Abstract
This study aimed to assess the efficacy and safety of esaxerenone (CS-3150) in treating primary hypertension. PubMed (Medline), Cochrane Central Register of Controlled Trials (CENTRAL), and Embase databases were searched for articles published until April 18, 2023. The outcomes included were diastolic blood pressure (DBP), systolic blood pressure (SBP), 24 h DBP, 24 h SBP, and adverse events. The meta-analysis was conducted using RevMan 5.3. This study included three trials. CS-3150 5 mg had a greater effect on lowering the SBP, DBP, 24 h SBP, and 24 h DBP than either CS-3150 2.5 mg or eplerenone 50 mg. In contrast, CS-3150 2.5 mg and eplerenone 50 mg showed no significant difference in lowering DBP, SBP, 24 h DBP, and 24 h SBP. Moreover, adverse events occurred at comparable rates in the three groups. CS-3150 (especially CS-3150 5 mg) is an effective and safe treatment for primary hypertension; which can reduce blood pressure and alleviate hypertensive symptoms.
Similar content being viewed by others
Introduction
Hypertension is a major global health challenge and contributor to stroke, cardiovascular disease, and chronic kidney disease [1]. Globally, hypertension affects approximately one billion people, and this number is expected to increase to over 1,500,000 by 2025 [2, 3]. The proportion of individuals with hypertension varies worldwide, affecting 37.3% and 22.9% of individuals in developed and developing countries, respectively [4]. Hypertension can be categorized as secondary or primary based on its etiology. Primary hypertension, which has no identifiable secondary cause, is the predominant type of hypertension and affects more than 90% of hypertensive patients [5]. This makes it a major chronic, non-communicable disease globally.
Mineralocorticoid receptor (MR) antagonists are highly effective group of drugs in managing Primary hypertension [6]. A previous study showed that O-linked β-N-acetylglucosamine modification of MR enhanced its protein expression and transcriptional activity in vitro and in vivo under high-glucose conditions [7], suggesting a potential advantage of MR antagonists in patients with resistant hypertension and diabetic nephropathy. Currently, spironolactone, eplerenone, and esaxerenone (CS-3150) are clinically used as MR antagonists. Spironolactone binds poorly to MR and may induce adverse events, such as gynecomastia, menstrual irregularities, and impotence [8]. Eplerenone binds more specifically to MR than spironolactone; however, hyperkalemia remains a clinical issue [9, 10]. Esaxerenone, CS-3150, is a new nonsteroidal MR antagonist that has at least 1000 times higher selectivity for MR than other MR antagonists and does not exhibit antagonism for androgen, progesterone, and glucocorticoid receptors even at high doses [11]. CS-3150 potently blocked the binding of [3H]-aldosterone to MR, with a median inhibitory concentration (IC50) of 9.4 nmol/L, better than eplerenone and spironolactone (IC50 of 713 nmol/L and 36 nmol/L, respectively) [12]. Moreover, CS-3150 2.5 mg or 5 mg significantly and dose-dependently lowered sitting diastolic and systolic blood pressures (DBP and SBP) compared to the placebo [13]. These results suggested that CS-3150 is a highly selective and orally effective MR antagonist that can be used to manage renal disease, cardiovascular disease, and hypertension. This study comprehensively assessed the efficacy and safety of CS-3150 for the treatment of Primary hypertension through a meta-analysis to provide evidence-based support for its clinical management.
Materials and methods
Search strategy
PubMed (Medline), Cochrane Central Register of Controlled Trials (CENTRAL), and Embase databases were searched for studies published until April 18, 2023, with no language restrictions. The search terms used were hypertension, Primary hypertension, Esaxerenone, and CS-3150. In addition, we conducted a manual search of relevant domestic and international journals as well as the EU and Japanese clinical trial registry databases.
Selection criteria
The inclusion criteria were: (1) people diagnosed with Primary hypertension (age ≥ 18), (2) studies with a randomized controlled trial (RCT) design, (3) the intervention group received CS-3150 and the comparison group received eplerenone or placebo, and (4) inclusion of at least one of the following outcome metrics: change from baseline in DBP, SBP, 24 h SBP, 24 h DBP, incidence of hyperkalemia, and any adverse events.
Exclusion criteria were as follows: (1) studies on secondary hypertension (2) non-RCT designs such as case reports, observational studies, reviews, conference abstracts, and protocols and (3) Incomplete data published.
Data extraction
Two independent investigators extracted the following information: (1) study information, such as first author, publication year, sample locations, sample size, age, and sex; (2) type of therapy, dosage, SBP and DBP at baseline, 24 h SBP, 24 h DBP at baseline; and (3) primary and secondary outcomes. The risk of bias was assessed using the Cochrane Collaboration’s risk-of-bias tool. Any disagreements regarding study selection, data extraction, and risk of bias assessment were resolved by a third investigator.
Statistical analysis
RevMan 5.3 was used to analyze the data. Continuous outcomes were reported as mean difference (MD) with a 95% confidence interval (CI), and categorical outcomes as relative risk (RR) with a 95% CI. Heterogeneity among the studies was assessed using the I2 value, where I2 > 50% indicated significant heterogeneity. A fixed-effect model or a random-effect model was applied depending on whether I2 ≤ 50% or not. To evaluate the robustness of the meta-analysis results, a sensitivity analysis was conducted.
Results
Characteristics of the included studies
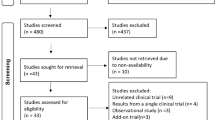
Initially, 52 potentially relevant articles were identified, of which 28 were duplicates and were excluded. Of the remaining 24 articles, 5 were excluded after screening their titles and abstracts. Of the 19 articles, 16 were excluded after assessing their full texts for eligibility for various reasons, including other types of interventions (n = 6), not RCTs (n = 9), and no blood pressure data (n = 1). Finally, three RCTs were selected for quantitative analysis (Fig. 1).
The three RCTs included 1591 patients and were published between 2012 and 2020. The participants were aged between 20 and above, 11.4% of the which were over 65 years old, and had baseline blood pressure blood pressure levels of DBP ≥ 90–110 mmHg, SBP ≥ 140–180 mmHg, and 24 h SBP/DBP ≥ 130/80 mmHg (supplementary table 1).
Quality assessment
Figure 2 presents the risk of bias assessment of the included studies. Overall, the quality of the included literature was moderate. Most studies used a double-blind approach, allocation concealment, and random allocation, but did not specify the methods used. two of the three included studies did not perform a sample size calculation. The three studies had a short observation period, only about 2 weeks, so long-term observation indicators such as mortality were not evaluated. In one of the studies, the results of the experiment were disclosed in the JRCT database, and the exact execution of the experiment was obtained by contacting Daiichi, the esaxerenone experimental institution, by email, and the description of how it was randomly and how it was allocated was not sufficient. Other sources of bias were unclear for all RCTs.
SBP
All RCTs compared the efficacy of eplerenone 50 mg and CS-3150 2.5 mg on SBP. Due to the significant heterogeneity among the studies (I2 = 66%), a random-effects model was chosen for the meta-analysis. The meta-analysis revealed that eplerenone 50 mg and CS-3150 2.5 mg showed no significant difference in SBP (MD = 0.23, 95% CI: −3.73, 4.19; P = 0.91). All RCTs compared the efficacy of CS-3150 5 mg with that of eplerenone 50 mg and CS-3150 2.5 mg on SBP. No heterogeneity was observed among the studies (I2 = 0% and I2 = 21%); therefore, a fixed-effects model was used. The meta-analysis found that CS-3150 5 mg reduced SBP more than eplerenone 50 mg (MD = −4.43, 95% CI: −6.07, −2.79; P < 0.00001) and CS-3150 2.5 mg (MD = −4.13, 95% CI: −6.32, −1.93; P = 0.0002) (Fig. 3).
DBP
All studies reported DBP. The studies were homogeneous (I2 = 0%), and fixed-effects models were used for the analysis. DBP did not significantly differ between CS-3150 2.5 mg and eplerenone 50 mg (MD = −0.39, 95% CI: −1.28, 0.50; P = 0.39). However, CS-3150 5 mg resulted in a significantly greater decrease in DBP compared with CS-3150 2.5 mg (MD = −1.84, 95% CI: −2.76, −0.92; P < 0.0001) and eplerenone 50 mg (MD = −2.10, 95% CI: −3.48, −0.72; P = 0.003) (Fig. 4).
24 h SBP
Figure 5 shows the meta-analysis of 24 h SBP for all RCTs comparing CS-3150 2.5 mg with eplerenone 50 mg, and CS-3150 5 mg with eplerenone 50 mg and CS-3150 2.5 mg. A random-effects model was employed to account for the significant heterogeneity among the studies (I2 = 68% and I2 = 59%) when comparing CS-3150 2.5 mg with eplerenone 50 mg, and CS-3150 5 mg and CS-3150 2.5 mg on 24 h SBP. A fixed-effects model was employed to analyze the effects of eplerenone 50 mg and CS-3150 5 mg on 24 h SBP, as the studies were homogeneous (I2 = 0%). 24 h SBP did not significantly differ between CS-3150 2.5 mg and eplerenone 50 mg (MD = −0.27, 95% CI: −4.84, 4.29; P = 0.91). However, the result showed that CS-3150 5 mg could significantly lower 24 h SBP compared with CS-3150 2.5 mg (MD = -5.65, 95% CI: −9.61, −1.68; P = 0.005) and eplerenone 50 mg (MD = −6.32, 95% CI: −8.00, −4.63; P < 0.00001).
24 h DBP
All RCTs evaluated the efficacy of CS-3150 2.5 mg versus eplerenone 50 mg, and CS-3150 5 mg versus eplerenone 50 mg and CS-3150 2.5 mg on 24 h DBP. Because of the significant heterogeneity among the studies (I2 = 63% and I2 = 57%, respectively), a random-effects model was used to compare the effects of CS-3150 2.5 mg, eplerenone 50 mg, and CS-3150 5 mg on 24 h DBP; whereas a fixed-effects model was used to compare the effects of eplerenone 50 mg and CS-3150 5 mg on 24 h DBP (I2 = 0%). Compared to eplerenone 50 mg, CS-3150 2.5 mg had no significant effect on 24 h DBP (MD = 0.20, 95% CI: −2.12, 2.52; P = 0.87). However, CS-3150 5 mg significantly reduced 24 h DBP compared with both CS-3150 2.5 mg (MD = -3.16, 95% CI: −5.29, −1.03; P = 0.004) and eplerenone 50 mg (MD = −3.06, 95% CI: −3.93, −2.18; P < 0.00001) (Fig. 6).
Adverse events
All the RCTs reported adverse events. A fixed-effects model was employed for all comparisons, as there was no significant heterogeneity among the studies (I2 = 25%, I2 = 0%, and I2 = 49%, respectively). No statistical difference in any adverse events was found between CS-3150 2.5 mg and eplerenone 50 mg (RR = 1.01, 95% CI:0.85, 1.20; P = 0.88), CS-3150 5 mg and eplerenone 50 mg (RR = 1.15, 95% CI:0.97, 1.35; P = 0.10), and CS-3150 5 mg and CS-3150 2.5 mg (RR = 1.13, 95% CI:0.96, 1.33; P = 0.13) in terms of any adverse effects (Fig. 7).
Two RCTs reported hyperkalemia. Since the studies that assessed the outcomes of these interventions on hyperkalemia showed no significant heterogeneity (I2 = 0% for all comparisons), a fixed-effects model was applied. The meta-analysis indicated no statistically significant difference in the risk of hyperkalemia between CS-3150 2.5 mg/5 mg and eplerenone (RR = 2.76, 95% CI:0.88, 8.58; P = 0.08 for CS-3150 2.5 mg vs eplerenone; RR = 2.44, 95% CI:0.77, 7.71; P = 0.13 for CS-3150 5 mg vs eplerenone), or between CS-3150 2.5 mg and 5 mg (RR = 0.88, 95% CI:0.38, 2.06; P = 0.77 for CS-3150 5 mg vs CS-3150 2.5 mg) (Fig. 8).
Sensitivity analysis
Sensitivity analyses were performed by omitting one study at a time to evaluate the reliability of the meta-analysis. These results were consistent and did not significantly change when a single study was omitted.
Discussion
Early cardiovascular disease can be caused by Primary hypertension, which is a major modifiable risk factor for early death and disability around the world [14, 15]. According to global statistics, approximately 100 million people are affected by hypertension, which results to approximately 70,000 deaths annually [16].
Current first-line treatments for hypertension include diuretics, angiotensin-converting enzyme (ACE) inhibitors, and/or angiotensin receptor blockers (ARBs). For patients who do not respond to first-line treatment, combination treatment with two or three agents is recommended, and for those who still do not respond, treatment with mineralocorticoid receptor blockers (MRBs) are recommended. MRBs are currently considered a fourth-line treatment. A few years after discovery of aldosterone, its excess was associated with elevated blood pressure. The current reports of the correlation between renin-aldosterone system activity and primary hypertension are gradually increasing, and clinical and biochemical data indicate that there is a gray area between primary hypertension, and primary aldosteronism [17]. Therefore, antihypertensive therapy for MR can also be effective.
MR antagonists are a class of drugs that effectively lower blood pressure and slow the progression of kidney disease by inhibiting excessive activation of aldosterone on MR, thereby reducing oxidative stress, inflammation, fibrosis, and vascular remodeling [9]. The MR antagonists currently used in clinical practice are spironolactone and eplerenone, both of which belong to the steroid class of MR antagonists with certain efficacy and safety but also some limitations [18]. Spironolactone, although having a strong antagonistic effect on MR, also cross-reacts with other steroid receptors, causing adverse reactions, such as breast development and sexual dysfunction [19, 20]. Eplerenone was found to improve all-cause mortality and cardiovascular mortality in patients with heart failure [21]. Eplerenone has a relatively higher selectivity for MR, but its antagonistic effect on MR is weak and may still cause adverse reactions, such as hyperkalemia [22]. However, there is currently no available evidence to determine the effect of eplerenone on clinically meaningful outcomes such as mortality or morbidity in hypertensive patients [23]. A new non-steroidal MR antagonist, CS-3150, has been approved for the treatment of hypertension. Studies have found that CS-3150 has comparable safety to eplerenone, with no serious hyperkalemia or renal impairment [24, 25].
This study compared the anti-hypertensive effects of CS-3150 and eplerenone in patients with Primary hypertension. We found that CS-3150 5 mg significantly lowered SBP, DBP, 24 h SBP and DBP, whereas CS-3150 2.5 mg and eplerenone 50 mg showed no significant BP reduction. These results suggested that CS-3150 has a dose-dependent antihypertensive effect and is more effective than eplerenone. These results are consistent with those of previous clinical studies. A long-term phase 3 study with a multicenter, open-label design reported that CS-3150 2.5 mg/day or 5 mg/day for 12 or 28 weeks significantly decreased sitting SBP and DBP in patients with Primary hypertension [26]. A post-hoc analysis also demonstrated that CS-3150 improved night-time BP and N-terminal pro-B-type natriuretic peptide, which is a cardiovascular risk marker, based on different nocturnal BP dipping patterns (dippers, extreme dippers, non-dippers, and risers) [27].
Compared to steroid MR antagonists, CS-3150 has better safety and tolerability. Steroid MR antagonists, such as spironolactone and eplerenone, may cause adverse reactions such as hyperkalemia, breast development, and sexual dysfunction. CS-3150 did not cause these adverse reactions in clinical studies, nor did it cause serious hyperkalemia or renal impairment. In this study, we found no statistically significant differences between CS-3150 and eplerenone in terms of adverse events and hyperkalemia.
Some limitations of this study should be acknowledged before translating these results to clinical settings. First, owing to the recent launch of CS-3150 in Japan last 2019, few clinical trials were available, hence only three articles were included in this study. The limited number of literatures may have introduced selection bias and statistical error. Second, this study did not perform a subgroup analysis for different factors such as age, sex, baseline blood pressure, and comorbidities, which may have affected the efficacy and safety of CS-3150. Third, this meta-analysis analyzed adverse events as a safety outcome without separately assessing the incidence of hyperkalemia among the three groups (CS-3150 2.5 mg, CS-3150 5 mg, and eplerenone). Finally, all three RCTs included in this meta-analysis were conducted with the support of the same pharmaceutical company, which may have potential publication bias. Therefore, more large-sample, multicenter, long-term RCTs are needed to validate the benefits and indications of CS-3150 in patients with Primary hypertension. In addition, there are new aldosterone synthase inhibitors, Lorundrostat and Baxdrostat, which have potential advantages over spironolactone and eplerenone, but the difference in efficacy and safety between aldosterone synthase inhibitors and esaxerenone requires further research in the future [28, 29].
Conclusion
In our study, CS-3150 (especially CS-3150 5 mg) was found to have a significant antihypertensive effect in lowering blood pressure in patients with Primary hypertension. To better assess the efficacy of CS-3150 in treating hypertension, larger high-quality RCTs are required.
Summary
What is known about topic
-
Esaxerenone, CS-3150, is a new nonsteroidal mineralocorticoid receptor antagonist that has at least 1000 times higher selectivity for mineralocorticoid receptor than other mineralocorticoid receptor antagonists.
-
CS-3150 is a highly selective and orally effective MR antagonist that can be used to manage renal disease, cardiovascular disease, and hypertension.
What this study adds
-
This study comprehensively assessed the efficacy and safety of CS-3150 for the treatment of primary hypertension through a meta-analysis to provide evidence-based support for its clinical management.
-
CS-3150 5 mg had a greater effect on lowering the SBP, DBP, 24 h SBP, and 24 h DBP than either CS-3150 2.5 mg or eplerenone 50 mg. CS-3150 2.5 mg and eplerenone 50 mg showed no significant difference in lowering DBP, SBP, 24 h DBP, and 24 h SBP. Moreover, adverse events occurred at comparable rates in the three groups.
Data availability
The data analyzed in study is available from the corresponding author upon reasonable request.
References
Buckley LF, Dixon DL, Wohlford GFT, Wijesinghe DS, Baker WL, Van Tassell BW. Effect of intensive blood pressure control in patients with type 2 diabetes mellitus over 9 years of follow-up: A subgroup analysis of high-risk ACCORDION trial participants. Diabetes Obes Metab. 2018;20:1499–502.
Mohammed Nawi A, Mohammad Z, Jetly K, Abd Razak MA, Ramli NS, Wan Ibadullah WAH, et al. The prevalence and risk factors of hypertension among the urban population in southeast asian countries: a systematic review and meta-analysis. Int J Hypertens. 2021;2021:6657003.
Kearney PM, Whelton M, Reynolds K, Muntner P, Whelton PK, He J. Global burden of hypertension: analysis of worldwide data. Lancet. 2005;365:217–23.
Mittal BV, Singh AK. Hypertension in the developing world: challenges and opportunities. Am J Kidney Dis. 2010;55:590–8.
Bruno RM, Masi S, Taddei M, Taddei S, Virdis A. Essential hypertension and functional microvascular ageing. High Blood Press Cardiovasc Prev. 2018;25:35–40.
Yugar-Toledo JC, Modolo R, de Faria AP, Moreno H. Managing resistant hypertension: focus on mineralocorticoid-receptor antagonists. Vasc Health Risk Manag. 2017;13:403–11.
Gurel Z, Sheibani N. O-Linked β-N-acetylglucosamine (O-GlcNAc) modification: a new pathway to decode pathogenesis of diabetic retinopathy. Clin Sci (Lond). 2018;132:185–98.
Manolis AA, Manolis TA, Melita H, Manolis AS. Eplerenone versus spironolactone in resistant hypertension: an efficacy and/or cost or just a men’s issue? Curr Hypertens Rep. 2019;21:22.
Sato A. Mineralocorticoid receptor antagonists: their use and differentiation in Japan. Hypertens Res. 2013;36:185–90.
Lainscak M, Pelliccia F, Rosano G, Vitale C, Schiariti M, Greco C, et al. Safety profile of mineralocorticoid receptor antagonists: Spironolactone and eplerenone. Int J Cardiol. 2015;200:25–9.
Yamada M, Mendell J, Takakusa H, Shimizu T, Ando O. Pharmacokinetics, Metabolism, and Excretion of [(14)C]Esaxerenone, a novel mineralocorticoid receptor blocker in humans. Drug Metab Dispos. 2019;47:340–49.
Arai K, Homma T, Morikawa Y, Ubukata N, Tsuruoka H, Aoki K, et al. Pharmacological profile of CS-3150, a novel, highly potent and selective non-steroidal mineralocorticoid receptor antagonist. Eur J Pharmacol. 2015;761:226–34.
Ito S, Itoh H, Rakugi H, Okuda Y, Yamakawa S. Efficacy and safety of esaxerenone (CS-3150) for the treatment of essential hypertension: a phase 2 randomized, placebo-controlled, double-blind study. J Hum Hypertens. 2019;33:542–51.
Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, Baddour LM, et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 study. J Am Coll Cardiol. 2020;76:2982–3021.
Yu J, Wei Y, Jing Y, Gao Y. Effect of acupuncture on essential hypertension: A protocol for systematic review and meta-analysis. Medicine. 2021;100:e25572.
Capewell S, Ford ES, Croft JB, Critchley JA, Greenlund KJ, Labarthe DR. Cardiovascular risk factor trends and potential for reducing coronary heart disease mortality in the United States of America. Bull World Health Organ. 2010;88:120–30.
Kapan NM. The current epidemic of primary aldosteronism: causes and consequences. J Hypertens. 2004;22:863–9.
Bădilă E. The expanding class of mineralocorticoid receptor modulators: New ligands for kidney, cardiac, vascular, systemic and behavioral selective actions. Acta Endocrinol (Buchar). 2020;16:487.
Williams TA, Verhovez A, Milan A, Veglio F, Mulatero P. Protective effect of spironolactone on endothelial cell apoptosis. Endocrinology. 2006;147:2496–2505.
Gao W, Bohl CE, Dalton JT. Chemistry and structural biology of androgen receptor. Chem Rev. 2005;105:3352–70.
Zannad F, McMurray JJ, Krum H, van Veldhuisen DJ, Swedberg K, Shi H, et al. Eplerenone in patients with systolic heart failure and mild symptoms. The New England journal of medicine. 2011;364:11–21.
Funder JW. Mineralocorticoid receptor antagonists: emerging roles in cardiovascular medicine. Integr Blood Press Control 2013:129–38.
Pradhan A, Vohra S, Sethi R. Eplerenone: the multifaceted drug in cardiovascular pharmacology. J Pharm Bioall Sci. 2020;12:381–90.
Rakugi H, Yamakawa S, Sugimoto K. Management of hyperkalemia during treatment with mineralocorticoid receptor blockers: findings from esaxerenone. Hypertens Res. 2021;44:371–85.
Ito S, Itoh H, Rakugi H, Okuda Y, Iijima S. Antihypertensive effects and safety of esaxerenone in patients with moderate kidney dysfunction. Hypertens Res. 2021;44:489–97.
Kifle ZD, Adugna M, Chanie GS, Mohammed A. Prevalence and associated factors of hypertension complications among hypertensive patients at University of Gondar Comprehensive Specialized Referral Hospital. Clin Epidemiol Glob. 2022;13:100951.
Kario K, Ito S, Itoh H, Rakugi H, Okuda Y, Yamakawa S. Effect of esaxerenone on nocturnal blood pressure and natriuretic peptide in different dipping phenotypes. Hypertens Res. 2022;45:97–105.
Laffin LJ, Rodman D, Luther JM, Vaidya A, Weir MR, Rajicic N, et al. Aldosterone Synthase Inhibition With Lorundrostat for Uncontrolled Hypertension: The Target-HTN Randomized Clinical Trial. Jama. 2023;330:1140–50.
Freeman MW, Bond M, Murphy B, Hui J, Isaacsohn J. Results from a phase 1, randomized, double-blind, multiple ascending dose study characterizing the pharmacokinetics and demonstrating the safety and selectivity of the aldosterone synthase inhibitor baxdrostat in healthy volunteers. Hypertens Res: Offic J Jpn Soc Hypertens. 2023;46:108–18.
Funding
This work was a study supported by the Shanxi Institute of Traditional Chinese Medicine. The project title is Effective, safe and affordable antihypertensive and lipid-lowering drugs and treatment modalities for Chinese people (2018YFC1312400).
Author information
Authors and Affiliations
Contributions
RS conducted a statistical analysis and drafted the manuscript; YL contributed to data extraction and revised the manuscript; XG designed the study and helped to revise the manuscript; LL was involved in data extraction; WZ was involved in literature quality evaluation. RS and YL contributed equally to this work. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Ethical approval was not required, because this is a meta-analysis and no human involved.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sun, R., Li, Y., Lv, L. et al. Efficacy and safety of esaxerenone (CS-3150) in primary hypertension: a meta-analysis. J Hum Hypertens 38, 102–109 (2024). https://doi.org/10.1038/s41371-023-00889-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41371-023-00889-9
- Springer Nature Limited
This article is cited by
-
Intellectual landscapes and emerging trends of non-steroidal mineralocorticoid receptor antagonists: a bibliometric and visual analysis
International Urology and Nephrology (2024)