Abstract
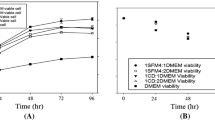
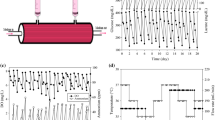
The kinetics and long-term stability of continuous production of monoclonal antibody IgG2b by hybridoma HD-24 cells immobilized in a fibrous-bed bioreactor (FBB) were studied for a period of ∼8 months. The cells were immobilized in the fibrous bed by surface attachment of cells and entrapment of large cell clumps in the void space of the fibrous matrix. A high viable cell density of 1.01 × 108/ml was attained in the bioreactor, which was about 63 times higher than those in conventional T-flask and spinner flask cultures. The continuous FBB produced IgG at a concentration of ∼0.5 g/l, with reactor productivity of ∼7 mg/h·l, which was about 23 times higher than those from conventional T-flask and spinner flask cultures. The IgG concentration can be further increased to ∼0.67 g/l by using higher feed (glucose and glutamine) concentrations and running the reactor at a recycle batch or fed-batch mode. The long-term performance of this bioreactor was also evaluated. For a period of 36 days monitored, the MAb produced in the continuous well-mixed bioreactor at 50 h retention time (0.02/h dilution rate) was maintained at a steady concentration level of ∼0.3 g/l with less than 8% drift. At the end of the study, it was found that ∼25% of the cells were strongly attached to the fiber surfaces and the other ∼75% entrapped or weakly immobilized in the fibrous matrix. The strongly attached cells had a high viability of ∼90%, compared to ∼75% for cells weakly immobilized and only ∼1.4% for freely suspended cells, suggesting that the fibrous matrix preferentially retained and protected the viable (productive) cells. The FBB thus was able to maintain its long-term productivity because nonviable and dead cells were continuously washed off from the fibrous matrix. The high MAb concentration and production rate and excellent stability for continuous long-term production obtained in this study compare favorably to other bioreactor studies reported in the literature. The reactor performance can be further improved by providing better pH and aeration controls at higher feed concentrations. The FBB is easy to operate and scale-up, and thus can be used economically for industrial production of MAb.
Similar content being viewed by others
References
Altshuler G.L., Dziewulski D.M., Sowek J.A. and Belfort G. 1987. Continuous hybridoma growth and monoclonal; antibody production in hollow fiber reactor-separator. Biotechnol. Bioeng. 28: 646–658.
Backer M.P., Metzger L.S., Slaber P.L., Nevitt K.L. and Boder G.B. 1988. Large-scale production of monoclonal antibodies in suspension culture. Biotechnol. Bioeng. 32: 993–1000.
Banik G.G. and Heath C.A. 1995. Hybridoma growth and antibody production as a function of cell density and specific growth rate in perfusion culture. Biotechnol. Bioeng. 48: 289–300.
Batt B.C., Davis R.H. and Kompala D.S. 1990. Inclined sedimentation for selective retention of viable hybridoma in a continuous suspension bioreactor. Biotechnol. Prog. 6: 458–464.
Bibila T.A. and Robinson D.K. 1995. In pursuit of the optimal fed-batch process for monoclonal antibody production. Biotechnol. Prog. 11: 1–13.
Bierau H., Perani A., Al-Rubeai M. and Emery A.N. 1998. A comparison of intensive cell culture bioreactors operating with hybridomas modified for inhibited apoptotic response. J. Biotechnol. 62: 195–207.
Broise D.D.L., Noiseux M., Massie B. and Lemieux R. 1992. Hybridoma perfusion systems: A comparison study. Biotechnol. Bioeng. 40: 25–32.
Brotherton J.D. and Chau P.C. 1995. Protein-free humanhuman hybridoma cultures in an intercalated-spiral alternate-dead-ended hollow fiber bioreactor. Biotechnol. Bioeng. 47: 384–400.
Cadic C., Dupuy B., Piamat I., Merle M., Margerin C. and Bezian J.H. 1992. In vitro culture hybridoma cells in agarose beads producing antibody secretion for two weeks. Biotechnol. Bioeng. 39: 108–112.
Chen C., Chen K. and Yang S.T. 2003. Effects of threedimensional culturing on osteosarcoma cells grown in a fibrous matrix: Analyses of cell morphology, cell cycle and apoptosis. Biotechnol. Prog. 19: 1574–1582.
Chen C., Huang Y.L. and Yang S.T. 2002. A fibrous-bed bioreactor for continuous production of developmental endothelial locus-1 by osteosarcoma cells. J. Biotechnol. 97: 23–39.
Chen K., Liu Q., Xie L., Sharp P.A. and Wang D.I.C. 2001. Engineering of a mammalian cell line for reduction of lactate formation and high monoclonal antibody production. Biotechnol. Bioeng. 72: 55–61.
Cherlet M. and Marc A. 2002. Stimulation of monoclonal antibody production of hybridoma cells by butyrate: evaluation of a feeding strategy and characterization of cell behavior. Cytotechnol. 32: 17–29.
Chiou T.W., Murakami S. and Wang D.I.C. 1991. A fiber-bed bioreactor for anchorage-dependent animal cell cultures: Part I. Bioreactor design and operations. Biotechnol. Bioeng. 37: 755–761.
Dalili M. and Ollis D.F. 1990. A flow-cytometric analysis of hybridoma growth and monoclonal antibody production. Biotechnol. Bioeng. 36: 64–73.
Emery A.N., Lavery M., Williams B. and Handa A. 1987. Large Scale Hybridoma Culture: Animal and Plant Cells Process Possibilities, Ellis Horwood Ltd., Chichester, pp. 137–146.
Fong W., Zhang Y. and Yung P. 1997. Optimization of monoclonal antibody production: combined effects of potassium acetate and perfusion in a stirred tank bioreactor. Cytotechnol. 24: 47–54.
Gramer M.J. and Britton T.L. 2002. Antibody production by a hybridoma cell line at high cell density is limited by two independent mechanisms. Biotechnol. Bioeng. 79: 277–283.
Gramer M.J. and Poeschl D.M. 2000. Comparison of cell growth in T-flasks, in micro hollow fiber bioreactors, and in an industrial scale hollow fiber bioreactor system. Cytotechnol. 34: 111–119.
Hansen H.A. and Emborg C. 1994. Influence of ammonium on growth, metabolism, and productivity of a continuous suspension Chinese hamster ovary cell culture. Biotechnol. Prog. 10: 121–124.
Hayter P.M., Kirkby N.F. and Spier R.E. 1992. Relationship between hybridoma growth and monoclonal antibody production. Enz. Microb. Technol. 14: 454–461.
Hülscher M., Scheilber U. and Onken U. 1992. Selective recycle of viable animal cells by coupling of airlift reactor and cell settler. Biotechnol. Bioeng. 39: 442–446.
Jackson L.R., Trudel L.J., Fox J.G. and Lipman N.S. 1996. Evaluation of hollow fiber bioreactor as an alternative to murine ascites production for small scale monoclonal antibody production. J. Immunol. Methods. 189: 217–231.
Jan D.C.H., Petch D.A., Huzel N. and Butler. 1997. The effect of dissolved oxygen on the metabolic profile of a murine hybridoma grown in serum-free medium in continuous culture. Biotechnol. Bioeng. 54: 153–164.
Junker B.H., Chiou T., Wang D.I.C. and Buckland B.C. 1993. Cultivation of virus antigen in fibroblast cells using a glass fiber bed reactor. Biotechnol. Bioeng. 42: 635–642.
Katinger H. 1987. Principles of animal cell fermentation. Dev. Biol. Stand. 66: 195–209.
Klement G., Scheier W. and Katinger H. 1987. Construction of a large membrane reactor system with different compartment for cells, medium and products. Dev. Biol. Stand. 66: 221–226.
Köhler G. and Milstein C. 1975. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256: 495–497.
Lambert K.J., Boraston R., Thompson P.W. and Birch J.R. 1987. Production of monoclonal antibodies using large scale culture. Dev. Ind. Microbiol. 27: 101–106.
Lee G.M. and Palsson B.O. 1990. Immobilization can improve the stability of hybridoma antibody productivity in serumfree media. Biotechnol. Bioeng. 36: 1049–1055.
Lee G.M., Varma A. and Palsson B.O. 1991. Production of monoclonal antibody using free-suspended and immobilized hybridoma cells: Effect of serum. Biotechnol. Bioeng. 38: 821–830.
Lewis V.P. and Yang S.T. 1992. Continuous propionic acid fermentation by immobilized Propionibacterium acidipropionici in novel packed-bed bioreactor. Biotechnol. Bioeng. 40: 465–474.
Linardos T.I., Kalogerakis N., Behie L.A. and Lamontagne L.R. 1992. Monoclonal antibody production in dialyzed continuous suspension culture. Biotechnol. Bioeng. 39: 504–510.
Lu Z., Thompson B.G., Sureah M.R. and Gray M.R. 1995. Cultivation of hybridoma cells in an inclined bioreactor. Biotechnol. Bioeng. 45: 176–186.
Luo J. 2002. Three Dimensional Culture of Animal Cells in a Fibrous-bed Bioreactor. Ph.D. Dissertation, The Ohio State University, Columbus, Ohio, USA.
Matsumura M., Nayve F. and Rey P. Jr. 1995. Effects of ammonium ion removal on growth and MAb production of hybridoma cells. Cytotechnol. 18: 35–50.
Needham H.P., Ting-Beall and Tran-Son-Tay R. 1991. A physical characterization of GAP A3 hybridoma cells: morphology, geometry, and mechanical properties. Biotechnol. Bioeng. 38: 838–852.
Nikolai T.J. and Hu W.-S. 1992. Cultivation of mammalian cells on macroporous microcarriers. Enz. Microb. Technol. 14: 203–208.
Nilsson K., Sheirer W., Merten O.W., Östberg L., Liehl E., Katinger H.W.D. and Mosbach K. 1983. Entrapment of animal cells for the production of monoclonal antibodies and other biomolecules. Nature 302: 629–630.
Ozturk S.S. and Palsson B. 1990. Effects of dissolved oxygen on hybridoma cell growth, metabolism, and antibody production kinetics in continuous culture. Biotechnol. Prog. 6: 437–446.
Ozturk S.S. and Palsson B.O. 1991. Growth, metabolic, and antibody production kinetics of hybridoma cell culture: 2. Effects of serum concentration, dissolved oxygen concentration, and medium pH in a batch reactor. Biotechnol. Prog. 7: 481–494.
Perry S.D. and Wang D.I.C. 1989. Fiber bed reactor design for animal cell culture. Biotechnol. Bioeng. 34: 1–9.
Pörtner R., Roessing S., Koop M. and Luedemann I. 1997. Kinetic studies on hybridoma cells immobilized in fixed bed reactors. Biotechnol. Bioeng. 55: 535–541.
Ramirez O.T. and Mutharasan R. 1990. Cell cycle-and growth phase-dependent variations in size distribution, antibody productivity, and oxygen demand in hybridoma cultures. Biotechnol. Bioeng. 36: 839–848.
Rupp R.G. 1985. Use of Cellular Micro-encapsulation in Largescale Production of Monoclonal Antibodies. Large-Scale Mammalian Cell Culture. Academic Press, Orlando, pp. 19–38.
Scheirer W. 1988. High-density growth of animal cells within cell retention fermentors equipped with membranes. In: Spier R.E. and Griffiths J.B. (eds), Animal Cell Biotechnology, Vol. 3, Academic Press, London, pp. 263–281.
Shi Y., Dewey D.Y. and Park H. 1992. Performance of mammalian cell culture bioreactor with a new impeller design. Biotechnol. Bioeng. 40: 260–270.
Simpson N.H., Sigh R.P., Emery A.N. and Al-Rubeai M. 1999. Bcl-2 over-expression reduces growth rate and prolongs G1 phase in continuous chemostat cultures of hybridoma cells. Biotechnol. Bioeng. 64: 187–193.
Sinacore M.S. 1984. Gel entrapment: Applications in production of biological and mass culturing animal cells. Karyon Technology News I, Karyon Technology Inc., 333 Provident Highway, Norwood, MA 02062.
Silva E.M. and Yang S.T. 1995. Continuous production of lactic acid from acid whey by Lactobacillus helveticus in a fibrousbed bioreactor. J. Biotechnol. 41: 59–70.
Stoll T., Perregaux C., Von Stockar U. and Marison I.W. 1995. Production of immunoglobulin A in different reactor configurations. Cytotechnol. 17: 53–63.
Tharakan J.P. and Chau P.C. 1986. A radial flow hollow fiber bioreactor for the large-scale culture of mammalian cells. Biotechnol. Bioeng. 28: 329–342.
Valdes R., Ibarra N., Gonzalez M., Alvarez T., Garcia J., Llambias R., Perez C.A., Quintero O. and Fischer R. 2001. CB.Hep-1 hybridoma growth and antibody production using protein-free medium in a hollow fiber bioreactor. Cytotechnol. 35: 145–154.
Wang G., Zhang W., Jacklin C., Freedman D., Eppstein L. and Kadouri A. 1992. Modified CelliGen-packed bed bioreactors for hybridoma cell cultures. Cytotechnol. 9: 41–49.
Wen Z.-Y., Teng X.-W. and Chen F. 2000. A novel perfusion system for animal cell cultures by two step sequential sedimentation. J. Biotechnol. 79: 1–11.
Xie L. and Wang D.I.C. 1996. High cell density and high monoclonal antibody production through medium design and rational control in a bioreactor. Biotechnol. Bioeng. 51: 725–729.
Yang J.-D., Angelillo Y., Chaudhry M., Goldenberg C. and Goldenberg D.M. 2000. Achievement of high cell density and high antibody productivity by a controlledfed perfusion bioreactor process. Biotechnol. Bioeng. 69: 74–82.
Yang S.T. and Shu C.H. 1996. Kinetics and stability of GM-CSFproduction by recombinant yeast cells immobilized in a fibrous-bed bioreactor. Biotechnol. Prog. 12: 449–456.
Yang S.T., Zhu H., Lewis V.P. and Tang I.C. 1992. Calcium magnesium acetate (CMA) production from whey permeate: process and economic analysis. Resources, Conserv. Recycling 7: 181–200.
Yang S.T., Zhu H., Li Y. and Hong G. 1994. Continuous propionate production from whey permeate using a novel fibrous bed bioreactor. Biotechnol. Bioeng. 43: 1124–1130.
Zhu H. 1995. Kinetics and Analysis of a Fibrous Bed Bioreactor for Mammalian Cell Cultures. Ph.D. dissertation, Ohio State University, Columbus, OH.
Rights and permissions
About this article
Cite this article
Zhu, H., Yang, ST. Long-term Continuous Production of Monoclonal Antibody by Hybridoma Cells Immobilized in a Fibrous-Bed Bioreactor. Cytotechnology 44, 1–14 (2004). https://doi.org/10.1023/B:CYTO.0000043395.36188.bc
Issue Date:
DOI: https://doi.org/10.1023/B:CYTO.0000043395.36188.bc