Abstract
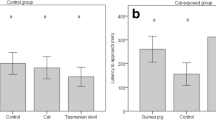
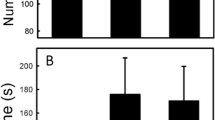
Squamate reptiles (snakes, lizards, amphisbaenians) rely heavily on chemosensory cues to identify, locate and choose between suitable prey items, but comparatively little research has focused on the chemical ecology of threatened squamate species. Such knowledge highlights ecologically important aspects of their survival. Due to gape limitations, squamates often demonstrate ontogenetic shifts in their diet where they consume larger prey as they grow older and their gape size increases. This shift enables squamates—especially snakes—to exploit new resources in their environments, usually mammalian prey. To test for ontogenetic variation in prey odor responses of a threatened snake species, the Northern pine snake (Pituophis melanoleucus melanoleucus), we presented food-naïve neonates and food-experienced adults with potential prey and non-prey animal scents and quantified their behavioral responses. Our data indicate a strong response to rodent scents from both neonates and adults. Further, neonates showed more frequent investigative probing and retreating behaviors from scented swabs and a higher rate of tongue-flicking than adults. We also developed a new metric for measuring snake responses to prey odor, a tongue-flick reaction score (TFRS), that incorporates investigative behaviors that may be unique to constrictor-type snakes. The TFRS did not differ between age classes and was highest when rodent odors were tested. A canonical discriminant analysis confirmed the relationship between TFRS behavioral components and tested chemical signal reactions. Based on our data, P. melanoleucus may fall into a category of snakes that exhibit an ontogenetic telescope rather than a general ontogenetic shift in their prey odor responses.






Similar content being viewed by others
References
Amo L, López P, Martín J (2004) Chemosensory recognition of its lizard prey by the ambush smooth snake, Coronella austriaca. J Herpetol 38(3):451–454
Arnold SJ (1978) Some effects of early experience on feeding responses in the common garter snake, Thamnophis sirtalis. Anim Behav 26:455–462
Aubret F, Burghardt GM, Maumelat S, Bonnet X, Bradshaw D (2006) Feeding preferences in 2 disjunct populations of tiger snakes, Notechis scutatus (Elapidae). Behav Ecol 17(5):716–725
Bealor MT, Krekorian CO (2006) Chemosensory response of desert iguanas (Dipsosaurus dorsalis) to skin lipids from a lizard-eating snake (Lampropeltis getula californiae). Ethology 112:503–509
Bevelander G, Smith T, Kardong K (2006) Microhabitat and prey odor selection in the foraging pigmy rattlesnake. Herpetologica 62(1):47–55
Burger J (1991) Response to prey chemical cues by hatchling pine snakes (Pituophis melanoleucus): effects of incubation temperature and experience. J Chem Ecol 17:1069–1078
Burger J, Zappalorti RT (1992) Philopatry and nesting phenology of pine snakes Pituophis melanoleucus in the New Jersey Pine Barrens. Behav Ecol Sociobiol 30(5):331–336
Burger J, Zappalorti R, Gochfeld M (1987) Developmental effects of incubation temperature on hatchling pine snakes Pituophis melanoleucus. Comp Biochem 87:727–732
Burger J, Boarman W, Kurzava L, Gochfeld M (1991) Effect of experience with pine (Pituophis melanoleucus) and king (Lampropeltis getulus) snake odors on Y-maze behavior of pine snake hatchlings. J Chem Ecol 17:79–87
Burghardt G (1969) Comparative prey-attack studies in newborn snakes of the genus Thamnophis. Behaviour 33:77–114
Burghardt G (1970) Intraspecific geographical variation in chemical food cue preferences of newborn garter snakes. Behaviour 36:246–257
Burghardt G (1993) The comparative imperative: genetics and ontogeny of chemoreceptive prey response in Natricine snakes. Brain Behav Evol 41(138–1):46
Burghardt GM, Hess EH (1968) Factors influencing the chemical release of prey attack in newborn snakes. J Comp Physiol Psychol 66(2):289–295
Burghardt G, Krause M (1999) Plasticity of foraging behavior in garter snakes (Thamnophis sirtalis) reared on different diets. J Comp Psychol 113(3):277–285
Burghardt G, Layne D, Konigsberg L (2000) The genetics of dietary experience in a restricted natural population. Psychol Sci 11:69–72
Chiszar D, Scudder K, Knight L (1976) Rate of tongue-flicking by garter snakes (Thamnophis radix haydeni) and rattlesnakes (Crotalus v. viridis, Sistrurus catenatus tergeminus, and S. c. edwardsi) during prolonged exposure to food Odors. Behav. Biol 283(5233):273–283
Clark RW (2004) Kin recognition in rattlesnakes. Proc Biol Sci 271:243–245
Clark RW (2006) Post-strike behavior of timber rattlesnakes (Crotalus horridus) during natural predation events. Ethology 112:1089–1094
Cooper W, Burghardt G (1990) A comparative analysis of scoring methods for chemical discrimination of prey by squamate reptiles. J Chem Ecol 16(1):45–65
Cooper W, Garstka W (1987) Lingual responses to chemical fractions of urodaeal glandular pheromone of the skink Eumeces laticeps. J Exp Zool 242:249–253
Cooper WE, Pérez-Mellado V (2001) Chemosensory responses to sugar and fat by the omnivorous lizard Gallotia caesaris: with behavioral evidence suggesting a role for gustation. Physiol Behav 73(4):509–516
Cooper W, Secor S (2007) Strong response to anuran chemical cues by an extreme dietary specialist, the eastern hog-nosed snake (Heterodon platirhinos). Can J Zool 85(5):619–625
Cooper WE, Buth DG, Vitt LJ (1990) Prey odor discrimination by ingestively naive coachwhip snakes (Masticophis flagellum). Chemoecology 1:86–91
de Queiroz A (1984) Effects of prey type on the prey-handling behavior of the bullsnake, Pituophis melanoleucus. J Herpetol 18:333–336
Diller L, Wallace R (1996) Comparative ecology of two snake species (Crotalus viridis and Pituophis melanoleucus) in Southwestern Idaho. Herpetologica 52:343–360
Dunbar GL (1979) Effects of early feeding experience on chemical preference of the Northern water snake, Natrix s. sipedon. J Herpetol 13:165–169
Forsman A (1996) Body size and net energy gain in gape-limited predators: a model. J Herpetol 30:307–319
Garrett C, Card W (1993) Chemical discrimination of prey by naive neonate Gould’s monitors Varanus gouldii. J Chem Ecol 19:2599–2604
Gerald G, Bailey M, Holmes J (2006) Movements and activity range sizes of Northern pine snakes (Pituophis melanoleucus melanoleucus) in Middle Tennessee. J Herpetol 40:503–510
Gove D, Burghardt G (1975) Responses of ecologically dissimilar populations of the water snake Natrix s. sipedon to chemical cues from prey. J Chem Ecol 1:25–40
Graves BM, Halpern M (1988) Neonate plains garter snakes (Thamnophis radix) are attracted to conspecific skin extracts. J Comp Psychol 102:251–253
Graves B, Halpern M, Friesen J (1991) Snake aggregation pheromones: source and chemosensory mediation in western ribbon snakes (Thamnophis proximus). J Comp Psychol 105:140–144
Greene H (1983) Dietary correlates of the origin and radiation of snakes. Am Zool 23:431–441
Greene H, Burghardt G (1978) Behavior and phylogeny: constriction in ancient and modern snakes. Science 200(4337):74–77
Halpern M, Frumin N (1979) Roles of the vomeronasal and olfactory systems in prey attack and feeding in adult garter snakes. Physiol Behav 22:1183–1189
Hampton PM (2014) Allometry of skull morphology, gape size and ingestion performance in the banded watersnake (Nerodia fasciata) feeding on two types of prey. J Exp Biol 217(Pt 3):472–478
Hampton PM, Moon BR (2013) Gape size, its morphological basis, and the validity of gape indices in western diamond-backed rattlesnakes (Crotalus atrox). J Morphol 274:194–202
IBM Corp. Released (2013) IBM SPSS Statistics for Windows, Version 22.0. IBM Corp., Armonk, NY. http://www-01.ibm.com/support/docview.wss?uid=swg21476197
Hayes W (1995) Venom metering by juvenile prairie rattlesnakes, Crotalus v. viridis: effects of prey size and experience. Anim Behav 50:33–40
Jackrel S, Reinert H (2011) Behavioral responses of a dietary specialist, the queen snake (Regina septemvittata), to potential chemoattractants released by its prey. J Herpetol 45:272–276
Kaas JH (2009) Evolutionary neuroscience. Academic Press, Oxford, pp 428–431
Kardong KV (1986) Predatory strike behavior of the rattlesnake, Crotalus viridis oreganus. J Comp Psychol 100:304–314
King R (2002) Predicted and observed maximum prey size–snake size allometry. Funct Ecol 16:766–772
Kramer M, Weldon PJ, Carroll JF (2009) Composite scores for concurrent behaviours constructed using canonical discriminant analysis. Anim Behav 77:763–768
LeMaster MP, Mason RT (2001) Evidence for a female sex pheromone mediating male trailing behavior in the red-sided garter snake, Thamnophis sirtalis parietalis. Chemoecology 11:149–152
Lind A, Welsh HH (1994) Ontogenetic changes in foraging behaviour and habitat use by the Oregon garter snake, Thamnophis atratus hydrophilus. Anim Behav 48(6):1261–1273
Loop M (1970) The effects of feeding experience on the response to prey-object extracts in rat snakes. Psychon Sci 21:189–190
López P, Amo L, Martín J (2006) Reliable signaling by chemical cues of male traits and health state in male lizards, Lacerta monticola. J Chem Ecol 32(2):473–488
López P, Ortega J, Martín J (2014) Chemosensory prey detection by the Amphisbaenian Trogonophis wiegmanni. J Herpetol 48:514–517. doi:10.1670/12-268
Mason RT (1992) Reptilian pheromones. In: Gans C, Crews D (eds) Biology of the reptilia, vol 18. University of Chicago Press, Chicago, pp 114–228
Mason RT, Parker MR (2010) Social behavior and pheromonal communication in reptiles. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 196(10):729–749
Mehta RS (2008) Early experience shapes the development of behavioral repertoires of hatchling snakes. J Ethol 27(1):143–151
Mori A (1994) Prey-handling behavior of newly hatched snakes in two species of the genus Elaphe with comparison to adult behavior. Ethology 97:198–214
Parker MR, Kardong KV (2005) Rattlesnakes can use airborne cues during post-strike prey relocation. In: Mason R, LeMaster MP, Mueller-Schwarze D (eds) Chemical signals in vertebrates 10. Springer Press, New York, pp 397–402
Persson L, Andersson J, Wahlström E, Eklöv P (1996) Size specific interactions in lake systems: predator gape limitation and prey growth rate and mortality. Ecology 77:900–911
Radcliffe C, Chiszar D, O’Connell B (1980) Effects of prey size on poststrike behavior in rattlesnakes (Crotalus durissus, C. enyo, and C. viridis). Bull Psychon Soc 16:449–450
Reformato LS, Kirschenbaum DM, Halpern M (1983) Preliminary characterization of response-eliciting components of earthworm extract. Pharmacol Biochem Behav 18:247–254
Rodríguez-Robles J (2002) Feeding ecology of North American gopher snakes (Pituophis catenifer, Colubridae). Biol J Linn Soc 77:165–183
Saviola A, Chiszar D, Mackessy S (2012) Ontogenetic shift in response to prey derived chemical cues in prairie rattlesnakes Crotalus viridis viridis. Curr Zool 58:549–555
Schubert SN, Houck LD, Feldhoff PW, Feldhoff RC, Woodley SK (2008) The effects of sex on chemosensory communication in a terrestrial salamander (Plethodon shermani). Horm Behav 54(2):270–277
Schwenk K (1993) The evolution of chemoreception in squamate reptiles: a phylogenetic approach. Brain Behav Evol 41:124–137
Scudder K, Stewart N, Smith H (1980) Response of neonate water snakes (Nerodia s. sipedon) to conspecific chemical cues. J Herpetol 14(2):196–198
Shepard D, Phillips C, Dreslik M, Jellen B (2004) Prey preference and diet of neonate eastern massasaugas (Sistrurus c. catenatus). Am Midl 152:360–368
Shine R, Mason RT (2012) An airborne sex pheromone in snakes. Biol Lett 8:183–185
Slip DJ, Shine R (1988) Habitat use, movements, and activity patterns of free-ranging diamond pythons, Morelia s. spilota (Serpentes: Boidae): a radiotelemetric study. Aust Wildl Res l5:515–553
Stark C, Tiernan C, Chiszar D (2011) Effects of deprivation of vomeronasal chemoreception on prey discrimination in rattlesnakes. Psychol Rec 61:363–370
Terrick T, Mumme R, Burghardt G (1995) Aposematic coloration enhances chemosensory recognition of noxious prey in the garter snake Thamnophis radix. Anim Behav 49:857–866
Troost T, Kooi BW, Dieckmann U (2008) Joint evolution of predator body size and prey-size preference. Evol Ecol 22:771–799
Urban M (2007) The growth-predation risk trade-off under a growing gape-limited predation threat. Ecology 88:2587–2597
Urban M (2008) Salamander evolution across a latitudinal cline in gape limited predation risk. Oikos 117:1037–1049
Waters RM (1993) Odorized air current trailing by garter snakes, Thamnophis sirtalis. Brain Behav Evol 41:219–223
Waters RM, Burghardt GM (2013) Prey availability influences the ontogeny and timing of chemoreception-based prey shifting in the striped crayfish snake, Regina alleni. J Comp Psychol 127(1):49–55
Weldon P, Schell F (1984) Responses by king snakes (Lampropeltis getulus) to chemicals from colubrid and crotaline snakes. J Chem Ecol 10:1509–1520
Acknowledgments
This study was conducted under NJDEP state permits (Permit No. SC 2012-085, SC 2013-085, SC 2014-085) and Drexel University IACUC (18924 and 20129). Thank you to the New Jersey Air National Guard and New Jersey Conservation Foundation for access to research sites. A special thanks to the Laboratory of Pinelands Research at Drexel University for financial support. KPWS would especially like to thank Dr. Emile DiVito, Raffaella Marano, Emily Ostrow, Kathryn Bendon and members of the Laboratory of Pinelands Research for assistance with field work and experiments, Dr. James R. Spotila for aid in manuscript revisions, and Kevin Redding at Monell Chemical Senses Center for supplying rodent scents for tests. The authors would also like to thank an anonymous reviewer for comments that significantly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Michael Heethoff.
Taxa
Class: Reptilia—Order: Squamata—Family: Colubridae—Genus: Pituophis—Species: Melanoleucus—Subspecies: Melanoleucus.
Rights and permissions
About this article
Cite this article
Smith, K.P.W., Parker, M.R. & Bien, W.F. Behavioral variation in prey odor responses in northern pine snake neonates and adults. Chemoecology 25, 233–242 (2015). https://doi.org/10.1007/s00049-015-0193-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-015-0193-6