Abstract
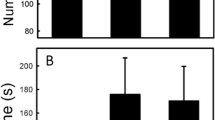
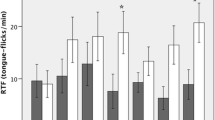
Chemicals left by organisms moving through the environment are used by other organisms to mediate interspecific interactions. Most studies of chemical eavesdropping focus on prey responding to chemical cues from predators,Despite the fact that chemical cues are frequently used by predators as a source of information about prey. Crotalus horridususes a foraging strategy that is widespread among sedentary predators: the snake chooses a site where it is likely to encounter prey and remains immobile for many hours. I investigated this ambush hunting behavior in captive-raised timber rattlesnakes and provide evidence that sit-and-wait predators may discriminate among prey chemical cues, even when they have no prior experience with the prey. Snakes explored chemical cues with chemosensory behaviors, and more frequently adopted a stereotyped ambush foraging posture toward chemical cues from prey sympatric with their population of origin than either allopatric prey or sympatric nonprey species that are eaten by other viperids. These results support the notion that intra- and interspecific variation in diet may be mediated proximally by innate recognition of cues from particular prey items. This system alsoDescribes a bioassay that may be used in the isolation and identification of prey-derived kairomones. Studies such as this can be used toDetermine more realistic parameters for models of predator–prey interaction and foraging behavior that involve secretive, less active predators.
Similar content being viewed by others
References
Arnold, S. J. 1978. Some effects of early experience on feeding responses in the common garter snake, Thamnophis sirtalis. Anim. Behav. 26:455–462.
Arnold, S. J. 1981. The microevolution of feeding behavior, pp. 409–453, in A. Kamil and T. Sargent (eds.). Foraging Behavior: Ecology, Ethological, and Psychological Approaches. Garland STPM Press, New York.
Bouskila, A. 2001. A habitat selection game of interactions between rodents and their predators. Ann. Zool. Fenn. 38:55–70.
Brown, W. L., Eisner, T., and Whitaker, R. H. 1970. Allomones and kairomones: Transspecific chemical messengers. Bioscience 20:21–22.
Burghardt, G. M. 1990. Chemically mediated predation in vertebrates, pp. 475–499, in D. McDonald D. Muller-Schwarze, and S. Natynzukf (eds.). Chemical Signals in Vertebrates 5. University Press, Oxford, UK.
Burghardt, G. M. 1999. Plasticity of foraging behavior in garter snakes (Thamnophis sirtalis) reared on different diets. J. Comp. Psych. 12:277–285.
Carroll, J. F. 2000. Responses of adult Ixodes scapularis(Acari: Ixodidae) to urine produced by white-tailedDeer of various reproductive conditions. J. Med. Entom. 37:472–475.
Chiszar, D. and Scudder, K. M. 1980. Chemosensroy searching by rattlesnakes during predatory episodes, pp. 125–139, in D. Muller-Schwarze and R. M. Silverstein (eds.). Chemical Signals in Vertabrates and Aquatic Invertebrates. Plenum Press, New York.
Chiszar, D., Lee, R., Smith, H., and Radcliffe, C. 1992. Searching behaviors by rattlesnakes following predatory strikes, pp. 369–382, in J. A. Campbell and E. D. Brodie, Jr. (eds.). Biology of the Pitvipers. Selva, Tyler, TX.
Clark, R. W. 2002. Dietary composition of the timber rattlesnake, Crotalus horridus. J. Herpetol. 36:494–499.
Cooper, W. E., Jr. 1995. Foraging mode, prey chemical discrimination, and phylogeny in lizards. Anim. Behav. 50:973–985.
Cooper, W. E., Jr. 1998. Evaluation of swab and related tests as a bioassay for assessing responses by squamate reptiles to chemical stimuli. J. Chem. Ecol. 24:841–866.
Cooper, W. E., Jr. and Burghardt, G. M. 1990. A comparative analysis of scoring methods for chemical discrimination of prey by squamate reptiles. J. Chem. Ecol. 16:45–65.
Downes, S. 1999. Prey odor influences retreat-site selection by naive broadheaded snakes (Hoplocephalus bungaroides). J. Herpetol. 33:156–159.
Duvall, D., Chiszar, D., Hayes, W. K., Leonhardt, J. K., and Goode, M. J. 1990. Chemical and behavioral ecology of foraging in prairie rattlesnakes (Crotalus viridis viridis). J. Chem. Ecol. 16:87–101.
Finelli, C. M., Pentcheff, N. D., Zimmer, R. K., and Wethey, D. S. 2000. Physical constraints on ecological processes: A field test of odor-mediated foraging. Ecology 81:784–797.
Graves, B. M. and Duvall, D. 1983. Occurrence and function of prairie rattlesnake mouth gaping in a non-feeding context. J. Exper. Zool. 227:471–474.
Greene, H. W. 1992. The ecological and behavioral context for pitviper evolution, pp. 107–118, in J. A. Campbell and E. D. Brodie Jr. (eds.). Biology of the Pitvipers. Selva, Tyler, TX.
Hall, E. R. 1981. The Mammals of North America. Wiley, New York.
Halpern, M. and Kubie, J. L. 1984. The role of the ophidian vomeronasal system in species-typical behavior. Trends Neurosci. 7:472–477.
Kats, L. E. and Dill, L. M. 1998. The scent ofDeath: Chemosensory assessment of predation risk by prey animals. Ecoscience 5:361–394.
Keenlyne, K. D. and Beer, J. R. 1973. Food habits of Sistrurus catenatus catenatus. J. Herpetol. 7:382–384.
Koivula, M. and Korpimaki, E. 2001. Do scent marks increase predation risk of microtine rodents? Oikos 95:275–281.
Nishimura, K. 1991. Optimal patch residence time of a sit-and-wait forager. Behav. Ecol. 2:283–294
Persons, M. H. and Rypstra, A. L. 2000. Preference for chemical cues associated with recent prey in the wolf spider Hogna helluo(Araneae: Lycosidae). Ethology 106:27–35.
Reinert, H. K., Cundall, D., and Bushar, L. M. 1984. Foraging behavior of the timber rattlesnake, Crotalus horridus. Copeia 1984:976–981.
Roth, E. D., May, P. G., and Farrell, T. M. 1999. Pigmy rattlesnakes use frog-derived chemical cues to select foraging sites. Copeia 1999:772–774.
Savage, T. 1967. The diet of rattlesnakes and copperheads in the Great Smoky Mountains National Park. Copeia 1967:226–227.
Schwenk, K. 1995. Of tongues and noses: Chemoreception in lizards and snakes. Trends Ecol. Evol. 10:7–12.
Stowe, M. K., Turlings, T. C. J., Lougrin, J. H., Lewis, W. J., and Tumlinson, J. H. 1995. The chemistry of eavesdropping, alarm, andDeceit. Proc. Natl. Acad. Sci. U.S.A 92:23–28.
Theodoratus, D. H. and Chiszar, D. 2000. Habitat selection and prey odor in the foraging behavior of western rattlesnakes (Crotalus viridis). Behaviour 137:119–135.
Uhler, F. M., Cottam, C. and Clarke, T. E. 1939. Food of snakes of the George Washington National Forest, Virginia. Trans. N. Am. Wildl. Conf. 605–622.
Wang, D., Jiang, X. C., Chen, P., Inouchi, J., and Halpern, M. 1993. Chemical and immunological analysis of prey-derived vomeronasal stimulants. Brain Behav. Evol. 41:246–254.
Wattiez, R., Remy, C., Falmagne, P., and Toubeau, G. 1994. Purification and preliminary characterization of a frog-derived proteinaceous chemoattractant eliciting prey attack by checkered garter snakes (Thamnophis marcianus). J. Chem. Ecol. 20:1143–1160.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Clark, R.W. Timber Rattlesnakes (Crotalus horridus) Use Chemical Cues to Select Ambush Sites. J Chem Ecol 30, 607–617 (2004). https://doi.org/10.1023/B:JOEC.0000018632.27010.1e
Issue Date:
DOI: https://doi.org/10.1023/B:JOEC.0000018632.27010.1e