Abstract
Applying biochar amendment and manure in tea plantation ecosystems can diminish soil acidification and degradation. However, the impact of these practices on soil respiration and associated mechanisms remains unclear. In this study, we combined a two-year field experiment and laboratory analyses based on soil properties, functional genes, and microbial co-occurrence networks to explore the determinants of soil respiration intensity in a subtropical tea plantation with biochar amendment and manure application. The results showed that the effect of biochar amendment on soil respiration was unconspicuous. Although biochar amendment increased bacterial richness and Shannon index, biochar amendment did not alter the abundance of species associated with C-cycling functional genes. Besides directly adding recalcitrant C to the soil, biochar also indirectly enhanced C sequestration by weakly increasing soil carbon dioxide (CO2) emissions. However, replacing mineral fertilizer with manure significantly stimulated soil respiration in the tea plantation, resulting in a 36% increase in CO2 emissions over two years. The increase in CO2 emissions under the manure treatment was mainly attributed to the increased soil labile C pool, the activity of hydrolytic enzymes (e.g., cellobiohydrolase and acetylglucosaminidase), and the relative abundance of functional genes associated with the C-cycle. This may also be related to the application of manure that increased the abundance of Gemmatimonadetes and altered ecological clusters in bacterial co-occurrence networks. Our correlation network analysis suggested that Gemmatimonadetes might be the potential hosts for C-cycling genes due to their strong positive correlation with the abundance of C-cycling genes. Overall, these findings provide new insights into soil respiration under biochar amendment and manure application in tea plantations and broaden the options for carbon sequestration in soils.
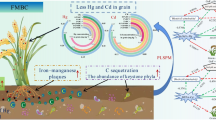
Graphical Abstract

Highlights
-
Manure application rather than biochar amendment increased acidic soil respiration.
-
Biochar application promoted soil C sequestration in the tea plantation.
-
Manure application stimulated CO2 emissions by altering ecological clusters’ abundance.
-
Gemmatimonadetes might be the dominant hosts for C-cycling genes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Carbon dioxide (CO2) emissions through soil respiration are estimated to be approximately 58 Pg C yr−1 (Barba et al. 2018), which is known as the second greatest C exchange between terrestrial ecosystems and the atmosphere (Schlesinger and Andrews 2000). Thus, a minor change in the magnitude of soil respiration will considerably affect the concentration of atmospheric CO2 and the global C cycling (Jones et al. 2003; Crowther et al. 2016). For improving future climate-C cycle feedback projections, unraveling the controlling factors and mechanisms underlying soil respiration changes is imperative.
Tea plantations are diffusely cultivated in subtropical and tropical regions of China, with a total area that approached 3.11 Mha in 2019, most of which are distributed in subtropical regions (NBSC 2020). These tea plantations have been proven to be important soil organic carbon (SOC) pools and contain approximately 83.3–93.5 Tg C (Li et al. 2011; Zhang et al. 2017). The considerable C storage of tea plantations also provides a plentiful potential substrate for releasing CO2 via soil respiration. To improve the quality of tea and enhance local farmers’ incomes, excessive loading of chemical fertilizer has generated soil nutrient leaching, structural damage, and severe acidification in the tea plantation ecosystems (Ni et al. 2019; Yan et al. 2020). With growing environmental awareness and demand for green and low-carbon development, there is an urgent necessity to restrain soil degradation without stimulating soil respiration in the tea plantation ecosystems using current knowledge-based strategies.
Biochar is an alternative soil ameliorative strategy to enhance soil fertility and alleviate environmental deterioration in the long term (Wang et al. 2016; Yang et al. 2022). Biochar amendment may have disparate effects on soil respiration by mediating biotic and abiotic factors. Increased soil respiration rate following biochar application was principally attributed to incremental soil labile C fractions (Smith et al. 2010; Jones et al. 2011). Conversely, the decreased soil respiration rate may be attributed to biochar-induced decreases in soil C availability because biochar particles can absorb the dissolved organic C (Lu et al. 2014) or promote the chemical stabilization of organic C (Weng et al. 2017). Besides, there was no measurable effect of amendment with switchgrass-derived biochar on soil CO2 emissions, as observed in a short-term laboratory incubation trial (Nguyen et al. 2014). Previous findings suggested that knowledge gaps exist in understanding how biotic and abiotic variables drive soil respiration following biochar application. Understanding the intensity and main drivers of soil respiration and the effects of biochar amendment is crucial for developing policies to increase SOC storage and achieve low-carbon development.
Manure substitution for mineral fertilizer has been suggested to be an efficient strategy for building up C stock (Maillard and Angers 2014; Poulton et al. 2018). However, inputs of fresh organic C along with manure can also affect the rate of soil respiration, which may largely affect the carbon storage capacity of soil (Blagodatskaya and Kuzyakov 2008; Guenet et al. 2018). The influences of replacing mineral fertilizer with manure on soil respiration hinge on the peculiarity of exogenous organic substrates, which determines the distinction between soil nutrient availability, SOC chemistry structure, and organic C pool (Yang et al. 2017; Chen et al. 2019; Zhang et al. 2021). Moreover, there might be various CO2 emissions over changes in microbial community diversity and composition after adding organic substances (Blagodatskaya and Kuzyakov 2008; Whitman et al. 2016). For example, diverse C qualities in manure can alter the soil nutrient levels and microbial co-occurrence network, which may affect the relative contribution of keystone taxa-guided groups to the SOC mineralization (Bian et al. 2022). Nevertheless, the previous investigation of manure application on soil respiration was primarily conducted in cropland soils (Guo et al. 2017; Chen et al. 2018), and few opinions are available regarding the influences of replacing mineral fertilizer with manure on soil respiration in subtropical tea plantation soils.
To meet these research needs, we combined a two-year field experiment and laboratory analyses in a subtropical tea plantation to measure CO2 fluxes, environmental factors, functional genes, as well as microbial community diversity and composition under different fertilizer treatments. We hypothesized that manure substitution for mineral fertilizer would promote soil respiration and CO2 emissions, but biochar amendment might not significantly affect soil respiration. The study aims to (i) quantify annual CO2 emissions of different fertilizer practices; (ii) assess the impact of explanatory factors such as soil properties, functional-gene abundance related to C-cycling and microbial structure; and (iii) improve understanding of keystone species in bacterial co-occurrence network controlling soil respiration.
2 Materials and methods
2.1 Site description and the field experiment
From August 2018 to August 2020, the two-year field experiment was performed in a subtropical tea plantation located in Zhenjiang City, Jiangsu Province, China (31°58′N, 119°08′E). The experimental region has a mean annual air temperature of 16.2 °C and a mean annual precipitation of 1192 mm. The soil is classified as a Planosol (FAO 1981), with 35.9 ± 2.1% clay, 36.1 ± 2.0% sand, and 28.0 ± 4.0% silt.
Five treatments with three replicates of the field experiment were settled in a randomized complete block design: no fertilizer (Control), chemical fertilizer (N: P2O5: K2O = 16:16:16; CF), CF in combination with 20 t ha−1 biochar (CF + Bc), manure (full replacement of mineral fertilizer with cattle manure; OF), and OF in combination with 20 t ha−1 biochar (OF + Bc). Each treatment was applied in an 18 m2 plot. Tea trees were planted in ridges, and the distance between rows was 0.3 m. Fertilizers were applied in the soil between rows at a rate of 450 kg N ha−1 yr−1, with one-third as base fertilizer in autumn and the rest as topdressing in spring. However, due to the impact of the COVID-19 pandemic, it was not possible to go out for sampling, and only basal fertilizer was applied in the second experiment period. The biochar was applied in combination with base fertilizer in autumn. Fertilizers and biochar were incorporated into the surface soil to a depth of 0.1 m. The pH value of cattle manure was 7.85, the total N was 2.76%, and the C/N ratio was 7.6. The pyrolysis temperature of biochar was 400 °C, with a total C of 71.3%, a pH of 9.61, and a total N of 0.63%.
2.2 Measurement of CO2 fluxes
CO2 fluxes were measured using the static chamber-gas chromatography (GC) (Zou et al. 2005). Before starting the flux measurement, circular (diameter 0.3 m) polyvinyl chloride resin collars were inserted into the soil between rows to a depth of 0.15 m. The chamber was 0.8 m in height and 0.3 m in diameter. The sampling method for gas samples was reported in our previous study (Han et al. 2021). CO2 concentrations were determined using a gas chromatograph equipped with a flame ionization detector to analyze CO2 concentrations at 330 °C (Agilent 7890A). We used a standard CO2 gas with a concentration of 336.7 ppm to calibrate CO2 concentration. A nonlinear fitting approach was adopted to determine the CO2 fluxes (Kroon et al. 2008). The trapezoid rule on time intervals between measured flux rates was used to calculate cumulative CO2 emissions.
2.3 Soil sample collection
On August 21, 2020, three soil samples were collected using stainless steel corers in each plot. Fresh soil were pooled and passed through a 2-mm sieve to remove plant residues and visible gravel. One portion of the soil sample was kept at − 80 °C to analyze soil microbiota properties, and the rest was stored at 4 °C to quantify soil C pools and determine soil chemical properties. The soil nutrient contents and enzyme activities were measured according to the previous description by Han et al. (2022).
2.4 Analytic method of C composition
The nuclear magnetic resonance spectra of solid-state 13C were determined using a Bruker spectrometer (AVANCE III 600 MHz, Germany). The soil powder samples were put in a 4-mm diameter zirconia rotor and spun at 10 kHz. To measure all spectra, a contact time of 3 ms and pulse delay of 5 s were set, and 10,000 scans were accumulated. The chemical shift (δ) regions of carboxyl and carbonyl C, aromatic C, O-alkyl C, and alky C are 160–190, 110–160, 45–110, and 0–45 ppm, respectively (Wang et al. 2015).
2.5 Incubation experiment
A microcosm incubation experiment was conducted to determine soil C pools and their dynamics (Carrillo et al. 2011). To measure CO2 fluxes, 25 g of fresh soil was packed into 50-ml polyethylene specimen cups, and then deionized water was added to condition the soil to 60% moisture capacity. The soil samples were incubated for 33 days at 25 °C. The syringe was used to extract gas samples at 1, 2, 4, 6, 10, 16, 22, and 33 d. On each date, we collected 10 ml of initial headspace gas immediately after closing the polyethylene sample cups and drew a final 10 ml air sample 2 h later. The CO2 concentrations were measured by the method described in 2.3. Daily fluxes of CO2 were determined according to the difference in CO2 concentration in the final and initial samples. The cumulative CO2 emissions were calculated according to the method described in 2.3. The C kinetic parameters were estimated by fitting a simplified two-order model (Dijkstra et al. 2005; Carrillo et al. 2011):
where F, Ri, k, and Rr represent the daily CO2 flux at time t (mg C kg−1 d−1), the initial decomposition rate of the labile C pool (mg C g−1 d−1), the decay C rate of the labile pool (d−1), and the decomposition rate of C of the resistant pool (mg C g−1 d−1), respectively; the size of the labile C pool was calculated by Ri/k.
2.6 DNA extraction, quantitative PCR, and Illumina sequencing
Following the manufacturer’s standard instructions of the DNeasy PowerSoil Kit (Qiagen Inc., CA, USA), 0.25 g of fresh soil was used to extract total DNA. To determine the copy number of the genes involved in C cycling, real-time quantitative PCR was performed on each DNA sample with three analytical replicates by HT-qPCR QMEC (Zheng et al. 2018). The PCR conditions and primers for these genes are described in Additional file 1: Table S1. High-through paired-end sequencing of DNA samples was performed via Illumina Miseq at Shanghai Tianhao Biological Technology Co. Ltd (Shanghai, China). 16S rRNA gene was amplified by bacterial primers 515F (5′-GTGCCAGCMGCCGCGG-3′) and 907R (5′-CCGTCAATTCMTTTRAGTTT-3′) in the V4–V5 hypervariable regions. The raw sequence data were deposited in the NCBI SRA database using the Accession number PRJNA780408.
2.7 Statistical analyses
Curve-fitting of C kinetic parameters was performed in SPSS 21 (SPSS, Chicago, IL, USA). All statistical analyses were performed in R version 4.2.2 (R Core Team 2021). One-way ANOVA was performed to examine the differences among treatments. Principal coordinates analysis (PCoA) was conducted to determine the Bray–Curtis dissimilarities of bacterial community composition and gene abundance by using the ‘vegan’ package. Procrustes analysis was performed using the ‘vegan’ package. The correlation analysis was performed using the ‘psych’ package. The random forest analysis was performed using the ‘randomForest’ package. The co-occurrence network was constructed based on Illumina sequencing data in the light of the Molecular Ecological Network Analyses Pipeline (http://129.15.40.240/mena/). The construction parameters of the co-occurrence network were based on the method by Han et al. (2022). Network visualization was performed using the Gephi 0.9.2 software. The abundance of each module was determined by adding the abundance of species that belong to it.
3 Results
3.1 CO2fluxes
The CO2 fluxes from subtropical tea plantations were highly episodic, displaying large intra-annual variation (Fig. 1a). Substantial CO2 emissions among the treatments were observed from May through September. Over the 2-year field experiment period, the total CO2 emissions ranged from 2.31 Mg C ha−1 for the CF + Bc treatment to 3.15 Mg C ha−1for the OF treatment (Fig. 1b). Regardless of biochar amendment, manure application stimulated soil CO2 emissions remarkably by 36% across both years. Notably, there was no measurable influence of biochar amendment on CO2 emissions.
Dynamics of soil CO2 emissions (a) and cumulative emissions of CO2 (b) from the field during the 2018–2020 experimental period in a subtropical tea plantation. The arrows indicate fertilizer application events. Control, no fertilizer; CF, mineral fertilizer; CF + Bc, CF treated with biochar; OF, manure; OF + Bc, OF treated with biochar. Values represent mean ± SEM (standard error, n = 3). Different lower-case letters indicate significant differences at P = 0.05 among treatments
3.2 Soil properties
Soil properties were changed significantly along with manure application and biochar amendment (Table 1). In detail, the SOC contents increased strongly following biochar amendment and manure application compared to the CF treatment (P < 0.05). Soil pH and total N contents in the OF plots were higher than those in the CF plots. On the contrary, in comparison with mineral fertilization, manure application and biochar amendment decreased the NH4+ concentration (P < 0.001). Without biochar amendment, manure application increased the activities of cellobiohydrolase and acetylglucosaminidase compared with mineral fertilization (P < 0.05). In contrast, there was no detectable variation in enzyme activities between fertilization treatments and corresponding biochar amendments.
The C functional groups of soils were distinguished using solid-state 13C NMR (Table 1; Additional file 1: Fig. S1). Fertilizer management significantly influenced the SOC chemical composition. Irrespective of fertilizer type, biochar amendments tended to have fewer contents of O-alkyl-C (P < 0.05). Lower levels of Carboxyl and carbonyl C were observed in the OF treatment when compared with the CF treatment (P < 0.05).
3.3 Carbon pool sizes and decomposition rates
The CO2 fluxes of all treatments decreased gradually with the incubation time (Fig. 2a). The cumulative emissions of CO2 among five treatments followed a similar tendency to that in the field experiment (Fig. 2b). Manure substitution for mineral fertilizer significantly increased the total CO2 emissions compared to the CF treatment during the period of incubation experiment, while biochar had no apparent influence on the cumulative CO2 emission regardless of fertilizer type.
Dynamics of soil CO2 emissions (a) and cumulative emissions of CO2 (b) during the incubation period. Labile C pool size (c), its initial decomposition rate (d), and the decomposition rate of the resistant C pool (e) in soils. See Fig. 1 for detailed treatment notes. Values represent mean ± SEM (n = 3). Different lower-case letters indicate significant differences at P = 0.05 among treatments
The simplified two-order model (R2 = 0.61, RMSE = 30.62) indicated that manure application and biochar amendment observably affected the C pool sizes and their decomposition rates. Replacing mineral fertilizer with manure increased the labile C pool by five times compared with the CF treatment (Fig. 2c). In contrast, biochar amendment decreased the labile C pool by an average of 55%, irrespective of fertilizer type. Compared with the Control treatment, fertilization treatments reduced the rate of labile C decomposition (Fig. 2d). The OF treatment decreased the labile C decomposition rate by 60% compared to the CF treatment. Regardless of fertilizer type, biochar amendment enhanced the labile C decomposition rate by 96%. There was no noticeable difference in the rate of resistant C decomposition between the OF and the CF treatments and between the CF and the CF + Bc treatments (Fig. 2e). However, biochar amendment combined with manure remarkably enhanced the rate of resistant C decomposition as compared to the OF treatment.
3.4 Functional-gene abundance related to C-cycling
Replacing mineral fertilizer with manure stimulated the total abundance of functional C-cycling genes, especially those involved in C fixation and starch hydrolysis (Fig. 3a, d). CF + Bc treatment increased the total abundance of functional genes compared to the CF treatment (Fig. 3a, b). On the contrary, OF + Bc treatment reduced the total abundance of functional genes compared to the OF treatment. The result of PCoA explained about 66% of the total variation (P = 0.002; Fig. 3c), which demonstrated that functional genes clustered strongly based on fertilizer treatments.
Summary of functional-gene abundance (a). The ratio of functional-gene abundance between treatments with and without biochar amendment (b). Principal coordinate analysis (PCoA) plot of Bray–Curtis distance of functional genes (c). The ratio of functional-gene abundance between organic fertilization and mineral fertilization (d). See Fig. 1 for detailed treatment notes. Values represent mean ± SEM (n = 3)
3.5 Bacterial community diversity and composition
The richness and Shannon index of bacteria in the OF plots showed higher values than in the CF plots (Fig. 4a, b; P < 0.05). Biochar amendment also increased the bacteria diversity irrespective of fertilizer type (P < 0.05). The PCoA explained the total variation for the bacterial community by 49% among five treatments (P < 0.001; Fig. 4c), which indicated the bacterial community strongly clustered according to treatments. The Procrustes analysis demonstrated that the microbial community composition has a strong correlation with soil conditions (P < 0.001; Fig. 4d). Acidbacteria, Actinobacteria, Bacteroidetes, Chloroflexi, and Proteobacteria were primarily members of the bacterial phyla, accounting for ~ 80% of the relative abundance (Fig. 4e).
Richness (a) and Shannon index (b) of soil bacterial alpha-diversity. Principal coordinate analysis (PCoA) plot of Bray–Curtis distance of bacteria (c). Procrustes analysis of the correlation between bacterial microbial community and soil properties based on non-metric multidimensional scaling (NMDS) analysis (M2 = 0.3349, P < 0.001, 999 permutations) (d). The relative abundance of the dominant bacterial phyla in the soil (e). See Fig. 1for detailed treatment notes. Different lower-case letters indicate significant differences at P = 0.05 among treatments
3.6 Keystone species associated with the functional gene related to C-cycling
As shown in Fig. 5a, based on the NMDS analysis of both OTUs and functional gene abundance, the Procrustes analysis showed an obvious correlation between microbial community and functional genes among all the plots (M = 0.668; P = 0.009). Meanwhile, network analysis was used to explore the pattern of co-occurrence between the species and genes based on their significant correlations (P < 0.05; Fig. 5b). Among the 20 phyla, Gemmatimonadetes had a positive correlation with most of the functional genes related to C cycling, indicating that Gemmatimonadetes might be the potential host of the genes. Notably, manure application remarkably increased the relative abundance of Gemmatimonadetes compared to the mineral fertilization, irrespective of biochar addition (Fig. 5d). Inversely, the network indicated that WPS_2 and TM6 were negatively linked to most of the genes related to C-cycling (Fig. 5b). In comparison with mineral fertilization plots, the relative abundance of WPS-2 and TM6 was lower in manure application plots (Fig. 5c, e). Biochar amendment generally enhanced the relative abundance of WPS-2 and TM6, regardless of fertilizer type.
Procrustes analysis of the correlation between bacterial microbial community and functional genes related to C-cycling based on NMDS results of all OTUs and gene abundances (a). The network correlation between the dominant bacterial phyla and functional genes (b). The relative abundance of WPS-2 (c), Gemmatimonadetes (d), and TM6 under different treatments (e). See Fig. 1 for detailed treatment notes. Values represent mean ± SEM (n = 3). Different lower-case letters indicate significant differences at P = 0.05 among treatments
3.7 Linking soil respiration to microbial co-occurrence network
We built a correlation network to reveal the co-occurrence among bacterial community composition (Fig. 6a). Generally, 6 modules with more than 5 nodes were obtained in the network. Random forest model indicated that CO2 emissions were greatly affected by module1 and Module 5 (R2 = 0.13, P < 0.01; Fig. 6c). Fertilization treatments dramatically influenced the relative abundance of Module 1 and Module 5 (Fig. 6d). Specifically, manure substitution for mineral fertilizer stimulated the relative abundance of Module 1 and Module 5. Besides, the common members of the phyla contained in Module 1 and Module 5 were Acidobacteria, Proteobacteria, Chloroflexi, Actinobacteria, and Gemmatimonadetes (Fig. 6e), indicating that Module 1 and Module 5 had the same ecological clusters. Correlation analysis showed that Module 1 and Module 5 were significantly correlated with soil NH4+ content, pH, and enzyme activities, including phenol oxidase and β-1,4-glucosidase (Fig. 6b).
Network of bacteria based on RMT analysis from OTU profiles (a). Heat maps showing significant correlations between modules and environmental variables (b). The relative abundance of modules in bacteria network (c). Main modules affecting CO2 emissions by random forest modeling analysis (d). The relative abundance of phyla in modules 1 and 5 (e). See Fig 1 for detailed treatment notes. Values represent mean ± SEM (n = 3). Different lower-case letters indicate significant differences at P = 0.05 among treatments
4 Discussion
4.1 Biochar amendment had unconspicuous effect on soil respiration
In this study, field and incubation experiments suggested that biochar application had unconspicuous effect on CO2 emissions in subtropical tea plantations. In accordance with our results, a previous study has also found such a phenomenon in temperate forest soils (Santos et al. 2012). A recent meta-analysis based on 116 observations demonstrated that the influence of biochar amendment on CO2 emissions was associated with various factors such as the experimental duration, soil properties, and biochar characteristics (Wang et al. 2016). The short experimental duration may be an important reason for the unconspicuous effect of biochar on soil carbon turnover rate in our study. A stable C pool of biochar does not decompose quickly in a short period (Wang et al. 2016; Kuzyakov et al. 2014) although the soil-resistant C pool decomposition rate increased within biochar amendment (Fig. 2e). Besides, application rate (20 t ha−1) and pyrolysis temperature (400 °C) of biochar in our study could be another two factors inducing the unnoticeable response of soil respiration to biochar amendment. The meta-analysis showed no effect on soil respiration under biochar amendment when the amount of biochar applied is less than 40 t ha−1 and the pyrolysis temperature is higher than 350 °C (Liu et al. 2016). Based on our observations, more studies on the effect of biochar application amount and its pyrolysis temperature on soil respiration in tea plantations should be designed in the future.
Soil extracellular enzyme activities reflect the labile organic C turnover. For example, the activity of β-1,4-glucosidase corresponded to soil CO2 production. However, biochar amendment insignificantly influenced hydrolytic enzyme activities such as cellobiohydrolase, β-1,4-glucosidase, and acetylglucosaminidase in our study (Table 1), which may cause minor changes in soil CO2 emissions. In addition, although biochar amendment increased bacterial richness and Shannon index, biochar amendment did not alter the abundance of species associated with C-cycle functional genes (Figs. 3a, b; 5c, d). The increased bacterial abundance did not lead to a promotion of CO2 emissions in our study. This finding suggested that the C use efficiency of microorganisms in biochar-amended soils was significantly improved without affecting soil respiration (Lehmann et al. 2015). Meanwhile, the slight changes in soil microbial composition, especially the abundance of Gemmatimonadetes and ecological clusters after biochar amendment, may also have little effect on soil respiration. The relative abundances of Module 1 and Module 5 were not changed under biochar-amended soils (Fig. 6d), although the Random forest model indicated that the rate of CO2 emissions was greatly affected by module1 and module 5 (R2 = 0.13, P < 0.01; Fig. 6c). Furthermore, biochar addition also did not affect soil C functional groups, such as the content of alkyl-C, aromatic C and carboxyl and carbonyl C (Table 1). Notably, the abundance of functional genes related to C-cycling increased when biochar was mixed with mineral fertilizer, while it decreased when combined with organic fertilizer (Fig. 3a, c). This phenomenon could be supported by lower soil NO3− and NH4+ concentrations in the OF + Bc plots because soil nutrients such as NO3− and NH4+ availability shaped the abundance of soil microbial functional genes (Zhang et al. 2019). Overall, the findings obtained in this study indicated that biochar addition could promote soil carbon sequestration in subtropical tea plantations. Besides directly adding recalcitrant C to the soil, biochar indirectly improved C sequestration by weakly affecting soil respiration rate, at least for several years after application.
4.2 Replacing mineral fertilizer with manure promoted soil respiration
Based on a 2-year field experimental observation in the subtropical tea plantation, we demonstrated that manure substitution for mineral fertilizer induced an apparent positive effect on soil respiration. Our incubation trial supported this finding. Specifically, alteration in the quantity and quality of soil C substrate may be one of the main reasons for increased CO2 emissions after replacing mineral fertilizer with manure (Table 1; Fig. 2). First, the increase in SOC content by manure application could enhance the rate of C mineralization and CO2 production. The content of SOC was a latent factor for stimulating the C mineralization (Xie et al. 2017; Xu et al. 2020) because a high quantity of substrate could promote microbial utilization and CO2 emissions (Jiang et al. 2017). Second, manure application provided more DOC content as energy for microbial growth, thereby improving CO2 production derived from microbial respiration (Xiao et al. 2017). It should be noted that soil DOC is also the typical labile fraction of SOC, which may be another reason for contributing to the higher CO2 emissions under manure application treatments, as the high mineralization of SOC was generally consistent with high proportion and low structural complexity of labile organic C (Yang et al. 2021). Third, replacing mineral fertilizer with manure changed the chemistry fractions of SOC. Solid-state 13C NMR spectroscopy indicated that manure application remarkably decreases the content of carbonyl and carboxyl C when compared to mineral fertilization. The low content of carboxyl and carbonyl C under manure treatments might be related to the rapid decomposition of organic acids, lipids, and proteins (Wang et al. 2015). Furthermore, manure application stimulated the labile C pool (Fig. 2), which has been proven to have a strong positive effect on SOC mineralization (Carrillo et al. 2011). Support for this explanation also comes from a grassland field experiment where the proportion of labile C is essential in SOC mineralization and correlates significantly with CO2 production (Dijkstra et al. 2005).
As expected, manure substitution for mineral fertilizer observably affected the biotic factors involved in soil respiration. Our results suggested that the increased enzyme activities may be responsible for stimulating CO2 production after replacing mineral fertilizer with manure (Table 1). SOC turnover is regulated by soil enzymes, such as hydrolase and oxidases, which are vital in converting non-bioavailable to bioavailable SOC (Schimel and Weintraub 2003). For instance, higher activities of cellobiohydrolase and β-1,4-glucosidase in manure plots promoted the hydrolysis of cellulose, which has an active role in the first phase of SOC decomposition and contributes to future biodegradation (Jiang et al. 2017). Besides, the PCoA results indicated that the composition of functional genes was separated by fertilization (Fig. 3c), and the abundance of total functional genes was higher in the manure treatments than in the mineral fertilization treatments (Fig. 3a, d). These findings might be another reason to explain manure-induced increments of CO2 because the functional-gene diversity and abundance are key for regulating biogeochemical processes (Crowther et al. 2016; Graham et al. 2016). Furthermore, manure application significantly increased the bacterial community diversity (Fig. 4), which was positively correlated to CO2 emissions, as a great diversity of bacteria can promote the rapid breakdown of litter derived from tea plantations, thereby enhancing soil organic matter content (Van Der Heijden et al. 1998). Changes in bacterial community composition by manure application also influenced CO2 production. This explanation could be supported by the strong correlation between species and functional genes related to C-cycling (Fig. 5). The correlation network demonstrated that the abundance of functional genes was positively correlated with the Gemmatimonadetes abundance and negatively related to WPS_2 and TM6 abundance (Fig. 5b). This finding consisted with the previous study that Gemmatimonadetes was positively governed by total C and soil organic matter (Liu et al. 2021). Meaningfully, our study indicated that Gemmatimonadetes might be the potential host of C-cycling genes.
We distinguished the extent to which management types and fertilization regimes impacted co-occurrence patterns in the bacteria community. There were six ecological clusters of species strongly co-occurring with each other (Modules1-6, Fig. 6a). Manure application induced a noticeable change in the relative abundance of these modules (Fig. 6d). Specifically, the major factors influencing the modules were soil pH and the activity of phenol oxidase (Fig. 6b), implying that these properties, known to be influenced by the fertilization regime, might not simply result in great changes in bacterial community composition, but might impart a remarkable effect on the network topology (Ling et al. 2016; Fan et al. 2019). In addition, our results identified a subset of associations between CO2 emissions and the co-occurrence patterns in the bacteria community. For instance, the results of the random forest model provided evidence that CO2 emissions in tea plantations were significantly associated with the relative abundance of modules 1 and 5 (Fig. 6c; R2 = 0.13, P < 0.01). This result is consistent with Hartman et al. (2018), who found that land management type and tillage intensity had a marked impact on dominant or well-connected bacteria in the soil, which are related to the function of an ecosystem. Furthermore, common taxa within the dominant phyla contained in modules 1 and 5 include Gemmatimonadetes, which validates the result of a strong positive correlation between the abundance of functional genes and Gemmatimonadetes. Overall, our results indicated that ecological clusters of species might have a proportional role in soil respiration, which could explain why replacing mineral fertilizer with manure affected CO2 emissions.
5 Conclusions
Replacing mineral fertilizer with manure stimulated soil respiration and increased CO2 emissions in subtropical tea plantations. The incubation experiment further supported this finding and suggested that the increased soil labile C pool, hydrolase activity, and the abundance of functional genes mainly contributed to improving C decomposition. Our results also highlight the importance of ecological clusters in the co-occurrence network on soil respiration. Regardless of fertilizer type, biochar amendment had no significant influence on soil respiration and CO2 emissions, indicating that biochar can improve C sequestration by feebly influencing soil respiration. Our study provides new evidence for better understanding the C cycle dynamics and their microbiological mechanisms in acidic agricultural soils.
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Barba J, Cueva A, Bahn M et al (2018) Comparing ecosystem and soil respiration: review and key challenges of tower-based and soil measurements. Agric for Meteorol 249:434–443. https://doi.org/10.1016/j.agrformet.2017.10.028
Bian Q, Wang X, Bao X et al (2022) Exogenous substrate quality determines the dominant keystone taxa linked to carbon mineralization: evidence from a 30-year experiment. Soil Biol Biochem 169:108683. https://doi.org/10.1016/j.soilbio.2022.108683
Blagodatskaya E, Kuzyakov Y (2008) Mechanisms of real and apparent priming effects and their dependence on soil microbial biomass and community structure: critical review. Biol Fertil Soils 45:115–131. https://doi.org/10.1007/s00374-008-0334-y
Carrillo Y, Pendall E, Dijkstra FA et al (2011) Response of soil organic matter pools to elevated CO2 and warming in a semi-arid grassland. Plant Soil 347:339–350. https://doi.org/10.1007/s11104-011-0853-4
Chen Z, Xu Y, He Y et al (2018) Nitrogen fertilization stimulated soil heterotrophic but not autotrophic respiration in cropland soils: a greater role of organic over inorganic fertilizer. Soil Biol Biochem 116:253–264. https://doi.org/10.1016/j.soilbio.2017.10.029
Chen Z, Xu Y, Castellano MJ et al (2019) Soil respiration components and their temperature sensitivity under chemical fertilizer and compost application: the role of nitrogen supply and compost substrate quality. J Geophys Res Biogeosci 124:556–571. https://doi.org/10.1029/2018JG004771
Crowther TW, Todd-Brown KEO, Rowe CW et al (2016) Quantifying global soil carbon losses in response to warming. Nature 540:104–108. https://doi.org/10.1038/nature20150
Dijkstra FA, Hobbie SE, Reich PB, Knops JMH (2005) Divergent effects of elevated CO2, N fertilization, and plant diversity on soil C and N dynamics in a grassland field experiment. Plant Soil 272:41–52. https://doi.org/10.1007/s11104-004-3848-6
Fan K, Delgado-Baquerizo M, Guo X et al (2019) Suppressed N fixation and diazotrophs after four decades of fertilization. Microbiome 7:1–10. https://doi.org/10.1186/s40168-019-0757-8
FAO (1981) Digital soil map of the world and derived soil properties (version 3.5)
Graham EB, Knelman JE, Schindlbacher A et al (2016) Microbes as engines of ecosystem function: when does community structure enhance predictions of ecosystem processes? Front Microbiol 7:1–10. https://doi.org/10.3389/fmicb.2016.00214
Guenet B, Camino-Serrano M, Ciais P et al (2018) Impact of priming on global soil carbon stocks. Glob Change Biol 24:1873–1883. https://doi.org/10.1111/gcb.14069
Guo H, Ye C, Zhang H et al (2017) Long-term nitrogen and phosphorus additions reduce soil microbial respiration but increase its temperature sensitivity in a Tibetan alpine meadow. Soil Biol Biochem 113:26–34. https://doi.org/10.1016/j.soilbio.2017.05.024
Han Z, Wang J, Xu P et al (2021) Differential responses of soil nitrogen-oxide emissions to organic substitution for synthetic fertilizer and biochar amendment in a subtropical tea plantation. GCB Bioenergy. https://doi.org/10.1111/gcbb.12842
Han Z, Xu P, Li Z et al (2022) Microbial diversity and the abundance of keystone species drive the response of soil multifunctionality to organic substitution and biochar amendment in a tea plantation. GCB Bioenergy 14:481–495. https://doi.org/10.1111/gcbb.12926
Hartman K, van der Heijden MGA, Wittwer RA et al (2018) Cropping practices manipulate abundance patterns of root and soil microbiome members paving the way to smart farming. Microbiome 6:1–14. https://doi.org/10.1186/s40168-017-0389-9
Jiang L, Zhu J, Qi Y et al (2017) Increasing molecular structural complexity and decreasing nitrogen availability depress the mineralization of organic matter in subtropical forest soils. Soil Biol Biochem 108:91–100. https://doi.org/10.1016/j.soilbio.2017.01.028
Jones CD, Cox P, Huntingford C (2003) Uncertainty in climate’ carbon-cycle projections associated with the sensitivity of soil respiration to temperature. Tellus B Chem Phys Meteorol 55:642–648. https://doi.org/10.3402/tellusb.v55i2.16760
Jones DL, Murphy DV, Khalid M et al (2011) Short-term biochar-induced increase in soil CO2 release is both biotically and abiotically mediated. Soil Biol Biochem 43:1723–1731. https://doi.org/10.1016/j.soilbio.2011.04.018
Kroon PS, Hensen A, Van Den Bulk WCM et al (2008) The importance of reducing the systematic error due to non-linearity in N2O flux measurements by static chambers. Nutr Cycl Agroecosyst 82:175–186. https://doi.org/10.1007/s10705-008-9179-x
Kuzyakov Y, Bogomolova I, Glaser B (2014) Biochar stability in soil: Decomposition during eight years and transformation as assessed by compound-specific 14C analysis. Soil Biol Biochem 70:229–236. https://doi.org/10.1016/j.soilbio.2013.12.021
Lehmann J, Abiven S, Kleber M (2015) Persistence of biochar in soil. In: Lehmann J, Joseph S (eds) Biochar for environmental management, chapter 10. Earthscan, Oxford, pp 235–281
Li S, Wu X, Xue H et al (2011) Quantifying carbon storage for tea plantations in China. Agr Ecosyst Environ 141:390–398. https://doi.org/10.1016/j.agee.2011.04.003
Ling N, Zhu C, Xue C et al (2016) Insight into how organic amendments can shape the soil microbiome in long-term field experiments as revealed by network analysis. Soil Biol Biochem 99:137–149. https://doi.org/10.1016/j.soilbio.2016.05.005
Liu X, Zheng J, Zhang D et al (2016) Biochar has no effect on soil respiration across Chinese agricultural soils. Sci Total Environ 14:554–555. https://doi.org/10.1016/j.scitotenv.2016.02.179
Liu M, Li X, Zhu R et al (2021) Vegetation richness, species identity and soil nutrients drive the shifts in soil bacterial communities during restoration process. Environ Microbiol Rep 13:411–424. https://doi.org/10.1111/1758-2229.12913
Lu W, Ding W, Zhang J et al (2014) Biochar suppressed the decomposition of organic carbon in a cultivated sandy loam soil: a negative priming effect. Soil Biol Biochem 76:12–21. https://doi.org/10.1016/j.soilbio.2014.04.029
Maillard É, Angers DA (2014) Animal manure application and soil organic carbon stocks: a meta-analysis. Glob Change Biol 20:666–679. https://doi.org/10.1111/gcb.12438
NBSC (2020) China statistical yearbook. China Statistical Press, Beijing
Nguyen BT, Koide RT, Dell C et al (2014) Turnover of soil carbon following addition of switchgrass-derived biochar to four soils. Soil Sci Soc Am J 78:531–537. https://doi.org/10.2136/sssaj2013.07.0258
Ni K, Liao WY, Yi XY et al (2019) Fertilization status and reduction potential in tea gardens of China. J Plant Nutr Fertil 25:421–432. https://doi.org/10.11674/zwyf.18078
Poulton P, Johnston J, Macdonald A et al (2018) Major limitations to achieving “4 per 1000” increases in soil organic carbon stock in temperate regions: Evidence from long-term experiments at Rothamsted Research, United Kingdom. Glob Change Biol 24:2563–2584. https://doi.org/10.1111/gcb.14066
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing. https://www.r-project.org/
Santos F, Torn MS, Bird JA (2012) Biological degradation of pyrogenic organic matter in temperate forest soils. Soil Biol Biochem 51:115–124. https://doi.org/10.1016/j.soilbio.2012.04.005
Schimel JP, Weintraub MN (2003) The implications of exoenzyme activity on microbial carbon and nitrogen limitation in soil: a theoretical model. Soil Biol Biochem 35:549–563. https://doi.org/10.1016/S0038-0717(03)00015-4
Schlesinger WH, Andrews JA (2000) Soil respiration and the global carbon cycle. Biogeochemistry 48:7–20. https://doi.org/10.1023/A:1006247623877
Smith JL, Collins HP, Bailey VL (2010) The effect of young biochar on soil respiration. Soil Biol Biochem 42:2345–2347. https://doi.org/10.1016/j.soilbio.2010.09.013
Van Der Heijden MGA, Klironomos JN, Ursic M et al (1998) Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396:69–72. https://doi.org/10.1038/23932
Wang K, He C, You S et al (2015) Transformation of organic matters in animal wastes during composting. J Hazard Mater 300:745–753. https://doi.org/10.1016/j.jhazmat.2015.08.016
Wang J, Xiong Z, Kuzyakov Y (2016) Biochar stability in soil: meta-analysis of decomposition and priming effects. GCB Bioenergy 8:512–523. https://doi.org/10.1111/gcbb.12266
Weng Z, Van Zwieten L, Singh BP et al (2017) Biochar built soil carbon over a decade by stabilizing rhizodeposits. Nat Clim Chang 7:371–376. https://doi.org/10.1038/nclimate3276
Whitman T, Pepe-Ranney C, Enders A et al (2016) Dynamics of microbial community composition and soil organic carbon mineralization in soil following addition of pyrogenic and fresh organic matter. ISME J 10:2918–2930. https://doi.org/10.1038/ismej.2016.68
Xiao W, Feng S, Liu Z et al (2017) Interactions of soil particulate organic matter chemistry and microbial community composition mediating carbon mineralization in karst soils. Soil Biol Biochem 107:85–93. https://doi.org/10.1016/j.soilbio.2016.12.025
Xie J, Hou M, Zhou Y et al (2017) Carbon sequestration and mineralization of aggregate-associated carbon in an intensively cultivated anthrosol in north China as affected by long term fertilization. Geoderma 296:1–9. https://doi.org/10.1016/j.geoderma.2017.02.023
Xu P, Liu Y, Zhu J et al (2020) Influence mechanisms of long-term fertilizations on the mineralization of organic matter in Ultisol. Soil Tillage Res 201:104594. https://doi.org/10.1016/j.still.2020.104594
Yan P, Wu L, Wang D et al (2020) Soil acidification in Chinese tea plantations. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2020.136963
Yang M, Li Y, Li Y et al (2017) Effects of inorganic and organic fertilizers on soil CO2 efflux and labile organic carbon pools in an intensively managed Moso Bamboo (Phyllostachys pubescens) plantation in subtropical China. Commun Soil Sci Plant Anal 48:332–344. https://doi.org/10.1080/00103624.2016.1269802
Yang Y, Liu H, Dai Y et al (2021) Soil organic carbon transformation and dynamics of microorganisms under different organic amendments. Sci Total Environ 750:141719. https://doi.org/10.1016/j.scitotenv.2020.141719
Yang Y, Sun K, Han L et al (2022) Biochar stability and impact on soil organic carbon mineralization depend on biochar processing, aging and soil clay content. Soil Biol Biochem 169:108657. https://doi.org/10.1016/j.soilbio.2022.108657
Zhang M, Chen Y, Fan D et al (2017) Temporal evolution of carbon storage in Chinese tea plantations from 1950 to 2010. Pedosphere 27:121–128. https://doi.org/10.1016/S1002-0160(15)60098-4
Zhang K, Li X, Cheng X et al (2019) Changes in soil properties rather than functional gene abundance control carbon and nitrogen mineralization rates during long-term natural revegetation. Plant Soil 443:293–306. https://doi.org/10.1007/s11104-019-04212-9
Zhang G, Bai J, Zhao Q et al (2021) Soil carbon storage and carbon sources under different Spartina alterniflora invasion periods in a salt marsh ecosystem. CATENA 196:104831. https://doi.org/10.1016/j.catena.2020.104831
Zheng W, Zhao Z, Gong Q et al (2018) Responses of fungal–bacterial community and network to organic inputs vary among different spatial habitats in soil. Soil Biol Biochem 125:54–63. https://doi.org/10.1016/j.soilbio.2018.06.029
Zou J, Huang Y, Jiang J et al (2005) A 3-year field measurement of methane and nitrous oxide emissions from rice paddies in China: effects of water regime, crop residue, and fertilizer application. Glob Biogeochem Cycles 19:1–9. https://doi.org/10.1029/2004GB002401
Acknowledgements
Not applicable.
Funding
This work was supported by the Jiangsu Funding Program for Excellent Postdoctoral Talent (2022ZB324), China Postdoctoral Science Foundation (2023M731725), the National Natural Science Foundation of China (42177285, 42007072), the Jiangsu Provincial Special Project for Carbon Peak Carbon Neutrality Science and Technology Innovation (BE2022423, BE2022308), and the Startup Foundation for Introducing Talent of Nanjing Agricultural University (030/804028).
Author information
Authors and Affiliations
Contributions
ZH: conceptualization, formal analysis, methodology, experimental operation, visualization, writing-original draft. PX: lab measurement. ZL: lab measurement. SG: lab measurement. SW: review and editing. SL: review and editing. JW: funding acquisition, supervision, conceptualization, writing. JZ: funding acquisition, supervision, conceptualization, writing.
Corresponding authors
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Handling editor: Xiaoyuan Yan
Supplementary Information
Additional file 1:
Table S1. Primers used for quantitative PCR and thermal profile for the target genes. Figure S1. 13C NMR of soils under different fertilization treatments. CF, mineral fertilizer; CF + Bc, CF treated with biochar; OF, organic fertilizer; OF + Bc, OF treated with biochar.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Han, Z., Xu, P., Li, Z. et al. Divergent effects of biochar amendment and replacing mineral fertilizer with manure on soil respiration in a subtropical tea plantation. Biochar 5, 73 (2023). https://doi.org/10.1007/s42773-023-00273-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42773-023-00273-3