Key Points
-
The ability of a mature cell to convert into a different cell type is called cell plasticity
-
Originally described in cultured cells, cell plasticity is now a recognized feature of organisms, particularly in response to injury
-
Epithelial dedifferentiation occurs after kidney injury, and can be considered as a limited form of cellular plasticity
-
No evidence exists to support the notion that epithelial dedifferentiation confers multipotency, or the ability of a cell from one tubule segment to differentiate into a cell from another segment
-
The potential existence of a fixed population of epithelial progenitors versus the existence of cells with universal dedifferentiation capacity is still controversial
-
The molecular pathways underlying epithelial cell plasticity are likely to be different from developmental pathways, and must be understood in order to identify novel therapeutic targets
Abstract
Terminally differentiated cells can be reprogrammed to pluripotency or directly to another differentiated cell type in vitro, a capacity termed cellular plasticity. Plasticity is not limited to in vitro manipulations but rather represents an important aspect of the regenerative response to injury in organs. Differentiated adult cells retain the capacity to dedifferentiate, adopting a progenitor-like phenotype after injury or, alternatively, to transdifferentiate, directly converting to a different mature cell type. Emerging concepts on cellular plasticity have relevance to our understanding of repair after kidney injury, including epithelial regeneration. Here we discuss work published in the past 5 years on the cellular hierarchies and mechanisms underlying kidney injury and repair, with a particular focus on potential roles for cellular plasticity in this response.




Similar content being viewed by others
References
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Du, Y. et al. Human hepatocytes with drug metabolic function induced from fibroblasts by lineage reprogramming. Cell Stem Cell 14, 394–403 (2014).
Huang, P. et al. Direct reprogramming of human fibroblasts to functional and expandable hepatocytes. Cell Stem Cell 14, 370–384 (2014).
Tata, P. R. & Rajagopal, J. Cellular plasticity: 1712 to the present day. Curr. Opin. Cell Biol. 43, 46–54 (2016).
Merrell, A. J. & Stanger, B. Z. Adult cell plasticity in vivo: de-differentiation and transdifferentiation are back in style. Nat. Rev. Mol. Cell Biol. 17, 413–425 (2016).
Steen, T. P. Stability of chondrocyte differentiation and contribution of muscle to cartilage during limb regeneration in the axolotl (Siredon mexicanum). J. Exp. Zool. 167, 49–78 (1968).
Kragl, M. et al. Cells keep a memory of their tissue origin during axolotl limb regeneration. Nature 460, 60–65 (2009).
Stange, D. E. et al. Differentiated Troy+ chief cells act as reserve stem cells to generate all lineages of the stomach epithelium. Cell 155, 357–368 (2013).
Tata, P. R. et al. Dedifferentiation of committed epithelial cells into stem cells in vivo. Nature 503, 218–223 (2013).
van Es, J. H. et al. Dll1+ secretory progenitor cells revert to stem cells upon crypt damage. Nat. Cell Biol. 14, 1099–1104 (2012).
Rompolas, P., Mesa, K. R. & Greco, V. Spatial organization within a niche as a determinant of stem-cell fate. Nature 502, 513–518 (2013).
Johnston, A. P. et al. Dedifferentiated Schwann cell precursors secreting paracrine factors are required for regeneration of the mammalian digit tip. Cell Stem Cell 19, 433–448 (2016).
Susantitaphong, P. et al. World incidence of AKI: a meta-analysis. Clin. J. Am. Soc. Nephrol. 8, 1482–1493 (2013).
Thakar, C. V., Christianson, A., Himmelfarb, J. & Leonard, A. C. Acute kidney injury episodes and chronic kidney disease risk in diabetes mellitus. Clin. J. Am. Soc. Nephrol. 6, 2567–2572 (2011).
Ishani, A. et al. Acute kidney injury increases risk of ESRD among elderly. J. Am. Soc. Nephrol. 20, 223–228 (2009).
Coca, S. G., Yusuf, B., Shlipak, M. G., Garg, A. X. & Parikh, C. R. Long-term risk of mortality and other adverse outcomes after acute kidney injury: a systematic review and meta-analysis. Am. J. Kidney Dis. 53, 961–973 (2009).
Koyner, J. L. et al. Tissue inhibitor metalloproteinase-2 (TIMP-2)IGF-binding protein-7 (IGFBP7) levels are associated with adverse long-term outcomes in patients with AKI. J. Am. Soc. Nephrol. 26, 1747–1754 (2015).
de Caestecker, M. et al. Bridging translation by improving preclinical study design in AKI. J. Am. Soc. Nephrol. 26, 2905–2916 (2015).
Linde, P. G. et al. Overcoming barriers in kidney health-forging a platform for innovation. J. Am. Soc. Nephrol. 27, 1902–1910 (2016).
Amdur, R. L., Chawla, L. S., Amodeo, S., Kimmel, P. L. & Palant, C. E. Outcomes following diagnosis of acute renal failure in U.S. veterans: focus on acute tubular necrosis. Kidney Int. 76, 1089–1097 (2009).
Hsu, R. K. & Hsu, C. Y. The role of acute kidney injury in chronic kidney disease. Semin. Nephrol. 36, 283–292 (2016).
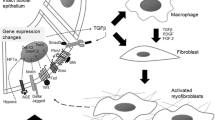
Basile, D. P. et al. Progression after AKI: understanding maladaptive repair processes to predict and identify therapeutic treatments. J. Am. Soc. Nephrol. 27, 687–697 (2016).
Kramann, R., Kusaba, T. & Humphreys, B. D. Who regenerates the kidney tubule? Nephrol. Dial. Transplant. 30, 903–910 (2015).
Witzgall, R., Brown, D., Schwarz, C. & Bonventre, J. V. Localization of proliferating cell nuclear antigen, vimentin, c-Fos, and clusterin in the postischemic kidney. Evidence for a heterogenous genetic response among nephron segments, and a large pool of mitotically active and dedifferentiated cells. J. Clin. Invest. 93, 2175–2188 (1994).
Bonventre, J. V. Dedifferentiation and proliferation of surviving epithelial cells in acute renal failure. J. Am. Soc. Nephrol. 14 (Suppl. 1), S55–S61 (2003).
Zhuang, S., Yan, Y., Han, J. & Schnellmann, R. G. p38 kinase-mediated transactivation of the epidermal growth factor receptor is required for dedifferentiation of renal epithelial cells after oxidant injury. J. Biol. Chem. 280, 21036–21042 (2005).
Hallman, M. A., Zhuang, S. & Schnellmann, R. G. Regulation of dedifferentiation and redifferentiation in renal proximal tubular cells by the epidermal growth factor receptor. J. Pharmacol. Exp. Ther. 325, 520–528 (2008).
Li, Y., Yang, J., Luo, J. H., Dedhar, S. & Liu, Y. Tubular epithelial cell dedifferentiation is driven by the helix-loop-helix transcriptional inhibitor Id1. J. Am. Soc. Nephrol. 18, 449–460 (2007).
Villanueva, S., Cespedes, C. & Vio, C. P. Ischemic acute renal failure induces the expression of a wide range of nephrogenic proteins. Am. J. Physiol. Regul. Integr. Comp. Physiol. 290, R861–R870 (2006).
Imgrund, M. et al. Re-expression of the developmental gene Pax-2 during experimental acute tubular necrosis in mice 1. Kidney Int. 56, 1423–1431 (1999).
Torres, M., Gomez-Pardo, E., Dressler, G. R. & Gruss, P. Pax-2 controls multiple steps of urogenital development. Development 121, 4057–4065 (1995).
Torban, E. et al. PAX2 activates WNT4 expression during mammalian kidney development. J. Biol. Chem. 281, 12705–12712 (2006).
Terada, Y. et al. Expression and function of the developmental gene Wnt-4 during experimental acute renal failure in rats. J. Am. Soc. Nephrol. 14, 1223–1233 (2003).
Dirocco, D. P., Kobayashi, A., Taketo, M. M., McMahon, A. P. & Humphreys, B. D. Wnt4/β-catenin signaling in medullary kidney myofibroblasts. J. Am. Soc. Nephrol. 24, 1399–1412 (2013).
Vogetseder, A., Karadeniz, A., Kaissling, B. & Le Hir, M. Tubular cell proliferation in the healthy rat kidney. Histochem. Cell Biol. 124, 97–104 (2005).
Vogetseder, A. et al. Proliferation capacity of the renal proximal tubule involves the bulk of differentiated epithelial cells. Am. J. Physiol. Cell Physiol. 294, C22–C28 (2008).
Humphreys, B. D. et al. Intrinsic epithelial cells repair the kidney after injury. Cell Stem Cell 2, 284–291 (2008).
Humphreys, B. D. et al. Repair of injured proximal tubule does not involve specialized progenitors. Proc. Natl Acad. Sci. USA 108, 9226–9231 (2011).
Kusaba, T., Lalli, M., Kramann, R., Kobayashi, A. & Humphreys, B. D. Differentiated kidney epithelial cells repair injured proximal tubule. Proc. Natl Acad. Sci. USA 111, 1527–1532 (2014).
Kusaba, T. & Humphreys, B. D. Controversies on the origin of proliferating epithelial cells after kidney injury. Pediatr. Nephrol. 29, 673–679 (2014).
Lombardi, D., Becherucci, F. & Romagnani, P. How much can the tubule regenerate and who does it? An open question. Nephrol. Dial. Transplant. 31, 1243–1250 (2016).
Sagrinati, C. et al. Isolation and characterization of multipotent progenitor cells from the Bowman's capsule of adult human kidneys. J. Am. Soc. Nephrol. 17, 2443–2456 (2006).
Bussolati, B. et al. Isolation of renal progenitor cells from adult human kidney. Am. J. Pathol. 166, 545–555 (2005).
Lindgren, D. et al. Isolation and characterization of progenitor-like cells from human renal proximal tubules. Am. J. Pathol. 178, 828–837 (2011).
Angelotti, M. L. et al. Characterization of renal progenitors committed toward tubular lineage and their regenerative potential in renal tubular injury. Stem Cells 30, 1714–1725 (2012).
Smeets, B. et al. Proximal tubular cells contain a phenotypically distinct, scattered cell population involved in tubular regeneration. J. Pathol. 229, 645–659 (2013).
Oliver, J. A., Maarouf, O., Cheema, F. H., Martens, T. P. & Al-Awqati, Q. The renal papilla is a niche for adult kidney stem cells. J. Clin. Invest. 114, 795–804 (2004).
Adams, D. C. & Oxburgh, L. The long-term label retaining population of the renal papilla arises through divergent regional growth of the kidney. Am. J. Physiol. Renal Physiol. 297, F809–F815 (2009).
Oliver, J. A. et al. Proliferation and migration of label-retaining cells of the kidney papilla. J. Am. Soc. Nephrol. 20, 2315–2327 (2009).
Oliver, J. A. et al. A subpopulation of label-retaining cells of the kidney papilla regenerates injured kidney medullary tubules. Stem Cell Rep. 6, 757–771 (2016).
Berger, K. et al. Origin of regenerating tubular cells after acute kidney injury. Proc. Natl Acad. Sci. USA 111, 1533–1538 (2014).
Romagnani, P., Rinkevich, Y. & Dekel, B. The use of lineage tracing to study kidney injury and regeneration. Nat. Rev. Nephrol. 11, 420–431 (2015).
Wang, B., Zhao, L., Fish, M., Logan, C. Y. & Nusse, R. Self-renewing diploid Axin2+ cells fuel homeostatic renewal of the liver. Nature 524, 180–185 (2015).
Rinkevich, Y. et al. In vivo clonal analysis reveals lineage-restricted progenitor characteristics in mammalian kidney development, maintenance, and regeneration. Cell Rep. 7, 1270–1283 (2014).
Barker, N. et al. Lgr5+ve stem/progenitor cells contribute to nephron formation during kidney development. Cell Rep. 2, 540–552 (2012).
Kumar, S. et al. Sox9 activation highlights a cellular pathway of renal repair in the acutely injured mammalian kidney. Cell Rep. 12, 1325–1338 (2015).
Kang, H. M. et al. Sox9-positive progenitor cells play a key role in renal tubule epithelial regeneration in mice. Cell Rep. 14, 861–871 (2016).
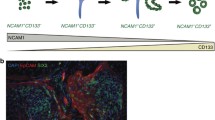
Bussolati, B. et al. Hypoxia modulates the undifferentiated phenotype of human renal inner medullary CD133+ progenitors through Oct4/miR-145 balance. Am. J. Physiol. Renal Physiol. 302, F116–F128 (2012).
Pode-Shakked, N. et al. Dissecting stages of human kidney development and tumorigenesis with surface markers affords simple prospective purification of nephron stem cells. Sci. Rep. 6, 23562 (2016).
Buzhor, E. et al. Reactivation of NCAM1 defines a subpopulation of human adult kidney epithelial cells with clonogenic and stem/progenitor properties. Am. J. Pathol. 183, 1621–1633 (2013).
Metsuyanim, S. et al. Expression of stem cell markers in the human fetal kidney. PLoS ONE 4, e6709 (2009).
Li, J. et al. Collecting duct-derived cells display mesenchymal stem cell properties and retain selective in vitro and in vivo epithelial capacity. J. Am. Soc. Nephrol. 26, 81–94 (2014).
Abbate, M., Brown, D. & Bonventre, J. V. Expression of NCAM recapitulates tubulogenic development in kidneys recovering from acute ischemia. Am. J. Physiol. 277, F454–F463 (1999).
Dor, Y., Brown, J., Martinez, O. I. & Melton, D. A. Adult pancreatic β-cells are formed by self-duplication rather than stem-cell differentiation. Nature 429, 41–46 (2004).
Thorel, F. et al. Conversion of adult pancreatic α-cells to β-cells after extreme β-cell loss. Nature 464, 1149–1154 (2010).
Chera, S. et al. Diabetes recovery by age-dependent conversion of pancreatic δ-cells into insulin producers. Nature 514, 503–507 (2014).
Waddington, C. H. The Strategy of the Genes. A Discussion of Some Aspects of Theoretical Biology (George Allen & Unwin, 1957).
Takahashi, K. & Yamanaka, S. A developmental framework for induced pluripotency. Development 142, 3274–3285 (2015).
Rajagopal, J. & Stanger, B. Z. Plasticity in the adult: how should the Waddington diagram be applied to regenerating tissues? Dev. Cell 36, 133–137 (2016).
Acknowledgements
This work was supported by the NIH/NIDDK DK107274, DK103740 and DK103050 and by an Established Investigator Award of the American Heart Association (to B.D.H.), and by F32 DK103441 (to M.C.-P.).
Author information
Authors and Affiliations
Contributions
M.C-P and B.D.H. researched the data for the article, contributed equally to discussions of the content, wrote the article and reviewed or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Cell plasticity
-
Ability of a mature cell to convert to a different cell type; dedifferentiation and transdifferentiation are two examples of cell plasticity.
- Blastema
-
Mass of undifferentiated cells that can develop into an organ or body part during embryogenesis, or regenerate an organ or body part after injury in an adult.
- Gastric chief cells
-
Differentiated epithelial cells located deep in the mucosal layer of the stomach lining that secretes pepsinogen and gastric lipase.
- Secretory cells
-
Cells present in the lumen of airways that have both secretory and detoxifying functions. Secretory cells can also differentiate into ciliated airway cells that propel mucus up the airway.
- Basal cells
-
Self-renewing airway stem cells that can differentiate into multiple epithelial cell types.
- Mesenchymal cells
-
Cells that develop in the lymphatic and circulatory systems and connective tissues, including bone and cartilage. Many mesenchymal cells have stem and progenitor cell characteristics such as the ability to differentiate into bone, cartilage or fat.
- Label retaining cells
-
(LRCs). Cells with a slow cell cycle, a property shared by many stem cells. The assay to identify them involves a DNA-analogue pulse, followed by a chase period. Slow-cycling cells retain the DNA label whereas fast-cycling cells dilute it during the chase period.
- Rainbow reporter mice
-
Transgenic mice that express a fluorescent reporter system that enables the genetic labelling of individual cells with each cell expressing a different reporter colour stochastically. This technique enables researchers to distinguish adjacent cells according to the colour of their fluorescent reporter and follow their expansion and fate clonally.
Rights and permissions
About this article
Cite this article
Chang-Panesso, M., Humphreys, B. Cellular plasticity in kidney injury and repair. Nat Rev Nephrol 13, 39–46 (2017). https://doi.org/10.1038/nrneph.2016.169
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2016.169
- Springer Nature Limited
This article is cited by
-
Galectins in epithelial-mesenchymal transition: roles and mechanisms contributing to tissue repair, fibrosis and cancer metastasis
Biological Research (2024)
-
Traditional Chinese Medicine and renal regeneration: experimental evidence and future perspectives
Chinese Medicine (2024)
-
Cellular senescence of renal tubular epithelial cells in acute kidney injury
Cell Death Discovery (2024)
-
Predicting proximal tubule failed repair drivers through regularized regression analysis of single cell multiomic sequencing
Nature Communications (2024)
-
Epithelial cell states associated with kidney and allograft injury
Nature Reviews Nephrology (2024)