Abstract
Coexistence curves in mixtures of limited miscibility with the lower critical consolute temperature (LCT), of 3-picoline with deuterium oxide (D2O), and D2O/H2O have been determined. They were tested with respect to the order parameter, the diameter of the binodal, and coordinates of the critical consolute point (critical temperature \({T}_{\mathrm{C}}\) and critical concentration \({\phi }_{\mathrm{C}}\)). Studies were carried out using the innovative method based on the analysis of relative volumes occupied by coexisting phases, yielding high-resolution data. The clear violation of the Cailletet-Mathew law of rectilinear diameter for the LCT mixtures of limited miscibility is evidenced. For the order parameter, the new distortion-sensitive analysis method yielded the evidence for the model-value of the order parameter critical exponent \(\beta =0.326\pm 0.003\), up to ca. 1 K from \({T}_{\mathrm{C}}\). Finally, the simple & easy method for determining the critical concentration by testing relative volumes of coexisting phases (or alternatively fractional meniscus heights, h) is presented. The significance of the invariant value \({h}_{\mathrm{C}}=h\left({T}_{\mathrm{C}},{\phi }_{\mathrm{C}}\right)=1/2\) is highlighted. The appearance of the milky & bluish critical opalescence is also shown.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Soft Matter denotes systems with enormous sensitivity to external perturbations, governed by mesoscale species, and associated with continuous or semi-continuous phase transitions [1,2,3,4,5,6]. These features are highly represented in the surrounding of the gas–liquid (G-L) critical point in one-component systems and the critical consolute point in binary mixtures of limited miscibility [7]. Nota that they belong to the same universality class, described by the space and order parameter dimensionalities \(d=3\) and \(n=1\), within the Physics of Critical Phenomena [8]. It means that physical properties on approaching the critical point are described by power relations with the same values of critical exponents for basic physical magnitudes. The primary role is played by precritical fluctuations of size (\(\xi \left(T\right)\)) and lifetime (\({\tau }_{fl.}\left(T\right)\)), which we can describe as follows [7, 8]:
where \(T>{T}_{\mathrm{C}}\), \({\xi }_{0}\) and \({\tau }_{0}\) are critical amplitudes’ regarding critical exponents \(\nu \approx 0.625\) for \(d=3, n=1\) universality class and the dynamic exponent \(z=3\) (the value for conserved order parameter).
It is worth recalling that the Physics of Critical Phenomena was one of the grand universalistic successes of twentieth-century physics, explaining the description in the surrounding of the critical point, avoiding microscopic specification of a given system [7,8,9]. As for the first evidence for critical phenomena in liquids, one can indicate the critical opalescence in the surrounding of the gas–liquid critical point and state-of-the-art P–V–T studies for CO2 by Thomas Andrews (1869) [10], serving as the basic reference for van der Waals equation of state (1873/1875) [7]. Soon afterward, Alekseev presented the first communicate regarding binary mixtures of limited miscibility [11]. Notable that he worked at Sankt Petersburg Mining University, and his interests in two-phase equilibria were stimulated by developing liquid–liquid extraction technology for hydro-metallurgy and hydro-mineralogy applications. Remarkable that the surrounding of the gas–liquid critical point in CO2 also found a significant technological application in supercritical fluids (SCF) technologies as the essential reference for the selective extraction technology [12].
The fundamental characterization of the gas–liquid critical point surrounding in one-component systems and the critical consolute point in binary mixtures constitute the coexistence curve, showing the location of the two-phase domain, in the homogenous surroundings. The gas–liquid critical point it is always located at the top of the coexistence curve [7, 8, 13]. The SCF domain located above was first considered homogeneous. Recently, less—and more dense SCF domains were detected, separated by ‘smooth & stretched’ Widom [14] or Frenkel [15] lines. For binary mixtures of limited miscibility, a direct equivalent is the case of the Upper Critical Point (UCP, also recalled as Upper Critical Consolute Temperature (UCT)) [8, 13]. For such mixtures, the Widom line parallel in the homogeneous liquid (\(T>{T}_{\mathrm{C}}\)) was detected [16].
The most extensive fundamental studies of limited miscibility mixtures are related to UCP-type systems. For such systems, the critical consolute/demixing temperature is located at the top of the coexistence curve: the two-phase region exists for \(T<{T}_{\mathrm{C}}\) and the homogeneous region for \(T>{T}_{\mathrm{C}}\). The analysis of coexisting curves clearly showed the characterization in line with predictions of the Physics of Critical Phenomena [8, 13, 17, 18]:
where \(<{T}_{\mathrm{C}}\), \(t=\left|{T}_{\mathrm{C}}-T\right|\) or \(t=\left|{T}_{\mathrm{C}}-T/{T}_{\mathrm{C}}\right|\) describes the distance from the critical consolute point; Indexes ‘U’ and ‘L’ are for the upper and lower coexisting phases. The order parameter exponent\(\beta \approx 0.325\), the heat-capacity related exponent \(\alpha \approx 0.125,\) and the first correction-to-scaling exponent\(\Delta \approx 0.5\); ‘ϕ’ denotes the concentration, in the given case, in volume fractions. Alternatively, one can consider the mole (x), mass/weight (w) fractions or simply density (ρ).
Equation (3) is related to the description of the order parameter, and Eq. (4) is for the diameter of the coexistence curve. Two conceptions for describing the latter are developed. The first one stresses the impact of correction-to-scaling terms on shifting away from the critical point (Eq. 4a) [8, 13] The second approach recalls Pérez-Sánchez et al. (2010, [18]) scaling concept of the asymmetric criticality in binary mixtures of limited miscibility (Eq. 4b). Notable that till the mid-eighties (twentieth century) a simplified version of Eq. (4) known as the Cailletet-Mathias (CM) law of rectilinear diameter [19] was used, avoiding the critical anomaly:
For decades, the CM law was used to determine critical concentration or density. However, the ultimate evidence for the diameter critical anomaly showed that it leads to strongly biased estimations [20, 21]. The alternative way of testing binodal’s properties is based on measurements of selected physical properties in coexisting phases. However, a comparison of results related to refractive index, dielectric constant or electric conductivity measurements revealed method-dependent binodal anisotropy [8, 13, 18, 22,23,24,25].
There is state-of-the-art evidence validating the characterization of coexistence curves in UCP-type binary mixtures via Eqs. (3) and (4) [7, 8, 13, 18, 22,23,24,25]. To the best of the authors’ knowledge, there are no such results for the second basic type of binary mixtures of limited miscibility associated with the Lower Critical Point (LCP, also called lower critical consolute temperature (LCT) system). For such systems, the critical consolute/demixing temperature is located at the bottom of the coexistence curve: the two-phase region exists for \(T>{T}_{\mathrm{C}}\) and the homogeneous region for \(T<{T}_{\mathrm{C}}\) [13, 26, 27].
This cognitive gap is particularly noteworthy when taking into account that LCP binary mixture is commonly referred to as an example of limited miscibility in monographs on the basics of physical chemistry or phase equilibria [8, 13, 26, 27]. It is especially true for systems based on low molecular weight liquids. This can probably be explained by the fact that the number of systems in which to identify the appearance of LCP is qualitatively smaller than that of UCP. Often these are systems with relatively troublesome preparation and a range of required temperatures which are problematic for traditional binodal determination methods. [13]
This report shows the preliminary communicate aiming at filling this cognitive gap, focusing on the characterization of the limited miscibility in 3-picoline + deuterium oxide (D2O)) mixtures. It is based on innovative experimental method which uses the analysis of temperature changes of fractional meniscus heights.
2 Experimental
The ‘standard’ method for determining phase equilibria in mixtures of limited miscibility is based on the preparation of the sequence of mixtures with different (total) concentrations and placing them within glass ampules, subsequently closed using a torch flame. Ampoules are located in a thermostated vessel, with a transparent ‘window’ enabling the observation of temperatures for which the phase separation and meniscus appear on cooling (UCP systems) or heating (LCP systems) [8, 13, 25]. This primary method suffers from some inherent experimental problems. First, the offer of commercial thermostats enabling the optical ‘eye-view’ observation is (very) limited and most often associated with small windows, restricting observations. There are also problems with the observational temperature range, particularly if the temperature stabilization and control better than 0.02 K is required. Second, it is a long-time experiment, often lasting days and requiring permanent attention. Third, possible supercooling of mixtures with non-critical concentrations can lead to the biasing scatter of detected binodal demixing temperatures (\({T}_{B}\)).
Results presented in this report were obtained using the innovative automatized visual method, described in detail in ref. [28] and tested there for the UCP type mixture of limited miscibility. The set-up is composed of a large volume, thermostated and transparent vessel (V = 20 L), linked to a large volume (V = 25 L) thermostat with external circulation, Julabo apparatus enables the high-precision temperature control. The transparent vessel was home-made and contained a transparent wall, 40 cm x 80 cm, made from a triple-walled glass element to ensure thermal insulation. Additionally, water (with a small amount of glycol) in this vessel was stirred 'remotely' using magnetic stirrers. The temperature was monitored with Pt 100 sensor.
Ampoules with an active height of ~ 25 mm and a diameter of 5 mm, ending in a thin tube with a diameter of 1 mm, were placed in the vessel. They were capped with a torch flame after placing solutions of various total concentrations in ampoules. The complete isolation from environmental influences enabled a long-lasting measurement. The appearance of menisci separating coexisting phases in initially homogeneous solutions was recorded using a high-resolution digital camera. The ampoules were illuminated from above with white, evenly distributed light to support the observation and recording. The temperature was changed, and the images were recorded using the temperature control without the ongoing participation of the researcher. This made it possible to conduct up to 7 days of precise observations without the researcher's active participation, whose role was limited to monitoring the condition of the apparatus and the experiment, possibly both 'in situ' and remotely using a computer or a smartphone.
A photo of the main elements of the described system is presented in Fig. 1. Figure 2 upper shows tested mixtures in the homogeneous phase, 0.2 K above the critical temperature, with critical opalescence signaling approaching transition. Usually, critical opalescence is recalled as the ‘milky-like’ appearance near the critical point. Figure 2 shows that it can exhibit a bluish background. This phenomenon is discussed in ref. [28]. Components for the preparation of tested mixtures were purchased in Fluka, with the highest available purity level and used without further purification.
Upper photograph of 3-picoline—deuterium oxide mixtures in the surrounding of the critical concentration, near the critical temperature. The white-bluish critical opalescence, signaling the vicinity of the critical point, is visible. Lower image is a collection of single ampule photographs with lowering temperature (from left to right) depicting opalescence onset (milky white color near the critical temperature) then clearly visible meniscus and its slight movement
3 Results and discussion
The coexistence curve was determined using the analysis of relative volumes occupied by coexisting phases in the two-phase domain: \({v}^{\mathrm{L}}={V}^{\mathrm{L}}/\left({V}^{\mathrm{L}}+{V}^{\mathrm{U}}\right)\) and \({v}^{\mathrm{U}}={V}^{\mathrm{U}}/\left({V}^{\mathrm{L}}+{V}^{\mathrm{U}}\right)\). For samples placed in normalized, cylindrical ampoules, one obtains the following relation [28]
where V is for the volume, S for the surface of the cylindric ampoule, and H for its height. Indexes ‘U’ and ‘L’ are for the upper and lower coexisting phases, respectively.
The normalized value of the height from the bottom of the ampule to the meniscus is called the fractional meniscus height (f.m.h.):\(h={h}^{\mathrm{L}}={H}^{\mathrm{L}}/H\). In the two-phase domain for \(T = const\), concentrations of coexisting phases are the same, independently of the total concentration of the mixture. However, relative volumes \(V^{{\text{L}}} ,V^{{\text{U}}}\) occupied by coexisting phases and fractional meniscus heights (h) change for different total concentrations. Consequently, one can consider the following pair of equations [23]:
where indexes ‘i’ and ‘j’ stand for ampoules with different total concentrations, \(\varphi\) denotes concentration in volume fraction.
When observing a set N ampoules containing mixtures with different total concentrations, one can consider \(\left(\genfrac{}{}{0pt}{}{N}{2}\right)\) pairs of equations, which for the selected value \(T=const\) are associated with the same values of \({\phi }^{\mathrm{L}}\) and \({\phi }^{\mathrm{U}}\). For instance, for 10 mixtures, one obtains even 45 pairs of relations, such as Eq. (7). It yields 45 values of \({\phi }^{\mathrm{L}}\) and \({\phi }^{\mathrm{U}}\), supports obtaining the high-accuracy final values of concentrations when using the supporting statistical analysis.
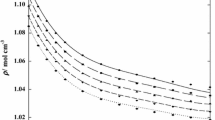
Such action is repeated for subsequent temperatures to determine related values \({\phi }^{\mathrm{L}}\left(T\right)\) and \({\phi }^{\mathrm{U}}\left(T\right)\) values, and finally, the coexistence curve. When increasing the number of observed ampoules with different concentrations of tested mixtures, the number of \({\phi }^{\mathrm{L}}\) and \({\phi }^{\mathrm{U}}\) values strongly increases. For instance, for 20 ampoules, one obtains 190 values. It enables the notable reduction of the experimental error. The obtained coexistence curves are shown in Fig. 3. The figure includes mixtures of picoline with D2O and H2O/D2O mixtures. The latter shows the shift up of LCST and the tendency to decrease the miscibility gap. The reference results by Andon and Cox [29] are also shown. It is still the essential reference for the tested system. The determination of the coexistence curve enabled the test of its shape via Eqs. 3 and 4, not tested for LCP-mixtures, so far (Fig. 4).
The obtained coexistence curves in 3-picoline—D2O and 3-picoline (H2O + D2O) mixtures. The ‘best’ available reference experimental data by Andon and Cox [24] is also shown. The line linking the latter is the ‘guide for eyes’, as in Ref. [29]. Based on the authors new results, the plot also contains the first evidence for the LCT binodal diameter and it’s pretransitional anomaly. Note that the addition of H2O shifts the (lower) critical consolute temperature ad narrows the binodal
These results related to the order parameter are presented in Fig. 5. The inset shows these data using the log–log scale analysis to test the ‘effective’ approximation of Eq. (3):
Changes of the order parameter in 3-picoline—deuterium oxide mixture of limited miscibility. The inset shows the test via Eq. (8), yielding the exponent \({\beta }_{\mathrm{eff}.}=0.365\pm 0.02\) using the log-log-scale with the adjustable critical temperature
Such description extends up to ca. 20 K above the lower critical consolute temperature. In the analysis, the value of \({T}_{C}\) was changed until reaching the linear behavior. However, such treatment creates some arbitrariness. The authors propose an alternative method, limiting this problem. Namely, based on Eq. (8) one obtains:
where coefficients \(B={\beta }^{-1}\) and \(A={T}_{\mathrm{C}}/\beta \).
The results of such analysis are shown in Fig. 6. It enables the subtle-distortions sensitive indication of the domain in which Eq. (8) is really valid. As visible in Fig. 6 this domain terminates at\(\sim {T}_{\mathrm{C}}+1K\). The slope of the line determines the order parameter critical exponent, which value is in fair agreement with model-value for \(d=3\) and \(n=1\) universality class. The condition \({\left(\mathrm{dln}\Delta \phi /\mathrm{d}T\right)}^{-1}=0\) enables reliable and straightforward estimation of \({T}_{C}\), indicated by the blue vertical arrow in the inset.
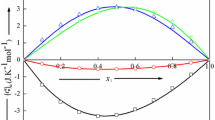
Figure 7 shows the focused insight into the coexistence curve diameter, only roughly visible in Fig. 4. It can be portrayed both by Eqs. (4a) and (4b) as indistinguishable. It can be associated with multi-parameter nonlinear fitting via dependences containing two power functions. The plot also shows that the linear behavior suggested by the CM ‘law’ cannot reliable portray experimental data, even in the limited range of temperatures. It is worth noting that Eqs. (4a) and (4b) contains the linear term ‘\({\phi }_{\mathrm{C}}+ct\)’, but it cannot be consider as the hallmark of the CM behavior or a specific background effect appearing away from \({T}_{C}\). Pre-transitional effect analysis via Eqs. (4a) and (4b) show that for each distance from the critical point, all terms in these relations have a significant influence on its resultant value.
The focused presentation of 3-picoline – D2O binodal curve diameter, given in Fig. 4. The curves are related to Eq. 4a, with exponents \(\beta =0.325\), and \(\alpha =0.12\) (red curve) and Eq. 4b, where ‘2 \(\beta \)’ anomaly is introduced (grey curve, slightly broadened). Both curves overlap. The green line terminated by the arrow shows the hypothetical Cailletet – Mathias law of rectilinear diameter [19,20,21], illustrating its essential inadequacy
Notable that using Eq. (7), one can introduce the relation describing isothermal changes of fractional meniscus heights:
where indexes ‘i’ and ‘j’ stand for ampoules with different total concentrations.
Following Eq. (10), f.m.h. are described by linear dependences vs. each isotherm's ‘total’ concentration. Experimentally, such behavior was validated in ref. [28], for nitrobenzene—decane mixture with the upper critical consolute temperature (UCT). Figure 8 shows its occurrence for the LCT mixture of limited miscibility. Notable, the intersection of lines related to different isotherms determines the critical concentration. This intersection occurs for the invariant value of f.m.h.:
Isothermal behavior of fractional meniscus height (f.m.h.) for different 3-picoline— D2O mixtures: the values of selected temperatures are given in the plot. The linear behavior validates the description via Eq. (10). The intersection of lines yields the critical concentration (\({\phi }_{\mathrm{C}}\)) and the coupled critical value of f.m.h. (\({h}_{\mathrm{C}}\)). The plot is for data given in Fig. 3
When entering from the homogeneous liquid (i.e., with the same concentration of component within the whole sample) into the two-phase domain, when passing LCT or UCT, the meniscus must appear in the center of the cylindrical ampoule to preserve the same concentration in the upper and lower part of the system. It indicates that the critical concentration can be determined by the analysis as in Fig. 6, using the f.m.h analysis for a minimum of 2–3 isotherms described by Eq. (10), or using only a single isotherm and the condition expressed by Eq. (11).
The experimental estimation of the critical temperature does not constitute a significant experimental problem due to its "flatness" of the binodal near\({\phi }_{\mathrm{C}}\), i.e., there is only a slight change of temperature, lesser than 0.1 K, for \({\phi }_{\mathrm{C}}\pm \Delta \phi \). However, it causes a considerable problem in determining the critical concentration, which explains the popularity of the Caillatet-Mathias ‘law’ (Eq. 5) [19,20,21]. It was invalidated by the clear evidence for the precritical anomaly of the diameter (Eq. 4). The results presented in this work (LCT case) and in ref. [23] (UCT case) introduce new methods based on the analysis of f.m.h (Fig. 6 and Eq. 10), eventually supported by the ‘invariant condition’ in Eq. (11). Notable that such analysis avoids the burdening impact of the diameter pretransitional anomaly (Table 1).
Concluding, this report shows the implementation of the recently developed innovative method for determining the coexistence curve via the analysis of the fractional meniscus heights for mixtures showing the lower critical consolute temperature. The obtained high-resolution experimental data enables precise analysis of the binodal curve regarding the order parameter and diameter. To the best of the authors' knowledge, it is the first example of such analysis for the LCT type mixture of limited miscibility. The authors stress the new concept for the simple and fast estimation of the critical concentration, finally solving the puzzling situation that appeared four decades ago after invalidating the Cailletet-Mathias law of rectilinear diameter [19,20,21]. This report focuses on the binary mixtures characterized via the critical concentration. Notwithstanding results of this report can be re-calculated using mole fractions, molar concentration, weight (mass) fraction, or even density [28].
Results presented also show the critical opalescence in a LCT-type critical mixture, while previously there was hardly, if at all, any evidence. Worth indicating is the appearance, in the broad range of concentrations, of ‘bluish’ accents in the hypothetically milky-white opalescence.
Data Availability Statement
The data that support the findings of this study are not openly available and can be provided by the corresponding author upon reasonable request.
References
R.A.L. Jones, Soft Condensed Matter (Oxford University Press, Oxford, 2002)
P.G. de Gennes, J. Badoz, Fragile Objects: Soft Matter, Hard Science, and the Thrill of Discovery (Copernicus, NY, 1995)
S.J. Rzoska, A. Drozd-Rzoska, Dual field nonlinear dielectric spectroscopy on glass forming EPON 828 epoxy resin. J. Phys. Condes. Matt. 25, 025101 (2011)
S.J. Rzoska, J. Zioło, A. Drozd-Rzoska, Stretched relaxation after switching off the strong electric field in a near-critical solution under high pressure. Phys. Rev. E 56, 2578 (1997)
A. Drozd-Rzoska, S.J. Rzoska, S. Pawlus, J. Zioło, Complex dynamics of supercooling n-butylcyanobiphenyl (4CB). Phys. Rev. E 72, 031501 (1997)
S.J. Rzoska, M. Paluch, A. Drozd-Rzoska, J. Zioło, P. Janik, K. Czupryński, Glassy and fluidlike behavior of the isotropic phase of n-cyanobiphenyls in broad-band dielectric relaxation studies. Eur. Phys. J. E. 7, 387–392 (2001)
H. E. Stanley, Introduction to Phase Transition and Critical Phenomena, (Clarendon Press, Oxford, 1971 & 1998)
M.A. Anisimov, Critical Phenomena in Liquids and Liquid Crystals (Gordon and Breach, Reading, 1994)
A. Drozd-Rzoska, S.J. Rzoska, J. Kalabiński, The impact of pressure on low molecular weight near-critical mixtures of limited miscibility. ACS Omega 5, 20141–20152 (2020)
B. Berche, M. Henkel, R. Kenna, Critical phenomena: 150 years since Cagniard de la Tour. J. Phys. Stud. 13(3), 3001 (2009)
A.G. Morachevkii, Professor Vladimir Fedorovich Alekseev (on 150th anniversary of his birthday). Rus. J. Appl. Chem. 75, 1541–1542 (2002)
Y. Arai, T. Sako, Y. Takebayashi, Supercritical Fluids: Molecular Interactions, Physical Properties, and New Applications (Springer, Berlin, 2001)
A. Kumar, H.R. Krishnamurthy, E.S.R. Gopal, Equilibrium critical phenomena in binary liquid mixtures. Phys. Rep. 98, 57–148 (1983)
M. Raju, D.T. Banuti, P.C. Ma, M. Ihme, Widom lines in binary mixtures of supercritical fluids. Sci. Rep. 7, 3027 (2017)
K. Ghosh, C.V. Krishnamurthy, Frenkel line crossover of confined supercritical fluids. Sci. Rep. 9, 14872 (2019)
S.J. Rzoska, A. Drozd-Rzoska, S. Starzonek, Nonlinear dielectric effect in critical liquids, in Nonlinear Dielectric Spectroscopy: Springer Series Advanced Dielectrics. ed. by R. Richert (Springer, Berlin, 2018), pp.187–218
M.A. Anisimov, J. Wang, The nature of asymmetry in fluid criticality. Phys. Rev. Lett. 97, 025703 (2006)
G. Pérez-Sánchez, P. Losada-Pérez, C.A. Cerdeiriña, J.V. Sengers, M.A. Anisimov, Asymmetric criticality in weakly compressible liquid mixtures. J. Chem. Phys. 132, 154502 (2010)
L. Cailletet, E. Mathias, Recherches sur les densités de gaz liquéfies et de leurs vapeurs saturées. Compt. Acad. Sci. 102, 1202–1207 (1886)
S. Reif-Acherman, The history of the rectilinear diameter law. Quim. Nova 33, 2003–2010 (2009)
J.S. Rowlinson, Physics of liquids: Are diameters rectilinear? Nature 319, 362 (1986)
P. Malik, S.J. Rzoska, A. Drozd-Rzoska, J. Jadżyn, Dielectric permittivity and electric conductivity studies in the one and in the two-phase region of nitrobenzene – dodecane critical point. J. Chem. Phys. 118, 9357–9363 (2003)
J. Chrapeć, S.J. Rzoska, J. Zioło, The tricritical point in an ethanediol - nitroethane - cyclohexane - methanol solution. Phase Transitions 4, 73–78 (1983)
S.J. Rzoska, J. Chrapeć, An isothermic coexistence curve in the nitrobenzene - decane - benzene solution. J. Chem. Phys. 90, 2783–2785 (1989)
S.J. Rzoska, Visual methods for determining of coexistence curves in liquid mixtures. Phase Trans. 27, 1–13 (1990)
J.R. Rumble, CRC Handbook of Physical Chemistry and Physics. 102 edn 2021–2022 (CRC Press, Boca Raton, 2022)
P. Atkins, J. de Paula, J. Keeker, Atkin’s Chemical Physics (Oxford University Press, Oxford, 2021)
S.J. Rzoska, A. Drozd-Rzoska, J. Kalabiński, Critical concentration in binary mixtures of limited miscibility. Fluid Phase Equilib. 540, 112979 (2021)
R.J.L. Andon, J.D. Cox, Phase relationships in the pyridine series homologues with water. J. Chem. Soc. 91, 4601–4606 (1952)
Acknowledgements
The authors were supported by the National Centre for Science (Poland): ref. UMO-2017/25/B/ ST3/02458, headed S. J. Rzoska. The paper is associated with the International Seminar on Soft Matter & Food—Physico-Chemical Models & Socio-Economic Parallels, 1st Polish-Slovenian Edition, Celestynów, Poland, 22–23 Nov., 2021; directors: Dr. hab. Aleksandra Drozd-Rzoska (Institute of High Pressure Physics PAS, Warsaw, Poland) and Prof. Samo Kralj (Univ. Maribor, Maribor, Slovenia).
Author information
Authors and Affiliations
Contributions
J.K. performed phase equilibria measurements, raw data analysis. A.D-R. and S.J.R. performed derivative analysis, result discussion.
Corresponding author
Ethics declarations
Conflict of interest
There are no competing interests for the authors of this report.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kalabiński, J., Drozd-Rzoska, A. & Rzoska, S.J. New insight into 3-picoline—deuterium oxide (D2O) mixtures of limited miscibility with the lower critical consolute temperature. Eur. Phys. J. E 45, 73 (2022). https://doi.org/10.1140/epje/s10189-022-00224-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epje/s10189-022-00224-z