Abstract
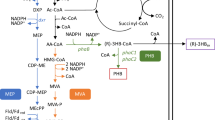
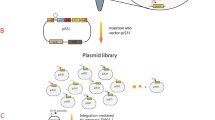
The uncoordinated expression of a target biosynthetic pathway often results in low growth and productivity for a producing strain due to the accumulation of toxic metabolic intermediates. In this study, we report an increase in isoprene accumulation by an engineered strain of Pantoea ananatis that possesses foreign, artificial mevalonate-pathway genes. This was achieved due to the simultaneous, φ80-Int-dependent integration of a mixture of conditionally replicated plasmids carrying genes for the upper and lower mevalonate pathways and mevalonate kinase, followed by selection according to the isoprene accumulation of clones with optimal copy numbers of the integrative cassettes. A previously developed method, Dual In/Out, was modified to accelerate the construction of recipient strains containing several attB sites of the φ80 phage in their genomes.



Similar content being viewed by others
REFERENCES
Siebert, D. and Wendisch, V.F., Metabolic pathway engineering for production of 1,2-propanediol and 1-propanol by Corynebacterium glutamicum,Biotechnol. Biofuels., 2015, vol. 8, pp. 91. https://doi.org/10.1186/s13068-015-0269-0
Atsumi, S., Cann, A.F., Connor, M.R., et al., Metabolic engineering of Escherichia coli for 1-butanol production, Metab. Eng., 2008, vol. 10, no. 6, pp. 305–311. https://doi.org/10.1016/j.ymben.2007.08.003
Qian, Z.G., Xia, X.X., and Lee, S.Y., Metabolic engineering of Escherichia coli for the production of putrescine: a four carbon diamine, Biotechnol. Bioeng., 2009, vol. 104, no. 4, pp.651–662. https://doi.org/10.1002/bit.22502
Bentley, F.K., Zurbriggen, A., and Melis, A., Heterologous expression of the mevalonic acid pathway in cyanobacteria enhances endogenous carbon partitioning to isoprene, Mol. Plant, 2014, vol. 7, no. 1, pp.71–86. https://doi.org/10.1093/mp/sst134
Hayes, F., Transposon-based strategies for microbial functional genomics and proteomics, Annu. Rev. Genet., 2003, vol. 37, pp. 3–29. https://doi.org/10.1146/annurev.genet.37.110801.142807
Harshey, R.M. and Transposable Phage Mu, Microbiol. Spectr., 2014, vol. 2, no. 5. https://doi.org/10.1128/microbiolspec.MDNA3-0007-2014
Link, A.J., Phillips, D., and Church, G.M., Methods for generating precise deletions and insertions in the genome of wild-type Escherichia coli: application to open reading frame characterization, J. Bacteriol., 1997, vol. 179, no. 20, pp.6228–6237.
Gu, P., Yang, F., Su, T., et al., A rapid and reliable strategy for chromosomal integration of gene(s) with multiple copies, Sci. Rep., 2015, vol. 5, p. 9684. https://doi.org/10.1038/srep09684
Haldimann, A. and Wanner, B.L., Conditional-replication, integration, excision, and retrieval plasmid-host systems for gene structure-function studies of bacteria, J. Bacteriol., 2001, vol. 183, pp. 6384–6393. https://doi.org/10.1128/JB.183.21.6384-6393.2001
Huang, L.-C., Wood, E.A., and Cox, M.M., Convenient and reversible site-specific targeting of exogenous DNA into a bacterial chromosome by use of the FLP recombinase: the FLIRT system, J. Bacteriol., 1997, vol. 179, no. 19, pp. 6076–6083.
Cregg, J.M. and Madden, K.R., Use of site-specific recombination to regenerate selectable markers, Mol. Gen. Genet. 1989, vol. 219, pp. 320–323. https://doi.org/10.1007/BF00261194
Albert, H., Dale, E.C.LeeE., and Ow, D.W., Site-specific integration of DNA into wild-type and mutant lox sites placed in the plant genome, Plant J., 1995, vol. 7, pp. 649–659. https://doi.org/10.1046/j.1365-313X.1995.7040649.x
Hoess, R.H., Wierzbicki, A., and Abremski, K., The role of the loxP spacer region in PI site-specific recombination, Nucleic Acids Res., 1986, vol. 14, no. 5, pp. 2287–2300. https://doi.org/10.1093/nar/14.5.2287
Lambert, J.M., Bongers, R.S., and Kleerebezem, M., Cre-lox-based system for multiple gene deletions and selectable-marker removal in Lactobacillus plantarum,Appl. Environ. Microbiol., 2007, vol. 73, pp. 1126–1135. https://doi.org/10.1128/AEM.01473-06
Peredelchuk, M.Y. and Bennett, G.N., A method for construction of E. coli strains with multiple DNA insertions in the chromosome, Gene, 1997, vol. 187, pp. 231–238. https://doi.org/10.1016/S0378-1119(96)00760-3 https://doi.org/10.1111/j.1365-2958.2009.06976.x
Katashkina, Zh.I., Skorokhodova, A.Yu., Zimenkov, D.V., et al., Tuning the Expression level of a gene located on a bacterial chromosome, Mol. Biol. (Moscow), 2005, vol. 39, no. 5, pp. 719–726.
Minaeva, N.I., Gak, E.R., Zimenkov, D.V., et al., Dual-In/Out strategy for genes integration into bacterial chromosome: a novel approach to step-by-step construction of plasmid-less marker-less recombinant E. coli strains with predesigned genome structure, BMC Biotechnol., 2008, vol. 8, p. 63. https://doi.org/10.1186/1472-6750-8-63
Hara, Y., Kadotani, N., Izui, H., et al., The complete genome sequence of Pantoea ananatis AJ13355, an organism with great biotechnological potential, Appl. M-icrobiol. Biotechnol., 2012, vol. 93, pp. 331–341. https://doi.org/10.1007/s00253-011-3713-5
Katashkina, J.I., Hara, Y., Golubeva, L.I., et al., U. Use of the lambda Red-recombineering method for genetic engineering of Pantoea ananatis,BMC Mol. Biol., 2009, vol. 10, p. 34. https://doi.org/10.1186/1471-2199-10-34
Izui, H., Hara, Y., Sato, M., and Akiyoshi, N., Method for producing L-glutamic acid, US Patent no. 6596517, 2003.
Takumi, K., Ziyatdinov, M.K., Samsonov, V., and Nonaka, G., Fermentative production of cysteine by Pantoea ananatis,Appl. Environ. Microbiol., 2017, vol. 83, no. 5. https://doi.org/10.1128/AEM.02502-16
Yoon, S.H. and Lee, S.H., Das A., et al., Combinatorial expression of bacterial whole mevalonate pathway for the production of beta-carotene in E. coli,J. Biotechnol., 2009, vol. 140, nos. 3–4, pp. 218–226. https://doi.org/10.1016/j.jbiotec.2009.01.008
Zurbriggen, A., Kirst, H., and Melis, A., Isoprene production via the mevalonic acid pathway in Escherichia coli (bacteria), Bioenerg. Res., 2012. https://doi.org/10.1007/s12155-012-9192-4
Tajima, Y., Nishio, Y., Katashkina, J.Y., and Bylino, O.V., Method of producing isoprene compound, WO Patent no. WO2016084963 (A1), 2016.
Mihara, Y., Rachi, H., Nishio, Y., et al., Method of producing isoprene monomer, US Patent no. US2015275233 (A1), 2015.
Mashko, S.V., Skorokhodova, A.Yu., Zimenkov, D.V., et al., The use of metabolic regulation to optimize gene expression in bacterial cells: a new direction in biotechnology of the 21st century, Biotekhnologiya, 2002, vol. 4, pp. 3–14.
Martin, V.J., Pitera, D.J., Withers, S.T., et al., Engineering a mevalonate pathway in Escherichia coli for production of terpenoids, Nat. Biotechnol., 2003, vol. 21, no. 7, pp. 796–802. https://doi.org/10.1038/nbt833
Kizer, L., Pitera, D.J., Pfleger, B.F., and Keasling, J.D., Application of functional genomics to pathway optimization for increased isoprenoid production, Appl. Environ. Microb., 2008, vol. 74, no. 10, pp. 3229–3241.
Primak, Y.A., Du, M., Miller, M.C., et al., Characterization of a feed-back resistant mevalonate kinase from the archaeon Methanosarcina mazei,Appl. Env. Microbiol., 2011, vol. 77, no. 21, pp. 7772–7778. https://doi.org/10.1128/AEM.05761-11
Kazieva, E., Yamamoto, Y., Tajima, Y., et al., Characterization of feedback-resistant mevalonate kinases from the methanogenic archaeons Methanosaeta concilii and Methanocella paludicola,Microbiology, 2017, vol. 163, no. 9, pp. 1283–1291. https://doi.org/10.1099/mic.0.000510
Sanchez, M.C., Krasnec, K.V., Parra, A.S., et al., Effect of praziquantel on the differential expression of mouse hepatic genes and parasite ATP binding cassette transporter gene family members during Schistosoma mansoni infection, PLoS Negl. Trop. Dis., vol. 11, no. 6. e0005691. https://doi.org/10.1371/journal.pntd.0005691
George, K.W., Thompson, M.G., Kim, J., et al., Integrated analysis of isopentenyl pyrophosphate (IPP) toxicity in isoprenoid-producing Escherichia coli,Metab. Eng., 2018, vol. 47, pp. 60–72. https://doi.org/10.1016/j.ymben.2018.03.004
Berthelot, K., Estevez, Y., Deffieux, A., and Peruch, F., Isopentenyl diphosphate isomerase: a checkpoint to isoprenoid biosynthesis, Biochimie, 2012, vol. 94, no. 8, pp. 1621–1634. https://doi.org/10.1016/j.biochi.2012.03.021
Arraiano, C.M. and Maquat, L.E., Post-transcriptional control of gene expression: effectors of mRNA decay, Mol. Microbiol., 2003, vol. 49, pp. 267–276. https://doi.org/10.1046/j.1365-2958.2003.03545.x
de Smit, M.H. and van Dui, J., Control of translation by mRNA secondary structure in Escherichia coli. A quantitative analysis of literature data, J. Mol. Biol., 1994, vol. 244, pp. 144–150.
Bertani, G., Studies on lysogenesis. I. The mode of phage liberation by lysogenic Escherichia coli,J. Bacteriol., 1951, vol. 62, pp. 293–300.
Hayashi, Y., Harada, M., Takaoka, S., et al., Isoprene synthase and polynucleotide encoding same, and method for producing isoprene monomer, WO Patent no. WO2013179722A1, 2013.
Herrero, M., de Lorenzo, V., and Timmis, K.N., Transposon vectors containing non-antibiotic resistance selection markers for cloning and stable chromosomal insertion of foreign genes in gram-negative bacteria, J. Bacteriol., 1990, vol. 172, no. 11, pp. 6557–6567.
Andreeva, I.G., Golubeva, L.I., Kuvaeva, T.M., et al., Identification of Pantoea ananatis gene encoding membrane pyrroloquinoline quinone (PQQ)-dependent glucose dehydrogenase and pqqABCDEF operon essential for PQQ biosynthesis, FEMS Microbiol. Lett., 2011, vol. 318, no. 1, pp. 55–60. https://doi.org/10.1111/j.1574-6968.2011.02240.x
Zimenkov, D.V., Skorokhodova, A.Yu., Katashkina, Zh. I., et al., E. coli chromosome regions that are more preferable for gene insertion, when the phage mu-driven system is used for integration, Biotekhnologiya, 2004, vol. 6, pp. 3–18.
Block, D.H.S., Hussein, R., Liang, L.W., and Lim, H.N., Regulatory consequences of gene translocation in bacteria, Nucleic Acids Res., 2012, vol. 40, no. 18, pp. 8979–8992. https://doi.org/10.1093/nar/gks694
Schute, H., Flossdorf, J., Sahm, H., and Kula, M.R., Purification and properties of formaldehyde dehydrogenase and formate dehydrogenase from Candida boidinii,Eur. J. Biochem., 1976, vol. 62, no. 1, pp. 151–160.
Dalwadi, M.P., Garavaglia, M., Webb, J.P., et al., Applying asymptotic methods to synthetic biology: modelling the reaction kinetics of the mevalonate pathway, J. Theor. Biol., 2018, vol. 439, pp. 39–49. https://doi.org/10.1016/j.jtbi.2017.11.022
Studier, F.W., Rosenberg, A.H., Dunn, J.J., and Dubendorff, J.W., Use of T7 RNA polymerase to direct expression of cloned genes, Methods Enzymol., 1990, vol. 185, pp. 60–89.
Olins, P.O. and Rangwala, S.H., A novel sequence element derived from bacteriophage T7 mRNA acts as an enhancer of translation of the lacZ gene in Escherichia coli,J. Biol. Chem., 1989, vol. 264, no. 29, pp. 16973–16976.
ACKNOWLEDGMENTS
The authors are extremely grateful to A.D. Kivero (AGRI) for valuable comments during the work with analytical equipment. The authors express their appreciation to Uehara (Nagai) Yuri for the fruitful cooperation and coordination of the joint work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
This article does not contain any studies involving animals or human participants performed by any of the authors.
SUPPLEMENTARY MATERIALS
The electronic version of the article contains the Supplementary Materials available free of charge on the journal website http://www.biotechnology-journal.ru.
Additional information
Translated by I. Gordon
Abbreviations: Bλ—attB site of λ phage; Bφ80—attB site of φ80 phage; cat—chloramphenicol resistance gene; Cm—chloramphenicol; CmR—Cm-resistance; Int—phage integrase; IPTG—isopropyl-β-D-thiogalactopyranoside; kan—kanamycin resistance gene; Km—kanamycin; KmR—kanamycin resistance; MES—4-morpholineethanesulfonic acid; MOPS—4-morpholinepropanesulfonic acid; LB medium—lysogeny broth medium; Lλ—attL site of λ phage; Lφ80—attL site of φ80 phage; Rλ—attR site of λ phage; Rφ80—attR site of φ80 phage; Tc—tetracyclin; TcR—tetracyclin resistance; tetAR—tetracyclin resistance genes; Xis—excisionase.
Rights and permissions
About this article
Cite this article
Katashkina, J.I., Kazieva, E.D., Tajima, Y. et al. Increased Isoprene Production by the Recombinant Pantoea ananatis Strain due to the Balanced Amplification of Mevalonate Pathway Genes. Appl Biochem Microbiol 55, 850–860 (2019). https://doi.org/10.1134/S0003683819090023
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683819090023