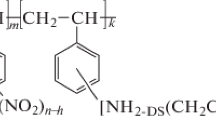
Abstract
The sorption kinetics of selected metal ions from multicomponent solutions by sorbents with taurine functions based on aminopolymers, namely, on chitosan, polyallylamine, and polyethyleneimine, has been studied. Sulfoethylated polyallylamine sorbents are distinguished by the highest silver(I) sorption selectivity and the shortest equilibration time. Sulfoethylated polyethyleneimines, unlike the other sorbents studied, can be used, depending on the pH of ammonium acetate buffer solution, for the group recovery of transition-metal ions or for co-recovering silver(I), copper(II), and nickel(II). The chemical structure of the polymer matrix and the degree of its modification do not significantly affect the initial silver(I) sorption rate from multicomponent solutions.




Similar content being viewed by others
REFERENCES
C. Ghiorghita, K. Borchert, A. Vasiliu, et al., Colloids Surf., A: Physicochem. 607, 125504 (2020). https://doi.org/10.1016/j.colsurfa.2020.125504
A. V. Pestov, Yu. O. Privar, A. V. Mekhaev, et al., Eur. Polym. J. 115, 356 (2019). https://doi.org/10.1016/j.eurpolymj.2019.03.049
L. K. Neudachina, Yu. S. Petrova, A. S. Zasukhin, et al., Anal. Kontrol 15, 88 (2011).
R. Kh. Khamizov, Russ. J. Phys. Chem. 94, 171 (2020). https://doi.org/10.1134/S0036024420010148
R. Kh. Khamizov, D. A. Sveshnikova, A. E. Kucherova, et al., Russ. J. Phys. Chem. 92, 1782 (2018). https://doi.org/10.1134/S0036024418090121
M. V. Maslova, V. I. Ivanenko, and L. G. Gerasimova, Russ. J. Phys. Chem. 93, 1245 (2019). https://doi.org/10.1134/S0036024419060219
N. S. Jalbani, A. R. Solangi, S. Memon, et al., J. Mol. Liq. 339, 116741 (2021). https://doi.org/10.1016/j.molliq.2021.116741
T. Manobala, S. Shukla, T. S. Rao, et al., Chemosphere 269, 128722 (2021). https://doi.org/10.1016/j.chemosphere.2020.128722
S. B. Yarusova, N. V. Makarenko, P. S. Gordienko, et al., Russ. J. Phys. Chem. 92, 559 (2018). https://doi.org/10.1134/S0036024418030354
A. V. Dolganov, A. V. Balandina, D. B. Chugunov, et al., Russ. J. Inorg. Chem. 65, 1770 (2020). https://doi.org/10.1134/S0036023620110030
G. Duca, I. Zinicovscaia, and D. Grozdov, Russ. J. Gen. Chem. 90, 2546 (2020). https://doi.org/10.1134/S1070363220130034
Yu. S. Petrova, A. V. Pestov, M. K. Usoltseva, et al., J. Hazard. Mater 299, 696 (2015). https://doi.org/10.1016/j.jhazmat.2015.08.001
L. M. K. Alifkhanova, K. Y. Lopunova, A. V. Pestov, et al., Sep. Sci. Technol. 56, 1303 (2021). https://doi.org/10.1080/01496395.2020.1781175
E. I. Kapitanova, E. O. Zemlyakova, A. V. Pestov, et al., Russ. Chem. Bull. 68, 1252 (2019). https://doi.org/10.1007/s11172-019-2549-5
G. E. Boyd, A. W. Adamson, and L. S. Myers, J. Am. Chem. Soc. 69, 2836 (1947). https://doi.org/10.1021/ja01203a066
Sh. Sharma and N. Rajesh, J. Environ. Chem. Eng. 4, 4287 (2016). https://doi.org/10.1016/j.jece.2016.09.028
Y. S. Ho and G. McKay, Process Biochem. 34, 451 (1999). https://doi.org/10.1016/S0032-9592(98)00112-5
T. L. Lin and H. L. Lien, Int. J. Mol. Sci. 14, 9834 (2013). https://doi.org/10.3390/ijms14059834
S. Y. Elovich and O. G. Larinov, Izv. Akad. Nauk SSSR 2, 209 (1962).
R. Han, W. Zou, Z. Zhang, et al., J. Hazard. Mater. 137, 384 (2006). https://doi.org/10.1016/j.jhazmat.2006.02.021
P. A. Perminov, N. R. Kil’deeva, L. M. Timofeeva, et al., Izv. Vyssh. Ucheb. Zav., Ser. Khim. Khim. Tekhnol. 50, 53 (2007).
N. R. Kildeeva, P. A. Perminov, L. V. Vladimirov, et al., Russ. J. Bioorg. Chem. 35, 360 (2009).
M. Ozacar, Process Biochem. 40, 565 (2005). https://doi.org/10.1016/j.procbio.2004.01.032
L. M. k. Alifkhanova, O. I. Merezhnikova, Yu. S. Petrova, et al., Russ. J. Appl. Chem. 93, 1392 (2020). https://doi.org/10.1134/S1070427220090128
Funding
The work was supported by the Russian Science Foundation (project No. 21-73-00052), https://rscf.ru/project/21-73-00052.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by O. Fedorova
Rights and permissions
About this article
Cite this article
Petrova, Y.S., Alifkhanova, L.M., Kuznetsova, K.Y. et al. Sorbents with a Taurine Function: Kinetics of Interaction with Singly and Doubly Charged Metal Ions in Ammonium Acetate Buffer Solution. Russ. J. Inorg. Chem. 67, 1080–1087 (2022). https://doi.org/10.1134/S003602362207018X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003602362207018X