximal expiratory flow at 50 and 25% of FVC (MEF50 and MEF25, respectively), ratio of MEF50 to MEF25 (MEF50/MEF25).
Methods
Spirometric parameters were extracted from the records of patients aged 15 years or older who underwent pulmonary function testing as a routine preoperative examination before non-cardiac surgery at the University of Tokyo Hospital. RCEXP was calculated in each patient from the slope of the descending limb of the MEFV curve using two points corresponding to MEF50 and MEF25. Airway obstruction was defined as an FEV1/FVC and FEV1 below the statistically lower limit of normal.
Results
We retrospectively analyzed 777 spirometry records, and 62 patients were deemed to have airway obstruction according to Japanese spirometric reference values. The cut-off value for RCEXP was 0.601 s with an area under the receiver operating characteristic curve of 0.934 (95% confidence interval = 0.898–0.970). RCEXP was strongly associated with FEV1/FVC, and was moderately associated with MMF and MEF50. However, RCEXP was less associated with MEF25 and MEF50/MEF25.
Conclusions
Our findings suggest that an RCEXP of longer than approximately 0.6 s can be linked to the presence of airway obstruction. Application of the concept of RCEXP to spontaneously breathing subjects was feasible, using our simple calculation method.
Similar content being viewed by others
Background
The expiratory time constant (RCEXP) is the parameter that refers to the length of time required for a lung unit to fill or empty. In mechanically ventilated patients, expiration is a passive process that depends on the time constant of the respiratory system, and RCEXP is thus useful for assessing the lung condition to optimize the ventilator settings [1]. Given that RCEXP is defined as the product of airway resistance and lung compliance [2], the variable serves as a dynamic measurement that can reflect the mechanical properties of the respiratory system [3, 4].
The concept of RCEXP is not common in spontaneously breathing patients, but it is possible to calculate it from the maximal expiratory flow-volume (MEFV) curve. According to the equal pressure point theory, the descending limb of the MEFV curve where the maximal expiratory flow is less than 75% of the forced vital capacity (FVC) corresponds to the effort-independent portion [5, 6]. In theory, RCEXP is obtained when maximal expiratory flows at low lung volumes are accurately ascertained, as the slope of the effort-independent portion is known to be expressed as the reciprocal of the time constant of the respiratory system [1, 7]. The maximal expiratory flows measured at a specified point of the MEFV curve can be susceptible to individual variability [8]. Unlike flow-based parameters, however, the value of RCEXP would contain qualitatively different information closely related to respiratory mechanics, given its ability to describe the features of the lung unit with its unique elasticity and capacity to resist airflow.
Standard spirometry, with all its limitations, remains an indispensable tool for detecting airway obstruction, and it can be used as a preoperative screening in patients scheduled for surgery under general anesthesia [9, 10]. Among a range of spirometric parameters, forced expiratory volume in 1 s (FEV1)/FVC, maximal mid-expiratory flow rate (MMF), maximal expiratory flows at 50 and 25% of FVC (MEF50 and MEF25, respectively), and the ratio of MEF50 to MEF25 (MEF50/MEF25) are, to one degree or another, linked to the pathology of small airways [8, 11, 12]. On the assumption that RCEXP is also associated with small airways obstruction, it could serve as a marker indicative of intra- or post-operative pulmonary conditions. However, there is no standard for RCEXP calculated from the effort-independent portion of the MEFV curve. The relationship between RCEXP and other spirometric parameters has not been investigated either.
Our study was designed to determine the reference value for RCEXP derived from spirometry, as well as to examine the association between RCEXP and the markers of small airway disease, including FEV1/FVC, MMF, MEF50, MEF25, and MEF50/MEF25. We then performed a retrospective analysis of our database that contains the records of preoperative spirometry testing obtained from patients scheduled for non-cardiac surgery at the University of Tokyo Hospital, Japan.
Materials and methods
Study subjects
Patients scheduled for surgery in Japan are supposed to undergo pulmonary function testing as part of routine preoperative examinations to reveal any undiagnosed respiratory dysfunction. Basically, all patients scheduled for surgery at the University of Tokyo Hospital undergo spirometry testing prior to general anesthesia, under the instruction of the attending doctor. Preoperative pulmonary function measures are occasionally screened in some patients undergoing regional anesthesia to assess their suitability to undergo general anesthesia in case of any sudden change in the type of anesthesia performed. Informed consent was obtained from each patient in advance on the use of data for scientific research.
With the approval of the institutional review board of the University of Tokyo (IRB #11108), we created a database containing information on the pulmonary function of patients scheduled for surgery in order to compare the flow-volume curves obtained prior to and during general anesthesia [13]. This database contains the records of preoperative MEFV curves that were available from patients aged 15 years or older, who were scheduled for non-cardiac surgery under general or regional anesthesia during the period between April 5 and May 31, 2016. A portion of the data, including baseline characteristics of the patients and respiratory function parameters derived from spirometry, had previously been reported [13].
Quality control of spirometry
In accordance with the guidelines issued by the Japanese Respiratory Society [14], spirometry testing was performed by experienced technicians at our institution to ensure measurement accuracy by diminishing the variability of the results. The acceptability criteria include (1) a continuous maximal effort throughout the maneuver without artefacts, (2) a satisfactory start of expiration with an extrapolated volume of less than 5% of FVC or 150 mL, whichever is larger, and (3) an adequate exhalation with a plateau in the volume-time curve of longer than 2 s, exhalation times of longer than 15 s, or exhalation times of longer than 6 s if the subject cannot continue further exhalation. Acceptable repeatability is achieved when the difference between the largest and the next largest FEV1 is within 200 mL of each other and the difference between the largest and the next largest FVC is within 200 mL of each other, after a minimum of three acceptable spirograms have been obtained. The best curve that meets all the criteria above is selected from the usable curves. It also requires that the sum of FEV1 and FVC be adequately large in the best curve.
Study design and methods
Our database of preoperative spirometry testing was retrospectively analyzed to clarify the association between RCEXP and other spirometric parameters, including FEV1/FVC, MMF, MEF50, MEF25, and MEF50/MEF25, and to estimate the reference value for RCEXP. The measured values of FEV1/FVC, MMF, MEF50, MEF25, and MEF50/MEF25 were extracted from our database. Emphasis was also placed on clarifying the relationship between MMF and other respiratory function parameters sensitive to the degree of airway obstruction in small airways.
Calculation of RCEXP
Based on respiratory mechanics, knowledge on the descending limb of the MEFV curve is described using the following equations:
where R is airway resistance, C is lung compliance, P is pressure, V is gas volume, and \( \dot{\mathrm{V}} \) is air flow. By definition, RCEXP is the product of airway resistance and lung compliance, and is expressed via the eqs. (1) and (2) as:
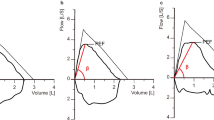
The eq. (3) refers to RCEXP as the reciprocal of the slope of the descending limb. RCEXP is theoretically obtained when two points along the effort-independent part of the descending limb, MEF50 and MEF25 for instance, are ascertained. In the present study, the value of RCEXP was calculated as the reciprocal of the slope of the line passing through the two points corresponding to MEF50 and MEF25 (Fig. 1) by using the following equation:
Visual representation of maximal expiratory flow at 50% (MEF50) and 25% (MEF25) of forced vital capacity (FVC). The MEF50 and MEF25 are shown as red points located along the descending limb of the maximum expiratory flow-volume (MEFV) curve. The expiratory time constant (RCEXP) is calculated as the reciprocal of the slope of the line passing through MEF50 and MEF25
Statistical analysis
Data are expressed as mean ± standard deviation, median and interquartile range, or n (%). The R2 value was calculated for the relationships between the spirometric parameters examined. The receiver operating characteristic (ROC) curve was generated for RCEXP to select the cut-off value in accordance with the presence of airway obstruction that was defined as a FEV1/FVC ratio and FEV1 below the statistically lower limit of normal (LLN) [15, 16]. The area under the ROC curve (AUC) was also obtained for RCEXP.
All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria), which is precisely described as a modified version of R commander designed to add statistical functions used in biostatistics [17]. A P value of < 0.001 was considered statistically significant.
Results
Overall, 777 patients aged 15 years or older were scheduled for surgery and underwent preoperative spirometry testing between April 5 and May 31, 2016. Of the patients scheduled for non-cardiac surgery, 689 underwent general anesthesia and 88 underwent regional anesthesia. The characteristics of the patients are summarized in Table 1. Airway obstruction was defined as an FEV1/FVC and FEV1 below their respective LLN values. When using Japanese spirometric reference values [18], 62 patients were deemed to have airway obstruction in the present study.
The relationships between RCEXP and the spirometric parameters, including FEV1/FVC, MMF, MEF50, MEF25, and MEF50/MEF25, are displayed as scatter plots (Fig. 2, Additional file 1: Figures S1 – S4), each with an R2 value of 0.8204, 0.3154, 0.4933, 0.1172, and 0.0144, respectively. The cut-off value for RCEXP was 0.601 s with an AUC of 0.934 (95% confidence interval = 0.898–0.970) (Fig. 3). The relationships between MMF and expiratory flow at lower lung volumes are also displayed as scatter plots (Additional file 1: Figures S5 and S6). MMF was closely associated with both MEF50 and MEF25 with an R2 value of 0.9005 and 0.8885, respectively.
The relationship between expiratory time constant (RCEXP) and forced expiratory volume in 1 s/forced vital capacity (FEV1/FVC). The value of RCEXP, which is calculated based on the effort-independent part of the MEFV curves, is closely associated with FEV1/FVC, with a high R2 value of 0.8204 (P < 0.001). Notably, there is a substantial increase in RCEXP with an FEV1/FVC ratio being less than approximately 0.70
The receiver operating characteristic (ROC) curve for RCEXP. With airway obstruction being defined as an FEV1/FVC and FEV1 below the statistically lower limit of normal, the cut-off value for RCEXP is 0.601 s. The area under the receiver operating characteristic curve (AUC) is calculated as 0.934 (95% confidence interval [CI] = 0.898–0.970)
Discussion
Main findings
Our retrospective analysis of 777 patients who underwent pulmonary function testing prior to surgery at our institution revealed that the cut-off value for RCEXP calculated from the MEFV curves was 0.601 s with an AUC of greater than 0.9. Among the spirometric parameters that are presumed to predict peripheral airways dysfunction, RCEXP was strongly or moderately associated with FEV1/FVC, MMF, and MEF50, whereas it was less associated with MEF25 and MEF50/MEF25. Even in spontaneously breathing subjects, calculation of RCEXP was possible using the descending limb of the MEFV curve, and our findings imply that a prolonged RCEXP, especially if it is longer than 0.601 s, could be associated with airway obstruction.
Physiological interpretation of expiratory time constant
To our knowledge, this study is the first attempt to provide the reference value for RCEXP that was calculated from the effort-independent portion of the MEFV curve. In theory, RCEXP can be altered depending on the degree of airway obstruction in spontaneously breathing patients. The finding that most patients without airway obstruction had an RCEXP of shorter than 0.6 s would be comparable to that of a previous study by McIlroy et al., who reported an average time constant of 0.38 s (ranging from 0.28 to 0.51 s) in their healthy, non-intubated subjects [7]. As might be expected, however, our reference value for RCEXP did not exceed the time constant values in mechanically ventilated patients with acute respiratory distress syndrome, which was reported to be in the range of 0.60 to 0.70 s [19, 20].
McIlroy et al. employed the slope of the line drawn using exhaled tidal volume and flow to determine values of the time constant of a relaxed expiration [7]. The reason why their findings were in agreement with those obtained during forced expiration in our study population could be attributable to the mechanism by which forced expiration is governed. As demonstrated in the comparison between relaxed and forced expirations in the same subject, the time rate of change in volume was similar under the relaxed and forced conditions [21]. Even when a greater expiratory flow is achieved, RCEXP will not be shorter than that during relaxed expiration, as the ratio of volume to flow is similar because of the difference in volume expired during forced and relaxed expirations [7]. As long as the linearity of the expiratory flow-volume curves validates the assumption that the linear portion is indicative of the mechanical properties of the respiratory system, namely its resistance and compliance, the value of RCEXP remains theoretically unchanged irrespective of whether the phase of expiration ends at the residual volume or at the functional residual volume.
In contrast, the finding that RCEXP gradually increased in tandem with the decrease in FEV1/FVC, MMF, and MEF50, especially when they were decreased below a certain level, could be interpreted as collateral evidence for the uneven distribution of RCEXP in patients with airway obstruction [22]. In a model resembling a lung unit where a single elastic element passively empties through a tube open to the atmosphere, the amount of ventilation depends on the compliance of the element and the resistance of the tube. When a particular portion of the lung unit is inadequately ventilated because of the narrowing of its airway, the increase in its airway resistance results in a prolonged RCEXP [23]. This is because the expiratory flow of emptying such a unit is determined using its time constant, the product of its airway resistance and lung compliance. The inequality of ventilation would therefore be a possible mechanism underlying the decreased rate of emptying of lung units with a larger airway resistance, the degree of which could be expressed as a longer RCEXP observed with an increase in the proportion of poorly ventilated regions. On the basis of our previous finding that patients with an FEV1/FVC ratio of less than 0.70 showed a substantial increase in the calculated value of airway resistance prior to general anesthesia [13], it could be inferred that elevated airway resistance was a major contributor to the increase in RCEXP.
In the present study, we calculated RCEXP by dividing a quarter of the FVC by the gap between MEF50 and MEF25. Even in healthy subjects, a degree of variability can exist in the parameters available from spirometry, partly because of the variability in FVC values that are possibly influenced by expiratory effort [8]. The advantage of our calculation method would lie in minimizing the variability in FVC, MEF50, and MEF25, thereby leading to decreased standard deviations of RCEXP. Even then, it would still be difficult to simply extrapolate the concept of the linearity of the flow-volume relationship to curvilinear MEFV curves scooping in toward the volume axis, considering that the MEF50/MEF25 ratio, which could be related to non-homogeneous emptying of the lung, was not constant regardless of the degree of airway obstruction.
MMF and expiratory flow at lower lung volumes
Given the phenomenon of maximal expiratory flow in which the equal pressure point shifts along the downstream segment to more peripheral airways and is eventually established in non-cartilaginous airways that are easily collapsible [24], the maximal expiratory flows measured at the lower range of FVC are likely sensitive to increased peripheral airway resistance where expiratory flow limitation occurs [25, 26]. For this reason, the measures derived from the middle or latter aspect of the MEFV curve, including MMF, MEF50, and MEF25, has been regarded as surrogate markers of peripheral airways obstruction.
The finding that there was a highly positive correlation between MMF and MEF50 is in close agreement with the finding of Bar-Yishay et al., who analyzed MEFV curves obtained from a large sample of children [27]. MMF is a time-weighted average flow over the mid-vital capacity range, and it is, by definition, likely that MMF contains information that is responsible for the physiological events occurring at the middle aspect of the MEFV curve. On the assumption that the lung empties non-homogeneously with more than a single time constant, the difference between MMF and MEF50 would theoretically reflect the degree of airway obstruction [28]. However, Bar-Yishay et al. presented the evidence that the ratio of MEF50 to MMF was not affected by peripheral airways obstruction, suggesting the possibility that this ratio is less reflective of the curvilinearity MEFV curve [27]. Their conclusion was that reporting both MMF and MEF50 was redundant, considering the close correlation between them. There would nevertheless be value in reporting MEF25, as it appeared from our study that the relationship between MMF and MEF25 was rather quadratic than simply linear. This might be because of the qualitative difference between MEF50 and MEF25 in the ability to detect airway obstruction, although both are supposed to be surrogate markers of early small airway disease. The finding that RCEXP was less associated with MEF25 than with MEF50 might also be related to the different property of MEF25.
MEFV curve evaluation using the slope-ratio (SR) index, which quantifies the instantaneous slope at any point along the MEFV curve, allows for assessment of special changes in curvature over a range of lung volumes [2]. It also provides additional information that is overlooked by the evaluation of MEFV curves based on absolute and relative values of volume and flow [29]. In elderly healthy subjects, there is a steady increase in SR with the progression of expiration [30], and consequently the decrease in expiratory flow occurs mainly at lower lung volumes [31]. The SR analysis used to detect difference in MEFV curves due to mild chronic obstructive pulmonary disease has demonstrated that the late scooping observed in these subjects is indicative of the normative aging process [29]. The interpretation of decreased MEF50 and MEF25 should thus be made with caution especially in older subjects.
MEF50/MEF25
MEF50/MEF25 is occasionally used in Japan to evaluate the degree of airway obstruction [12, 32]. Patients with airway obstruction frequently exhibit a marked decrease in MEF25 compared with MEF50, resulting in an increase in MEF50/MEF25 [32]. Some studies have suggested that an elevated MEF50/MEF25 is associated with the pathology of small airways, especially when it is greater than 4.0 [32, 33], but whether MEF50/MEF25 functions as a marker of small airway disease is still obscure because of the lack of sufficient epidemiological data for this parameter.
A Japanese study reported that MEF50/MEF25 was greater than 4.0 in many healthy subjects aged 40 years or older, with no difference in MEF50/MEF25 between smokers and non-smokers, suggesting that it could be difficult to detect the presence of airway obstruction using only MEF50/MEF25 [8]. This tendency was consistent with our results in which MEF50/MEF25 exceeded 4.0 in more than one-third of the study population without airflow obstruction. The limited utility of MEF50/MEF25 may be explained by the qualitative difference between MEF50 and MEF25 in the degree of association with small airway pathology. MEF50/MEF25 could nevertheless be useful in younger subjects, as healthy adults aged 30 years or younger generally have a MEF50/MEF25 of less than 3.0 [32].
Limitations
Several limitations of our study should be mentioned. First, it was not clarified whether RCEXP was more sensitive than other spirometric parameters in detecting the pathology of small airways. Our results showed that the value of RCEXP quantified from spirometry was associated with airway obstruction, but it was unclear whether RCEXP could provide more useful clinical information than standard spirometric measures. It would be necessary to explore the extent to which RCEXP reflects the different level of severity of airway obstruction because there was a limited number of patients with airway obstruction in our study population. Second, this is a retrospective study and the quality of spirometry testing performed in our patients may be questioned. Improved quality and standardization of forced expiratory maneuver is required to properly interpret the results. Every possible attempt was made to ensure quality-assured and standardized spirometry at our institution. Third, it was difficult to assess the effect of cigarette smoking on lung function because current and former smokers were included in our study. As reported before, an age-related decline has been noted in the maximal expiratory flows in the smoking population aged 40 years or older [8]. Finally, we included only Japanese patients scheduled for surgery under general or regional anesthesia. Although our results cannot be simply applied to different races other than Asians, our reference value for RCEXP could still be theoretically useful in assessing the degree of airway obstruction if it reflects the properties of the respiratory system.
Conclusions
Our study shed light on the calculated value of RCEXP that was derived from the effort-independent portion of the MEFV curve, suggesting that an RCEXP of longer than approximately 0.6 s can be linked to the presence of airway obstruction in spontaneously breathing patients. While monitoring of RCEXP allows us to assess the overall respiratory mechanics in critical care practice, it would be feasible to apply the concept of RCEXP to non-intubated subjects, using our simple method of calculating RCEXP from the MEFV curve. Further studies are warranted to confirm the ability of RCEXP to detect the presence of airway obstruction.
Availability of data and materials
The datasets used and analyzed during the current study may be made available from the corresponding author on reasonable request.
Abbreviations
- FEV1 :
-
forced expiratory volume in 1 s
- FVC:
-
forced vital capacity
- MEF25 :
-
maximal expiratory flows at 25% of FVC
- MEF50 :
-
maximal expiratory flows at 50% of FVC
- MEF50/MEF25 :
-
ratio of MEF50 to MEF25
- MEFV:
-
maximal expiratory flow-volume
- MMF:
-
maximal mid-expiratory flow rate
- RCEXP :
-
expiratory time constant
- VC:
-
vital capacity
References
Brunner JX, Laubscher TP, Banner MJ, Iotti G, Braschi A. Simple method to measure total expiratory time constant based on the passive expiratory flow-volume curve. Crit Care Med. 1995;17:1117–22.
Mead J. Analysis of the configuration of maximum expiratory flow-volume curves. J Appl Physiol Respir Environ Exerc Physiol. 1978;44:156–65.
Lourens MS, van den Berg B, Aerts JG, Verbraak AF, Hoogsteden HC, Bogaard JM. Expiratory time constants in mechanically ventilated patients with and without COPD. Intensive Care Med. 2000;26(11):1612–8.
Arnal JM, Garnero A, Saoli M, Chatburn RL. Parameters for simulation of adult subjects during mechanical ventilation. Respir Care. 2018;63(2):158–68.
Arora VK, Raghu S. Flow volume curves: clinical significance. Lung India. 1996;14:169–71.
Hyatt RE, Schilder DP, Fry DL. Relationship between maximum expiratory flow and degree of lung inflation. J Appl Physiol. 1958;13:331–6.
McIlroy MB, Tierney DF, Nadel JA. A new method of measurement of compliance and resistance of the lungs and thorax. J Appl Physiol. 1963;17:424–7.
Nemoto T, Shibata Y, Osaka D, Abe S, Inoue S, Tokairin Y, Igarashi A, Yamauchi K, Kimura T, Kishi H, Nishiwaki M, Aida Y, Nunomiya K, Sato M, Watanabe T, Konta T, Kawata S, Kato T, Kayama T, Kubota I. Impact of cigarette smoking on maximal expiratory flows in a general population: the Takahata study. Intern Med. 2011;50:2547–55.
Qaseem A, Snow V, Fitterman N, Hornbake ER, Lawrence VA, Smetana GW, Weiss K, Owens DK, Aronson M, Barry P, Casey DE Jr, Cross JT Jr, Fitterman N, Sherif KD, Weiss KB. Clinical Efficacy Assessment Subcommittee of the American College of Physicians. Risk assessment for and strategies to reduce perioperative pulmonary complications for patients undergoing noncardiothoracic surgery: a guideline from the American college of physicians. Ann Intern Med. 2006;144(8):575–80.
Phunmanee A, Tuntisirin C, Zaeoue U. Preoperative spirometry to predict postoperative complications in thoracic surgery patients. J Med Assoc Thail. 2000;83(10):1253–9.
McFadden ER Jr, Linden DA. A reduction in maximum mid-expiratory flow rate. A spirographic manifestation of small airway disease. Am J Med. 1972;52:725–7.
The Committee of Pulmonary Physiology JRS. Forced expiratory curve, flow-volume curve and peak flow. In: The Clinical Respiratory Function Test 7th Edition. Medical Review, Tokyo, 2008 (in Japanese).
Ikeda T, Uchida K, Yamauchi Y, Oba K, Nagase T, Yamada Y. Relationship between pre-anesthetic and intra-anesthetic airway resistance in patients undergoing general anesthesia: a prospective observational study. PLoS One. 2012;12(2):e0172421.
The Japanese Respiratory Society. Spirometry and flow-volume curve. In: Guidelines for Pulmonary Function Testing. Medical Review, Tokyo, 2004 (in Japanese).
Ferguson GT, Enright PL, Buist AS, Higgins MW. Office spirometry for lung health assessment in adults: a consensus statement from the National Lung Health Education Program. Respir Investig. 2014;52(4):242–50.
Swanney MP, Ruppel G, Enright PL, Pedersen OF, Crapo RO, Miller MR, Jensen RL, Falaschetti E, Schouten JP, Hankinson JL, Stocks J, Quanjer PH. Using the lower limit of normal for the FEV1/FVC ratio reduces the misclassification of airway obstruction. Thorax. 2008;63(12):1046–51.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Kubota M, Kobayashi H, Quanjer PH, Omori H, Tatsumi K, Kanazawa M. Clinical Pulmonary Functions Committee of the Japanese Respiratory Society. Reference values for spirometry, including vital capacity, in Japanese adults calculated with the LMS method and compared with previous values. Respir Investig. 2014;52(4):242–50.
Guttmann J, Eberhard L, Fabry B, Bertschmann W, Zeravik J, Adolph M, Eckart J, Wolff G. Time constant/volume relationship of passive expiration in mechanically ventilated ARDS patients. Eur Respir J. 1995;17:114–20.
Kondilli E, Prinianakis H, Athanasakis D, Georgopoulos D. Lung emptying in patients with acute respiratory distress syndrome: effect of positive end expiratory pressure. Eur Respir J. 2002;17:811–9.
Pierce JA. Studies of free collapse in the intact human lung. J Lab Clin Med. 1959;54:96–106.
West JB. Pulmonary pathophysiology: the essentials, 8th ed. Philadelphia: Lippincott Williams & Wilkins; 2012. p. 10–2.
West JB. Respiratory physiology: the essentials, 9th ed. Lippincott Williams & Wilkins, Philadelphia. 2012:118–9.
Mead J, Turner JM, Macklem PT, Little JB. Significance of the relationship between lung recoil and maximal expiratory flow. J Appl Physiol. 1967;22:95–108.
Dawson SV, Elliott EA. Wave-speed limitation on expiratory flow-a unifying concept. J Appl Physiol Respir Environ Exerc Physiol. 1977;43:498–515.
Elliott EA, Dawson SV. Test of wave-speed theory of flow limitation in elastic tubes. J Appl Physiol Respir Environ Exerc Physiol. 1977;43:516–22.
Bar-Yishay E, Amirav I, Goldberg S. Comparison of maximal midexpiratory flow rate and forced expiratory flow at 50% of vital capacity in children. Chest. 2003;123:731–5.
McNamara JJ, Castile RG, Glass GM, Fredberg JJ. Heterogeneous lung emptying during forced expiration. J Appl Physiol. 1987;63:1648–57.
Dominelli PB, Foster GE, Guenette JA, Haverkamp HC, Eves ND, Dominelli GS, Henderson WR, O'Donnell DE, Sheel AW. Quantifying the shape of the maximal expiratory flow-volume curve in mild COPD. Respir Physiol Neurobiol. 2015;219:30–5.
Fowler RW, Pluck RA, Hetzel MR. Maximal expiratory flow-volume curves in Londoners aged 60 years and over. Thorax. 1987;42:173–82.
Knudson RJ, Clark DF, Kennedy TC, Knudson DE. Effect of aging alone on mechanical properties of the normal adult human lung. J Appl Physiol Respir Environ Exerc Physiol. 1977;43:1054–62.
Takishima T, Sasaki T, Takahashi K, Sasaki H, Nakamura T. Direct-writing recorder of the flow-volume curve and its clinical application. Chest. 1972;61:262–6.
Sakamoto O, Saita N, Ando M, Kohrogi H, Suga M, Ando M. Two cases of Sjögren's syndrome with multiple bullae. Intern Med. 2002;41:124–8.
Acknowledgements
We thank Aranya Bagchi, MBBS, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital, for critically reading the manuscript and suggesting substantial improvements.
Funding
The current study was not supported by any funding.
Author information
Authors and Affiliations
Contributions
TI developed the idea for this research and collected all data regarding spirometry testing. TI, along with YY1, KU, and KO, analyzed and interpreted the data. YY2 and TN supervised the study. TI wrote the manuscript. All authors read and approved the final manuscript. Note that YY1 corresponds to the author furthest up on the author list.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
We have obtained the approval of the institutional review board of the University of Tokyo (IRB #11108). Informed consent was obtained from all surgery patients in advance on the use of data for scientific research and publication. The current study focuses on preoperative pulmonary function measures, and the institutional review board of the University of Tokyo decided that additional informed consent was not required, considering that all patients at our institution who are scheduled for surgery under general anesthesia are supposed to undergo pulmonary function testing as part of routine preoperative examinations.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Additional file 1: Figure S1.
The relationship between RCEXP and maximal mid-expiratory flow rate (MMF). To a certain extent, RCEXP is sociated with MMF with an R2 value of 0.3154 (P < 0.001). There is a gradual increase in RCEXP, especially when MMF is below approximately 1.0 L/s. Figure S2. The relationship between RCEXP and maximal expiratory flow at 50% of FVC (MEF50). MEF50 is one of the spirometric parameters used to calculate RCEXP. RCEXP is moderately associated with MEF50 with an R2 value of 0.4933 (P < 0.001). When MEF50 is below approximately 1.5 L/s, RCEXP increases with a reduction in MEF50. Figure S3. The relationship between RCEXP and maximal expiratory flow at 25% of FVC (MEF25). MEF25 is also one of the spirometric parameters used to calculate RCEXP. As compared with MEF50, RCEXP is less associated with MEF25, and the R2 value was estimated to be 0.1172 (P < 0.001). Figure S4. The relationship between RCEXP and maximal expiratory flow at 50% of FVC divided by maximal expiratory flow at 25% of FVC (MEF50/MEF25). Overall, RCEXP is almost constant regardless of the value of MEF50/MEF25. As compared with MEF50 and MEF25, RCEXP is less associated with MEF50/MEF25 with an R2 value of 0.0144 (P = 0.001331). Figure S5. The relationship between MMF and MEF50. Both MMF and MEF50 are parameters that quantify flow in the middle portion of the descending limb of the MEFV curve. MMF is linearly associated with MEF50 with a high R2 value of 0.9005. Figure S6. The relationship between MMF and MEF25. MMF is also closely associated with MEF25, and there is an almost linear relationship when MMF is below approximately 3.0 L/s.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Ikeda, T., Yamauchi, Y., Uchida, K. et al. Reference value for expiratory time constant calculated from the maximal expiratory flow-volume curve. BMC Pulm Med 19, 208 (2019). https://doi.org/10.1186/s12890-019-0976-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12890-019-0976-6