Abstract
Background
Gastroesophageal reflux disease (GERD) and cholecystitis share overlapping symptoms, including belching, acid reflux, and heartburn. Despite this, the causal relationship between these two conditions remains unclear. This study aimed to investigate the causal link between GERD and cholecystitis using a Mendelian randomization (MR) approach.
Methods
A two-sample MR analysis was conducted using the inverse variance weighted (IVW), weighted median, weighted mode, and MR-Egger method to assess the causal effects of GERD on the cholecystitis risk. Genome-wide association studies (GWASs) on GERD (N cases = 129080; N controls = 473524) and cholecystitis (N cases = 1930; N controls =359264) were obtained from the IEU Open GWAS project. Various techniques were employed to assess pleiotropy and heterogeneity.
Results
Seventy-seven single nucleotide polymorphisms from GERD GWASs were selected as instrumental variables (IVs). The primary IVW method revealed a significant association between GERD and an increased risk of cholecystitis (odds ratio = 1.004; 95% confidence interval = 1.003–1.005, p = 2.68 × 10− 9). The absence of heterogeneity and pleiotropy in the data supports the reliability of the results.
Conclusions
GERD was positively associated with the risk of cholecystitis. This study provides insights into potential avenues for the development of prevention strategies and treatment options for cholecystitis in patients with GERD. These findings contribute to our understanding of the complex interplay between GERD and cholecystitis.
Similar content being viewed by others
Background
Cholecystitis, a prevalent gallbladder disease, stands as a common cause of abdominal pain in Western countries. In the United States alone, gallbladder disease affects approximately 20 million people annually, with approximately 200,000 new diagnoses, imposing substantial physical and financial burdens on affected individuals [1]. Cystic ductal obstruction, mainly associated with gallstones, accounts for 90–95% of acute cholecystitis cases. The overall prevalence of gallstones is estimated at 10–15%, with 20–40% of patients facing gallstone-related complications and an annual incidence rate of 1–3% [2,3,4]. Typical clinical manifestations of cholecystitis include right upper abdominal pain, nausea, and malaise. Laparoscopic cholecystectomy stands as the preferred treatment for patients with symptomatic cholelithiasis due to its ability to reduce postoperative pain and shorten hospital stays [5, 6].
Various risk factors contribute to cholecystitis exist, including female sex, rapid weight loss, pregnancy, advanced age, obesity, consumption of high-fat or low-fiber diets, diabetes, and receipt of intravenous nutrition [7,8,9]. In clinical practice, gastroesophageal reflux disease (GERD) often coexists with cholecystitis, potentially leading to diagnostic challenges due to overlapping symptoms [10,11,12]. However, evidence regarding the causal relationship between GERD and cholecystitis remains inconclusive, underscoring the need for further investigation to prevent misdiagnosis and ensure timely interventions.
GERD, characterized by the backflow of gastric contents into the esophagus, presents distressing symptoms such as acid regurgitation, heartburn, and belching [13]. Over recent years, GERD has emerged as a prevalent clinical condition, affecting approximately 20% of adults in the Western world [14, 15]. The symptomatic similarities between GERD and cholecystitis exceed chance expectations, increasing the likelihood of diagnostic errors and compromising patient treatment and outcomes. Vagus nerve abnormalities lead to dyskinesia and paresthesia in the gastroesophageal and biliary systems, and some studies have reported an increased incidence of GERD after cholecystectomy, suggesting that there may be a connection between GERD and cholecystitis, or due to neurological or endocrine factors [16, 17]. Additionally, the direction and causative association between GERD and cholecystitis remain uncertain. Therefore, the objective of this study is to elucidate the causal relationship between these two conditions, and we have employed a Mendelian randomization (MR) analysis.
MR offers an alternative approach in epidemiology for inferring causality, utilizing single nucleotide polymorphisms (SNPs) as instrumental variables (IVs) to explore causal relationships between exposures and outcomes [18, 19]. Given the random inheritance of alleles during gamete formation, genetic variants may serve as a means of controlling for reverse causation bias and confounding [20]. As a result, MR designs can be employed to address causal assumptions, with outcomes deemed comparable to those of randomized trials [21].
Therefore, we conducted a two-sample MR study utilizing single SNPs obtained from genome-wide association studies linked to exposure factor GERD as IVs in this study, in order to examine the causal impact of GERD on outcome factor cholecystitis risk, marking the first such attempt. And we found that GERD was positively associated with the risk of cholecystitis. These findings can serve as a fundamental basis for understanding the intricate interactions between GERD and cholecystitis, which can further provide new ideas for the prevention and treatment of cholecystitis in GERD patients.
Methods
Study design
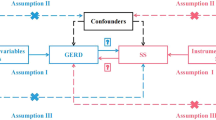
MR is an epidemiological method that utilizes IVs which are highly linked with the exposures (e.g., GERD), such as SNPs, to establish causal relationships in statistical analyses [22]. Figure 1 presents a flowchart that provides a concise overview of the two-sample MR analysis employed in this study to investigate the causal associations between GERD and cholecystitis, utilizing genetic variants as IVs. Three key assumptions underlie MR analysis [23]: the IV must be strongly correlated with the exposure of interest, must not be associated with any confounders, and must exclusively influence outcomes through the exposure variable. Ethical approval was deemed unnecessary for the present study, which utilized aggregate-level genetic data sourced from publicly accessible repositories of extensive genome-wide association studies (GWASs).
Schematic representation of Mendelian randomization (MR) analysis. (a-b) Flow chart for the analytical assumptions (a) and how MR analysis was performed step-by-step (b). The application of MR in this context relies on three fundamental assumptions: [1] Assumption 1, the genetic variants must exhibit associations with the exposures [2], Assumption 2, the genetic variants must not demonstrate associations with confounding factors, and [3] Assumption 3, the genetic variants must solely influence outcomes through the exposures, excluding alternative pathways. Abbreviations: MR, Mendelian randomization; SNP, single nucleotide polymorphism; LD, linkage disequilibrium; GERD, gastroesophageal reflux disease; IVW, inverse-variance weighted
Data source and IV selection
Publicly-available data were used for this study. Specifically, exposure data for GERD [24] were obtained from GWASs with larger sample sizes, comprising 602,604 individuals (N cases = 129,080 cases; N control = 473,524) of European ancestry, which were accessible through the IEU open GWAS project (https://gwas.mrcieu.ac.uk/datasets/ebi-a-GCST90000514/). Additionally, the pooled statistics GWASs for cholecystitis (N cases = 1,930; N control = 359,264) were obtained from https://gwas.mrcieu.ac.uk/datasets/ukb-d-K81/. Table 1 provides a detailed overview of all GWASs that were included in this investigation.
Our study identified SNPs linked to GERD at a genome-wide significant level (p < 5 × 10− 8), while also considering factors such as linkage disequilibrium (LD, R2 ≤ 0.001) and genetic clumping distance < 10,000 kb. If a specific exposure SNP did not exist in the outcome GWAS, a proxy SNP in linkage disequilibrium with the exposure SNP (minimum LD R2 value of 0.8) was used. To avoid bias caused by weak proxies, the R2 and F-statistics for each SNP were computed. The R2 value denotes the fraction of variability in a risk factor accounted for by the IVs. The F-statistic, commonly employed as an indicator of the potency of the relationship between instruments and the exposure of interest, is deemed weak if it falls below a threshold of F < 10; such weak instruments are precluded from the MR analysis. The R2 and F-statistics were derived using the following formulae [19]:
Where N represents sample size, β represents the genetic estimation of each SNP on the exposure, k represents the number of SNPs, and EAF refers to effect allele frequency.
MR statistical methods
Seventy-seven SNPs were utilized as IVs following the coordination of effect alleles (EAs) from GWAS in both GERD and cholecystitis. Several MR analysis methods were employed to determine the causal relationship between GERD and cholecystitis, including the inverse-variance weighted (IVW), MR-Egger, weighted median, and weighted mode approaches. The random-effects model of IVW was the primary statistical method used in the MR analysis [25]. The IVW method, which combines Wald ratio estimates through inverse variance weighting, was employed to evaluate the causal effect of exposure on the outcomes. MR-Egger regression was used to address potential issues of the causal effects and the directional pleiotropy, which are robust to invalid IVs. Additionally, the weighted median method was employed to provide a consistent estimate of causality, even when up to 50% of the genetic IVs used were invalid [26]. The robustness of the significant findings was further tested by applying the weighted mode method, which accommodated more than 50% of the invalid IVs.
Sensitivity analysis
The potential pleiotropic effects of the instrumental SNPs were evaluated using MR-Egger regression. To assess heterogeneity among the various genetic variations, the Cochran’s test and the I2 test [27] were employed. Furthermore, a leave-one-out sensitivity analysis was conducted to verify the conformity of each SNP. Funnel plots were used to present the heterogeneity of the results, and asymmetric plots indicated the absence of heterogeneity.
Statistical analysis
Statistical analyses were conducted with R software version 4.2.1, utilizing the R-based package “TwoSampleMR (0.5.5)” for MR analysis. For the methods of MR Egger, weighted median, IVW, weighted model, Cochran’s Q test, and I2 test, the results with a P value of less than 0.05 were significant.
Results
Detail information on the IVs
This study incorporated 77 SNPs in GERD as IVs. To mitigate the risk of weak instrumental bias, SNPs with an F-statistic lower than 10 were also removed, and all these SNPs were significantly associated with GERD at the genome-wide level (p < 5 × 10− 8). Additional file 1–2 provide detailed information on each SNP, including the EA and effect allele frequency (EAF), as well as estimations of their associations with GERD and cholecystitis, including the beta, SE, and P values. The details of the SNPs we have covered in this article can be found in the https://gwas.mrcieu.ac.uk/.
Causal effect of GERD on cholecystitis
Table 2 presents the results of the IVW analysis, indicating an elevated risk of cholecystitis in patients with GERD (odds ratio [OR] = 1.004; 95% confidence interval [95%CI] 1.003–1.005, p = 2.68 × 10− 9). Meanwhile, the estimates derived from the weighted median (OR = 1.005; 95%CI 1.003–1.006; P = 2.35 × 10− 6) and weighted mode (OR = 1.007; 95%CI 1.002–1.012; p = 0.01044) aligned with the direction of the IVW estimates. However, the MR-Egger analysis (OR = 1.005; 95%CI 0.998–1.013; p = 0.1841) suggested no causal relationship between GERD and cholecystitis. These findings are further supported by the forest plot (Fig. 2) and scatter diagram (Fig. 3).
Forest plot of the causal effects of SNPs associated with GERD on cholecystitis. The logarithmic odds ratio (OR) for cholecystitis per standard deviation (SD) increase in GERD is denoted by black dots. The combined causal estimate using all SNPs together as a single instrument is represented by red dots, generated via two different methods: the inverse variance weighted (IVW) method and MR-Egger. The MR results of the MR-Egger test and IVW method are indicative of the significance of red lines. The 95% confidence intervals of the estimate are represented by the horizontal line segments
Scatter plots of SNPs associated with GERD and cholecystitis. The causal associations for each Mendelian randomization (MR) method, namely the IVW method, weighted median estimator, weighted mode estimator, and MR-Egger, are represented by the slopes of each line. The plot illustrates the effect sizes of the SNP-cholecystitis association (y-axis, log (OR)) and the SNP-GERD association (x-axis, SD units) along with their respective 95% confidence intervals
Sensitivity test
Subsequently, the Cochran’s Q test did not detect the existence of heterogeneity in the causality of cholecystitis, and the I2 values in our results showed low heterogeneity, indicating increased reliability of the MR estimates. Table 3 summarizes the results of the sensitivity analysis. In addition, there was no evidence that the result was affected by horizontal pleiotropy (MR-Egger regression intercept = -3.6 × 10− 5; SE = 0.00012; directionality p = 0.772). Asymmetry in the funnel plot indicated directional horizontal pleiotropy; however, there was no evidence of asymmetry (Fig. 4). Moreover, the leave-one-out analysis results were stable when each SNP was discarded one by one (Fig. 5).
Leave-one-out of SNPs associated with GERD and their risk of cholecystitis. Each black dot represents the outcome of the IVW MR method employed to evaluate the causal impact of GERD on cholecystitis. Conversely, each red dot illustrates the IVW estimate obtained by incorporating all SNPs. Notably, no SNPs were identified as having a significant influence on the relationship between GERD and cholecystitis in this leave-one-out sensitivity analysis
Discussion
Cholecystitis and GERD are prevalent pathological conditions that result in dyspepsia of the upper gastrointestinal tract. These diseases exhibit comparable clinical symptoms, including epigastric pain, heartburn, abdominal distension, and early satiety [12, 28, 29]. Consequently, a significant number of patients remain undiagnosed or misdiagnosed with stomach disease, and the causal relationship between GERD and cholecystitis remains uncertain. In this study, we conducted a two-sample MR analysis to evaluate the causal association between GERD and cholecystitis. Our analysis demonstrated that genetically-predicted GERD was significantly associated with an increased risk of cholecystitis.
We conducted the study utilizing four distinct estimation methods for MR analysis: the IVW, weighted mode, MR-Egger, and weighted median analyses. Our analysis, which employed large-scale GWASs data, demonstrated that genetically predicted GERD was significantly associated with an increased risk of cholecystitis. While the results of the four MR analysis methods were inconsistent, the IVW, weighted mode, and weighted median analyses provided evidence of a causal link between GERD and cholecystitis. Unlike observational studies, MR analyses are less likely to be affected by confounding bias and reverse causality. Our investigation, being the first of its kind on a large scale, is less prone to bias and reverse causation, thereby enhancing our comprehension of the potential risk factors for Cholecystitis among patients with GERD.
The etiology of cholecystitis is complex and it is an independent risk factor for extrahepatic biliary tract malignancies, including gallbladder cancer [30]. Given the unfavorable prognosis associated with gallbladder cancer, with an overall 5-year survival rate of less than 10% [31], proactive measures to prevent cholecystitis assume paramount importance in the prevention of gallbladder cancer. Numerous risk factors associated with cholecystitis in the general population have been identified, including prolonged fasting, rapid weight loss, total parenteral nutrition, and somatostatin (-analog) treatment, which significantly increase the risk of developing the condition [32]. Additionally, female sex, estrogen levels, and obesity have also been identified as risk factors [33,34,35]. In clinical practice, patients diagnosed with cholecystitis frequently present with varying degrees of belching, acid reflux, heartburn, and other reflux symptoms. Belching, acid reflux, and heartburn are also recognized as typical symptoms of GERD. Therefore, the correlation between GERD and cholecystitis has attracted considerable interest.
The roles of gastric acid and bile as direct causative factors in the pathogenesis of GERD have been widely acknowledged. Typically, the esophagus maintains a state of equilibrium between its anti-reflux defense mechanism and the erosive effects of reflux substances on the esophageal mucosa. However, this balance is disrupted when the defense mechanisms of the esophagus are weakened, or when the harmful effects of reflux substances are intensified, ultimately resulting in the development of GERD. The principal pathophysiological mechanism underlying GERD is the compromised function of the anti-reflux barrier, primarily characterized by reduced pressure in the lower esophageal sphincter, transient relaxation of the lower esophageal sphincter, dysfunction of the crural diaphragm, presence of hiatal hernia, and impaired esophageal clearance, which is mainly attributed to ineffective esophageal motility and multiple rapid swallowing [15]. GERD and cholecystitis are both common motility disorders, and the relationship between GERD and cholecystitis is intricate, involving complex mechanisms.
Research has demonstrated that compared to healthy individuals, patients with esophagitis and Barrett’s esophagus exhibit substantially elevated levels of bile acid, which is linked to acid reflux [36, 37]. Bile reflux-induced chemical damage has been identified as a significant contributor to esophageal mucosal rupture and refractory GERD. Abnormal gallbladder function and anatomical structure contribute to cholestasis, resulting in impaired gallbladder function in terms of bile concentration and biliary pressure regulation. This, in turn, can accelerate bile enterohepatic circulation, elevate bile acid content, and ultimately compromise the regulatory function of the sphincter of Oddi, leading to bile reflux into the stomach [38]. Stimulation of mucosal receptors at the distal end of the esophagus by GERD can expedite the vagus nerve reflex, leading to anomalous peristalsis of the digestive tract [39]. The motor function of the gastroesophageal and biliary tract systems is regulated by nervous and humoral factors, among which the enteric nervous system (ENS), which is mainly composed of peptitineural nerves, plays the most important role. The ENS-interstitial cells of Cajal (ICC)-smooth muscle cells (SMC) network structure is the basic functional unit of gastrointestinal motility [40,41,42,43]. Therefore, GERD and cholecystitis are closely related to vagus nerve abnormalities and ICC abnormalities. This may exacerbate cholestasis, resulting in the onset of cholecystitis, accompanied by intractable dyspepsia. Patients with cholecystitis are prone to bile reflux, and the main damage caused by bile reflux to the esophageal mucosa is bile acids. When the concentration of bile acids exceeds the physiological level or the action time is prolonged, the intracellular bile acid concentration is too high, which destroys the intracellular membrane system and leads to cell necrosis, while the stimulation of bile salts can cause the expression of oxidative stress-related genes, induce DNA damage, and eventually lead to mucosal erosion and ulceration [44]. From the summary of clinical practice experience, the application of ursodeoxycholic acid can be used to treat GERD [45]. Bile reflux is present in GERD, and esophageal inflammation becomes more severe as the incidence of bile reflux increases. High concentrations of bile, deoxycholic acid, and trypsin can reduce the expression of cadherin, an interepithelial tight junction protein, and disrupt the esophageal epithelial barrier [46]. Treatment of GERD with ursodeoxycholic acid in combination with PPIs and/or prokinetic agents is associated with a higher rate of symptom remission [47]. Ursodeoxycholic acid has been reported to be effective in reducing the frequency and duration of reflux and significantly alleviating heartburn and acid reflux [48]. In addition, cholecystitis can be treated with gastrointestinal motility drugs, and Dhiman et al. [49]. found that cisapride can inhibit stone formation. By promoting the movement of the gallbladder and intestines, cisapride enhances the intestinal and hepatic circulation, normalizes the lipid composition of bile, increases the secretion of bile salts, prolongs the nucleation time, and reduces the concentration of cholesterol in bile [50, 51]. There may be a positive synergistic effect between acid reflux and bile reflux, and in-depth study of the relationship between GERD and cholecystitis is of great value for the diagnosis and treatment of clinical diseases. In this study, the causal relationship between GERD and cholecystitis was explored from the perspective of genetics, and a number of SNPs of great significance were screened. Therefore, GERD should be considered as a risk factor for cholecystitis in clinical practice, and early intervention in GERD may reduce the avoidance of gallbladder resection. Understanding the underlying mechanisms may be valuable in preventing and treating cholecystitis symptoms in patients with GERD, therefore, further research should be conducted to better elucidate the mechanisms, and intervention targets can be studied through the relevant SNPs that we have screened.
MR was employed to address the inherent limitations of observational studies, thereby reducing the likelihood of confounding or inherent biases [52]. One of the fundamental assumptions of MR is that the genetic variants utilized as instruments do not affect the outcome other than via the exposure of interest [53]. The incorporation of pleiotropic variation in the MR analysis leads to distorted causal effect estimates and a higher likelihood of type I errors when testing of causal hypotheses [54]. To identify any potential imbalances in pleiotropy, the weighted median approach was used to generate reliable estimates in instances where over 50% of the weight was derived from valid IVs [55]. The MR-Egger regression method was used to produce estimates after accounting for horizontal pleiotropy. In contrast to the outcomes obtained from the other three methods, the MR-Egger approach resulted in reduced in precision and statistical power. However, our weighted median estimator findings aligned with the IVW estimator, thereby offering supplementary support for a causal association. The present MR study provides compelling evidence that the mechanisms underlying the effects of GERD on cholecystitis risk are noteworthy. Further investigations are warranted to gain a more comprehensive understanding of these mechanisms. However, we continue to emphasize the need for independent MR studies, large prospective trials, and basic experiments in relevant laboratories to validate our findings using more accurate population data on cholecystitis and GERD.
However, this study had several limitations. First, the reliance on self-reported GERD diagnosis may have affected the reliability of the MR results. The lack of a detailed classification of GERD in the current GWAS study limits our ability to investigate the causal relationship between GERD and cholecystitis. To address this issue, future investigations should use GWAS data for GERD diagnosis based solely on the ICD criteria to validate the findings presented herein. In addition, due to the lack of sufficient IVs for GERD subtypes, the inverse causal relationship between GERD subtypes and GM needs to be further explored in the future. Second, the summary GWAS data for GERD and cholecystitis were obtained exclusively from individuals of European descent, thereby limiting the generalizability of our findings to the broader population. Considering the potential influence of race and selection bias on causality, additional MR studies involving diverse populations are warranted. Furthermore, genetic variants may have a negligible impact on a given exposure (GERD), because they may account for only a small proportion of the variance. Therefore, this study may have been underpowered in terms of detection of such associations. Third, two-sample MR studies may involve over-identification, leading to an overestimation of the association between SNP and exposure. Additionally, the causal effects of GERD on cholecystitis appeared modest, indicating a relatively low risk of cholecystitis as a secondary outcome of GERD. The absence of pleiotropy in the modest MR estimates provides reassurance of the robustness of the MR estimates, nevertheless, our understanding of the biological functions of genetic tools is still evolving, which introduces the potential risk of violating the assumptions of independence and exclusion restrictions of MR, especially with regard to pleiotropy. Lastly, while this study has identified SNPs that have a causal effect of GERD on the risk of cholecystitis, these findings are primarily based on statistical analyses. Experimental research is required to elucidate the exact roles of these SNPs in the pathogenesis of cholecystitis and to validate our results further. Notably, this study is the first to investigate of the causal relationship between GERD and cholecystitis.
Conclusions
In summary, we found that genetically predicted GERD is causally associated with a higher risk of cholecystitis from the general population. GERD can stimulate the mucosal receptors distal to the esophagus and accelerate the vagus nerve reflex to cause abnormal peristalsis of the digestive tract, thereby aggravating cholestasis, and cholecystitis with intractable dyspepsia symptoms may occur. There may be a positive synergistic effect between acid reflux and bile reflux, and in-depth study of the relationship between the two is of great value for the diagnosis and treatment of clinical diseases. Clinicians should pay more attention to the development of cholecystitis in patients with GERD. The pathophysiological mechanism of cross-talk between GERD and cholecystitis needs to be further studied.
Data availability
The datasets utilized in this study, namely the GWAS for GERD and cholecystitis, were obtained from publicly accessible databases of large-scale genome-wide association studies.
Abbreviations
- GERD:
-
Gastroesophageal reflux disease
- SE:
-
Standard error
- CI:
-
Confidence interval
- GWAS:
-
Genome-wide association study
- IV:
-
Instrumental variable
- IVW:
-
Inverse-variance weighted
- LD:
-
Linkage disequilibrium
- MR:
-
Mendelian randomization
- MR-PRESSO:
-
MR pleiotropy residual sum and outlier
- OR:
-
Odds ratio
- SNP:
-
Single nucleotide polymorphism
References
Gallaher JR, Charles A. Acute Cholecystitis: a review. JAMA. 2022;327:10.
Pisano M, Allievi N, Gurusamy K, Borzellino G, Cimbanassi S, Boerna D, et al. 2020 World Society of Emergency Surgery updated guidelines for the diagnosis and treatment of acute calculus cholecystitis. World J Emerg Surg. 2020;15:1.
Stinton LM, Myers RP, Shaffer EA. Epidemiology of gallstones. Gastroenterol Clin North Am. 2010;39:2.
Kimura Y, Takada T, Strasberg SM, Pitt HA, Gouma DJ, Garden OJ, et al. TG13 current terminology, etiology, and epidemiology of acute cholangitis and cholecystitis. J Hepatobiliary Pancreat Sci. 2013;20:1.
Johansson M, Thune A, Nelvin L, Stiernstam M, Westman B, Lundell L. Randomized clinical trial of open versus laparoscopic cholecystectomy in the treatment of acute cholecystitis. Br J Surg. 2005;92:1.
Lee PC, Lo C, Lai PS, Chang JJ, Huang SJ, Lin MT, et al. Randomized clinical trial of single-incision laparoscopic cholecystectomy versus minilaparoscopic cholecystectomy. Br J Surg. 2010;97:7.
Knab LM, Boller A-M, Mahvi DM, Cholecystitis. Surg Clin North Am. 2014;94:2.
Lammert F, Gurusamy K, Ko CW, Miquel J-F, Méndez-Sánchez N, Portincasa P et al. Gallstones Nat Reviews Disease Primers. 2016;2.
Doty JE, Pitt HA, Porter-Fink V, Denbesten L. Cholecystokinin prophylaxis of parenteral nutrition-induced gallbladder disease. Ann Surg. 1985;201:1.
Dzhulai GS, Sekareva EV, Chervinets VM, Mikhailova ES, Dzhulai TE. [Gastroesophageal reflux disease associated with duodenogastroesophageal reflux in patients with biliary pathology: the specific features of the course and esophagogastroduodenal microbial biocenosis]. Ter Arkh. 2014;86:2.
Santos MLC, de Brito BB, da Silva FAF, Sampaio MM, Marques HS, Oliveira E, Silva N, et al. Helicobacter pylori infection: beyond gastric manifestations. World J Gastroenterol. 2020;26:28.
Robinson M. Dyspepsia: challenges in diagnosis and selection of treatment. Clin Ther. 2001;23:8.
Katzka DA, Kahrilas PJ. Advances in the diagnosis and management of gastroesophageal reflux disease. BMJ. 2020;371.
Maret-Ouda J, Markar SR, Lagergren J. Gastroesophageal Reflux Disease JAMA. 2020;324:24.
Zheng Z, Shang Y, Wang N, Liu X, Xin C, Yan X, et al. Current Advancement on the dynamic mechanism of gastroesophageal reflux disease. Int J Biol Sci. 2021;17:15.
Shah Gilani SN, Bass GA, Kharytaniuk N, Downes MR, Caffrey EF, Tobbia I, et al. Gastroesophageal mucosal Injury after Cholecystectomy: an indication for Surveillance? J Am Coll Surg. 2017;224:3.
Pozo F, Giganto F, Raodrigo L. Non-complicated cholelithiasis associated with GERD. Results of combined laparoscopic surgery in low risk patients. Rev Esp Enferm Dig. 2004;96:4.
Smith GD, Ebrahim S. Mendelian randomization’: can genetic epidemiology contribute to understanding environmental determinants of disease? Int J Epidemiol. 2003;32:1.
Burgess S, Thompson SG. Avoiding bias from weak instruments in mendelian randomization studies. Int J Epidemiol. 2011;40:3.
Zoccali C, Testa A, Spoto B, Tripepi G, Mallamaci F. Mendelian randomization: a new approach to studying epidemiology in ESRD. Am J Kidney Dis. 2006;47:2.
Burgess S, Thompson SG. Multivariable mendelian randomization: the use of pleiotropic genetic variants to estimate causal effects. Am J Epidemiol. 2015;181:4.
Birney E. Mendelian randomization. Cold Spring Harb Perspect Med. 2022;12:4.
Spiga F, Gibson M, Dawson S, Tilling K, Davey Smith G, Munafò MR, et al. Tools for assessing quality and risk of bias in mendelian randomization studies: a systematic review. Int J Epidemiol. 2023;52:1.
Ong J-S, An J, Han X, Law MH, Nandakumar P, Schumacher J, et al. Multitrait genetic association analysis identifies 50 new risk loci for gastro-oesophageal reflux, seven new loci for Barrett’s oesophagus and provides insights into clinical heterogeneity in reflux diagnosis. Gut. 2022;71:6.
Burgess S, Butterworth A, Thompson SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol. 2013;37:7.
Burgess S, Bowden J, Fall T, Ingelsson E, Thompson SG. Sensitivity analyses for robust causal inference from mendelian randomization analyses with multiple genetic variants. Epidemiology. 2017;28:1.
Higgins JPT, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:11.
Martin WT, Stewart K, Sarwar Z, Kennedy R, Quang C, Albrecht R et al. Clinical diagnosis of cholecystitis in emergency department patients with cholelithiasis is indication for urgent cholecystectomy: a comparison of clinical, ultrasound, and pathologic diagnosis. Am J Surg. 2022;224:1 Pt A.
Fass R, Boeckxstaens GE, El-Serag H, Rosen R, Sifrim D, Vaezi MF. Gastro-oesophageal reflux disease. Nat Reviews Disease Primers. 2021;7:1.
Goetze TO. Gallbladder carcinoma: prognostic factors and therapeutic options. World J Gastroenterol. 2015;21:43.
de Bitter TJJ, de Reuver PR, de Savornin Lohman EAJ, Kroeze LI, Vink-Börger ME, van Vliet S, et al. Comprehensive clinicopathological and genomic profiling of gallbladder cancer reveals actionable targets in half of patients. NPJ Precis Oncol. 2022;6:1.
Venneman NG, van Erpecum KJ. Gallstone disease: primary and secondary prevention. Best Pract Res Clin Gastroenterol. 2006;20:6.
Svoboda M, Sellner F, Ekmekcioglu C, Klimpfinger M, Jaeger W, Thalhammer T. Expression of estrogen-metabolizing enzymes and estrogen receptors in cholelithiasis gallbladder. Volume 62. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie; 2008. p. 10.
Dua A, Dua A, Desai SS, Kuy S, Sharma R, Jechow SE, et al. Gender based differences in management and outcomes of cholecystitis. Am J Surg. 2013;206:5.
Littlefield A, Lenahan C. Cholelithiasis: presentation and management. J Midwifery Womens Health. 2019;64:3.
Eusebi LH, Telese A, Cirota GG, Haidry R, Zagari RM, Bazzoli F, et al. Effect of gastro-esophageal reflux symptoms on the risk of Barrett’s esophagus: a systematic review and meta-analysis. J Gastroenterol Hepatol. 2022;37:8.
Souza RF. The role of acid and bile reflux in oesophagitis and Barrett’s metaplasia. Biochem Soc Trans. 2010;38:2.
Krishnamurthy S, Krishnamurthy GT. Biliary dyskinesia: role of the sphincter of Oddi, gallbladder and cholecystokinin. J Nucl Med. 1997;38:11.
Ustaoglu A, Woodland P. Esophageal afferent innervation and its role in gastro-esophageal reflux disease symptoms. Curr Opin Gastroenterol. 2021;37:4.
Zhu G-Y, Jia D-D, Yang Y, Miao Y, Wang C, Wang C-M. The Effect of Shaoyao Gancao Decoction on Sphincter of Oddi Dysfunction in Hypercholesterolemic rabbits via protecting the enteric nervous system-interstitial cells of cajal-smooth muscle cells Network. J Inflamm Res. 2021;14.
De Giorgio R, Barbara G, Furness JB, Tonini M. Novel therapeutic targets for enteric nervous system disorders. Trends Pharmacol Sci. 2007;28:9.
Debas HT, Yamagishi T. Evidence for a pyloro-cholecystic reflex for gallbladder contraction. Ann Surg. 1979;190:2.
Groneberg D, Zizer E, Lies B, Seidler B, Saur D, Wagner M, et al. Dominant role of interstitial cells of Cajal in nitrergic relaxation of murine lower oesophageal sphincter. J Physiol. 2015;593:2.
Sharma P, Yadlapati R. Pathophysiology and treatment options for gastroesophageal reflux disease: looking beyond acid. Ann N Y Acad Sci. 2021;1486:1.
Maev IV, Yurenev GL, Mironova EM, Yureneva-Thorzhevskaya TV. Phenotype of obesity and gastroesophageal reflux disease in the context of comorbidity in patients with cardiovascular diseases. Ter Arkh. 2019;91:2.
Björkman EVC, Edebo A, Oltean M, Casselbrant A. Esophageal barrier function and tight junction expression in healthy subjects and patients with gastroesophageal reflux disease: functionality of esophageal mucosa exposed to bile salt and trypsin in vitro. Scand J Gastroenterol. 2013;48:10.
Krolevets TS, Livzan MA, Lapteva IV. Gastroesophageal reflux disease in overweight various degrees. Eksp Klin Gastroenterol. 2016:9.
Siddiqui A, Rodriguez-Stanley S, Zubaidi S, Miner PB. Esophageal visceral sensitivity to bile salts in patients with functional heartburn and in healthy control subjects. Dig Dis Sci. 2005;50:1.
Dhiman RK, Reddi R, Sharma A, Singh R, Kohli KK, Gupta S, et al. Cisapride improves gallbladder emptying and bile lipid composition in patients with gallstones. J Gastroenterol Hepatol. 2001;16:7.
Wang XD, Soltesz V, Andersson R. Cisapride prevents enteric bacterial overgrowth and translocation by improvement of intestinal motility in rats with acute liver failure. Eur Surg Res. 1996;28:6.
Xu QW, Shaffer EA. Cisapride improves gallbladder contractility and bile lipid composition in an animal model of gallstone disease. Gastroenterology. 1993;105:4.
Sekula P, Del Greco MF, Pattaro C, Köttgen A. Mendelian randomization as an Approach to assess causality using Observational Data. J Am Soc Nephrology: JASN. 2016;27:11.
Carter AR, Sanderson E, Hammerton G, Richmond RC, Davey Smith G, Heron J, et al. Mendelian randomisation for mediation analysis: current methods and challenges for implementation. Eur J Epidemiol. 2021;36:5.
Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol. 2015;44:2.
Yavorska OO, Burgess S. MendelianRandomization: an R package for performing mendelian randomization analyses using summarized data. Int J Epidemiol. 2017;46:6.
Acknowledgements
We extend our appreciation to the participants and research teams who facilitated the public accessibility of the GWAS findings. We acknowledge the language assistance provided by Editage.
Funding
This work was supported by grants from the Wuxi Health Commission Youth Project (Q202242), Top Talent Support Program for Young and Middle-Aged People of the Wuxi Health Committee (HB2023046), and the National Natural Science Foundation of China (82303117).
Author information
Authors and Affiliations
Contributions
CW: conceptualization, data curation, writing of the original draft, methodology, formal analysis. JW: investigation, data curation, and formal analysis. MF: methodology, writing, review and editing. BJF: conceptualization, methodology, writing – review, and editing. All the authors have read and approved the final version of this manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Written informed consent for publication was obtained from all participants.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12876_2024_3390_MOESM1_ESM.docx
Additional File 1: Instrument variables of the causal effect of GERD on cholecystitis in MR analysis (Description: Instrument variables of the causal effect of GERD on cholecystitis in MR analysis)
12876_2024_3390_MOESM2_ESM.csv
Additional File 2: Instrumental variables (IVs) used in MR analysis of the association between GER and Cholecystitis among European populations (Description: Instrumental variables (IVs) used in MR analysis of the association between GER and Cholecystitis among European populations)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, C., Wang, J., Fang, M. et al. Genetic evidence causally linking gastroesophageal reflux disease to cholecystitis: a two-sample mendelian randomization study. BMC Gastroenterol 24, 301 (2024). https://doi.org/10.1186/s12876-024-03390-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12876-024-03390-w