Abstract
Preeclampsia is a leading cause of maternal and fetal/neonatal mortality and morbidity worldwide. The early identification of patients with an increased risk for preeclampsia is therefore one of the most important goals in obstetrics. The availability of highly sensitive and specific physiologic and biochemical markers would allow not only the detection of patients at risk but also permit a close surveillance, an exact diagnosis, timely intervention (e.g. lung maturation), as well as simplified recruitment for future studies looking at therapeutic medications and additional prospective markers. Today, several markers may offer the potential to be used, most likely in a combinatory analysis, as predictors or diagnostic tools. We present here the current knowledge on the biology of preeclampsia and review several biochemical markers which may be used to monitor preeclampsia in a future, that, we hope, is not to distant from today.
Similar content being viewed by others
Background
Preeclampsia occurs in 2–5% of pregnancies in the Occident, but it complicates up to 10% of pregnancies in the developing countries, where emergency care is often inadequate or lacking. Therefore we are in need of a widely applicable and affordable test that could permit presymptomatic diagnosis in order to identify and monitor the patients at risk and thus provide the best prenatal care for these women and their child. Such a test would also be of benefit to confirm a confounding clinical diagnosis and for future studies investigating prophylactic treatments or temporizing therapies.
To be effective a screening test need to be sufficiently sensitive and specific and must provide an adequate postive predictive value [1]. Today, several promising markers have been described, alone or in combination, that might fulfill these criteria. However, these data came often from small case studies with selected populations. Therefore, there is a need for worldwile large scale prospective studies to confirm the sensitivity and specificity of these promising markers and assess their utility in different subtypes of preeclampsia before they could serve in clinically useful screening tests.
Furthermore, when evaluating new screening strategies, not only sensitivity, specificity and predictive values should be taken into account, but also costs, patient's acceptability and quality control [2]. Thus, the implementation of clinical tests will require close collaboration between the medical institutions, optimally in a worldwide network, together with the pharmacieutical industry in order to develop functional and, as best as possible, affordable tests which could profit to the pregnant women worldwide.
Preeclampsia
Preeclampsia is a multi-system disorder of pregnancy, which is characterized by new onset hypertension (systolic and diastolic blood pressure of ≥ 140 and 90 mm Hg, respectively, on two occasions, at least 6 hours apart) and proteinuria (protein excretion of ≥ 300 mg in a 24 h urine collection, or a dipstick of ≥ 2+), that develop after 20 weeks of gestation in previously normotensive women [3, 4]. Dependent on the systemic involvement, several other symptoms, such as edema, disturbance of hemostasis, renal or liver failure, and the HELLP syndrome (hemolysis, elevated liver enzymes, and low platelet counts) also complicate the clinical picture. Preeclampsia can have an early onset (preeclampsia starting before 34 weeks of gestation) or late onset (preeclampsia starting after 34 weeks of gestation), can show mild or severe symptoms (systolic blood pressure ≥ 160 mmHg or diastolic blood pressure ≥ 110 mmHg, proteinuria >5 g/24 hours, oliguria, neurological symptoms, other clinical symptoms such as deranged liver function, thrombocytopenia < 100 000 mm3, HELLP syndrome), and can evolve in eclampsia in the most severe cases. In addition, it can manifest as a maternal disorder only, with an appropriate fetal growing, or it can present itself with a growth restricted fetus (in utero growth restriction (IUGR)) or sudden fetal distress.
The disorder has a higher incidence among nulliparous women, in women who conceive with assisted reproduction techniques, and in women affected by autoimmune disorders, reflecting the probable influence of an "inexperienced" or dysregulated maternal immune system in its emergence [5, 6]. On the other hand, women with pre-existing metabolic, vascular or renal disease are especially at increased risk for superimposed preeclampsia [7], possibly due to their elevated sensitivity to the mere normal physiological changes imposed by pregnancy itself.
Despite extensive clinical trials, up to date, no therapeutic approaches are available for either treatment or prevention of preeclampsia. Anti-hypertensive drugs, corticosteroids for lung maturation or magnesium sulfate to prevent from eclampsia (RCOG Guideline No. 10(A)) are given to handle (or prevent the worsening of) the symptoms and can thus temporize over the short term to allow for safe delivery with a more mature fetus. However, the maternal risks must be carefully weighted against the possible fetal benefits in temporizing management, as the risk of fatal deterioration of the maternal and/or fetal health condition is high. Several prophylactic therapies (anti-oxidant vitamins, calcium or folic acid supplementation, Aspirin) have so far failed to prove efficacious in the prevention of preeclampsia in healthy, nulliparous subjects, although some benefit has been shown in high risk groups (see [8–12] for a review on the different trials). As a consequence, the sole, though radical, resolution of preeclampsia is the removal of the placenta, and in case of prematurity, with the adverse consequence of delivering a pre-term baby. Therefore, preeclampsia, with or without IUGR, remains a major cause of maternal and neonatal mortality and morbidity worldwide.
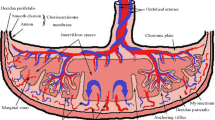
Placental pathophysiology in preeclampsia
The precise origin of preeclampsia remains elusive, but it is believed to be likely multifactorial. A certainty is the central role played by the placenta in its pathology [13–15]. A long standing hypothesis has been that preeclampsia develops as a consequence of some kind of immune maladaptation between the mother and the fetus during the very first weeks of pregnancy, leading to a 2-step disorder progression that can be summarized as following: in a first – asymptomatic – step, local aberrant feto-maternal immune interactions within the uterine wall lead to impaired tissue and arterial invasion by trophoblast cells. This results in failed transformation of the uterine spiral arteries and subsequently worsened placental perfusion. Chronic hypoxia or alternate periods of hypoxia/re-oxygenation within the intervillous space is expected to trigger tissue oxidative stress and increase placental apoptosis and necrosis [16, 17]. the clinical disorder arises, in a second step, when the maternal vascular and immune systems cannot handle any longer the increased shedding of placentally-produced debris and the aberrant expression of pro-inflammatory, anti-angiogenic and angiogenic factors, leading to a systemic endothelial cell dysfunction and an exaggerated inflammatory response [3, 18, 19]. Recently, this hypothesis has been challenged [20]. It was proposed instead that intrinsic failure in trophoblast differentiation at different time points of ontogeny may lead to either a mild disorder with late-onset appearance, or IUGR complicated or not with the maternal symptoms. However, the origin of preeclampsia might not be restricted to an alteration of trophoblast differentiation, but may also in some cases depend on an underlying maternal constitutional factors such as genetic, obesity, dysfunctional maternal clearance or inflammatory systems [21].
Potential benefits of biochemical markers in preeclampsia
Regardless of the lack of existing prophylactic and therapeutic means against preeclampsia, the search for non-invasive, blood-borne or urinary biomarkers that could predict the development or assist in the detection of this life-threatening pregnancy disorder is still of utmost importance. The availability of such markers could have decisive impact on the medical management of pregnant women and their child (e.g. refer to a tertiary centre) but also on the health costs associated with this poor medical condition. Since many years, different biophysical and biochemical markers have been investigated, based on pathophysiological observations that have been noted in case of preeclampsia, such as placental dysfunction, a generalized inflammatory response, endothelial dysfunction and activation of the coagulation system.
-
Miss-diagnosis is still an issue in hospital- or community midwifery care owing to the multiple clinical symptoms associated with the syndrome [22]. The availability of one or several reliable biochemical indicators might thus help to ascertain a clinical diagnosis.
-
Biochemical markers might allow the stratification of preeclamptic patients in different categories according to symptoms severity and/or pregnancy outcome and thus improve its clinical management [23].
-
Very importantly, biomarkers might ensure a reliable early disease assessment in asymptomatic pregnant women, in particular among target groups at increased risk based on their clinical history (preeclampsia or hypertension in a previous pregnancy) or pre-pregnancy state (hypertension, obesity, autoimmune disease are examples of the latter).
On account of the current understanding of the etiology of this life-threatening pregnancy disorder, a major focus of research has recently been the identification of placental factors showing abnormal expression in preeclamptic placentas and the assessment of their potential use for non-invasive early prediction or early detection. On the other hand, it appears that maternally-expressed proteins may also serve this purpose. We review below a selected choice of the most promising markers that have been identified up to now (Table 1), with an emphasis on the factors which have been the subject of large-scale studies and for whom the data are not disputed.
Biomarkers
Angiogenic factors
Angiogenesis requires the complex interplay between the pro-angiogenic factors vascular endothelial growth factor (VEGF) and placental growth factor (PlGF) with their cognate receptors VEGF receptor-1 (VEGFR-1, which is alternatively called fms-like tyrosine kinase (flt)-1) and VEGFR-2 (for a review on the function of these factors: [24]). Interestingly, the placenta is a rich source of these factors [25–28]. In addition to regulating blood vessel homeostasis, VEGF, PlGF and the flt-1 receptor have been shown to be key components in regulating trophoblast cell survival and function [25, 29–31].
Placental cells also secrete a soluble isoform of flt-1, which is generated through alternative splicing of the messenger RNA and acts as an anti-angiogenic factor by interacting with, and thereby neutralizing, PlGF and VEGF [32]. There is strong evidence for the occurrence of higher placental expression of sflt-1 and repeated findings of elevated circulating levels of sflt-1 and reduced free bioactive PlGF and VEGF in preeclamptic patients [18, 33–37]. It was thus suggested that a part of this excess of circulatory sflt-1 may stem from the placenta.
Maternal blood levels of sflt-1 were shown to correlate with the severity of preeclampsia, whereas, in an opposite manner, the quantities of bioactive VEGF and PlGF were further decreased in patients with severe symptoms compared to normal pregnant women or preeclamptic patients with mild symptoms [18, 33, 38]. Alterations in sflt-1 and PlGF are also more pronounced in early onset- in comparison to late onset preeclampsia [38, 39]. However, it was also shown that increased levels of sflt-1 were also associated with IUGR [40].
Remarkably, when introduced into pregnant rats, exogenous sflt-1 triggers hypertension and proteinuria, symptoms akin to those in preeclampsia [18]. Besides, condition medium of villous explants from preeclamptic placentas impaired vessel formation in vitro, which could be restored by prior immuno-depletion of sflt-1 from the conditioned medium [41]. It has therefore been proposed that the maternal endothelial dysfunction in preeclampsia was caused by the imbalance of the levels of circulatory angiogenic factors. Aberrant plasma or serum levels of sflt-1, VEGF and PlGF can indeed be measured prior the onset of the symptoms [42–47]. Increased amounts of sflt-1 are apparent in second trimester-, but not first trimester blood, in women destined to develop preeclampsia, whereas PlGF and VEGF levels already show alterations at the end of the first trimester of pregnancy in these patients. Three longitudinal studies comparing normotensive pregnancies and pregnancies with preeclampsia as an end-point, characterized the circulatory expression profile of these angiogenic factors [42, 43, 48]. In normotensive pregnancies, sflt-1 levels remain relatively stable until the last 2 months of gestation when they steadily increase. This increase is much more pronounced in pregnancies ending with preeclampsia and can discriminate this condition beginning approximately 5 to 8 weeks before the symptoms arise, in particular in cases with preterm (< 37 weeks) symptoms. In contrary to sflt-1, the levels of circulatory PlGF increase gradually and peak at mid gestation before declining again in uneventful pregnancies. PlGF concentration profile follows a similar pattern in women who later developed preeclampsia, however with decreased amplitude. PlGF concentrations are already significantly reduced at the end of the first trimester and remain lower throughout pregnancy. Yet, the difference in circulatory PlGF between normotensive pregnancies and those affected by preeclampsia is the highest within weeks of the onset of the clinical symptoms. As with sflt-1, the pre-symptomatic levels of circulatory PlGF seemed to correlate with the severity or time of onset of preeclampsia [49]. Urinary PlGF is likewise lower in preeclamptic patients before and at the time of symptoms [50, 51].
According to some studies, the presymptomatic alterations in sflt-1 levels appeared to be specific for preeclampsia as no changes are detected in women who later deliver SGA neonate or whose pregnancies are complicated by IUGR, compared to women with normal pregnancy outcome [48, 52]. However, others found that in a selected group of patients with abnormal uterine perfusion, similar alterations in sflt-1 and PlGF levels could be detected during the second trimester in cases with subsequent IUGR [53]. Nevertheless, owing to the evolving unbalance of angiogenic factors after 25 weeks of gestation in women with subsequent preeclampsia, the ratio sflt-1/PlGF has been advocated to be a reliable marker of overall preeclampsia risk. As a matter of fact, soluble flt-1 and PlGF have been launched by Roche as a screening test for preeclampsia in the second trimester in Europe and is expected to be submitted to the FDA soon.
It has recently been reported that patients with preeclampsia have lower plasma concentrations of soluble VEGF-R2 [54]. However, this biomarker may not be specific for preeclampsia as an equivalent decrease was observed in patients with SGA babies in the absence of preeclampsia.
Soluble Endoglin
Endoglin (Eng) is a co-receptor for transforming growth factor (TGF)-β1 and TGF-β3 that is highly expressed on cellular membranes of the vascular endothelium and on the syncytiotrophoblast [55, 56]. It functions as a modulator of TGF-β signaling and is involved in angiogenesis and the regulation of the vascular tone [57, 58]. A circulatory form of endoglin, which consists of the extra cellular part of the molecule that may be produced through the proteo-cleavage of the placental membrane-bound form, has been identified in normal pregnancy and in preeclampsia [59]. In vitro, sEng acts as a negative regulator of angiogenesis by competitive interaction with TGF-β, thereby impairing capillary formation by endothelial cells. Furthermore, it induces high arterial pressure and vascular permeability in pregnant rats in which the protein was over-expressed. Very interestingly, the combined introduction of sEng and sflt-1 in the pregnant animals induced renal, placental and hepatic changes reminiscent of the HELLP syndrome [59].
Soluble Eng is present in substantial excess in preeclamptic patients compared to normotensive controls, and its concentrations appear to increase with the severity of the symptoms and are the highest in preeclampsia complicated by the HELLP symptom [40, 59, 60]. Pregnancies with IUGR without the maternal syndrome may also be characterized by elevated levels of sEng, suggesting that this factor is not specific for preeclampsia, but may be a marker for clinical conditions associated with an underlying placental pathology [40, 60]. However, these results remain conflicting as others demonstrated no association between IUGR and the levels of sEng [60]. Moreover, a pilot study has suggested that sEng may prove useful in differentiating preeclampsia from other hypertensive diseases of pregnancy, such as gestational- or chronic hypertension [61]. Large scale studies will be needed in order to clarify these important issues.
Like sflt-1, sEng concentrations raise during the last 2 months of normal pregnancy. In pregnancies ending with preeclampsia, this increase occurs earlier and is steeper [48, 62–65]. The distinction becomes significant starting 9–11 weeks before the clinical symptoms, for both early and late onset preeclampsia, but is more prominent for preterm preeclampsia or in women in whom preeclampsia is complicated with SGA. Altered levels of this factor throughout gestation are also associated with SGA pregnancies without the maternal symptoms [48, 66]. Thus, a specific prediction can not be achieved with this analyte alone.
Several longitudinal case-control studies have therefore evaluated the potential of sEng in combination with the pro- and anti-angiogenic factors PlGF and sflt-1 for the prediction of preeclampsia [48, 62, 63]. The studies reported that the pattern of changes in the ratio of different combinations of these factors (PlGF/sEng; (sflt-1+sEng)/PlGF; etc), collected at 13 weeks and around 20 weeks, was more informative than the individual biomarkers at single time-point screening. One study suggested that a rigorous monitoring of the sequential changes in the profile of these three biomarkers between the first and the second trimesters permits sensitive and specific risk assessment [66]. A change in PlGF/sEng ratio that was below the median slope for controls conferred an odds ratio of 7.68 for the development of pre-term preeclampsia, and 2.46 for the development of term preeclampsia, and discriminated SGA pregnancies from preeclampsia. Further studies with large number of patients will be required to confirm these very promising preliminary results and assess the utility of analyzing these biomarkers in the clinical routine.
P-Selectin
P-selectin is a member of the selectin family of cell surface adhesion molecules. It is expressed by platelets and endothelial cells upon activation and plays crucial roles in inflammatory reactions by supporting the recruitment and activation of circulating leucocytes, and in coagulation through the generation of leukocyte-derived "blood-borne" tissue factor [67, 68]. P-selectin is rapidly shed from the cellular membrane of activated platelets and this release is suggested to contribute to most of the soluble isoform of the molecule that is found in the plasma [69].
Preeclampsia is associated with extensive platelet activation [70–72]. P-selectin-exposing micro particles with procoagulant activity, released from activated platelets, have been detected in the peripheral blood of preeclamptic women [73, 74]. In addition, soluble P-selectin has been repeatedly, though not constantly, observed in higher amounts in serum or plasma of patients with this disorder [75–78].
Interestingly, it has recently been shown that alterations in the levels of soluble P-selectin before 20 weeks of gestation antedate the symptoms [79–81]. This early up-regulation of soluble P-selectin has been suggested to reflect the early but still asymptomatic disturbances of the maternal vascular system. In one of these studies, P-selectin was identified as the marker with the highest discriminatory ability among three biomolecules evaluated between gestational weeks 11 to 15 [79]. However, the combination of P-selectin with the two other markers, namely Activin A and VEGFR, showed a detection rate of only 59% (with a false-positive rate of 5%), which is not sufficient for a possible routine clinical implementation as a screening test.
Cell-free fetal DNA
Since its detection in maternal plasma many approaches have been tested to use cell free fetal DNA for non-invasive diagnostic approaches. These include qualitative analyses like fetal sex analysis [82], determination of the fetal Rhesus status [83, 84] or the analysis of fetal point mutations [85] as well as the quantitative analysis as an indicator for several fetal anomalies, e.g. fetal growth restriction [86], polyhydramnios [87], trisomy [88–90] or preterm labor [91]. The value of cffDNA in maternal plasma as an indicator for preeclampsia has first been reported by Lo et al. in a small scale study in the plasma of 20 preeclamptic women and 20 gestational age matched controls in the third trimester, where cffDNA was increased approximately 5-fold in women with preeclampsia [92]. The same effect was observed in the second trimester in a study by Zhong et al. in 10 preeclamptic women and 40 controls [93]. The so far biggest study in that field was conducted by Levine et al. with 120 preeclamptic women and 120 controls: A two- to five-fold increase of cffDNA levels was monitored starting from week 17 until three weeks before the onset of preeclampsia [94]. As the amount of fetal DNA is routinely determined by quantifying Y-chromosome specific sequences, e.g. SRY (sex determining region Y) and DYS [95], alternative approaches have been tested to overcome this limitation: An increase of total cell free DNA was observed in women with preeclampsia at term [96–98] and before the onset of preeclampsia [98]. Furthermore, approaches to analyze cffDNA independent from fetal sex, using epigenetic differences between maternal and fetal DNA have been developed, e.g. the use of the maspin gene, which is hypomethylated in fetal tissue [99] or the hypermethylated fetal promoter sequence of RASSF1A [100]. Although these approaches are promising, only one study quantifying cffDNA with the RASFF1A approach in 10 women with preeclampsia and 20 controls has been published [101]. cffDNA has shown some predictive value for the prediction of preeclampsia between 20–25 weeks of gestation, however, higher sensitivities and specificities can be obtained by combining several markers as has been shown in a nested case-control study for cell free DNA combined with Inhibin A in the second (n = 15 at risk for PE), n = 68 controls) and third trimester (n = 34 preeclampsia, n = 44 controls) [102]. Currently, several multicenter studies are being performed to confirm the predictive value of cffDNA to predict and monitor preeclampsia in combination with other potential markers, e.g. P-selectin, PAPP-A, PP-13, sflt-1, sEng, PlGF).
ADAM12
ADAM12 (a disintegrin and metalloprotease 12) is a membrane bound zinc dependant protease and belongs to the ADAM protein family, a group of proteins involved in cell-cell and cell-matrix interactions in fertilization, muscle development and neurogenesis [103–105]. For this gene, two alternatively spliced transcripts are known, a short secreted form and a long membrane-bound form [106]. The plasma concentration of ADAM12 has been found to be altered in several pregnancy related disorders. Several studies have demonstrated that the plasma level of ADAM12 is decreased in women carrying a fetus with trisomy 21 and trisomy 18 [107–110]. It has also been shown that the ADAM12 concentration is decreased in women with other aneuploidies and in women with low for gestational age birth weights [111]. The first connection of ADAM12 serum levels to preeclampsia was demonstrated by Laigaard et al. in a study with 160 women with preeclampsia and 324 healthy controls in the first trimester [112]. The serum concentration of ADAM12 was significantly decreased in women that later developed preeclampsia. These results were confirmed by Spencer et al. in a study with two groups (1. n = 64 PE, n = 240 controls, 2: n = 24 cases, n = 144 controls) [113]. However another study failed to confirm these promising results but concluded that measurement of ADAM12 does not provide useful prediction of SGA, preeclampsia, or spontaneous preterm delivery [114].
PP-13
Placental protein 13 (PP-13, galectin-13) was first isolated in 1983 by Bohn et al. [115, 116]. It is a relatively small protein with 139 amino acids (16,118 kDa) which is highly homologous (69%) to the human eosinophil Charcot-Leyden Crystal protein, a phospholipase that belongs to the beta-galactoside binding S-type animal lectin super family. The homodimer which is linked by disulfide bonds probably has special haemostatic and immunobiological functions at the feto-maternal interface or a developmental role in the placenta [117]. The 600 bp mRNA transcript is only detectable in placental tissue but not in any other fetal or adult tissue [115, 116, 118, 119]. The serum levels of PP-13 slowly increase during a normal pregnancy but abnormally low levels of PP-13 were detected in first trimester serum samples of women subsequently developing fetal growth restriction and preeclampsia, in particular cases with early onset [120–124]. Elevated serum concentrations of PP-13 have been found in the second and third trimester in women with preeclampsia, IUGR and in preterm delivery [123]. For this study 514 controls, 69 cases with preeclampsia, 69 cases with IUGR, 52 cases with preterm delivery and 24 cases with preeclampsia developing before 34 weeks of gestation have been included. Another study concluded that first-trimester serum levels of PP-13 may serve as a suitable marker for preterm preeclampsia but are weak for the prediction of severe preeclampsia and ineffective for mild preeclampsia at term [125].
Here again the combination of several diagnostic tools results in improved predictive power as was shown by combined measuring of first trimester serum PP-13 levels and median uterine artery pulsatility index by ultrasound. This combination achieved a detection rate for preeclampsia of 90% with a false positive rate of 6% [126]. However, this combination of serum PP-13 levels and uterine artery pulsatility index loses its predictive power when late second trimester (22–24 weeks of gestation) serum is analyzed [127]. Currently a commercial PP-13 test kit is developed for the first trimester screening for preeclampsia by Diagnostic Technologies, Haifa. The test has already been approved in Europe and approval in the United States is expected in the near future.
PTX3
Pentraxin 3 (PTX3, tumor necrosis factor stimulated gene-14 [128]) belongs to the same family as C-reactive protein (CRP) or serum amyloid P component (SAP) and consists of 381 amino acids. The C-terminus is highly homologous to SAP and CRP whereas the N-terminus doesn't show any homology to other proteins. The according gene is organized into three exons [129] and is extremely evolutionarily conserved from horseshoe crab to human [130]. Responding to proinflammatory stimuli CRP, SAP and PTX3 are produced by various tissues. It is also expressed in tissues undergoing cell death. PTX3 then interacts with several growth factors, extra cellular matrix components and certain pathogens but is also involved in the activation of the complement system [131] and facilitates pathogen recognition by phagocytes [132]. During pregnancy, PTX3 is increasingly expressed in amniotic epithelium, chorionic mesoderm, trophoblast terminal villi, and perivascular stroma of placentae [132]. Cetin et al. and Rovere-Querini et al. showed that in case of a future preeclampsia and IUGR the PTX3 plasma levels are even more increased in all three trimesters [133, 134]. So far no studies that combine PTX3 with other potential markers have been performed.
PAPP-A
PAPP-A (pregnancy-associated plasma protein A, pappalysin 1, insulin-like growth factor binding protein-4 protease, EC 3.4.24.79) is a disulfide bond linked homodimeric peptidase of 1628 amino acids and a mass of 400 kDa [135]. It can be detected during pregnancy in maternal circulation mainly as a complex with the proform of the eosinophil major basic protein, an inhibitor of PAPP-A [136, 137]. Although the reaction products are not identified yet, insulin-like growth factor binding proteins are substrates for the hydrolytic activity of PAPP-A [138]. PAPP-A is supposedly involved in local proliferative processes, for example bone remodeling [139, 140]. In the recent years decreased plasma levels of PAPP-A have been reported in all trimesters in women with preeclampsia [141–150]. Furthermore, a correlation between birth weight and maternal PAPP-A plasma levels have been reported [151].
Recent candidates
Visfatin
Visfatin (nicotinamide phosphoribosyltransferase (Nampt) enzyme, EC 2.4.2.12) is an adipokine secreted by adipose tissue and involved in the biosynthesis of nicotinamide adenine dinucleotide as it catalyzes the condensation of nicotinamide with 5-phosphoribosyl-1-pyrophosphate to yield nicotinamide mononucleotide [152, 153]. Several studies have shown that this protein shows altered plasma levels in different disorders, e.g. type-2 diabetes mellitus [154], obesity [155], fetal growth retardation [156] and gestational diabetes mellitus [157]. A recent cross sectional study by Hu et al. with 27 preeclampsia cases, 28 pregnant women in the third trimester and 28 non-pregnant women demonstrated that the maternal plasma visfatin levels were significantly decreased in women with mild preeclampsia and even more decreased in women with severe preeclampsia [158]. However, the exact opposite was reported by Fasshauer et al. [159], indicating that further, larger studies are necessary to evaluate the potential of Visfatin as a marker for preeclampsia.
Adrenomedullin
Adrenomedullin is a peptide consisting of 52 amino acids that exhibits a long lasting hypotensive effect and was first isolated from pheochromocytoma by Kitamura et al. in 1993 [160]. It is expressed in all organs but predominantly in vascular endothelial cells and vascular smooth muscle cells where it regulates circulation and endothelial permeability [161, 162]. The plasma level of adrenomedullin is elevated in several disorders, mostly in those resulting from pathologic processes in vasculature [163]. A medium scale study with 90 women (each 15 normotensive women in the first, second and third trimester, 15 women with preeclampsia between 25–38 weeks of gestation, 15 normotensive non-pregnant women and 15 hypertensive non-pregnant women) by Senna et al. showed that the plasma adrenomedullin levels increase during normal pregnancies. In preeclampsia the AM plasma levels are higher compared to normal pregnancies [164]. These results agree with the finding that elevated AM mRNA levels were found in placental tissue [165]. As AM acts as vasodilator it makes sense to assume that increased AM levels are increased to act against the preeclamptic hypertension.
Auto antibodies against the angiotensin II type 1 (AT1) receptor
This is an old actor whose role in the pathogenesis of preeclampsia is currently being reevaluated. There is long time evidence for a role for the renin-angiotensin system in pregnancy. Exactly a decade ago, it was discovered that patients with preeclampsia develop agonistic autoantibodies against the second extracellular loop of the AT1 receptor [166]. Several in vitro studies confirmed that these antibodies can induce a profusion of responses related to hypertensive disorder in vascular cells and reduce the migration of trophoblast cells (reviewed in [167]). However, they are not specific to preeclampsia but rather mark pregnancies with abnormal uterine artery Doppler flow, such that they are also found in the circulation of normotensive women with IUGR [168]. Furthermore, they are present in recipients of unsuccessful kidney allografts who develop malignant hypertension [169]. At the time, these data suggested that the AT1 autoantibodies might be a mere secondary response to a hypoxic vascular insult. However, a recent study demonstrated a link between these autoantibodies and the overproduction of sflt-1 in human trophoblast cells or placental explants [170], raising a new interest for these molecules. Recently, a small prospective, nested, case control study has suggested that AT1 autoantibodies were common in patients with preeclampsia and, although sflt-1 was superior for early-onset preeclampsia, the AT1 autoantibodies may represent a better marker for preeclampsia developing at term [171]. Nevertheless, several questions remain to be answered before the AT1 autoantibodies could be used in a diagnosis strategy. First, no data exist as to the presence of these molecules before the pregnancy or prior the symptoms develop. Second, the detection still relies on a cumbersome bioassay, which prevents the initiation of large scale studies.
Doppler ultrasonography of the uterine arteries
The inadequate placental perfusion has lead to the use of Doppler ultrasonography to assess the velocity of the blood flow in the uterine arteries. A persistence of an early diastolic notch after 24 weeks of gestation or abnormal flow velocity ratio's has been associated with an inadequate trophoblast invasion. Pregnancies associated with an abnormal uterine Doppler after 24 weeks of gestation (high pulsatility index and/or presence of an early diastolic notch) are associated with a more than six fold increase in the rate of preeclampsia [172].
Among high-risk patients with a previous preeclampsia, doppler ultrasound of the uterine arteries has an excellent negative predictive value, thus it is an important tool in patient management and care which is of paramount benefit for patients with PE in a previous pregnancy. However effort must be made so that this sophisticated technology becomes available in centers throughout underdeveloped countries.
A recently published systematic review assessed the use of Doppler ultrasonography in case of preeclampsia [173]. A total of 74 studies (69 cohort studies, 3 randomized controlled trials and 2 case-control studies with a total of 79'547 patients, of whom 2498 developed preeclampsia, were included. The authors showed that Doppler ultrasonography of the uterine arteries were less accurate in the first trimester, than in the second trimester. The combined data showed that the pulsatility index, alone or in combination with a persistent notching after 24 weeks of gestation is the most predictive parameter of Doppler ultrasonography to predict preeclampsia. This parameter may be used, especially in combination with other biomarkers.
Current data do not support the use of Doppler ultrasonography for routine screening of patients for preeclampsia [174]. However several studies show that the combination of the measurement of uterine perfusion in the second trimester and analysis of angiogenic markers have a high detection rate, especially for early onset preeclampsia [53, 65, 175].
Identification of novel biomarkers
Although a panel of promising biomarkers already exists, a lot of effort is made to find novel candidates that bear a greater potential to identify women at risk for preeclampsia, in order to provide the best possible care for these mothers and children. Several approaches for the identification of novel potential biomarkers can be applied. Microarrays offer the possibility to rapidly screen the placental transcriptome for up- and down-regulated transcripts in preeclamptic samples compared to healthy controls hoping that the resulting proteins are excreted and detectable in maternal plasma. Comparative transcription analyses have been performed to some extent by several groups [17, 176–183], although those groups were studying the molecular mechanisms of preeclampsia rather than potential biomarkers. Recently, the microarray based screening for RNA molecules in maternal circulation that are transcribed in placenta but not maternally has been reported as a potential source for pregnancy related biomarkers [184–187], including preeclampsia [188]. As the microarray technology is evolving rapidly regarding feature size and the number of available and well characterized genes, these experiments are performed continuously.
A more direct approach is to compare the proteome in the maternal circulation as transcriptome and proteome do not comply with each other. To cope with the complexity of human plasma, high throughput methods need to be employed. The classic approach is comparative 2D-gelelectrophoresis of albumin depleted plasma and subsequent mass spectrometric analysis of the remaining proteins that show different quantities [189–191]. This approach is currently being replaced by a mass spectrometric technique based on the iTRAQ® reagent by Applied Biosystems [192]. Up to eight conditions can be simultaneously compared by labeling them with defined mass-tags. After fragmentation, mass analysis, identification and quantification the mass-tags allow the allocation of the proteins to each sample-pool. Therefore, this technique allows a global quantitative comparison of complex body fluids like plasma [192–195].
A new direction of research in the field of complex diseases is metabolomics, which consists in the global analysis of endogenous and secreted metabolites in a biological system. The first reports investigating the placental metabolome under varying oxygen tensions or in plasma of preeclamptic patients revealed novel redox biomarkers and suggest that this technology might bring new insights into placental function [196, 197].
However, the different -omics profiling techniques are not suited for simple, low-cost, and rapid routine clinical screenings. Their use is primarily for the identification of novel biomarkers within global analyte groups. The next step is then to develop straightforward methods specifically designed for use in a hospital environment.
Conclusion
Despite there exists many different potential markers for preeclampsia, the reliability of these markers in predicting preeclampsia has been inconsistent between different studies. Furthermore, preeclampsia is a multifaceted disorder, certain say it is not one but several diseases. Therefore, there is a need for high quality, large scale multi-center trials which enroll patients with different risks of developing the syndrome and throughout multi-ethnical background, in order to assess the predictive value of different markers and finally propose the best marker combination for a routine use in clinical settings.
References
Wilson JMG, Jungner G: Principles and Practice of Screening for Disease. Public Health Paper Number 34. 1968, Geneva: WHO
Audibert F: Screening for pre-eclampsia: the quest for the Holy Grail?. Lancet. 2005, 365: 1367-1369.
Redman CW, Sargent IL: Latest advances in understanding preeclampsia. Science. 2005, 308: 1592-1594.
Sibai B, Dekker G, Kupferminc M: Pre-eclampsia. Lancet. 2005, 365: 785-799.
Saito S, Shiozaki A, Nakashima A, Sakai M, Sasaki Y: The role of the immune system in preeclampsia. Mol Aspects Med. 2007, 28: 192-209.
Sargent IL, Borzychowski AM, Redman CW: Immunoregulation in normal pregnancy and pre-eclampsia: an overview. Reprod Biomed Online. 2006, 13: 680-686.
Catov JM, Ness RB, Kip KE, Olsen J: Risk of early or severe pre-eclampsia related to pre-existing conditions. Int J Epidemiol. 2007, 36: 412-419.
Wen SW, Chen XK, Rodger M, White RR, Yang Q, Smith GN, Sigal RJ, Perkins SL, Walker MC: Folic acid supplementation in early second trimester and the risk of preeclampsia. Am J Obstet Gynecol. 2008, 198: 45-47.
Bucher HC, Guyatt GH, Cook RJ, Hatala R, Cook DJ, Lang JD, Hunt D: Effect of calcium supplementation on pregnancy-induced hypertension and preeclampsia: a meta-analysis of randomized controlled trials. JAMA. 1996, 275: 1113-1117.
Coomarasamy A, Honest H, Papaioannou S, Gee H, Khan KS: Aspirin for prevention of preeclampsia in women with historical risk factors: a systematic review. Obstet Gynecol. 2003, 101: 1319-1332.
Duley L, Henderson-Smart DJ, Meher S, King JF: Antiplatelet agents for preventing pre-eclampsia and its complications. Cochrane Database Syst Rev. 2007, CD004659-
Rumiris D, Purwosunu Y, Wibowo N, Farina A, Sekizawa A: Lower rate of preeclampsia after antioxidant supplementation in pregnant women with low antioxidant status. Hypertens Pregnancy. 2006, 25: 241-253.
Hahn S, Gupta AK, Troeger C, Rusterholz C, Holzgreve W: Disturbances in placental immunology: ready for therapeutic interventions?. Springer Semin Immunopathol. 2006, 27: 477-493.
Huppertz B: The feto-maternal interface: setting the stage for potential immune interactions. Semin Immunopathol. 2007, 29: 83-94.
Jauniaux E, Poston L, Burton GJ: Placental-related diseases of pregnancy: Involvement of oxidative stress and implications in human evolution. Hum Reprod Update. 2006, 12: 747-755.
Hung TH, Skepper JN, Charnock-Jones DS, Burton GJ: Hypoxia-reoxygenation: a potent inducer of apoptotic changes in the human placenta and possible etiological factor in preeclampsia. Circ Res. 2002, 90: 1274-1281.
Soleymanlou N, Jurisica I, Nevo O, Ietta F, Zhang X, Zamudio S, Post M, Caniggia I: Molecular evidence of placental hypoxia in preeclampsia. J Clin Endocrinol Metab. 2005, 90: 4299-4308.
Maynard SE, Min JY, Merchan J, Lim KH, Li J, Mondal S, Libermann TA, Morgan JP, Sellke FW, Stillman IE, Epstein FH, Sukhatme VP, Karumanchi SA: Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest. 2003, 111: 649-658.
Roberts JM, Taylor RN, Musci TJ, Rodgers GM, Hubel CA, McLaughlin MK: Preeclampsia: an endothelial cell disorder. Am J Obstet Gynecol. 1989, 161: 1200-1204.
Huppertz B: Placental origins of preeclampsia: challenging the current hypothesis. Hypertension. 2008, 51: 970-975.
Roberts JM, Hubel CA: The two stage model of preeclampsia: variations on the theme. Placenta. 2009, 30 (Suppl A): S32-S37.
Schutte JM, Schuitemaker NW, van RJ, Steegers EA: Substandard care in maternal mortality due to hypertensive disease in pregnancy in the Netherlands. BJOG. 2008, 115: 732-736.
von Dadelszen P, Magee LA, Roberts JM: Subclassification of preeclampsia. Hypertens Pregnancy. 2003, 22: 143-148.
Tjwa M, Luttun A, Autiero M, Carmeliet P: VEGF and PlGF: two pleiotropic growth factors with distinct roles in development and homeostasis. Cell Tissue Res. 2003, 314: 5-14.
Ahmed A, Li XF, Dunk C, Whittle MJ, Rushton DI, Rollason T: Colocalisation of vascular endothelial growth factor and its Flt-1 receptor in human placenta. Growth Factors. 1995, 12: 235-243.
Clark DE, Smith SK, Sharkey AM, Charnock-Jones DS: Localization of VEGF and expression of its receptors flt and KDR in human placenta throughout pregnancy. Hum Reprod. 1996, 11: 1090-1098.
Clark DE, Smith SK, Licence D, Evans AL, Charnock-Jones DS: Comparison of expression patterns for placenta growth factor, vascular endothelial growth factor (VEGF), VEGF-B and VEGF-C in the human placenta throughout gestation. J Endocrinol. 1998, 159: 459-467.
Khaliq A, Li XF, Shams M, Sisi P, Acevedo CA, Whittle MJ, Weich H, Ahmed A: Localisation of placenta growth factor (PIGF) in human term placenta. Growth Factors. 1996, 13: 243-50.
Crocker IP, Strachan BK, Lash GE, Cooper S, Warren AY, Baker PN: Vascular endothelial growth factor but not placental growth factor promotes trophoblast syncytialization in vitro. J Soc Gynecol Investig. 2001, 8: 341-346.
Desai J, Holt-Shore V, Torry RJ, Caudle MR, Torry DS: Signal transduction and biological function of placenta growth factor in primary human trophoblast. Biol Reprod. 1999, 60: 887-892.
Shore VH, Wang TH, Wang CL, Torry RJ, Caudle MR, Torry DS: Vascular endothelial growth factor, placenta growth factor and their receptors in isolated human trophoblast. Placenta. 1997, 18: 657-665.
Clark DE, Smith SK, He Y, Day KA, Licence DR, Corps AN, Lammoglia R, Charnock-Jones DS: A vascular endothelial growth factor antagonist is produced by the human placenta and released into the maternal circulation. Biol Reprod. 1998, 59: 1540-1548.
Chaiworapongsa T, Romero R, Espinoza J, Bujold E, Mee KY, Goncalves LF, Gomez R, Edwin S: Evidence supporting a role for blockade of the vascular endothelial growth factor system in the pathophysiology of preeclampsia. Young Investigator Award. Am J Obstet Gynecol. 2004, 190: 1541-1547.
Chung JY, Song Y, Wang Y, Magness RR, Zheng J: Differential expression of vascular endothelial growth factor (VEGF), endocrine gland derived-VEGF, and VEGF receptors in human placentas from normal and preeclamptic pregnancies. J Clin Endocrinol Metab. 2004, 89: 2484-2490.
Levine RJ, Karumanchi SA: Circulating angiogenic factors in preeclampsia. Clin Obstet Gynecol. 2005, 48: 372-386.
Torry DS, Wang HS, Wang TH, Caudle MR, Torry RJ: Preeclampsia is associated with reduced serum levels of placenta growth factor. Am J Obstet Gynecol. 1998, 179: 1539-1544.
Tsatsaris V, Goffin F, Munaut C, Brichant JF, Pignon MR, Noel A, Schaaps JP, Cabrol D, Frankenne F, Foidart JM: Overexpression of the soluble vascular endothelial growth factor receptor in preeclamptic patients: pathophysiological consequences. J Clin Endocrinol Metab. 2003, 88: 5555-5563.
Robinson CJ, Johnson DD, Chang EY, Armstrong DM, Wang W: Evaluation of placenta growth factor and soluble Fms-like tyrosine kinase 1 receptor levels in mild and severe preeclampsia. Am J Obstet Gynecol. 2006, 195: 255-259.
Wikstrom AK, Larsson A, Eriksson UJ, Nash P, Norden-Lindeberg S, Olovsson M: Placental growth factor and soluble FMS-like tyrosine kinase-1 in early-onset and late-onset preeclampsia. Obstet Gynecol. 2007, 109: 1368-1374.
Stepan H, Kramer T, Faber R: Maternal plasma concentrations of soluble endoglin in pregnancies with intrauterine growth restriction. J Clin Endocrinol Metab. 2007, 92: 2831-2834.
Ahmad S, Ahmed A: Elevated placental soluble vascular endothelial growth factor receptor-1 inhibits angiogenesis in preeclampsia. Circ Res. 2004, 95: 884-891.
Chaiworapongsa T, Romero R, Kim YM, Kim GJ, Kim MR, Espinoza J, Bujold E, Goncalves L, Gomez R, Edwin S, Mazor M: Plasma soluble vascular endothelial growth factor receptor-1 concentration is elevated prior to the clinical diagnosis of pre-eclampsia. J Matern Fetal Neonatal Med. 2005, 17: 3-18.
Levine RJ, Maynard SE, Qian C, Lim KH, England LJ, Yu KF, Schisterman EF, Thadhani R, Sachs BP, Epstein FH, Sibai BM, Sukhatme VP, Karumanchi SA: Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med. 2004, 350: 672-683.
Polliotti BM, Fry AG, Saller DN, Mooney RA, Cox C, Miller RK: Second-trimester maternal serum placental growth factor and vascular endothelial growth factor for predicting severe, early-onset preeclampsia. Obstet Gynecol. 2003, 101: 1266-1274.
Thadhani R, Mutter WP, Wolf M, Levine RJ, Taylor RN, Sukhatme VP, Ecker J, Karumanchi SA: First trimester placental growth factor and soluble fms-like tyrosine kinase 1 and risk for preeclampsia. J Clin Endocrinol Metab. 2004, 89: 770-775.
Tidwell SC, Ho HN, Chiu WH, Torry RJ, Torry DS: Low maternal serum levels of placenta growth factor as an antecedent of clinical preeclampsia. Am J Obstet Gynecol. 2001, 184: 1267-1272.
Tjoa ML, van Vugt JM, Mulders MA, Schutgens RB, Oudejans CB, van WI: Plasma placenta growth factor levels in midtrimester pregnancies. Obstet Gynecol. 2001, 98: 600-607.
Romero R, Nien JK, Espinoza J, Todem D, Fu W, Chung H, Kusanovic JP, Gotsch F, Erez O, Mazaki-Tovi S, Gomez R, Edwin S, Chaiworapongsa T, Levine RJ, Karumanchi SA: A longitudinal study of angiogenic (placental growth factor) and anti-angiogenic (soluble endoglin and soluble vascular endothelial growth factor receptor-1) factors in normal pregnancy and patients destined to develop preeclampsia and deliver a small for gestational age neonate. J Matern Fetal Neonatal Med. 2008, 21: 9-23.
Moore Simas TA, Crawford SL, Solitro MJ, Frost SC, Meyer BA, Maynard SE: Angiogenic factors for the prediction of preeclampsia in high-risk women. Am J Obstet Gynecol. 2007, 197: 244-248.
Aggarwal PK, Jain V, Sakhuja V, Karumanchi SA, Jha V: Low urinary placental growth factor is a marker of pre-eclampsia. Kidney Int. 2006, 69: 621-624.
Levine RJ, Thadhani R, Qian C, Lam C, Lim KH, Yu KF, Blink AL, Sachs BP, Epstein FH, Sibai BM, Sukhatme VP, Karumanchi SA: Urinary placental growth factor and risk of preeclampsia. JAMA. 2005, 293: 77-85.
Wathen KA, Tuutti E, Stenman UH, Alfthan H, Halmesmaki E, Finne P, Ylikorkala O, Vuorela P: Maternal serum-soluble vascular endothelial growth factor receptor-1 in early pregnancy ending in preeclampsia or intrauterine growth retardation. J Clin Endocrinol Metab. 2006, 91: 180-184.
Stepan H, Unversucht A, Wessel N, Faber R: Predictive value of maternal angiogenic factors in second trimester pregnancies with abnormal uterine perfusion. Hypertension. 2007, 49: 818-824.
Chaiworapongsa T, Romero R, Gotsch F, Espinoza J, Nien JK, Goncalves L, Edwin S, Kim YM, Erez O, Kusanovic JP, Pineles BL, Papp Z, Hassan S: Low maternal concentrations of soluble vascular endothelial growth factor receptor-2 in preeclampsia and small for gestational age. J Matern Fetal Neonatal Med. 2008, 21: 41-52.
Cheifetz S, Bellon T, Cales C, Vera S, Bernabeu C, Massague J, Letarte M: Endoglin is a component of the transforming growth factor-beta receptor system in human endothelial cells. J Biol Chem. 1992, 267: 19027-19030.
St-Jacques S, Forte M, Lye SJ, Letarte M: Localization of endoglin, a transforming growth factor-beta binding protein, and of CD44 and integrins in placenta during the first trimester of pregnancy. Biol Reprod. 1994, 51: 405-413.
Duff SE, Li C, Garland JM, Kumar S: CD105 is important for angiogenesis: evidence and potential applications. FASEB J. 2003, 17: 984-992.
Toporsian M, Gros R, Kabir MG, Vera S, Govindaraju K, Eidelman DH, Husain M, Letarte M: A role for endoglin in coupling eNOS activity and regulating vascular tone revealed in hereditary hemorrhagic telangiectasia. Circ Res. 2005, 96: 684-692.
Venkatesha S, Toporsian M, Lam C, Hanai J, Mammoto T, Kim YM, Bdolah Y, Lim KH, Yuan HT, Libermann TA, Stillman IE, Roberts D, D'Amore PA, Epstein FH, Sellke FW, Romero R, Sukhatme VP, Letarte M, Karumanchi SA: Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat Med. 2006, 12: 642-649.
Jeyabalan A, McGonigal S, Gilmour C, Hubel CA, Rajakumar A: Circulating and placental endoglin concentrations in pregnancies complicated by intrauterine growth restriction and preeclampsia. Placenta. 2008, 29: 555-563.
Salahuddin S, Lee Y, Vadnais M, Sachs BP, Karumanchi SA, Lim KH: Diagnostic utility of soluble fms-like tyrosine kinase 1 and soluble endoglin in hypertensive diseases of pregnancy. Am J Obstet Gynecol. 2007, 197: 28-6.
Levine RJ, Lam C, Qian C, Yu KF, Maynard SE, Sachs BP, Sibai BM, Epstein FH, Romero R, Thadhani R, Karumanchi SA: Soluble endoglin and other circulating antiangiogenic factors in preeclampsia. N Engl J Med. 2006, 355: 992-1005.
Rana S, Karumanchi SA, Levine RJ, Venkatesha S, Rauh-Hain JA, Tamez H, Thadhani R: Sequential changes in antiangiogenic factors in early pregnancy and risk of developing preeclampsia. Hypertension. 2007, 50: 137-142.
Robinson CJ, Johnson DD: Soluble endoglin as a second-trimester marker for preeclampsia. Am J Obstet Gynecol. 2007, 197: 174-175.
Stepan H, Geipel A, Schwarz F, Kramer T, Wessel N, Faber R: Circulatory soluble endoglin and its predictive value for preeclampsia in second-trimester pregnancies with abnormal uterine perfusion. Am J Obstet Gynecol. 2008, 198: 175-176.
Erez O, Romero R, Espinoza J, Fu W, Todem D, Kusanovic JP, Gotsch F, Edwin S, Nien JK, Chaiworapongsa T, Mittal P, Mazaki-Tovi S, Than NG, Gomez R, Hassan SS: The change in concentrations of angiogenic and anti-angiogenic factors in maternal plasma between the first and second trimesters in risk assessment for the subsequent development of preeclampsia and small-for-gestational age. J Matern Fetal Neonatal Med. 2008, 21: 279-287.
Andre P: P-selectin in haemostasis. Br J Haematol. 2004, 126: 298-306.
Polgar J, Matuskova J, Wagner DD: The P-selectin, tissue factor, coagulation triad. J Thromb Haemost. 2005, 3: 1590-1596.
Dunlop LC, Skinner MP, Bendall LJ, Favaloro EJ, Castaldi PA, Gorman JJ, Gamble JR, Vadas MA, Berndt MC: Characterization of GMP-140 (P-selectin) as a circulating plasma protein. J Exp Med. 1992, 175: 1147-1150.
Holthe MR, Staff AC, Berge LN, Lyberg T: Different levels of platelet activation in preeclamptic, normotensive pregnant, and nonpregnant women. Am J Obstet Gynecol. 2004, 190: 1128-1134.
Konijnenberg A, Stokkers EW, Post van der JA, Schaap MC, Boer K, Bleker OP, Sturk A: Extensive platelet activation in preeclampsia compared with normal pregnancy: enhanced expression of cell adhesion molecules. Am J Obstet Gynecol. 1997, 176: 461-469.
Yoneyama Y, Suzuki S, Sawa R, Kiyokawa Y, Power GG, Araki T: Plasma adenosine levels and P-selectin expression on platelets in preeclampsia. Obstet Gynecol. 2001, 97: 366-370.
Bretelle F, Sabatier F, Desprez D, Camoin L, Grunebaum L, Combes V, D'Ercole C, gnat-George F: Circulating microparticles: a marker of procoagulant state in normal pregnancy and pregnancy complicated by preeclampsia or intrauterine growth restriction. Thromb Haemost. 2003, 89: 486-492.
Lok CA, Nieuwland R, Sturk A, Hau CM, Boer K, Vanbavel E, Vanderpost JA: Microparticle-associated P-selectin reflects platelet activation in preeclampsia. Platelets. 2007, 18: 68-72.
Aksoy H, Kumtepe Y, Akcay F, Yildirim AK: Correlation of P-selectin and lipoprotein(a), and other lipid parameters in preeclampsia. Clin Exp Med. 2002, 2: 39-43.
Chaiworapongsa T, Romero R, Yoshimatsu J, Espinoza J, Kim YM, Park K, Kalache K, Edwin S, Bujold E, Gomez R: Soluble adhesion molecule profile in normal pregnancy and pre-eclampsia. J Matern Fetal Neonatal Med. 2002, 12: 19-27.
Halim A, Kanayama N, el ME, Nakashima A, Bhuiyan AB, Khatun S, Terao T: Plasma P selectin (GMP-140) and glycocalicin are elevated in preeclampsia and eclampsia: their significances. Am J Obstet Gynecol. 1996, 174: 272-277.
Heyl W, Handt S, Reister F, Gehlen J, Schroder W, Mittermayer C, Rath W: Elevated soluble adhesion molecules in women with pre-eclampsia. Do cytokines like tumour necrosis factor-alpha and interleukin-1beta cause endothelial activation. Eur J Obstet Gynecol Reprod Biol. 1999, 86: 35-41.
Banzola I, Farina A, Concu M, Sekizawa A, Purwosunu Y, Strada I, Arcelli D, Simonazzi G, Caramelli E, Rizzo N: Performance of a panel of maternal serum markers in predicting preeclampsia at 11–15 weeks' gestation. Prenat Diagn. 2007, 27: 1005-1010.
Bosio PM, Cannon S, McKenna PJ, O'Herlihy C, Conroy R, Brady H: Plasma P-selectin is elevated in the first trimester in women who subsequently develop pre-eclampsia. BJOG. 2001, 108: 709-715.
Chavarria ME, Lara-Gonzalez L, Garcia-Paleta Y, Vital-Reyes VS, Reyes A: Adhesion molecules changes at 20 gestation weeks in pregnancies complicated by preeclampsia. Eur J Obstet Gynecol Reprod Biol. 2008, 137: 157-164.
Lo YM, Corbetta N, Chamberlain PF, Rai V, Sargent IL, Redman CW, Wainscoat JS: Presence of fetal DNA in maternal plasma and serum. Lancet. 1997, 350: 485-487.
Hromadnikova I, Vechetova L, Vesela K, Benesova B, Doucha J, Kulovany E, Vlk R: Non-invasive fetal RHD exon 7 and exon 10 genotyping using real-time PCR testing of fetal DNA in maternal plasma. Fetal Diagn Ther. 2005, 20: 275-280.
Finning K, Martin P, Daniels G: A clinical service in the UK to predict fetal Rh (Rhesus) D blood group using free fetal DNA in maternal plasma. Ann N Y Acad Sci. 2004, 1022: 119-123.
Li Y, Holzgreve W, Hahn S: Size fractionation of cell-free DNA in maternal plasma and its application in noninvasive detection of fetal single gene point mutations. Methods Mol Biol. 2008, 444: 239-251.
Sekizawa A, Jimbo M, Saito H, Iwasaki M, Matsuoka R, Okai T, Farina A: Cell-free fetal DNA in the plasma of pregnant women with severe fetal growth restriction. Am J Obstet Gynecol. 2003, 188: 480-484.
Zhong XY, Holzgreve W, Li JC, Aydinli K, Hahn S: High levels of fetal erythroblasts and fetal extracellular DNA in the peripheral blood of a pregnant woman with idiopathic polyhydramnios: case report. Prenat Diagn. 2000, 20: 838-841.
Wataganara T, LeShane ES, Farina A, Messerlian GM, Lee T, Canick JA, Bianchi DW: Maternal serum cell-free fetal DNA levels are increased in cases of trisomy 13 but not trisomy 18. Hum Genet. 2003, 112: 204-208.
Farina A, LeShane ES, Lambert-Messerlian GM, Canick JA, Lee T, Neveux LM, Palomaki GE, Bianchi DW: Evaluation of cell-free fetal DNA as a second-trimester maternal serum marker of Down syndrome pregnancy. Clin Chem. 2003, 49: 239-242.
Vodicka R, Vrtel R, Dusek L, Prochazka M, Schneiderova E, Vrbicka D, Krejcirikova E, Dhaifalah I, Santava A, Santavy J: Refined fluorescent STR quantification of cell-free fetal DNA during pregnancy in physiological and Down syndrome fetuses. Prenat Diagn. 2008, 28: 425-433.
Farina A, LeShane ES, Romero R, Gomez R, Chaiworapongsa T, Rizzo N, Bianchi DW: High levels of fetal cell-free DNA in maternal serum: a risk factor for spontaneous preterm delivery. Am J Obstet Gynecol. 2005, 193: 421-425.
Lo YM, Leung TN, Tein MS, Sargent IL, Zhang J, Lau TK, Haines CJ, Redman CW: Quantitative abnormalities of fetal DNA in maternal serum in preeclampsia. Clin Chem. 1999, 45: 184-188.
Zhong XY, Holzgreve W, Hahn S: The levels of circulatory cell free fetal DNA in maternal plasma are elevated prior to the onset of preeclampsia. Hypertens Pregnancy. 2002, 21: 77-83.
Levine RJ, Qian C, LeShane ES, Yu KF, England LJ, Schisterman EF, Wataganara T, Romero R, Bianchi DW: Two-stage elevation of cell-free fetal DNA in maternal sera before onset of preeclampsia. Am J Obstet Gynecol. 2004, 190: 707-713.
Zimmermann BG, Maddocks DG, Avent ND: Quantification of circulatory fetal DNA in the plasma of pregnant women. Methods Mol Biol. 2008, 444: 219-229.
Farina A, Sekizawa A, Iwasaki M, Matsuoka R, Ichizuka K, Okai T: Total cell-free DNA (beta-globin gene) distribution in maternal plasma at the second trimester: a new prospective for preeclampsia screening. Prenat Diagn. 2004, 24: 722-726.
Sekizawa A, Farina A, Koide K, Iwasaki M, Honma S, Ichizuka K, Saito H, Okai T: beta-globin DNA in maternal plasma as a molecular marker of pre-eclampsia. Prenat Diagn. 2004, 24: 697-700.
Swinkels DW, de Kok JB, Hendriks JC, Wiegerinck E, Zusterzeel PL, Steegers EA: Hemolysis, elevated liver enzymes, and low platelet count (HELLP) syndrome as a complication of preeclampsia in pregnant women increases the amount of cell-free fetal and maternal DNA in maternal plasma and serum. Clin Chem. 2002, 48: 650-653.
Chim SS, Tong YK, Chiu RW, Lau TK, Leung TN, Chan LY, Oudejans CB, Ding C, Lo YM: Detection of the placental epigenetic signature of the maspin gene in maternal plasma. Proc Natl Acad Sci USA. 2005, 102: 14753-14758.
Chan KC, Ding C, Gerovassili A, Yeung SW, Chiu RW, Leung TN, Lau TK, Chim SS, Chung GT, Nicolaides KH, Lo YM: Hypermethylated RASSF1A in maternal plasma: A universal fetal DNA marker that improves the reliability of noninvasive prenatal diagnosis. Clin Chem. 2006, 52: 2211-2218.
Tsui DW, Chan KC, Chim SS, Chan LW, Leung TY, Lau TK, Lo YM, Chiu RW: Quantitative aberrations of hypermethylated RASSF1A gene sequences in maternal plasma in pre-eclampsia. Prenat Diagn. 2007, 27: 1212-1218.
Diesch CH, Holzgreve W, Hahn S, Zhong XY: Comparison of activin A and cell-free fetal DNA levels in maternal plasma from patients at high risk for preeclampsia. Prenat Diagn. 2006, 26: 1267-1270.
Cho C, Turner L, Primakoff P, Myles DG: Genomic organization of the mouse fertilin beta gene that encodes an ADAM family protein active in sperm-egg fusion. Dev Genet. 1997, 20: 320-328.
White JM: ADAMs: modulators of cell-cell and cell-matrix interactions. Curr Opin Cell Biol. 2003, 15: 598-606.
Yang P, Baker KA, Hagg T: A disintegrin and metalloprotease 21 (ADAM21) is associated with neurogenesis and axonal growth in developing and adult rodent CNS. J Comp Neurol. 2005, 490: 163-179.
Gilpin BJ, Loechel F, Mattei MG, Engvall E, Albrechtsen R, Wewer UM: A novel, secreted form of human ADAM 12 (meltrin alpha) provokes myogenesis in vivo. J Biol Chem. 1998, 273: 157-166.
Laigaard J, Spencer K, Christiansen M, Cowans NJ, Larsen SO, Pedersen BN, Wewer UM: ADAM 12 as a first-trimester maternal serum marker in screening for Down syndrome. Prenat Diagn. 2006, 26: 973-979.
Laigaard J, Christiansen M, Frohlich C, Pedersen BN, Ottesen B, Wewer UM: The level of ADAM12-S in maternal serum is an early first-trimester marker of fetal trisomy 18. Prenat Diagn. 2005, 25: 45-46.
Laigaard J, Sorensen T, Frohlich C, Pedersen BN, Christiansen M, Schiott K, Uldbjerg N, Albrechtsen R, Clausen HV, Ottesen B, Wewer UM: ADAM12: a novel first-trimester maternal serum marker for Down syndrome. Prenat Diagn. 2003, 23: 1086-1091.
Spencer K, Cowans NJ: ADAM12 as a marker of trisomy 18 in the first and second trimester of pregnancy. J Matern Fetal Neonatal Med. 2007, 20: 645-650.
Spencer K, Cowans NJ, Stamatopoulou A: Maternal serum ADAM12s as a marker of rare aneuploidies in the first or second trimester of pregnancy. Prenat Diagn. 2007, 27: 1233-1237.
Laigaard J, Sorensen T, Placing S, Holck P, Frohlich C, Wojdemann KR, Sundberg K, Shalmi AC, Tabor A, Norgaard-Pedersen B, Ottesen B, Christiansen M, Wewer UM: Reduction of the disintegrin and metalloprotease ADAM12 in preeclampsia. Obstet Gynecol. 2005, 106: 144-149.
Spencer K, Cowans NJ, Stamatopoulou A: ADAM12s in maternal serum as a potential marker of pre-eclampsia. Prenat Diagn. 2008, 28: 212-216.
Poon LC, Chelemen T, Granvillano O, Pandeva I, Nicolaides KH: First-trimester maternal serum a disintegrin and metalloprotease 12 (ADAM12) and adverse pregnancy outcome. Obstet Gynecol. 2008, 112: 1082-1090.
Bohn H, Kraus W, Winckler W: Purification and characterization of two new soluble placental tissue proteins (PP13 and PP17). Oncodev Biol Med. 1983, 4: 343-350.
Visegrady B, Than NG, Kilar F, Sumegi B, Than GN, Bohn H: Homology modelling and molecular dynamics studies of human placental tissue protein 13 (galectin-13). Protein Eng. 2001, 14: 875-880.
Than NG, Pick E, Bellyei S, Szigeti A, Burger O, Berente Z, Janaky T, Boronkai A, Kliman H, Meiri H, Bohn H, Than GN, Sumegi B: Functional analyses of placental protein 13/galectin-13. Eur J Biochem. 2004, 271: 1065-1078.
Than NG, Sumegi B, Than GN, Berente Z, Bohn H: Isolation and sequence analysis of a cDNA encoding human placental tissue protein 13 (PP13), a new lysophospholipase, homologue of human eosinophil Charcot-Leyden Crystal protein. Placenta. 1999, 20: 703-710.
Sekizawa A, Purwosunu Y, Yoshimura S, Nakamura M, Shimizu H, Okai T, Rizzo N, Farina A: PP13 mRNA expression in trophoblasts from preeclamptic placentas. Reprod Sci. 2009, 16: 408-413.
Huppertz B, Sammar M, Chefetz I, Neumaier-Wagner P, Bartz C, Meiri H: Longitudinal determination of serum placental protein 13 during development of preeclampsia. Fetal Diagn Ther. 2008, 24: 230-236.
Chafetz I, Kuhnreich I, Sammar M, Tal Y, Gibor Y, Meiri H, Cuckle H, Wolf M: First-trimester placental protein 13 screening for preeclampsia and intrauterine growth restriction. Am J Obstet Gynecol. 2007, 197: 35-37.
Spencer K, Cowans NJ, Chefetz I, Tal J, Meiri H: First-trimester maternal serum PP-13, PAPP-A and second-trimester uterine artery Doppler pulsatility index as markers of pre-eclampsia. Ultrasound Obstet Gynecol. 2007, 29: 128-134.
Burger O, Pick E, Zwickel J, Klayman M, Meiri H, Slotky R, Mandel S, Rabinovitch L, Paltieli Y, Admon A, Gonen R: Placental protein 13 (PP-13): effects on cultured trophoblasts, and its detection in human body fluids in normal and pathological pregnancies. Placenta. 2004, 25: 608-622.
Khalil A, Cowans NJ, Spencer K, Goichman S, Meiri H, Harrington K: First trimester maternal serum placental protein 13 for the prediction of pre-eclampsia in women with a priori high risk. Prenat Diagn. 2009,
Romero R, Kusanovic JP, Than NG, Erez O, Gotsch F, Espinoza J, Edwin S, Chefetz I, Gomez R, Nien JK, Sammar M, Pineles B, Hassan SS, Meiri H, Tal Y, Kuhnreich I, Papp Z, Cuckle HS: First-trimester maternal serum PP13 in the risk assessment for preeclampsia. Am J Obstet Gynecol. 2008, 199 (2): 122-
Nicolaides KH, Bindra R, Turan OM, Chefetz I, Sammar M, Meiri H, Tal J, Cuckle HS: A novel approach to first-trimester screening for early pre-eclampsia combining serum PP-13 and Doppler ultrasound. Ultrasound Obstet Gynecol. 2006, 27: 13-17.
Spencer K, Cowans NJ, Chefetz I, Tal J, Kuhnreich I, Meiri H: Second-trimester uterine artery Doppler pulsatility index and maternal serum PP13 as markers of pre-eclampsia. Prenat Diagn. 2007, 27: 258-263.
Souza DG, Soares AC, Pinho V, Torloni H, Reis LF, Teixeira MM, Dias AA: Increased mortality and inflammation in tumor necrosis factor-stimulated gene-14 transgenic mice after ischemia and reperfusion injury. Am J Pathol. 2002, 160: 1755-1765.
Breviario F, d'Aniello EM, Golay J, Peri G, Bottazzi B, Bairoch A, Saccone S, Marzella R, Predazzi V, Rocchi M: Interleukin-1-inducible genes in endothelial cells. Cloning of a new gene related to C-reactive protein and serum amyloid P component. J Biol Chem. 1992, 267: 22190-22197.
Alles VV, Bottazzi B, Peri G, Golay J, Introna M, Mantovani A: Inducible expression of PTX3, a new member of the pentraxin family, in human mononuclear phagocytes. Blood. 1994, 84: 3483-3493.
Inforzato A, Peri G, Doni A, Garlanda C, Mantovani A, Bastone A, Carpentieri A, Amoresano A, Pucci P, Roos A, Daha MR, Vincenti S, Gallo G, Carminati P, De SR, Salvatori G: Structure and function of the long pentraxin PTX3 glycosidic moiety: fine-tuning of the interaction with C1q and complement activation. Biochemistry. 2006, 45: 11540-11551.
Popovici RM, Krause MS, Jauckus J, Germeyer A, Brum IS, Garlanda C, Strowitzki T, von WM: The long pentraxin PTX3 in human endometrium: regulation by steroids and trophoblast products. Endocrinology. 2008, 149: 1136-1143.
Cetin I, Cozzi V, Pasqualini F, Nebuloni M, Garlanda C, Vago L, Pardi G, Mantovani A: Elevated maternal levels of the long pentraxin 3 (PTX3) in preeclampsia and intrauterine growth restriction. Am J Obstet Gynecol. 2006, 194: 1347-1353.
Rovere-Querini P, Antonacci S, Dell'Antonio G, Angeli A, Almirante G, Cin ED, Valsecchi L, Lanzani C, Sabbadini MG, Doglioni C, Manfredi AA, Castiglioni MT: Plasma and tissue expression of the long pentraxin 3 during normal pregnancy and preeclampsia. Obstet Gynecol. 2006, 108: 148-155.
Lawrence JB, Bale LK, Haddad TC, Clarkson JT, Conover CA: Characterization and partial purification of the insulin-like growth factor (IGF)-dependent IGF binding protein-4-specific protease from human fibroblast conditioned media. Growth Horm IGF Res. 1999, 9: 25-34.
Chen BK, Overgaard MT, Bale LK, Resch ZT, Christiansen M, Oxvig C, Conover CA: Molecular regulation of the IGF-binding protein-4 protease system in human fibroblasts: identification of a novel inducible inhibitor. Endocrinology. 2002, 143: 1199-1205.
Lawrence JB, Oxvig C, Overgaard MT, Sottrup-Jensen L, Gleich GJ, Hays LG, Yates JR, Conover CA: The insulin-like growth factor (IGF)-dependent IGF binding protein-4 protease secreted by human fibroblasts is pregnancy-associated plasma protein-A. Proc Natl Acad Sci USA. 1999, 96: 3149-3153.
Chelius D, Conover CA, Baldwin MA, Spencer EM: Characterization of the enzymatic specificity of the IGF-dependent insulin-like growth factor binding protein-4 (IGFBP-4) protease. Growth Horm IGF Res. 2000, 10: 360-366.
Jadlowiec J, Dongell D, Smith J, Conover C, Campbell P: Pregnancy-associated plasma protein-a is involved in matrix mineralization of human adult mesenchymal stem cells and angiogenesis in the chick chorioallontoic membrane. Endocrinology. 2005, 146: 3765-3772.
Miller BS, Bronk JT, Nishiyama T, Yamagiwa H, Srivastava A, Bolander ME, Conover CA: Pregnancy associated plasma protein-A is necessary for expeditious fracture healing in mice. J Endocrinol. 2007, 192: 505-513.
Cowans NJ, Spencer K: First-trimester ADAM12 and PAPP-A as markers for intrauterine fetal growth restriction through their roles in the insulin-like growth factor system. Prenat Diagn. 2007, 27: 264-271.
Dugoff L, Hobbins JC, Malone FD, Porter TF, Luthy D, Comstock CH, Hankins G, Berkowitz RL, Merkatz I, Craigo SD, Timor-Tritsch IE, Carr SR, Wolfe HM, Vidaver J, D'Alton ME: First-trimester maternal serum PAPP-A and free-beta subunit human chorionic gonadotropin concentrations and nuchal translucency are associated with obstetric complications: a population-based screening study (the FASTER Trial). Am J Obstet Gynecol. 2004, 191: 1446-1451.
Krantz D, Goetzl L, Simpson JL, Thom E, Zachary J, Hallahan TW, Silver R, Pergament E, Platt LD, Filkins K, Johnson A, Mahoney M, Hogge WA, Wilson RD, Mohide P, Hershey D, Wapner R: Association of extreme first-trimester free human chorionic gonadotropin-beta, pregnancy-associated plasma protein A, and nuchal translucency with intrauterine growth restriction and other adverse pregnancy outcomes. Am J Obstet Gynecol. 2004, 191: 1452-1458.
Ong CY, Liao AW, Spencer K, Munim S, Nicolaides KH: First trimester maternal serum free beta human chorionic gonadotrophin and pregnancy associated plasma protein A as predictors of pregnancy complications. BJOG. 2000, 107: 1265-1270.
Smith GC, Stenhouse EJ, Crossley JA, Aitken DA, Cameron AD, Connor JM: Early pregnancy levels of pregnancy-associated plasma protein a and the risk of intrauterine growth restriction, premature birth, preeclampsia, and stillbirth. J Clin Endocrinol Metab. 2002, 87: 1762-1767.
Spencer K, Cowans NJ, Chefetz I, Tal J, Meiri H: First-trimester maternal serum PP-13, PAPP-A and second-trimester uterine artery Doppler pulsatility index as markers of pre-eclampsia. Ultrasound Obstet Gynecol. 2007, 29: 128-134.
Spencer K, Cowans NJ, Nicolaides KH: Low levels of maternal serum PAPP-A in the first trimester and the risk of pre-eclampsia. Prenat Diagn. 2008, 28: 7-10.
Spencer K, Cowans NJ, Molina F, Kagan KO, Nicolaides KH: First-trimester ultrasound and biochemical markers of aneuploidy and the prediction of preterm or early preterm delivery. Ultrasound Obstet Gynecol. 2008, 31: 147-152.
Tul N, Pusenjak S, Osredkar J, Spencer K, Novak-Antolic Z: Predicting complications of pregnancy with first-trimester maternal serum free-betahCG, PAPP-A and inhibin-A. Prenat Diagn. 2003, 23: 990-996.
Yaron Y, Heifetz S, Ochshorn Y, Lehavi O, Orr-Urtreger A: Decreased first trimester PAPP-A is a predictor of adverse pregnancy outcome. Prenat Diagn. 2002, 22: 778-782.
Toop K, Klopper A: Concentration of pregnancy-associated plasma protein A (PAPP-A) in patients with pre-eclamptic toxaemia. Placenta Suppl. 1981, 3: 167-173.
Preiss J, Handler P: Enzymatic synthesis of nicotinamide mononucleotide. J Biol Chem. 1957, 225: 759-770.
Fukuhara A, Matsuda M, Nishizawa M, Segawa K, Tanaka M, Kishimoto K, Matsuki Y, Murakami M, Ichisaka T, Murakami H, Watanabe E, Takagi T, Akiyoshi M, Ohtsubo T, Kihara S, Yamashita S, Makishima M, Funahashi T, Yamanaka S, Hiramatsu R, Matsuzawa Y, Shimomura I: Visfatin: a protein secreted by visceral fat that mimics the effects of insulin. Science. 2005, 307: 426-430.
Lopez-Bermejo A, Chico-Julia B, Fernandez-Balsells M, Recasens M, Esteve E, Casamitjana R, Ricart W, Fernandez-Real JM: Serum visfatin increases with progressive beta-cell deterioration. Diabetes. 2006, 55: 2871-2875.
Pagano C, Pilon C, Olivieri M, Mason P, Fabris R, Serra R, Milan G, Rossato M, Federspil G, Vettor R: Reduced plasma visfatin/pre-B cell colony-enhancing factor in obesity is not related to insulin resistance in humans. J Clin Endocrinol Metab. 2006, 91: 3165-3170.
Fasshauer M, Bluher M, Stumvoll M, Tonessen P, Faber R, Stepan H: Differential regulation of visfatin and adiponectin in pregnancies with normal and abnormal placental function. Clin Endocrinol (Oxf). 2007, 66: 434-439.
Krzyzanowska K, Krugluger W, Mittermayer F, Rahman R, Haider D, Shnawa N, Schernthaner G: Increased visfatin concentrations in women with gestational diabetes mellitus. Clin Sci (Lond). 2006, 110: 605-609.
Hu W, Wang Z, Wang H, Huang H, Dong M: Serum visfatin levels in late pregnancy and pre-eclampsia. Acta Obstet Gynecol Scand. 2008, 87: 413-418.
Fasshauer M, Waldeyer T, Seeger J, Schrey S, Ebert T, Kratzsch J, Lossner U, Bluher M, Stumvoll M, Faber R, Stepan H: Serum levels of the adipokine visfatin are increased in preeclampsia. Clin Endocrinol (Oxf). 2007
Kitamura K, Kangawa K, Kawamoto M, Ichiki Y, Nakamura S, Matsuo H, Eto T: Adrenomedullin: a novel hypotensive peptide isolated from human pheochromocytoma. Biochem Biophys Res Commun. 1993, 192: 553-560.
Hinson JP, Kapas S, Smith DM: Adrenomedullin, a multifunctional regulatory peptide. Endocr Rev. 2000, 21: 138-167.
Hippenstiel S, Witzenrath M, Schmeck B, Hocke A, Krisp M, Krull M, Seybold J, Seeger W, Rascher W, Schutte H, Suttorp N: Adrenomedullin reduces endothelial hyperpermeability. Circ Res. 2002, 91: 618-625.
Kato J, Tsuruda T, Kita T, Kitamura K, Eto T: Adrenomedullin: a protective factor for blood vessels. Arterioscler Thromb Vasc Biol. 2005, 25: 2480-2487.
Senna AA, Zedan M, bd El Salam GE, El Mashad AI: Study of plasma adrenomedullin level in normal pregnancy and preclampsia. Medscape J Med. 2008, 10: 29-
Gratton RJ, Gluszynski M, Mazzuca DM, Nygard K, Han VK: Adrenomedullin messenger ribonucleic acid expression in the placentae of normal and preeclamptic pregnancies. J Clin Endocrinol Metab. 2003, 88: 6048-6055.
Wallukat G, Homuth V, Fischer T, Lindschau C, Horstkamp B, Jupner A, Baur E, Nissen E, Vetter K, Neichel D, Dudenhausen JW, Haller H, Luft FC: Patients with preeclampsia develop agonistic autoantibodies against the angiotensin AT1 receptor. J Clin Invest. 1999, 103: 945-952.
Herse F, Staff AC, Hering L, Muller DN, Luft FC, Dechend R: AT1-receptor autoantibodies and uteroplacental RAS in pregnancy and pre-eclampsia. J Mol Med. 2008, 86: 697-703.
Walther T, Wallukat G, Jank A, Bartel S, Schultheiss HP, Faber R, Stepan H: Angiotensin II type 1 receptor agonistic antibodies reflect fundamental alterations in the uteroplacental vasculature. Hypertension. 2005, 46: 1275-1279.
Dragun D, Muller DN, Brasen JH, Fritsche L, Nieminen-Kelha M, Dechend R, Kintscher U, Rudolph B, Hoebeke J, Eckert D, Mazak I, Plehm R, Schonemann C, Unger T, Budde K, Neumayer HH, Luft FC, Wallukat G: Angiotensin II type 1-receptor activating antibodies in renal-allograft rejection. N Engl J Med. 2005, 352: 558-569.
Zhou CC, Ahmad S, Mi T, Abbasi S, Xia L, Day MC, Ramin SM, Ahmed A, Kellems RE, Xia Y: Autoantibody from women with preeclampsia induces soluble Fms-like tyrosine kinase-1 production via angiotensin type 1 receptor and calcineurin/nuclear factor of activated T-cells signaling. Hypertension. 2008, 51: 1010-1019.
Herse F, Verlohren S, Wenzel K, Pape J, Muller DN, Modrow S, Wallukat G, Luft FC, Redman CW, Dechend R: Prevalence of agonistic autoantibodies against the angiotensin II type 1 receptor and soluble fms-like tyrosine kinase 1 in a gestational age-matched case study. Hypertension. 2009, 53: 393-398.
Chien PF, Arnott N, Gordon A, Owen P, Khan KS: How useful is uterine artery Doppler flow velocimetry in the prediction of pre-eclampsia, intrauterine growth retardation and perinatal death? An overview. BJOG. 2000, 107: 196-208.
Cnossen JS, Morris RK, ter Riet G, Mol BW, van der Post JA, Coomarasamy A, Zwinderman AH, Robson SC, Bindels PJ, Kleijnen J, Khan KS: Use of uterine artery Doppler ultrasonography to predict pre-eclampsia and intrauterine growth restriction: a systematic review and bivariable meta-analysis. CMAJ. 2008, 178: 701-711.
Barton JR, Sibai BM: Prediction and prevention of recurrent preeclampsia. Obstet Gynecol. 2008, 112: 359-372.
Savvidou MD, Noori M, Anderson JM, Hingorani AD, Nicolaides KH: Maternal endothelial function and serum concentrations of placental growth factor and soluble endoglin in women with abnormal placentation. Ultrasound Obstet Gynecol. 2008, 32: 871-876.
Enquobahrie DA, Meller M, Rice K, Psaty BM, Siscovick DS, Williams MA: Differential placental gene expression in preeclampsia. Am J Obstet Gynecol. 2008, 199 (5): e1-11.
Gack S, Marme A, Marme F, Wrobel G, Vonderstrass B, Bastert G, Lichter P, Angel P, Schorpp-Kistner M: Preeclampsia: increased expression of soluble ADAM 12. J Mol Med. 2005, 83: 887-896.
Reimer T, Koczan D, Gerber B, Richter D, Thiesen HJ, Friese K: Microarray analysis of differentially expressed genes in placental tissue of pre-eclampsia: up-regulation of obesity-related genes. Mol Hum Reprod. 2002, 8: 674-680.
Zhou R, Zhu Q, Wang Y, Ren Y, Zhang L, Zhou Y: Genomewide oligonucleotide microarray analysis on placentae of pre-eclamptic pregnancies. Gynecol Obstet Invest. 2006, 62: 108-114.
Hansson SR, Chen Y, Brodszki J, Chen M, Hernandez-Andrade E, Inman JM, Kozhich OA, Larsson I, Marsal K, Medstrand P, Xiang CC, Brownstein MJ: Gene expression profiling of human placentas from preeclamptic and normotensive pregnancies. Mol Hum Reprod. 2006, 12: 169-179.
Heikkila A, Tuomisto T, Hakkinen SK, Keski-Nisula L, Heinonen S, Yla-Herttuala S: Tumor suppressor and growth regulatory genes are overexpressed in severe early-onset preeclampsia–an array study on case-specific human preeclamptic placental tissue. Acta Obstet Gynecol Scand. 2005, 84: 679-689.
Herse F, Dechend R, Harsem NK, Wallukat G, Janke J, Qadri F, Hering L, Muller DN, Luft FC, Staff AC: Dysregulation of the circulating and tissue-based renin-angiotensin system in preeclampsia. Hypertension. 2007, 49: 604-611.
Pang ZJ, Xing FQ: Comparative profiling of metabolism-related gene expression in pre-eclamptic and normal pregnancies. Arch Gynecol Obstet. 2004, 269: 91-95.
Tsui NB, Lo YM: A microarray approach for systematic identification of placental-derived RNA markers in maternal plasma. Methods Mol Biol. 2008, 444: 275-289.
Ng EK, Tsui NB, Lau TK, Leung TN, Chiu RW, Panesar NS, Lit LC, Chan KW, Lo YM: mRNA of placental origin is readily detectable in maternal plasma. Proc Natl Acad Sci USA. 2003, 100: 4748-4753.
Ng EK, El-Sheikhah A, Chiu RW, Chan KC, Hogg M, Bindra R, Leung TN, Lau TK, Nicolaides KH, Lo YM: Evaluation of human chorionic gonadotropin beta-subunit mRNA concentrations in maternal serum in aneuploid pregnancies: a feasibility study. Clin Chem. 2004, 50: 1055-1057.
Tsui NB, Chim SS, Chiu RW, Lau TK, Ng EK, Leung TN, Tong YK, Chan KC, Lo YM: Systematic micro-array based identification of placental mRNA in maternal plasma: towards non-invasive prenatal gene expression profiling. J Med Genet. 2004, 41: 461-467.
Ng EK, Leung TN, Tsui NB, Lau TK, Panesar NS, Chiu RW, Lo YM: The concentration of circulating corticotropin-releasing hormone mRNA in maternal plasma is increased in preeclampsia. Clin Chem. 2003, 49: 727-731.
Herzog A, Kuntz S, Daniel H, Wenzel U: Identification of biomarkers for the initiation of apoptosis in human preneoplastic colonocytes by proteome analysis. Int J Cancer. 2004, 109: 220-229.
Reddy CS, Holzgreve W, Hahn S: Detection of new screening markers for fetal aneuploidies in maternal plasma: a proteomic approach. Methods Mol Biol. 2008, 444: 311-326.
Cho SY, Lee EY, Kim HY, Kang MJ, Lee HJ, Kim H, Paik YK: Protein profiling of human plasma samples by two-dimensional electrophoresis. Methods Mol Biol. 2008, 428: 57-75.
Gluckmann M, Fella K, Waidelich D, Merkel D, Kruft V, Kramer PJ, Walter Y, Hellmann J, Karas M, Kroger M: Prevalidation of potential protein biomarkers in toxicology using iTRAQ reagent technology. Proteomics. 2007, 7: 1564-1574.
Abdi F, Quinn JF, Jankovic J, McIntosh M, Leverenz JB, Peskind E, Nixon R, Nutt J, Chung K, Zabetian C, Samii A, Lin M, Hattan S, Pan C, Wang Y, Jin J, Zhu D, Li GJ, Liu Y, Waichunas D, Montine TJ, Zhang J: Detection of biomarkers with a multiplex quantitative proteomic platform in cerebrospinal fluid of patients with neurodegenerative disorders. J Alzheimers Dis. 2006, 9: 293-348.
Desouza LV, Grigull J, Ghanny S, Dube V, Romaschin AD, Colgan TJ, Siu KW: Endometrial carcinoma biomarker discovery and verification using differentially tagged clinical samples with multidimensional liquid chromatography and tandem mass spectrometry. Mol Cell Proteomics. 2007, 6: 1170-1182.
Ralhan R, Desouza LV, Matta A, Chandra TS, Ghanny S, Datta GS, Bahadur S, Siu KW: Discovery and verification of head-and-neck cancer biomarkers by differential protein expression analysis using iTRAQ labeling, multidimensional liquid chromatography, and tandem mass spectrometry. Mol Cell Proteomics. 2008, 7: 1162-1173.
Heazell AE, Brown M, Dunn WB, Worton SA, Crocker IP, Baker PN, Kell DB: Analysis of the metabolic footprint and tissue metabolome of placental villous explants cultured at different oxygen tensions reveals novel redox biomarkers. Placenta. 2008, 29: 691-698.
Kenny LC, Broadhurst D, Brown M, Dunn WB, Redman CW, Kell DB, Baker PN: Detection and identification of novel metabolomic biomarkers in preeclampsia. Reprod Sci. 2008, 15: 591-597.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
SG, CR and OL drafted and wrote the manuscript, RZD, ST, WH and SH: helped to draft the manuscript and gave important inputs. All authors read and approved the final manuscript.
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Grill, S., Rusterholz, C., Zanetti-Dällenbach, R. et al. Potential markers of preeclampsia – a review. Reprod Biol Endocrinol 7, 70 (2009). https://doi.org/10.1186/1477-7827-7-70
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1477-7827-7-70