Abstract
The occurrence and level of polycyclic aromatic hydrocarbons (PAHs) in mangrove sediment from Ifiekporo Creek in Warri, Delta State, Nigeria, were assessed in this study. The extraction of PAHs from the sediment samples was achieved using the PAH analytical test method (USEPA 8270). This method measures the individual concentrations of extractable PAHs in samples. The PAHs were quantified within the C11-C22 subgroup. The basic GC parameters for the analysis of polyaromatic hydrocarbons were analysed using a gas chromatography‒mass selective detector (GC‒MSD). The individual PAH concentrations in the mangrove sediment samples ranged from 850 µg/kg−1 to 3470 µg/kg−1. PAH source apportionment employing isomeric ratios and multivariate statistics indicated both pyrogenic and petrogenic source inputs in the sediment samples. The order of PAH homology in the sediment was 3-ring > 2-ring > 4-ring. The sediment samples were mostly dominated by LMW PAHs. The results of the ecological risk assessment in the study area indicate that there is low ecological risk associated with PAH exposure in sediment. These results provide data on the concentrations and compositional patterns of PAHs, which is useful for understanding the effects, sources, fates, and transport of PAHs in sediment in the study area, as well as for providing relevant information for environmental quality management and forensic studies.
Similar content being viewed by others
1 Introduction
Mangroves are halophytic forests found in wetlands between land and sea [1]. Mangrove forests are found in 105 countries on 5 continents [2], inhabiting relatively sheltered coastal areas at tropical and subtropical latitudes [3]. Nigeria’s mangrove forests are located mainly in the Niger Delta, covering 10,500 km2 and inhabited by a diversity of animal and plant species. Fish, birds, reptiles, and mammals all use mangroves as breeding and growth habitats; they also act as riparian buffers, species habitats, and sediment filters [4,5,6,7]. In terms of vegetation, mangrove forests are characterized by red mangroves, black mangroves, and white mangroves [8, 9]. The mangrove forests in Nigeria play essential roles in coastal ecosystems. It provides many services that are critical for the wellbeing of the environment and for human health, such as protection of shorelines [10], maintenance of water quality, prevention of erosion, and climate mitigation and adaptation through carbon sequestration [11]. The Forests provide benefits to local communities through the provision of food, economic opportunities, fuelwood, and logging services [12]. The loss of mangroves may have far-reaching effects on the environment as well as humans [13]. Several anthropogenic activities threaten mangrove forests in Nigeria [14]. The Ecosystems suffer from a number of devastating environmental stressors, including overexploitation of mangrove forests for wood and fuelwood, which may lead to habitat degradation and ecological devastation [15, 16]. The conversion of mangrove forests to other land uses, such as agricultural or urban development, results in the loss of essential ecosystem services [17,18,19]. Furthermore, climate change poses a danger to mangroves by altering their hydrological conditions, which are further exacerbated by sea level rise [20,21,22].
Paradoxically, the Niger Delta region of Nigeria is the hub of oil and gas production [23]. Over 22.5 billion barrels of oil reserves are stationed within an estimated 250 separate fields in the Niger Delta region [24]. The region is responsible for approximately 80% of government revenue and 95% of foreign exchange earnings [25, 26]. Unfortunately, pollution from industrial activities and frequent oil spills have become commonplace in this area, posing a significant threat to its delicate mangrove ecosystem [27,28,29]. The impact could be widespread. These factors not only affect water quality but also pose significant risks to the health of the various species that depend on these important wetlands [30]. Oil-induced contaminants, such as heavy metals and persistent organic pollutants (POPs), have been found to bioaccumulate in mangrove organisms, resulting in long-term ecological harm and posing serious health hazards for humans [31]. Moreover, research has shown that these toxic substances can interfere with crucial reproductive processes [32], impair the immune system, and even trigger cancerous growth in both marine and terrestrial organisms [33].
Polycyclic aromatic compounds, such as polycyclic aromatic hydrocarbons (PAHs), are a significant group of persistent organic pollutants (POPs) [34, 35]. PAHs are compounds that are among the range of environmental pollutants that are resistant to degradation and can remain in the environment for a long period [36,37,38]; they persist because of their high degree of conjugation and aromaticity [39]. PAHs can originate from three main sources in the environment: fossil fuels (petrogenic PAHs), organic matter (pyrogenic PAHs), or natural organic precursors in the environment generated by relatively rapid chemical/biological processes (biogenic PAHs) [40, 41]. Petrogenic PAHs are introduced into the aquatic environment through accidental oil spills, discharge from routine tanker operations, municipal and urban runoff, etc. [42]. Pyrogenic PAHs are released as byproducts of combustion and solid residues and are largely prevalent in aquatic environments [36, 43]. Biogenic PAHs are generated naturally from living organisms such as algae, phytoplankton, microorganisms and plants [44].
PAHs with high molecular weights are mainly pyrolytic and reach aquatic biota through direct atmospheric deposition or via contaminated soil [45]. However, low-molecular-weight pyrogenic PAHs are mainly introduced into the environment by rain washout [46]. PAHs containing (2–4) planar rings are termed light or low-molecular-weight (LMW) PAHs, while those containing more than four (4) planar rings are termed heavy or high-molecular weight (HMW) PAHs [47]. HMW particles (PAHs with 4 or more rings) have high resonance energies due to their dense clouds of π electrons surrounding the aromatic rings, which makes them persistent and recalcitrant to microbial degradation [48]. Its low aqueous solubility and high soil sorption also contribute to its persistence and recalcitrance [49]. PAHs that are heavy are stable and toxic, whereas light PAHs are more unstable, soluble and volatile, are highly soluble in aqueous media and are less lipophilic [47].
United States Environmental Protection Agency (US EPA) has identified sixteen PAHs as 'priority pollutants' (Table 1), although there are hundreds of PAH compounds that could be included in risk assessments [47, 50].
Of the 16 priority PAHs, the International Agency for Research on Cancer (IARC) further classified benzo(a)pyrene as group 1A (carcinogenic to humans) or dibenzo(a,h)anthracene (probably carcinogenic to humans) and benzo(a)anthracene, benzo(b)fluoranthene, benzo(k)fluoranthene, chrysene and Indeno (1,2,3-cd) perylene as group 3B (possibly carcinogenic to humans), while the other PAHs not listed are classified as not carcinogenic to humans [51].
PAHs are present as trace contaminants in freshwater and marine sediments worldwide [52]. PAH are recognized to contribute significantly to the contamination of mangrove sediments and threats to aquatic life is growing, especially in the area of high-density human and industrial activities [53]. Crude oil-related anthropogenic activities in mangrove swamps include oil spills from offshore drilling or transportation, where large quantities of oil are released into the water and can contaminate sediments [54]. industrial discharge of wastewater from oil refineries and other industrial processes can also contribute to PAH contamination. Improper waste management practices, such as the direct dumping of oil and other petroleum products into mangrove ecosystems, further exacerbates this issue [55]. Additionally, the activities of unchecked oil bunkering and unstandardized crude oil refining in the region also release significant amounts of crude oil into the environment [56]. These activities can introduce PAHs into mangrove ecosystems, where they can accumulate in sediments and be taken up by mangrove plants and organisms [57, 58].
The lack of sufficient data on PAHs from mangrove swamps in developing nations such as Nigeria highlights the need for further research to understand the extent of contamination and potential impacts on these valuable coastal ecosystems. Against this backdrop, this study investigated the incidence and level of PAH in mangrove sediments from Ifiekporo Creek, Warri, Delta State, Nigeria. The main goal of this study was to estimate the levels of PAHs in the sediments of Ifiekporo Creek by (i) investigating the distribution of PAHs in the Ifiekporo sediments, (ii) identifying possible PAH contamination sources using diagnostic ratios and principal component analysis (PCA), and (iii) assessing the potential ecological risk-related PAH assemblages in the sediments. The novelty of this study is the control of environmental pollution in the Ifiekporo mangrove ecosystem by using environmental standard criteria linked to PAHs.
2 Materials and methods
2.1 Study area
According to the World Bank (1995), Nigeria's mangrove ecosystem is the third largest in the world and covers approximately 975,000 km2 of the low-lying area that borders the coastal wetlands of southern Nigeria, stretching from Bakassi in the east to Badagry in the west. The mangroves of the Niger Delta are a broad confluence of wetlands with a web of creeks and rivers, which results in a significant inflow of nutrients and organic waste. This is where the ecosystem achieves its maximum extent. This abundant nutritional foundation is used by primary producers to boost biomass. Mangrove forests provide many ecosystem services, such as littoral protection, species habitats, and contaminant and sediment filtering [5]. As a result of the high primary productivity, high levels of organic compounds, abundance of detritus, low energy and reduced current flow, and anoxic and suboxic conditions that characterize mangrove ecosystems, they are favorable environments for the preservation, accumulation, and significant uptake of PAHs [59].
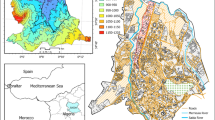
The study was carried out in Ifiekporo Creek in Warri, Warri South Local Government Area of Delta State in the Niger Delta region of Nigeria. The Ifiekporo community is populated by the Itsekiri people. The Community shares a landmass with the Ijala Community and its water-bounders the Warri River to the South. It is often referred to as “that community behind Warri Refinery”. It plays host to the Warri Refining Petrochemical Company’s (WRPC) jetty and several petroleum tank farms and depots. It is defined by latitudes 5°34′23.3"N and longitudes 5°41′14.5"E (Fig. 1).
2.2 Materials/reagents
For the analysis of the PAHs, an analytical test method (USEPA 8270) was used. This method measures the individual and total concentrations of extractable PAHs in samples. Polyaromatic hydrocarbons are quantified within C11–C22. The basic GC parameters for the analysis of polyaromatic hydrocarbons were as follows: Agilent 6890 5973 N gas chromatography‒mass selective detector (GC‒MSD). An Agilent 6890 gas chromatograph and Agilent 5973 N mass selective detector (GCMSD) manufactured by Agilent Technology (Palo, Alto CA, USA) and manufactured by Agilent Technology (Palo, Alto CA, USA) were used for this study. The PAH reference mixture (2000 ppm) contained 16 PAH components obtained from Accu Standard, Inc., New Haven, USA. Silica gel (60–120 mesh) and acetone were obtained from BDH laboratories, and dichloromethane and DCM (99% analytical grade) were obtained from Fisher Scientific (Loughborough, UK) and used for this study.
2.3 Sample collection and preparation
A total of 10 selected sampling points were identified within the Ifiekporo Creek in the Warri South Local Government Area of Delta State, Nigeria. As described by Decena et al. [60], at every sampling location, sediment samples were collected at depths of 0–15 cm with the aid of a Van Veen grab sediment sampler aboard a speedboat powered by a Yamaha 40 horsepower (HP) engine. At each collection point, a Garmin eTrex 20 handheld receiver was used to record the coordinates. The sediment samples were placed in new Zip-lock plastic bags, placed in a cooler with ice, transported to Jacio Environmental Limited, Warri, Delta State, Nigeria, and kept at 4 °C until analysis.
2.4 Extraction of sediment samples for PAHs
Non-sediment materials such as plant roots and stones were removed from the sediment sample and then mixed thoroughly for uniformity prior to the extraction process. A standard reference procedure for the analysis of PAHs in sediment was employed (US EPA 8240 method).
The extraction was carried out as described by the USEPA 3550C method (USEPA, 2007).
-
i.
Ten grams of well-mixed sediment sample was individually weighed and homogenized with 10 g of anhydrous sodium sulfate (Na2SO4) to produce a freely flowing finely divided slurry.
-
ii.
The sample homogenate was spread on foil paper to increase the surface area, which helps quickly remove moisture. The sample homogenate was subsequently crushed in a clean laboratory ceramic mortar to obtain a well mixture and representative sample prior to extraction.
-
iii.
The crushed/mixed sample homogenate was transferred to a clean 250 ml glass extraction flask. Then, 20 ml (1:1 v/v) of dichloromethane-acetone mixture was added, and the flask was tightly capped.
-
iv.
The flask was placed on a mechanical shaker (Searchtech instrument, HZ-300 model, USA) and shaken for 60 min.
-
v.
The flask was subsequently transferred to an ultrasonic apparatus (Omegasonic Quantum series model, Simi Valley, USA), where the sample was further extracted ultrasonically for 30 min at approximately 70 °C.
-
vi.
The solvent extract was gently decanted into a 250 ml Erlenmeyer flask, which was properly corked to prevent escape of the solvent extract.
-
vii.
The sample was extracted twice more using a 20 ml (1:1 v/v) portion of dichloromethane-acetone mixture following steps iii-vi. The extracts were subsequently combined.
-
viii.
The combined extract was concentrated using a rotary evaporator (Searchtech instrument, RE52-2 model, USA) until the volume was reduced to approximately 1–2 ml.
2.5 Clean-up procedure for PAHs
A column of approximately 15 cm (length) × 1 cm (internal diameter) was packed first with glass wool and then with approximately 10 g of activated silica gel prepared in a slurry form in dichloromethane. Approximately 10 g of anhydrous sodium sulfate was placed at the top of the column to absorb any water in the sample or the solvent. Preelution was performed with 30 mL of dichloromethane without exposing the sodium sulfate layer to air to prevent drying of the silica gel adsorbent. The reduced extract was passed through the column and allowed to sink below the sodium sulfate layer. Elution was performed with 3 × 10 mL portions of dichloromethane. The eluate was collected in a solvent-rinsed 100 ml conical flask. The eluate was transferred to a round bottom flask and concentrated to a 1 ml final volume using a rotary evaporator. The concentrate was pipetted into a clean 2 ml Teflon screw-cap vial and capped tightly, labelled and preserved to avoid oxidation prior to analysis via GC‒MS.
2.6 GC‒MS analysis
An Agilent 6890 gas chromatograph equipped with an autosampler connected to an Agilent 5973 N mass selective detector was used. One microliter of sample solution was injected in pulsed splitless mode onto a 30 m × 0.25 mm META X5-coated fused capillary column with a film thickness of 0.25 µm. Helium was used as the carrier gas, and the column head pressure was maintained at 13 psi to give a constant flow rate of 1.0 ml/min. Other operating conditions included a preset flow, a purge time of 2.00 min, a purge flow of 20.0 ml/min, a total flow of 23.7 ml/min, and an injection temperature of 250 °C. The column temperature was initially held at 70 °C for 2 min, increased to a final temperature of 300 °C at a rate of 12 °C/min and held for 8 min. The mass spectrometer (MS) conditions involved setting the transfer line at 280 °C. MS source temp. set at 23 °C, and the MS quad temperature was set at 55 °C. Scan acquisition mode ranged from 50 to 550 amu, a threshold of 150 and a sampling rate of 2 were used with a solvent delay of 3.5 min and a gain factor of 1.0. The MS tune file was saved as an atune. The PAH identification time was based on the retention time since each of the PAHs had a separate retention time in the column. Those with shorter retention times were identified first, followed by those with longer retention times.
2.7 Preparation of PAH standard solutions
Accu-Standard stock solutions of the sixteen (16) USEPA priority PAH calibration standards, including naphthalene (NaP), acenaphthene (Ace), acenaphthylene (Acy), fluorene (Flu), phenanthrene (Phe), anthracene (Ant), fluoranthene (Flt), pyrene (Pyr), benzo(a)anthracene (BaA), chrysene (Chr), benzo(b) fluoranthene (BbF), benzo(k) fluoranthene (BkF), benzo(a)pyrene (BaP), indeno(1,2,3-cd)pyrene (InP), dibenzo (a,h) anthracene (DaHa), and benzo(g,h,i)perylene (BghiP), were prepared and then serially diluted to produce different concentrations ranging from 0.5 ppm to 20.0 ppm. Stock standard solutions were stored in amber-colored bottles at 4 °C in a refrigerator, and working standard solutions were prepared fresh before use. Standard solutions of the PAHs were subjected to GC‒MS under the set chromatographic conditions, and the mean peak areas were plotted against the concentrations to construct calibration curves of the PAHs.
2.8 Quantification of the results
The Agilent Technologies Inc. MSD Chemsta] Mm tion G1701DA D.03.00.611 software was used to quantify the aromatic component results in ppm (part per million), and a suite of 16 priority polycyclic aromatic hydrocarbons recommended by the EPA (acu standard, 16 PAHs) (acu standard) was used.
2.9 Quality control/assurance
The performance of the analytical procedure was validated using matrix spike, procedure blank and surrogate compound recovery methods. Descriptions of these procedures are given in Iwegbue et al. [61]. The matrix spike method gave 83.4–98.9% recoveries for the PAH compounds, while the surrogate compound recovery method gave 78–101% recoveries for the PAH. The concentrations of PAHs in the procedural blank samples were below the limits of detection (LODs). The PAHs in the samples were quantified via external calibration methods. The calibration curves were obtained as plots of the peak areas of the PAHs versus known concentrations of PAHs, and the linear regression coefficient (r2) of the calibration curves ranged from 0.9994 to 0.9999. The intraday and interday precisions, expressed as the relative standard deviation (RSD), were 3.5–9.6% and 5.2% to 10.4%, respectively. The minimum detection limit for GCMS used for the analysis of the sediment samples on the chromatogram was 1 × 10–3 mg/kg.
2.10 Statistical analysis
Analysis of variance (ANOVA) was used to determine whether the PAH concentrations varied significantly within and among the sampling locations, with a p value less than 0.05 (p < 0.05) considered to indicate statistical significance. The statistical calculations were performed with SPSS version 19. The relationship between PAHs was established through the use of principal component analysis.
2.11 Source identification
The PAH isomeric ratios and their significance, as compiled by Yunker et al. [62], were adopted as tools to identify the PAH sources. The isomeric ratios used included BaA/(BaA + Chry), IndP/(IndP + BghiP), Ant/(Ant + Phen), Flt/(Flt + Pyr), LMW/HMW, COMB-PAHs/TPAHs, BaP/BghiP, PAH(4)/PAH(5 + 6) and the total index.
2.12 Ecological risk assessment of PAHs in sediments
In this research, risk quotients (RQs) were used to assess the ecological risk of PAHs in sediments. The quality values for negligible concentrations (NCs) and maximum permissible concentrations (MPCs) of PAHs in the media were determined from [63, 64]. The risk levels posed by certain PAHs were characterized by risk quotients (RQs), which were calculated as follows:
where CPAHs is the concentration of certain PAHs in the medium and CQV is the corresponding quality value of a certain PAH in the medium (Kalf, et al., [65]. The MPC refers to the concentration of pollutants above which the risk of adverse effects is considered unacceptable to ecosystems, and the NC refers to the concentration in the environment below which the occurrence of adverse effects is considered to be negligible [67]. Therefore, RQNCs and RQMPCs are defined as follows:
RQ (NCs) and RQ (MPCs) of individual PAHs that were not less than 1 were assumed to calculate the RQ∑PAHs (NCs) and RQ∑PAHs (MPCs) of ∑PAHs to fully consider the ecological risk of individual PAHs. A RQ (NCs) of 1.0 indicates that the contamination of a single PAH was more severe. RQ (NCs) > 1.0 and RQ(MPCs), where CQV(NCs) is the quality value of the NCs of the PAHs in the medium (Table 2) and CQV(MPCs) is the quality value of the MPCs of the PAHs in the medium. The RQ∑PAHs, RQ∑PAHs (NCs) and RQ∑PAHs (MPCs) are defined as follows:
3 Results
Table 3 shows the PAH concentrations found in sediment samples, whereas Table 4 shows the summary statistics of the PAH concentrations (µg·kg−1) in the sediment. A total of 10 PAH compounds were detected in the sediment samples, including naphthalene, acenaphthylene acenaphthene, fluorene, phenanthrene, anthracene, benzo(a)pyrene, pyrene, fluoranthene and chrysene. Across the sampling locations, the concentration of PAHs ranged from 810 µg/kg−1 to 3470 µg/kg−1. Compared to the other points sampled, sampling point 5 had the highest concentration of PAHs (3470 µg/kg−1). PAH source apportionment employing isomeric ratios and multivariate statistics indicated both pyrogenic and petrogenic source inputs (such as burning biomass, wood, charcoal, liquid fossil fuel, and crude oil) in the sediment samples (Table 5). The homologous distribution sequence of PAHs in the sediment revealed 3-ring > 2-ring > 4-ring PAHs (Fig. 2). The sediment samples were mostly dominated by LMW PAHs. Table 6 shows the results of the PCA for the sediments. The results of the ecological risk assessment for the study area indicate that there is low ecological risk associated with PAH exposure in sediment.
4 Discussions
Anthropogenic activities such as industrialization, urbanization, population explosion and modernization have obviously and unobviously affected mangrove ecosystems, especially sediments. PAHs are widespread pollutants in the environment, and their major sources are human activities generated from the partial combustion of carbon-based materials such as fossil fuels; wood; emissions from engines; smoke from cooking; industrial activities; and natural occurrences such as when volcanoes erupt [40, 51].
4.1 Concentrations of PAHs in sediment
The total concentration of each polycyclic aromatic hydrocarbon (PAH) at ten (10) sampling points in Ifiekporo Creek was reported, with the primary goal of providing an overview of the distribution of the PAHs in the mangrove sediments. The total concentration of PAHs analysed in the sediments ranged from 810 µg/kg to 3470 µg/kg (Table 3), with a mean value of 3134 µg/kg (Table 4). The highest PAH concentration (3470 µg/kg) was recorded at sample point 5. As far as individual PAHs are concerned, some of the analysed components were not detected across the sampling points. However, Nap (2-ring) was the most prevalent PAH species, with a mean value of 485 µg/kg detected in the sediments from all sampling points, with the exception of sample point 7. Other studies investigating PAH in sediments have also reported naphthalene as the compound that accumulates at the highest concentrations in aquatic organisms [41, 64]. A high concentration of Nap together with other LMWW PAH species is indicative of recent, recurrent and widespread industrial pollution from petrogenic sources [33]. Ace with a mean value of 539 µg/kg was detected at all sample points except sample points 3, 5, and 10. Other 3-ring PAHs, such as Acy, Flu, Ant, and Phe, were equally detected in the sediment, with mean values of 298, 110, 136, and 600 µg/kg, respectively. The results showed high concentrations of 2-ring, 3-ring and 4-ring PAH species in the sediment from Ifiekporo Creek. However, 5-ring and 6-ring PAHs (BbF, BkF, BaP, DahA, IndP and BghiP) were not detected in the sediment from Ifiekporo. Furthermore, the results indicate that Flt (4-ring) had the highest mean concentration of 616 µg/kg. The 3-ring PAHs were the highest (dominant) species in the sediment of the study area. The concentration of PAHs in the samples was P > 0.05, suggesting a spatially significant difference (Table 7). CV was less than 100%, while the F-calculated was less than the F-critical.
Compared with the results of other reported studies (Table 8), the PAH concentration in sediment from the Escravos River Basin, Calabar River, River Niger, Ase and Forcados River which fall under the same region of this study, revealed that the PAH concentration ranged between 750 and 213,000 µg/kg−1, 1,670.0 and 100.0 µg/kg−1, 2400 and 19000 µg/kg, 2930 and 16100 µg/kg−1 and 1620 and 19800 µg/kg−1 for the River Niger, Ase and Forcados Rivers, respectively [66]. The Ʃ16 PAH concentrations in the sediment from Ifiekporo were greater than those found in sediments from several lakes, rivers and estuaries worldwide: Lake Awassa, Ethiopia 24.9–413 µg/kg−1 [68]; the Pengkalan Chepa River, Malaysia, 36.7–560.7 µg/kg− [69]; and the Luan River, China, 20.9–287.0 µg/kg−1 [63].
The mean concentration (3134 µg/kg−1) of PAHs in sediments from Ifiekporo Creek exceeded the target value of 1000 μg/kg−1 but was less than the intervention of 40,000 μg/kg−1 (Department of Petroleum Resources DPR, [80]. Baumard et al. [81] described four categories of sediment contamination by PAHs (Table 9). Based on this classification, the sediments from the Ifiekporo Creek rivers can be classified as being in the “high” contamination category since the mean concentrations of PAHs (3134 µg/kg-1) were above 1000 µg/kg and below 5000 µg/kg-1−1. Taken together, these findings suggest that Ifiekporo Creek has experienced a high level of chemical contamination. However, stringent pollution control measures and sustainable management are needed to reduce PAH concentrations and associated impacts on mangrove ecosystems.
4.2 Composition and distribution patterns of PaHs in sediments
Figure 2 shows the compositional patterns of the PAHs detected in this study. The sediment samples were mostly dominated by LMW PAHs. The HMW PAHs ranged from 4 to 6 rings, and the LMW PAHs ranged from 2 to 3 rings. The settled HMW PAHs are not prone to surface exchange due to their low vapour pressure, and HMW PAHs exhibit increased resistance to degradation. In contrast, LMW PAHs, since they have vapour pressures, are associated with the gaseous phase. However, LMW PAHs are susceptible to weathering through oxidation, photodegradation and loss by leaching owing to their high-water solubility [65, 82, 83]. This study revealed that 5- and 6-ring PAHs, BbF, BkF, BaP, DahA, BghiP and IndP, are the most common recessive components because they are less common. The presence of Flu and Phen at sampling points 1, 2, 4 and 8 are also indicative of combustion sources at those sampling points [84, 85].
4.3 Source apportionment/diagnostic ratio
Isomeric ratio determination and multivariate statistical analysis were carried out to determine the sources of PAHs in the sediment samples. This tool is used to design control strategies and evaluate both ecological and human risks associated with the study area (Table 5). In previous studies [61, 66, 86], isomeric ratios such as Ant/(Ant + Phen), Flt/(Flt + Pyr), BaA/(BaA + Chry), IndP/(IndP + BghiP), and BaP/BghiP as well as other LMW/HMW, ƩCOMB/TPAHs, and PAH (4) + (5)/TPAHs have been employed for PAH analysis of input sources and transport properties.
Fossil fuel combustion and petroleum product leakage are pyrolytic and petrogenic sources of PAHs and are the principal sources of anthropogenic PAHs. To differentiate between petrogenic and pyrogenic sources of PAHs, ratios of Phe/Ant and Flu/Pyr have been commonly used. Phe/Ant values > 10 are typically associated with polycyclic aromatic hydrocarbons of petrogenic origin, whereas low Phe/Ant ratios 10 are frequently produced during combustion processes. Values > 1 have been utilized to denote pyrolytic origins, while values < 1 are assigned to petrogenic sources for the Fl/Py ratio [87, 88].
LMW/HMW can also be used to differentiate emissions from pyrogenic sources and petrogenic sources [86, 89]. An abundance of HMW PAH (e.g., 4-, 5- and 6-rings) is typically characteristic of pyrogenic origin whereas LMW PAHs (e.g. 2- and 3-ring species) are more abundant in petroleum sources; ratios of LMW/HMW were calculated to further distinguish petroleum from combustion source inputs. LMW/HMW ratios > 1 indicate petroleum source whereas values < 1 imply combustion [59, 102]. The LMW/HMW ratio from this study in Ifiekporo Creek ranged between 0.81 and 3.08, and most of the samples (94.4%) had LMW/HMW values of > 1, while only 5.6% had LMW/HMW < 1. Similar to the findings of previous studies, these findings strongly suggest that petrogenic sources are more prevalent than pyrogenic source inputs.
The Ant/(Ant + Phen) ratio for this study ranged from 0.37 to 1.00; this was found in sample stations one, two, and eight, indicating a prevalence of inputs from combustion sources. The BaA/(BaA + Chry) value of 0.75 in the sediment of Ifiekporo Creek was detected only in sampling station one. A Flt/Flt + Pyr < 0.4 indicates a petroleum source, values between 0.4 and 0.5 indicate petroleum combustion sources (especially carbon-based fuel, fumes from outboard engines and crude oil), and a ratio > 0.5 indicates contributions from biomass combustion, coal, and wood sources. A value > 0.5 indicates a dominant contribution from biomass combustion sources; < 0.2 indicates petroleum-related sources; values between 0.20 and 0.30 suggest sources related to the combustion of liquid fossil fuels; and values > 0.35 indicate coal/biomass combustion sources [62]. Both IndP/(IndP + BghiP) and BaP/BghiP were absent in the samples collected from this creek. This finding suggests that further studies should be carried out in the studied areas for confirmation or clarity. Although an IndP/(IndP + BghiP) ratio between 0.2 and 0.5 is an indication of the presence of biomass and liquid fossil fuel combustion as major sources of PAH contamination, BaP/BghiP from other studies has been utilized to distinguish between vehicular traffic and nonvehicular traffic sources. The COMBPAH/TPAH ratios are indicative of the relationship between PAH input sources and the combustion of typical organics [66, 90]. A ƩCOMB-PAH/Ʃ16 PAH value < 0.3 is an indication of petrogenic emission; a range between 0.3 and 0.7 indicates mixed sources of emission or deposition; and a ratio > 0.7 is usually suggestive of input from a burning process of high magnitude [61, 91]. In this research, 33.3% of the sediment samples had values between 0.3 and 0.7, which suggested the dominance of mixed sources, while the rest of the samples had values less than the equipment detection limits. This was detected in three sample stations; one station shows petrogenic, and the other two show mixed sources. Finally, the PAH (4)/PAH (5 + 6) ratio used to provide detailed information on the transport features of the PAHs from related studies was not shown in these studies. For example, a high PAH (4)/PAH (5 + 6) value indicates that the contamination is related to PAHs transported from a certain distance, while a low value suggests inputs from local or nearby emission sources [66].
4.4 Principal component analysis (PCA)
Principal component analysis (PCA) is an important tool for identifying the source of PAHs, and it can be used to determine the loading of different components/factors. For the sediments, PCA revealed 28.855% positive loadings for Flu, BaA and Chry for factor 1 (Table 6). BaA and Chry are markers for coal, wood, natural gas combustion, and gasoline emissions [92,93,94,95]. Flu is a 3-ring PAH that is characterized by a low-temperature combustion process involving the burning of wood, grass and other biomass [90]. Factor 2 accounted for 56.398% of the variance, with positive loadings comprising Nap, Acy, Phen and Carb. Nap is a 2-ring PAH, while Acy and Phen are 3-ring PAHs. The nap is an index for incomplete combustion processes [96]. Although Carb is not among the major or priority US EPA 16 PAHs, it is semi volatile, and Factor or Component 3 constitutes 70.997%, with Ant, Flt and Pyr as the main positive loadings. Flt and Pyr are tracers for the combustion of coals [97]. Flt and Ant are indicators of oil combustion [98, 99]. Finally, factor 4 constituted 84.860% of the total oil combustion with only Ace and Flt.
4.5 Ecological risk assessment of PAHs in sediments from Ifiekporo Creek
PAHs always occur in sediment as complex mixtures of compounds, leading to cumulative effects on the ecological environment [100]. Consequently, PAHs in sediment may pose a threat to aquatic ecosystems. Ecological risk assessment is a useful technique for evaluating the risk that polycyclic aromatic hydrocarbons (PAHs) pose to ecosystems [101]. The application of the risk quotient (RQ) to assess the ecological risk of organic compounds was first proposed by Kalf et al. [66] in 1997. In 2010, Cao et al. [63] improved the method by including components for toxic equivalency factors. This new method was proven to be more accurate and scientific [63] and was used here to assess the ecological risk of PAHs in sediment from Ifiekporo Creek.
Table 10 presents the RQ(NCs) and RQ(MPCs) data for PAHs in the sediments from Ifiekporo Creek. The RQΣPAHs(NCs) and the RQΣPAHs(MPCs) were calculated for different sampling locations in Ifiekporo Creek. All of the RQΣPAHs (NCs) except Nap (2.10 µg/kg-1) were found to be less than 1.00 in areas where the values were within the detection limits. In comparison, most of the RQΣPAHs (MPCs) were greater than 1.00 and above 800 (Table 2). The MPCs values for Nap (2100 µg/kg-1) and Acy (960 µg/kg-1) were considered to indicate high risk. All the other detected PAH species were of moderate or low risk, which suggests a low-risk or minimal ecological interaction with the organism’s exposure to PAH concentrations in the mangrove sediments.
5 Conclusion
In the present study, sediment samples contained both low- and high-molecular-weight PAHs. The total PAH concentrations in the sediment samples were below safe levels, indicating that at the time of the investigation, there may not have been any significant threats to the environment, human health or aquatic life around Ifiekporo Creek. PAH pollution in the Ifiekporo area may be natural in origin. The total PAH concentrations in the sediment samples were moderately high. With low-molecular-weight PAHs predominating, indicating a higher proportions of petroleum PAH inputs over their pyrolytic-derived counterparts. The results of the ecological health risk study in this area indicate that there was low ecological risk associated with PAH exposure in sediments from Ifiekporo Creek.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper. Should any raw data files be needed in another format, they are available from the corresponding author upon reasonable request.
References
FAO. The World’s Mangroves 2000–2020. Rome, Italy. 2023. https://doi.org/10.4060/cc7044en.
Implementation, U. N. E. P. D. O. E. P. Mangroves of Western and Central Africa. Nairobi: UNEP/Earthprint; 2007.
Usman U, Hilmi E, Iqbal A. The distribution of mangrove area, mangrove density, and species diversity on the North Coast of Jakarta. Omni-Akuatika. 2023;19(1):88. https://doi.org/10.20884/1.oa.2023.19.1.1061.
De Souza AS, Machado Torres JP, Meire RO, Neves RC, Couri MS, Serejo CS. Organochlorine pesticides and polychlorinated biphenyls in sediments and crabs (Chasmagnathus granulate, DANA, from mangroves Guanabara Bay, Rio De Janeiro State Brazil. Chemospher. 1851. https://doi.org/10.1016/j.chemosphere.2007.04.093.
Lee SY, Primavera JH, Dahdouh-Guebas F, McKee K, Bosire JO, Cannicci S, Diele K, Fromard F, Koedam N, Marchand C, Mendelssohn I, Mukherjee N, Record S. Ecological role and services of tropical mangrove ecosystems: a reassessment: reassessment of mangrove ecosystem services. Glob Ecol Biogeogr. 2014;23(7):726–43. https://doi.org/10.1111/geb.12155.
Aghomi SS. The implications of good governance and security for sustainable environment and development in the Niger Delta region of Nigeria. Peace Environ African J. 2019;5(1):66–90.
Eriegha OJ, Sam K. Characterization of crude oil impacts and loss of livelihood in the Niger delta, Nigeria: a fisheries perspective. Int J Interdisciplinary Res. 2020;1:255–73.
Faridah-Hanum I, Salleh MN. Tertiary forestry education beyond 2020: the case for Malaysia. J Trop For Sci. 2018;30(5):439–45.
Numbere AO, Camilo RG. Mangrove leaf herbivory along a hydrocarbon pollution gradient in a mangrove forest (Rhizophora racemosa) in the Niger River Delta, Nigeria. J Petroleum Environ Biotechnol. 2019. https://doi.org/10.35248/2157-7463.19.10.391.
Strain E, Kompas T, Boxshall A, Kelvin J, Swearer S, Morris R. Assessing the coastal protection services of natural mangrove forests and artificial rock revetments. Ecosyst Serv. 2022;55:101429. https://doi.org/10.1016/j.ecoser.2022.101429.
Kazemi A, Castillo L, Curet OM. Mangrove roots model suggest an optimal porosity to prevent erosion. Sci Reports. 2021. https://doi.org/10.1038/s41598-021-88119-5.
Akanni KA. Economic benefits of non-timber forest products among rural communities in Nigeria. Environ Natl Resour Res. 2013. https://doi.org/10.5539/enrr.v3n4p19.
Mayur SN, Prabhakar RP. Importance of mangroves to human well-being. Int J Zool Investigat. 2022;08(01):421–33. https://doi.org/10.33745/ijzi.2022.v08i01.047.
Akram H, Hussain S, Mazumdar P, Chua KO, Butt TE, Harikrishna JA. Mangrove health: a review of functions, threats, and challenges associated with mangrove management practices. Forests. 2023;14(9):1698. https://doi.org/10.3390/f14091698.
Adanguidi J, Padonou EA, Zannou A, Houngbo SB, Saliou IO, Agbahoungba S. Fuelwood consumption and supply strategies in mangrove forests—Insights from RAMSAR sites in Benin. Forest Policy Econ. 2020;116:102192. https://doi.org/10.1016/j.forpol.2020.102192.
Hendy IW, Michie L, Taylor BW. Habitat creation and biodiversity maintenance in mangrove forests: teredinid bivalves as ecosystem engineers. PeerJ. 2014;2:e591. https://doi.org/10.7717/peerj.591.
Fang X, Ghazali S, Azadi H, Skominas R, Scheffran J. Agricultural land conversion and ecosystem services loss: a meta-analysis. Environ Develop Sustain. 2023. https://doi.org/10.1007/s10668-023-03597-z.
Getzner M, Islam MS. Ecosystem services of mangrove forests: results of a meta-analysis of economic values. Int J Environ Res Public Health. 2020;17(16):5830. https://doi.org/10.3390/ijerph17165830.
Zhang L. Effects of urban land use transitions on ecosystem services: implication on Chinese urban planning. J Land Sci. 2022. https://doi.org/10.56388/land220730.
Ding X. Research on the ecological and economic impacts of rising sea levels on mangroves in southern florida in the context of climate change. E3S Web of Conf. 2023;424:02005. https://doi.org/10.1051/e3sconf/202342402005.
Kabir M, Habiba U, Iqbal MZ, Shafiq M, Farooqi R, Shah A, Khan W. Impacts of anthropogenic activities & climate change resulting from increasing concentration of Carbon dioxide on environment in 21st Century; a critical review. IOP Conf Series Earth Environ Sci. 2023;1194:012010. https://doi.org/10.1088/1755-1315/1194/1/012010.
Mackie E, Shuckburgh E, Jones D, Vaughan D. How climate change is affecting sea levels. Weather. 2020;75(9):280–280. https://doi.org/10.1002/wea.3716.
Paul AO. Oil production and the poverty situation in the Niger delta region of Nigeria: a case of selected communities. J World Econ Res. 2018;7(1):21. https://doi.org/10.11648/j.jwer.20180701.13.
Wosowei JB, Shastry C. An efficient intelligent oil well monitoring system for Niger delta oil fields. Int J Adv Res Sci Commun Technol. 2023. https://doi.org/10.48175/ijarsct-9563.
Idamoyibo H. Accounting for government performance in the development of the Niger delta region of Nigeria. Int J Account Business Soc. 2020;28(3):25–54. https://doi.org/10.21776/ub.ijabs.2020.28.3.2.
Khisamutdinov I, Litskevich E. Problems of evaluation efficiency of the national wealth region. Oil And Gas Business. 2014;2:368–82. https://doi.org/10.17122/ogbus-2014-2-368-382.
Ajibola JO. Mangrove ecosystem recovery and restoration from oil spill in the Niger delta: the GIS perspective. Geoinform Geostatist Overview. 2013. https://doi.org/10.4172/2327-4581.s1-017.
Makowski C, Finkl CW. Threats to mangrove forests. Berlin: Springer; 2018.
Onyena AP, Sam K. A review of the threat of oil exploitation to mangrove ecosystem: Insights from Niger Delta. Nigeria Global Ecol Conservat. 2020;22:e00961. https://doi.org/10.1016/j.gecco.2020.e00961.
Keller R, Justino JF, Cassini ST. Assessment of water and seafood microbiology quality in a mangrove region in Vitória. Brazil J Water Health. 2013;11(3):573–80. https://doi.org/10.2166/wh.2013.245.
Vyas DA. Assessing the chemical contaminants including heavy metals and persistent organic pollutants in urban wildlife and birds. Natl J Arts Commerce Sci Res Rev. 2023. https://doi.org/10.52458/23944870.2023.v10.iss1.kp.a1.
Nowakowski M, Rykowska I, Wolski R, Andrzejewski P. Polycyclic aromatic hydrocarbons (PAHs) and their derivatives (O-PAHs, N-PAHs, OH-PAHs): determination in suspended particulate matter (SPM)—a review. Environ Proc. 2021. https://doi.org/10.1007/s40710-021-00555-7.
Li XH, Ma LL, Liu XF, Fu S, Cheng HX, Xu XB. Polycyclic aromatic hydrocarbon in urban soil from Beijing. China J Environ Sci. 2006;18:944–50. https://doi.org/10.1016/S1001-0742(06)60019-3.
Farrington J, Takada H. Persistent organic pollutants (POPs), polycyclic aromatic hydrocarbons (PAHs), and plastics: examples of the status, trend, and cycling of organic chemicals of environmental concern in the ocean. Oceanography. 2014;27(1):196–213. https://doi.org/10.5670/oceanog.2014.23.
Kumar S, Choudhary M. Synthetic aromatic organic compounds bearing 4,4-Dimethyl-3-thiosemicarbazide moiety: theoretical and experimental approach. Polycyclic Aromat Compd. 2022;43(2):1735–57. https://doi.org/10.1080/10406638.2022.2036777.
Balmer JE, Hung H, Yu Y, Letcher RJ, Muir DC. Sources and environmental fate of pyrogenic polycyclic aromatic hydrocarbons (PAHs) in the Arctic. Emerging Contaminants. 2019;5:128–42. https://doi.org/10.1016/j.emcon.2019.04.002.
Ejiako EJ. Persistent organic pollutant: a review on the distribution of polycyclic aromatic hydrocarbons (PAHs) in aquatic ecosystem. Int J Environ Sci Natl Resour. 2022. https://doi.org/10.19080/ijesnr.2022.29.556272.
Peng J, Chen Y, Xia Q, Rong G, Zhang J. Ecological risk and early warning of soil compound pollutants (HMs, PAHs, PCBs and OCPs) in an industrial city, Changchun China. Environ Pollut. 2021;2021(272):116038. https://doi.org/10.1016/j.envpol.2020.116038.
Patel AB, Shaikh S, Jain KR, Desai C, Madamwar D. Polycyclic aromatic hydrocarbons: sources, toxicity, and remediation approaches. Front Microbiol. 2020;5(11):562813. https://doi.org/10.3389/fmicb.2020.562813.
Abdel-shafy HI, Manseur SMM. A review on polycyclic aromatic hydrocarbons: source, environmental impacts, effects on human health and remediation. Egypt J Pet. 2016;25(1):107–23.
Neff JM, Stout SA, Gunster DG. Ecological risk assessment of polycyclic aromatic hydrocarbons in sediments: identifying sources and ecological hazard. Integrated Environ Assess Manag. 2005;1(1):22–33.
Lara S, Villanueva F, Martín P, Salgado S, Moreno A, Sánchez-Verdú P. Investigation of PAHs, nitrated PAHs and oxygenated PAHs in PM10 urban aerosols. A Compr Data Anal Chemosphere. 2022;294:133745. https://doi.org/10.1016/j.chemosphere.2022.133745.
Mastrolonardo G, Francioso O, Di Foggia M, Bonora S, Forte C, Certini G. Soil pyrogenic organic matter characterization by spectroscopic analysis: a study on combustion and pyrolysis residues. J Soils Sediments. 2014;15(4):769–80. https://doi.org/10.1007/s11368-014-1034-x.
Wakeham SG, Canuel EA. Biogenic polycyclic aromatic hydrocarbons in sediments of the San Joaquin River in California (USA), and current paradigms on their formation. Environ Sci Pollut Res Int. 2016;23(11):10426–42. https://doi.org/10.1007/s11356-015-5402-x.
Chibwe L, Davie-Martin CL, Aitken MD, Hoh E, Massey Simonich SL. Identification of polar transformation products and high molecular weight polycyclic aromatic hydrocarbons (PAHs) in contaminated soil following bioremediation. Sci Total Environ. 2017;599–600:1099–107. https://doi.org/10.1016/j.scitotenv.2017.04.190.
Hussain K. Monitoring and risk analysis of PAHs in the environment. In: Hussain C, editor. Handbook of environmental materials management. Cham: Springer; 2019. https://doi.org/10.1007/978-3-319-73645-7_29.
Hamidi NE, Hajeb P, Selamat J, Razis AFA. Polycyclic aromatic hydrocarbon (PAHs) and their bioaccessibility in meat: a tool for assessing cancer risk. Asian Pac J Cancer Prev. 2015;17(1):15–23.
Johnsen AR, Wick LY, Harms H. Principles of microbial PAH-degradation in soil. Environ Pollut. 2005;133:71–84.
Parrish ZD, Banks MK, Schwab AP. Effectiveness of phytoremediation as a secondary treatment for polycyclic aromatic hydrocarbons (PAHs) in composted soil. Int J Phytorem. 2004;6(2):119–37.
Barron MG, Holder E. Are exposure and ecological risks of PAHs underestimated at petroleum contaminated sites? Hum Ecol Risk Assess. 2003;9:1533–45.
Ossai EK, Iwegbue CMA, Ajogungbe EA, Tesi GO. Polycyclic aromatic hydrocarbon and metal concentrations in imported canned maize. Turkish J Agric-Food Sci Technol. 2015;3(1):53–8.
Buell MC, Johannessen C, Drouillard K, Metcalfe C. Concentrations and source identification of PAHs, alkyl-PAHs and other organic contaminants in sediments from a contaminated harbor in the Laurentian Great Lakes. Environ Pollut. 2021;270:116058. https://doi.org/10.1016/j.envpol.2020.116058.
Bashir I, Lone FA, Bhat RA, Mir SA, Dar ZA, Dar SA. Concerns and threats of contamination on aquatic ecosystems. Bioremed Biotechnol Sustain Approaches Pollut Degradat. 2020. https://doi.org/10.1007/978-3-030-35691-0_1.
Odisu T, Okieimen CO, Ogbeide SE. Post oil spill petroleum hydrocarbon components transport and evaluation in parts of the Niger delta mangrove swamp of Nigeria: a case of hydrocarbon component transport through non-turbulent water. Eur J Sustain Develop Res. 2021;5(1):0152. https://doi.org/10.21601/ejosdr/9708.
Nazarov AM, Araslanova LK. New sorbents based on industrial waste for wastewater treatment from petroleum products. Problems Gathering Treat Trans Oil Oil Products. 2023. https://doi.org/10.17122/ntj-oil-2023-3-195-204.
Aghomi SS, Berezi OK. Environmental impact of illegal artisanal refinery in the Niger delta region. Af J Environ Sci Technol. 2023;17(1):68–90.
Nguyen-Tran TTM, Bui TA, Le TS, Pham QH. Evaluation of the carbon storage in the sediments in the mangrove ecosystem growing on abandoned salt pan in Can Gio District, Ho Chi Minh City, Vietnam. Res J Chem Environ. 2023;27(7):65–73. https://doi.org/10.25303/2707rjce065073.
Nguh BS. Anthropogenic stressors on mangrove ecosystem and the emergence of mangrove Refugia on parts of the eastern Atlantic coastline of Cameroon. Int J Contemporary Res Rev. 2017. https://doi.org/10.15520/ijcrr/2017/8/11/362.
Oyo-Ita OE, Offem JO, Ekpo BO, Adie PA. Anthropogenic PAHs in mangrove sediments of the Calabar River, SE Niger Delta, Nigeria. Appl Geochem. 2013;28(212):219. https://doi.org/10.1016/j.apgeochem.2012.09.011.
Decena SC, Arguilles M, Robel L. Assessing heavy metal contamination in surface sediments in an urban river in the Philippines. Pol J Environ Stud. 2018;27(5):1983–95. https://doi.org/10.15244/pjoes/75204.
Iwegbue CMA, Iteku-Atata EC, Odali EW, Egobueze FE. Distribution, sources and health risks of polycyclic aromatic hydrocarbons (PAHs) in household dusts from rural, Semiurban and Urban Areas in the Niger Delta Nigeria. Exposure Health. 2019. https://doi.org/10.1007/s12403-018-0276-z.
Yunker MB, Macdonald RW, Vingarzan R, Mitchell RH, Goyette D, Sylvestre S. PAHs in the Fraser river basin: a critical appraisal of PAH ratios as indicators of PAH source and composition. Org Geochem. 2002;2002(33):489–515.
Cao Z, Liu J, Luan Y, Li Y, Ma M, Xu J, Han S. Distribution and ecosystem risk assessment of polycyclic aromatic hydrocarbons in the Luan River. China Ecotoxicol. 2010;19:827–37.
Asagbra MC, Adebayo AS, Anumudu CI, Ugwumba OA, Ugwumba AAA. Polycyclic aromatic hydrocarbons in water, sediment and fish from the Warri River at Ubeji, Niger Delta Nigeria. Afr J Aquat Sci. 2015;40(2):193–9. https://doi.org/10.2989/16085914.2015.1035223.
Kalf DF, Mensink BJWG, Montforts MHMM Protocol for derivation harmonized maximum permissible concentrations (MPCs) RIVM report 601506 001. National Institute of Public Health and the Environment, Bilthoven, The Netherlands. 1999.
Iwegbue CMA, Ernest B, Obi G, Tesi O, Olisah C, Egobueze FE, Martincigh B. Distribution and sources of n-alkanes and polycyclic aromatic hydrocarbons in sediments around oil production facilities in the Escravos river basin, Niger delta, Nigeria. Archiv Environ Contaminat Toxicol. 2021. https://doi.org/10.1007/s00244-021-00810-w.
Crommentuijn T, Sijm D, De Bruijn J, Van Leeuwen K, Van De Plassche E. Maximum permissible and negligible concentrations for some organic substances and pesticides. J Environ Manag. 2000;58:297–312. https://doi.org/10.1006/jema.2000.0334.
Mekonnen KN, Chandravanshi BS, Redi-Abshiro M, Ambushe AA, McCrindle RI, Moyo R. Distribution of polycyclic aromatic hydrocarbons in sediments of Akaki River, Lake Awassa, and Lake Ziway, Ethiopia. Environ Monit Assess. 2015;187(7):474. https://doi.org/10.1007/s10661-015-4669-1.
Muslim NZ, Babaheidari SH, Zakaria MP. Comparison of polycyclic aromatic hydrocarbons level between suspended solid and sediment samples of Pengkalan Chepa River, Kelantan state, Malaysia. InAIP Conference Proceedings. 2015;1677(1). AIP Publishing.
Ahmed MM, Doumenq P, Awaleh MO, Syakti AD, Asia L, Chiron S. Levels and sources of heavy metals and PAHs in sediment of Djibouti-city (Republic of Djibouti). Mar Pollut Bull. 2017;120(1–2):340–6.
Edokpayi JN, Odiyo JO, Popoola OE, Msagati TAM. Determination and distribution of polycyclic aromatic hydrocarbons in rivers, sediments and wastewater effluents in Vhembe district, South Africa. Int J Environ Res Publ Health. 2016;13:387. https://doi.org/10.3390/ijerph13040387.
Adeniji AO, Okoh OO, Okoh AI. Levels of polycyclic aromatic hydrocarbons in the water and sediment of Buffalo River Estuary, South Africa and their health risk assessment. Arch Environ Contam Toxicol. 2019;76:657–69.
Kanzari F, Syakti AD, Asia L, et al. Aliphatic hydrocarbons, polycyclic aromatic hydrocarbons, polychlorinated biphenyls, organochlorine, and organophosphorous pesticides in surface sediments from the Arc river and the Berre lagoon, France”. Environm Sci Pollut Res. 2012;19(2):559–76.
Karaca G, Tasdemir Y. Temporal and spatial variations in PAH concentrations in the sediment from the Nilufer Creek in Bursa, Turkey. J Environ Sci Health Part A Toxic/Hazardous Substances Environ Eng. 2014;49(8):900–12.
Mcgroddy SE, Farrington JW. “Sediment porewater partitioning of polycyclic aromatic hydrocarbons in three cores from Boston Harbor, Massachusetts.” Environ Sci Technol. 1995;29(6):1542–50.
Potapova M, Desianti N, Enache M. Potential effects of sediment contaminants on diatom assemblages in coastal lagoons of New Jersey and New York States. Mar Pollut Bull. 2016;107(2):453–8.
Liu F, Liu J, Chen Q, Wang B, Cao Z. Pollution characteristics, ecological risk and sources of polycyclic aromatic hydrocarbons (PAHs) in surface sediment from Tuhai-Majia River system China. Procedia Environ Sci. 2012;13:1301–14.
Keshavarzifard M, Moore F, Keshavarzi B, Sharifi R. Polycyclic aromatic hydrocarbons (PAHs) in sediment and sea urchin (Echinometra mathaei) from the intertidal ecosystem of the northern Persian Gulf: distribution, sources, and bioavailability. Mar Pollut Bull. 2017;123:373–80.
Liu Y, Chen L, Jianfu Z, Qinghui H, Zhiliang Z, Hongwen G. Distribution and sources of polycyclic aromatic hydrocarbons in surface sediments of rivers and an estuary in Shanghai China. Environ Pollut. 2008;154(2):298–305.
Department of Petroleum Resources (DPR), Environmental Guidelines and Standards for the Petroleum Industry in Nigeria (EGASPIN). 2012. Department of Petroleum Resources, Abuja, Nigeria.
Baumard P, Budzinski H, Garrigues P, Sorbe JC, Burgeot T, Bellocq J. Concentrations of PAHs (polycyclic aromatic hydrocarbons) in various marine organisms in relation to those in sediments and to trophic level. Mar Pollut Bull. 1998;36(12):951–60.
Najmeddin A, Keshavarzi B, Moore F, Sadegh Z. Pollution, source apportionment and health risk of potentially toxic elements (PTEs) and polycyclic aromatic hydrocarbons (PAHs) in urban street dust of Mashhad, the second largest city of Iran. J Geochem Explorat. 2018. https://doi.org/10.1016/j.gexplo.2018.03.004.
Marynowski L, Kurkiewicz S, Rakociński M, Simoneit BRT. 20 Effects of weathering on organic matter: I. Changes in molecular composition of extractable organic compounds caused by palaeoweathering of a lower Carboniferous (Tournaisian) marine shale. Chem Geol. 2011;285:144–56.
Wang CH, Wu S, Zhou SL, Wang H, Li B, Chen H, Yu YN, Shi YX. Polycyclic aromatic hydrocarbons in soils from urban to rural areas in Nanjing: concentration, source, spatial distribution, and Page 13/19 potential human health risk. Sci Total Environ. 2015. https://doi.org/10.1016/j.scitotenv.2015.05.025.
Zhang Z, Huang J, Yu G, Hong H. Occurrence of PAHs, PCBs and organochlorine pesticides in the Tonghui River of Beijing, China. Environ Pollut. 2004;130(2):249–61.
Effiong IA, Bassey FI, Iwegbue CM, Ekpa OD, Williams SA, Oguntunde FC, Osabor VN. Martincigh BS polycyclic aromatic hydrocarbons in three commercially available fish species from the bonny and cross river estuaries in the Niger delta Nigeria. Environ Monit Assess. 2016;188(9):508.
Chen SC, Liao CM. Health risk assessment on human exposed to environmental polycyclic aromatic hydrocarbons pollution sources. Sci Total Environ. 2006;366(1):112–23. https://doi.org/10.1016/j.scitotenv.2005.08.047.
Magi E, Bianco R, Ianni C, Di Carro M. Distribution of polycyclic aromatic hydrocarbons in the sediments of the Adriatic Sea. Environ Pollut. 2002;119(1):91–8. https://doi.org/10.1016/s0269-7491(01)00321-9.
Socl HH, Garrigues P, Ewald M. Origin of polycyclic aromatic hydrocarbons (PAHs) in coastal marine sediments: case studies in Cotonou (Benin) and Aquitaino (France) area. Mar Pollut Bull. 2000;40:387–96.
Dong T, Lee B. Characteristics, toxicity, and source apportionment of polycyclic aromatic hydrocarbons (PAHs) in road dust of Ulsan Korea. Chemosphere. 2009;74(9):1245–53.
Ravindra K, Wauters E, Van Grieken R. Variation in particulate PAHs levels and their relation with the transboundary movement of the air masses. Sci Total Environ. 2008. https://doi.org/10.1016/j.scitotenv.2008.02.1.
Lin T, Hu L, Guo Z, Qin Y, Yang Z, Zhang G, Zheng M. Sources of polycyclic aromatic hydrocarbons to sediments of the Bohai and Yellow Seas in East Asia. J Geophys Res Atmospheres. 2011;116(D23):23305.
Larsen RK, Baker EJ. Source apportionment of polycyclic aromatic hydrocarbons in the urban atmosphere: a comparison of three methods. Environ Sci Technol. 2003;37(9):1873–81.
Simcik MF, Eisenreich SJ, Lioy P. Source apportionment and source/sink relationships of PAHs in the coastal atmosphere of Chicago and Lake Michigan. Atmos Environ. 1999;33(5071–5079):1999.
Duval MM, Friedlander SK. Source resolution of polycyclic aromatic hydrocarbons in Los Angeles atmosphere—application of CMB with first order decay. US EPA Report no EPA-600/2-81-161. 1981
Zhang X, Cheng S, Zhu C, Sun S. Microbial PAH-degradation in soil: degradation pathways and contributing factors. Pedosphere. 2006;16(5):5655–65.
Liu W, Wang Y, Chen Y, Tao S, Liu W. Polycyclic aromatic hydrocarbons in ambient air, surface soil and wheat grain near a large steel-smelting manufacturer in northern China. J Environ Sci. 2017;57:93–103.
Soltani N, Keshavarzi B, Moore F, Tavakol T, Lahijanzadeh AR, Jaafarzadeh N, Kermani MJ. Ecological and human health hazards of heavy metals and polycyclic aromatic hydrocarbons (PAHs) in road dust of Isfahan metropolis. Iran Sci Total Environ. 2015;505(1):712–23.
Harrison RM, Smith D, Luhana L. Source apportionment of atmospheric polycyclic aromatic hydrocarbons collected from an urban location in Birmingham. UK Environ Sci Technol. 1996;30:825–32.
Baran A, Tarnawski M, Urbański K, Klimkowicz-Pawlas A, Spałek I. Concentration, sources and risk assessment of PAHs in bottom sediments. Environ Sci Pollut Res Int. 2017;24(29):23180–95. https://doi.org/10.1007/s11356-017-9944-y.
Wu C, Bao L-J, Tao S, Zeng E. Mediated distribution pattern of organic compounds in estuarine sediment by anthropogenic debris. Sci Total Environ. 2016;565:132–9. https://doi.org/10.1016/j.scitotenv.2016.04.141.
Walker SE, Dickhut RM, Chisholm-Brause C, Sylva S, Reddy CM. Molecular and isotopic identification of PAH sources in a highly industrialized urban estuary. Org Geochem. 2005;36:619–32.
Acknowledgements
The authors thank the Tertiary Education Trust Fund (TETFUND), which provided funds for this research, and Nigeria Maritime University, Okerenkoko, for providing the enabling environment for the study.
Author information
Authors and Affiliations
Contributions
BOK Funding acquisition, , Resources, Supervision, review & editing S A Conceptualization, Investigation, Writing – original draft, review & editing EOJ Data curation, Formal analysis, Methodology, Software, review & editing
Corresponding author
Ethics declarations
Informed consent
Not applicable, as the research does not involve human subjects..
Competing interests
There are no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Berezi, O.K., Aghomi, S.S. & Eriegha, O.J. Characterization of polycyclic aromatic hydrocarbons in mangrove sediments from Ifiekporo Creek, Warri, Nigeria. Discov Environ 2, 25 (2024). https://doi.org/10.1007/s44274-024-00052-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44274-024-00052-2