Abstract
The mechanisms of acetylene vapour-phase hydrochlorination over chloro complexes of noble metals were reviewed. The nature of the active sites of heterogeneous catalysts, isotope effects of HCl/DCl, stereochemistry of reaction products, possible stepwise mechanisms, and mechanisms of individual steps were considered.






Similar content being viewed by others
Notes
In aqueous solutions, the PtII chloride complexes catalyze the hydrochlorination of acetylene, while the PtIV chloride complexes do not show any appreciable catalytic activity [29].
For terminal carbonyls, νСО is generally shifted toward long waves relative to the 2143 cm–1 of free СО and lies between 2125 and 1850 cm–1 [38].
The stretching vibrations of the С≡С triple bond generally show themselves in the range 2260–2190 cm–1 [41]. A small bathochromic shift is the consequence of the binding of acetylene in the π-complex.
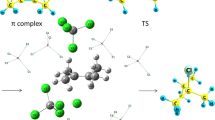
The stoichiometric consequence of the acetylene chloropalladation step is the formation of a PdII chlorovinyl derivative and adjacent palladium complex lying on the (100) crystallographic plane with a coordination vacancy that bears a local positive charge.
REFERENCES
Ciacci, L., Passarini, F., and Vassura, I., Resour., Conserv. Recycl., 2017, vol. 123, p. 108.
Johnston, P., Carthey, N., and Hutchings, G.J., J. Am. Chem. Soc., 2015, vol. 137, p. 14 548.
Flid, M.R., Katal.Prom-sti., 2009, vol. 1, p. 285.
Meng, X., Ding, Q., Wang, Q., and Duan, L., Energy Environ.Focus, 2014, vol. 3, no. 1, p. 37.
Polyvinyl Chloride (PVC) – Market Study Ceresana. http://www.ceresana.com/en/market-studies/plastics/polyvinyl-chloride/ (cited May 06, 2019).
Schellerer, K.-M., Kufner, Th., Mieden, O., and Vogel, E., Kunstoffe Int., 2016, vol. 10, p. 28.
Magistro, A.J. and Cowfer, J.A., J. Chem. Educ., 1986, vol. 63, p. 1056.
Zhang, J., Liu, N., Li, W., and Dai, B., Front. Chem. Sci. Eng., 2011, vol. 5, p. 514.
Dai, B., Wang, Q., Yu, F., and Zhu, M., Sci. Rep., 2015, vol. 5.
Zhou, K., Jia, J., Li, C., Xu, H., Zhou, J., Luo, G., and Wei, F., Green Chem., 2015, vol. 17, p. 356.
Ren, W., Duan, L., Zhu, Z., Du, W., An, Z., Xu, L., Zhang, C., Zhuo, Y., and Chen, C., Environ. Sci. Technol., 2014, vol. 48, p. 2321.
Temkin, O.N., Shestakov, G.K., and Treger, Yu.A., Atsetilen: khimiya, mekhanizmy reaktsii i tekhnologiya (Acetylene: Chemistry, Reaction Mechanisms and Technology), Moscow: Khimiya, 1991.
Liu, X., Conte, M., Elias, D., Lu, L., Morgan, D.J., Freakley, S.J., Johnston, P., Kiely, C.J., and Hutchings, G.J., Catal. Sci. Technol., 2016, vol. 6, p. 5144.
Minamata Convention on Mercury. http://www.mercuryconvention.org/ (Cited May 06, 2019).
Davies, C.J., Miedziak, P.J., Brett, G.L., and Hutchings, G.J., Chin. J. Catal., 2016, vol. 37, p. 1600.
Zhong, J., Xu, Y., and Liu, Z., Green Chem., 2018, vol. 20, p. 2412.
Trotuş, I.-T., Zimmermann, T., and Schüth, F., Chem. Rev., 2014, vol. 114, p. 1761.
Xu, H. and Luo, G., J. Ind. Eng. Chem., 2018, vol. 65, p. 13.
Eisenstein, O. and Hoffmann, R., J. Am. Chem. Soc., 1980, vol. 102, no. 19, p. 6148.
Germain, J.E., Catalytic Conversion of Hydrocarbons, London: Academic, 1969.
Kazanskii, B.V., Usp. Khim., 1988, vol. 57, no. 12, p. 1937.
Lunin, V.V. and Romanovskii, B.V., Vestn. Mosk. Un-ta, Ser. 2, 1999.
Mitchenko, S.A., Theor. Exp. Chem., 2007, vol. 43, no. 4, p. 211.
Mitchenko, S.A., Kinet. Catal., 1998, vol. 39, no. 6, p. 859.
Shubin, A.A., Mitchenko, R.S., and Vdovichenko, A.N., Russ. J. Org. Chem., 2006, vol. 42, no. 5, p. 768.
Mitchenko, S.A., Krasnyakova, T.V., and Zhikharev, I.V., Theor. Exp. Chem., 2008, vol. 44, no. 5, p. 316.
Melander, L. and Saunders, W.H., Reaction Rates of Isotopic Molecules, New York: Wiley, 1980.
Mitchenko, S.A, Ananikov, V.P., and Beletskaya, I.P., Russ. J. Org. Chem., 1998, vol. 34, no. 12, p. 1786.
Sil’chenko, L.A., Panova, S.A., Shestakov, G.K., and Temkin, O.N., Kinet. Catal., 1997, vol. 38, no. 6, p. 790.
Mitchenko, S.A., Khomutov, E.V., Shubin, A.A., and Shul’ga, Yu.M., Theor. Exp. Chem., 2003, vol. 39, no. 4, p. 255.
Mitchenko, S.A., Khomutov, E.V., Shubin, A.A., and Shul’ga, Yu.M., J. Mol. Catal. A: Chem., 2004, vol. 212, p. 345.
Barker, C., James, P.L., and Yarwood, J., Faraday Discuss. Chem. Soc., 1977, no. 64, p. 188.
Mitchenko, S.A., Krasnyakova, T.V., and Zhikharev, I.V., Theor. Exp. Chem., 2012, vol. 48, no. 3, p. 157.
Mitchenko, S.A. and Krasnyakova, T.V., Kinet. Catal., 2014, vol. 55, no. 6, p. 722.
Mitchenko, S.A., Krasnyakova, T.V., and Zhikharev, I.V., Kinet. Catal., 2009, vol. 50, no. 5, p. 734.
Mitchenko, S.A., Khomutov, E.V., Kovalenko, V.V., and Beletskaya, I.P., Kinet. Catal., 2002, vol. 43, no. 4, p. 469.
Mitchenko, S.A., Khomutov, E.V., and Kovalenko, V.V., Inorg. Chim. Acta, 2001, vol. 320, nos. 1–2, p. 31.
Collman, J.P., Hegedus, L.S., Norton, J.R., and Finke, R.G., Principles and Applications of Organotransition Metal Chemistry, Mill Valley: University Science Books, 1987, p. 859.
Mitchenko, R.S., Shubin, A.A., and Krasnyakova, T.V., Theor. Exp. Chem., 2006, vol. 42, no. 5, p. 314.
Mitchenko, S.A., Krasnyakova, T.V., Mitchenko, R.S., and Korduban, A.M., J. Mol. Catal. A: Chem., 2007, vol. 275, p. 101.
Nakanisi, K., Infrared Absorption Spectroscopy. Practical, San Francisco: Holden Day, 1962.
Mitchenko, S.A., Krasnyakova, T.V., and Zhikharev, I.V., Theor. Exp. Chem., 2010, vol. 46, no. 1, p. 32.
Krasnyakova, T.V., Zhikharev, I.V., Mitchenko, R.S., Burkhovetski, V.I., Korduban, A.M, Kryshchuk, T.V., and Mitchenko, S.A., J. Catal., 2012, vol. 288, p. 33.
Mitchenko, S.A., Krasnyakova, T.V., and Zhikharev, I.V., Kinet. Catal., 2009, vol. 50, no. 5, p. 734.
Mitchenko, R.S., Shubin, A.A., and Vdovichenko, A.N., Theor. Exp. Chem., 2006, vol. 42, no. 3, p. 186.
Herberhold, M., Metal Complexes: Complexes with Mono-Olefinic Liga, Amsterdam: Elsevier, 1974.
Mitchenko, S.A., Khomutov, E.V., Shubin, A.A., and Beletskaya, I.P., Kinet. Catal., 2004, vol. 45, no. 3, p. 391.
JCPDS: International Centre for Diffraction Data, no. 25-1203.
JCPDS: International Centre for Diffraction Data, no. 9-367.
JCPDS: International Centre for Diffraction Data, no. 12-412.
Mitchenko, S.A., Zamashchikov, V.V., and Shubin, A.A., Kinet. Catal., 1993, vol. 34, no. 3, p. 479.
Flid, M.R. and Treger, Yu.A., Vinilkhlorid: khimiya i tekhnologiya (Vinyl Chloride: Chemistry and Technology), Moscow: Kalvis, 2008, vol. 1.
Mitchenko, S.A., Khomutov, E.V., and Shubin, A.A., Theor. Exp. Chem., 2003, vol. 39, no. 2, p. 96.
Luinstra, G.A., Wang, L., Stahl, S.S., Labinger, J.A., and Bercaw, J.E., J. Organomet. Chem., 1995, vol. 504, p. 75.
Gordon, A.J. and Ford, R.A., The Chemist’s Companion: A Handbook of Practical Data, Techniques, and References, New York: Wiley, 1973.
Paulusse, J.M.J. and Sijbesma, R.P., Chem. Commun., 2008, p. 4416.
Ananikov, V.P., Musaev, D.G., and Morokuma, K., Organometallics, 2005, vol. 24, p. 715.
Gurvich, L.V., Karachevtseva, G.V., Kondrat’eva, N.V., and Lebedev, Yu.A., Energii razryva khimicheskikh svyazei. Potentsialy ionizatsii i srodstvo k elektronu (The Energy of Breaking Chemical Bonds. Ionization Potentials and Electron Affinity), Moscow: Nauka, 1974.
Hill, G.S., Rendina, L.M., and Puddephatt, R., Organometallics, 1995, vol. 14, p. 4966.
Simonov, P.A., Filimonova, S.V., Kryukova, G.N., Moroz, E.M., Likholobov, V.A., Kuretzky, T., and Boehm, H.P., Carbon, 1999, vol. 37, p. 591.
Simonov, P.A., Moroz, E.M., Chuvilin, A.L., Kolomiichuk, V.N., Boronin, A.I., and Likholobov, V.A., Stud. Surf. Sci. Catal., 1995, vol. 91, p. 977.
Moroz, E.M., Simonov, P.A., Bogdanov, S.V., and Chuvilin, A.L., Mater. Sci. Forum, 2000, vol. 321, p. 1074.
Ryndin, Yu.A., Alekseev, O.S., Simonov, P.A., and Likholobov, V.A., J. Mol. Catal., 1989, vol. 55, p. 109.
Simonov, P.A., Troitskii, S.Yu., and Likholobov, V.A., Kinet. Catal., 2000, vol. 41, no. 2, p. 255.
Simonov, P.A., Romanenko, A.V., Prosvirin, I.P., Moroz, E.M., Boronin, A.I., Chuvilin, A.L., and Likholobov, V.A., Carbon, 1997, vol. 35, no. 1, p. 73.
Simonov, P.A., Semikolenov, V.A., Likholobov, V.A., Voronin, A.I., and Ermakov, Yu.I., Izv. Akad. Nauk SSSR, Ser. Khim., 1988, no. 12, p. 2719.
Simonov, P.A., Chuvilin, A.L., and Likholobov, V.A., Izv. Akad. Nauk SSSR, Ser. Khim., 1989, no. 9, p. 1952.
Simonov, P.A., Moroz, E.M., Likholobov, V.A., and Plaksin, G.V., Izv. Akad. Nauk SSSR, Ser. Khim., 1990, no. 7, p. 1478.
Simonov, P.A., Romanenko, A.V., Prosvirin, I.P., Kryukova, G.N., Chuvilin, A.L., Bogdanov, S.V., Moroz, E.M., and Likholobov, V.A., Stud. Surf. Sci. Catal., 1998, vol. 118, p. 15.
Simakova, O.A., Simonov, P.A., Romanenko, A.V., and Simakova, I.L., React. Kinet. Catal., 2008, vol. 95, p. 3.
Song, Q.L., Wang, S.J., Shen, B.X., and Zhao, J.G., Pet. Sci. Technol., 2010, vol. 28, p. 1825.
Wang, L., Wang, F., and Wang, J., Catal. Commun., 2015, vol. 65, p. 41.
Wang, L., Wang, F., Wang, J., Tang, X., Zhao, Y., Yang, D., Jia, F., and Hao, T., React. Kinet., Mech. Catal., 2013, vol. 110, p. 187.
Wang, L., Wang, F., and Wang, J., Catal. Commun., 2016, vol. 83. P. 9.
Wang, F., Wang, L., Wang, J., Zhao, Y., Wang, Y., and Yang, D., React. Kinet., Mech. Catal., 2015, vol. 114, p. 725.
Krasnyakova, T.V., Nikitenko, D.V., Khomutova, E.V., and Mitchenko, S.A., Kinet. Catal., 2017, vol. 58, no. 5, p. 533.
Evers, J., Beck, W., Gobel, M., Jakob, S., Mayer, P., Oehlinger, G., Rotter, M., and Klapotke, T.M., Angew. Chem., 2010, vol. 49, p. 5677.
Nkosi, B., Coville, N.J., and Hutchings, G.J., J. Chem. Soc., Chem. Commun., 1988, p. 71.
Nkosi, B., Coville, N.J., and Hutchings, G.J., Appl. Catal., 1988, vol. 43, p. 33.
Nkosi, B., Coville, N.J., Hutchings, G.J., Adams, M.D., Friedl, J., and Wagner, F.E., J. Catal., 1991, vol. 128, p. 366.
Nkosi, B., Adams, M.D., Coville, N.J., and Hutchings, G.J., J. Catal., 1991, vol. 128, p. 378.
Hutchings, G.J., Gold Bull., 1996, vol. 29, p. 123.
Conte, M., Carley, A.F., Heirene, C., Willock, D.J., Johnston, P., Herzing, A.A., Kiely, C.J., and Hutchings, G.J., J. Catal., 2007, vol. 250, p. 231.
Hutchings, G.J., Top. Catal., 2008, vol. 48, p. 55.
Hutchings, G.J., Gold Bull., 2009, vol. 42, p. 260.
Conte, M., Davies, C.J., Morgana, D.J., Davies, T.E., Elias, D.J., Carley, A.F., Johnston, P., and Hutchings, G.J., J. Catal., 2013, vol. 297, p. 128.
Conte, M., Davies, C.J., Morgan, D.J., Carley, A.F., Johnston, P., and Hutchings, G.J., Catal. Lett., 2014, vol. 144, p. 1.
Hutchings, G.J., Top. Catal., 2014, vol. 57, p. 1265.
Johnston, P., Carthey, N., and Hutchings, G.J., J. Am. Chem. Soc., 2015, vol. 137, p. 14 548.
Malta, G., Freakley, S.J., Kondrat, S.A., and Hutchings, G.J., Catal. Commun., 2017, vol. 53, p. 11 733.
Malta, G., Kondrat, S.A., Freakley, S.J., Davies, C.J., Dawson, S., Liu, X., Lu, L., Dymkowski, K., Fernandez-Alonso, F., Mukhopadhyay, S., Gibson, E.K., Wells, P.P., Parker, S.F., Kiely, C.J., and Hutchings, G.J., ACS Catal., 2018, vol. 8, p. 8493.
Zhao, Y., Zhao, F., and Kang, L., J. Mol. Model., 2018, vol. 24, p. 61.
Gu, J., Du, Q., Han, Y., He, Z., Li, W., and Zhang, J., Phys. Chem. Chem. Phys., 2014, vol. 116, p. 25 498.
Zhang, J., He, Z., Lib, W., and Han, Y., RSC Adv., 2012, p. 4814.
Zhu, M., Kang, L., Su, Y., Zhang, S., and Dai, B., Can. J. Chem., 2013, vol. 91, p. 120.
Gong, W., Zhao, F., and Kang, L., Comput. Theor. Chem., 2018, vol. 1130, p. 83.
Zhao, F., Wang, Y., and Kang, L., Can. J. Chem., 2016, vol. 94, p. 842.
Wang, Y., Zhu, M., Kang, L., and Dai, B., RSC Adv., 2014, vol. 4, p. 38 466.
Xu, H., Meng, S., and Luo, G., Catal. Sci. Technol., 2018, vol. 8, p. 1176.
Wan, F., Chao, S., Guan, Q., Wang, G.C., and Li, W., Catal. Commun., 2017, vol. 101, p. 120.
Wegner, H.A. and Auzias, M., Angew. Chem., 2011, vol. 50, p. 8236.
Reutov, O.A., Kurts, A.L., and Butin, K.P., Organicheskaya khimiya (Organic Chemistry), Moscow: Moscow State University, 1999.
Funding
This study was financially supported by the Ministry of Education and Science of the Russian Federation under the government contract for R&D (application no. 10.2980.2017/4.6).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by L. Smolina
Abbreviations: VCM, vinyl chloride monomer; PVC, polyvinyl chloride; DCE, dichloroethane; KIE, kinetic isotope effect; γ, product isotope effect; TEMPO, 2,2,6,6-tetramethylpiperidin-1-yl)oxyl; DRIFT, diffuse reflection infrared Fourier transform spectroscopy; EDS SEM, energy dispersive X-ray spectroscopy with scanning electron microscopy; TOF, specific catalytic activity (turnover frequency); HAADF-STEM, high-angle annular dark-field scanning transmission electron microscopy; XAFS, X-ray absorption fine structure spectroscopy; XPS, X-ray photoelectron spectroscopy; EPR, electron paramagnetic resonance; NMR, nuclear magnetic resonance; HRTEM, high-resolution transmission electron microscopy; SAXS, small-angle X-ray scattering; WAXS, wide-angle X-ray scattering; RDF, radial distribution function; RED, radial electron density distribution; DFT, density functional theory.
Rights and permissions
About this article
Cite this article
Krasnyakova, T.V., Nikitenko, D.V. & Mitchenko, S.A. Mechanisms of the Catalytic Hydrochlorination of Acetylene: Active Sites, Isotope Effects, and Stereoselectivity. Kinet Catal 61, 58–79 (2020). https://doi.org/10.1134/S0023158420010036
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158420010036