Abstract
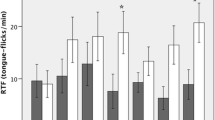
Chemical eavesdropping is a critical tool used by organisms to gain information about their environment and mediate interactions with other organisms within it, such as prey gaining information about predators and vice versa. However, most chemical eavesdropping studies focus on information used by prey species, not predators. The goal of this study was to assess whether or not a desert ambush predator, Crotalus ornatus (Eastern Black-tailed Rattlesnake), differentiates among chemical cues from nonnative familiar and novel native prey items when choosing ambush spots. Naive neonate C. ornatus were obtained from wild mothers, born and raised in captivity, and placed in an arena where they were presented with aquatic extracts from the integument from 9 known and suspected prey items, a familiar non-native prey item, and a tap water control. Their reactions to the chemical extracts were recorded under diurnal and nocturnal conditions, and they were given a tongue-flick ambush (TFAM) score based on their predatory behavior towards the chemical cue. Ten naive neonate C. ornatus were put through each of the 11 trials under diurnal and nocturnal conditions, and based on TFAM scores, snakes differentiated among the chemical cues, but there were no detectable differences based on photoperiod. Post hoc pairwise tests revealed that snakes preferred native small mammal and lizard prey. The snakes used in this study had never been exposed to wild prey stimuli in their native ecosystem and had fed exclusively on non-native mammalian prey in captivity, thus their preference for native small mammals and lizards indicates an innate basis for prey preference in C. ornatus.


Similar content being viewed by others
Data availability
The datasets generated and analyzed during the study presented within this manuscript are available from the corresponding author on reasonable request.
Code availability
N/A.
References
Anderson CG, Greenbaum E (2012) Phylogeography of northern populations of the Black-tailed Rattlesnake (Crotalus molossus, Baird and Girard, 1853), with the revalidation of C. Ornatus, Hallowell, 1854. Herpetol Monogr 26:19–57
Arnold SJ (1977) Polymorphism and geographic variation in feeding behavior of the garter snake Thamnophis elegans. Science 197:676–678
Arnold SJ (1981) The microevolution of feeding behavior. In: Kamil A, Sargent T (eds) Foraging behavior: ecology, ethological, and physiological approaches. Garland STPM Press, New York, pp 409–453
Aubret F, Burghardt GM, Maumelat S et al (2006) Feeding preferences in 2 disjunct populations of Tiger snakes, Notechis scutatus (Elapidae). Behav Ecol 16:716–725
Bates D, Maechler M, Bolker B et al (2015) Package ‘lme4’
Burghardt GM (1990) Chemically mediated predation in vertebrates. In: McDonald D, Muller-Schwarze D, Natynzuk S (eds) Chemical signals in vertebrates 5. Oxford University Press, Oxford, pp 475–499
Burghardt GM, Hess EH (1968) Factors influencing chemical release of prey Attack in newborn snakes. J Comp Psychol 1999:277–285
Carroll JF (2000) Responses of adult Ixodes scapularis (Acari: Ixodidae) to urine produced by White-tailed deer of various reproductive conditions. J Med Entomol 37:472–475
Clark RW (2004a) Timber rattlesnakes (Crotalus horridus) use chemical cues to select ambush sites. J Chem Ecol 30:607–617
Clark RW (2004b) Feeding experience modifies the assessment of ambush sites by the timber rattlesnake, a sit-and-wait predator. Ethology 110:471–483
Cooper WE Jr (2008) Tandem evolution of diet and chemosensory responses in snakes. Amphib-Reptil 29:393–398
Dinno A (2017) Package ‘dunn.test’
Downes S (1999) Prey odor influences retreat-site selection by naive Broadheaded snakes (Hoplocephalus bungaroides). J Herpetol 33:156–159
Drummond H, Burghardt G (1983) Geographic variation in the foraging behavior of the garter snake, Thamnophis elegans. Behav Ecol Sociobiol 12:43–48
Duvall D, Goode MJ, Hayes WK et al (1990) Prairie Rattlesnake vernal migration: field experimental analyses and survival value. Natl Geogr Res 6:457–469
Emerson JD, DeSantis DL, Wagler AE et al (2022) Movement, home range size, and habitat use of Eastern Black-tailed rattlesnakes (Crotalus ornatus) in the northern Chihuahuan Desert. Herpetol 78:110–118
Fuchs JL, Burghardt GM (1971) Effects of early feeding experiences on the responses of garter snakes to food chemicals. Learn Motiv 2:271–279
Garnett ST, Christidis L (2017) Taxonomy anarchy hampers conservation: the classification of complex organisms is in chaos. Nature 546:25–27
Gomulkiewicz R, Drown DM, Dybdahl MF et al (2007) Dos and don’ts of testing the geographic mosaic theory of coevolution. Heredity 98:249–258
Greene HW (1992) The ecological and behavioral context for pitviper evolution. In: Campbell JA, Brodie ED Jr (eds) Biology of the Pitvipers. Selva, Tyler, pp 107–118
Holding ML, Kern EH, Denton RD et al (2016) Fixed prey cue preferences among Dusky Pigmy rattlesnakes (Sistrurus miliarius barbouri) raised on different long-term diets. Evol Ecol 30:1–7
Kats LE, Dill LM (1998) The scent of death: chemosensory assessment of predation risk by prey animals. Ecoscience 5:361–394
Kuznetsova A, Brockhoff PB, Christensen RHB (2017) Package ‘lmerTest’
Ogle DH, Doll JC, Wheeler AP et al (2023) Package ‘FSA’
Persons MH, Rypstra AL (2000) Preference for chemical cues associated with recent prey in the wolf spider Hogna helluo (Araneae: Lycosidae). Ethology 106:27–35
R Core Team (2023) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria
Reinert HK, Cundall D, Bushar LM (1984) Foraging behavior of the timber rattlesnake, Crotalus horridus. Copeia 1984:976–981
Reynolds RP, Scott NJ Jr (1982) Use of a mammalian resource by a Chihuahuan snake community. In Scott NJ Jr (ed) Herpetological communities. U.S. Department of Interior, Fish and Wildlife Service, Wildlife Research Report 13, pp 99–118
Roth ED, May PG, Farrell TM (1999) Pigmy rattlesnakes use frog-derived chemical cues to select foraging sites. Copeia 1999:772–774
Theodoratus DH, Chiszar D (2000) Habitat selection and prey odor in the foraging behavior of western rattlesnakes (Crotalus viridis). Behaviour 137:119–135
Waters MR, Burghardt GM (2013) Prey availability influences the ontogeny and timing of chemoreception-based prey shifting in the Striped Crayfish Snake, Regina alleni. J Comp Psychol 127:49–55
Werler JE, Dixon JR (2000) Texas snakes: identification, distribution, and natural history. University of Texas Press, Austin
Acknowledgements
Funding for this project was provided by awards through the University of Texas at El Paso Graduate School and was also aided by a National Science Foundation grant and associated supplements for Indio Mountains Research Station facilities improvements. We thank other members of the JD Johnson lab, notably DL DeSantis, O De Cunha, and J Mead, for their assistance in conducting fieldwork.
Funding
University of Texas at El Paso Graduate School, through Dodson Research Grant and Dissertation Completion Fellowships awarded to JD Emerson. National Science Foundation, through grants and associated supplements for facilities improvements at Indio Mountains Research Station (FSML, DBI #09342721) awarded to JD Johnson.
Author information
Authors and Affiliations
Contributions
J.D.E. and J.D.J.: contributed to research conception and experimental design; J.D.E.: contributed to material preparation, data collection and analysis, and draft writing; J.D.J.: contributed to manuscript editing and logistics support, such as transportation, access to materials, and site access.
Corresponding author
Ethics declarations
Ethical approval
The authors state that the research represented by this manuscript has not been submitted elsewhere and is original research conducted by the authors. The University of Texas at El Paso Institutional Animal Care and Use Committee (Protocol #A-201806-1 and amendments) approved all protocols detailed herein from the period 17 July 2018 through 16 July 2021. Texas Parks and Wildlife (Scientific Permit Number SPR-0290-019) also authorized all relevant protocols pertaining to this study.
Consent to participate
N/A.
Consent for publication
The authors consent to publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Emerson, J.D., Johnson, J.D. Evidence for an innate basis of prey preference in a desert ambush predator. Evol Ecol (2023). https://doi.org/10.1007/s10682-023-10286-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10682-023-10286-w