Abstract
Root-knot nematodes (RKNs) are obligate endoparasites that feed on their host plants to complete its life cycle, representing a major threat to agriculture and economy worldwide. The development of new management strategies becomes essential as effective chemical nematicides are progressively being restricted. Hence, we analysed grape pomace-derived biochars, pyrolysed at 350 °C (BC350) and 700 °C (BC700), focusing on their potential for RKN control. The thermal treatment of grape pomace caused an increase in the concentration of carbon and plant macro- and micronutrients, which were largely present in a water-soluble form. Synchrotron radiation-based Fourier transform infrared microspectroscopy data showed a general loss of carboxylic functional groups during pyrolysis, partially contributing to the alkalinisation of both biochars, mostly in BC700. Scanning electron microscopy coupled with energy-dispersive X-ray spectroscopy analysis revealed a highly porous structure filled with different crystals composed of elements such as K, Ca, Mg, P, Si or Al, which could be a suitable environment for the growth of microorganisms. Biochar-derived aqueous extracts showed phytotoxicity to tomato seedlings at high concentrations, and disappeared upon dilution, but no toxic effect was observed on the nematode’s infective stage. However, the infective and reproductive traits of a Meloidogyne javanica population in tomato were significantly reduced (i.e. egg masses and eggs per plant) in washed-biochar-treated soil in pots (0.75%; BC350W). Therefore, the large amount of grape waste generated after wine production can be transformed into a valuable product such as biochar, effective for RKNs control, thus reducing the waste management problem and contributing to a circular economy.
Graphical abstract

Highlights
-
Grape pomace biochar had a porous structure and was enriched in C and other plant macro- and micronutrients.
-
Biochar produced at high temperatures showed a certain degree of phytotoxicity.
-
Biochar obtained at 350 °C at a 0.75% (w/w) application rate was effective for nematode control in pots.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Global population is rapidly increasing, which undoubtedly leads to an increase in the global food demand, expected to rise up to 56% by 2050 (van Dijk et al. 2021). In order to meet this global need, it is necessary to find new ways of improving agricultural productivity, where optimal pest and disease management will be essential. Plant-parasitic nematodes (PPNs) are among the most widespread pests and can be found in almost every crop worldwide, causing annual global crop losses of approximately $125 billion (Chitwood 2003). PPNs include two major groups: the cyst nematodes (Heterodera spp. and Globodera spp.) and the root-knot nematodes (RKNs; Meloidogyne spp.), which are the focus of this study. RKNs penetrate the roots through the elongation zone and migrate intracellularly towards the root apex, where they enter the vascular cylinder, establish in it, and induce a feeding site which would eventually turn into a knot or gall. Inside the galls, 4–8 vascular cells develop into feeding cells, termed giant cells (GCs); that will nourish the nematode until life cycle completion (reviewed in Escobar et al. 2015). RKNs control constitutes a major problem in tomato both in open fields and greenhouses, as the durability of resistant plants based on the Mi-1 gene, a sustainable control strategy, is being seriously questioned and high temperatures lead to resistance breakage (Verdejo-Lucas et al. 2009). In addition, given the progressive ban of the most effective and economically viable chemical nematicides (Directive 2009/128/EC of the European Parliament and of the Council 2009), it is important to develop alternative and more sustainable management strategies. Agricultural residues are rich in bioactive compounds and can be used as an alternative source to produce bionematicides. Lignocellulosic residues generated by forestry and wood-processing activities, along with lignin-rich agricultural wastes, may be processed through pyrolysis to generate biochar, which is a valorized biomass that can also be rich in natural-product-based biopesticides (Andrés and González-Coloma 2022), and it may therefore contain bioactive components effective for RKN pest control (Marra et al. 2018).
Biochar is a carbon-rich material derived from the decomposition of organic matter at high temperature in an oxygen (O) deficient environment (Lehmann and Joseph 2009). The pyrolysis conditions and the nature of the feedstock are the primary determinants of the biochar physicochemical characteristics (Chandra and Bhattacharya 2019; Yakout 2017). Among them, biochar yield, cation exchange capacity, electrical conductivity or hydrogen (H) and O content tend to decrease with increasing pyrolysis temperature (Cheng et al. 2017; Gai et al. 2014), while pH, specific surface area, fixed carbon (C), hydrophobicity, aromaticity and recalcitrance (defined as the ability of certain molecular structures to resist biodegradation), usually increase (Chandra and Bhattacharya 2019; Cheng et al. 2017; Kloss et al. 2012). Depending on the specific characteristics of each biochar, its application has the potential to change many of the soil characteristics, such as pH (Biederman and Harpole 2013), soil particle aggregation (Blanco-Canqui 2017), water holding capacity (Basso et al. 2013) or nutrient availability (Kamau et al. 2019; Song et al. 2019; Xu et al. 2019). Incorporation of biochar as a soil amendment has several beneficial environmental effects, such as C sequestration and reduction of greenhouse gases emissions (Ginebra et al. 2022), energy production (Bhatia et al. 2021), immobilization of organic and inorganic pollutants (Ali et al. 2019; Du et al. 2021) or contribution to waste management (reviewed by Kavitha et al. 2018). In addition, it has shown multiple benefits related to agriculture such as increased plant growth and crop productivity (Martínez-Gómez et al. 2022) or protecting plants against abiotic (Kim et al. 2016; Liu et al. 2019) and biotic stress (Poveda et al. 2021).
Biochar has been described to promote plant growth through different mechanisms, such as changes in the soil’s physicochemical properties (Huang et al. 2019; Liu et al. 2019) and microbial populations (Hansen et al. 2017; Zheng et al. 2018), increased nutrient availability and plant uptake (Cui et al. 2021; Xi et al. 2020; You et al. 2021; Zhao et al. 2019), changes in the expression of genes related to plant growth (Jaiswal et al. 2020; Mehmood et al. 2020) or increased tolerance to abiotic (Fahad et al. 2016; Kim et al. 2016; Ramzani et al. 2017) or biotic stress (reviewed in Poveda et al. 2021). In the particular case of tomato plants, different types of biochar have shown to be effective in promoting plant growth. Adebajo et al. (2022) observed an increased height, weight and number of flowers and fruits of tomato plants after a rice biochar application, the results that were in line with those of French and Iyer-Pascuzzi (2018), who described a biochar-mediated plant growth promotion, partially through the stimulation of the gibberellin pathway. Moreover, also in tomato plants, biochar application has shown to be effective in ameliorating the effects of abiotic stress such as salinity (She et al. 2018) or drought (Akhtar et al. 2014) and in the management of plant pathogens such as Ralstonia solanacearum (Gu et al. 2017; Lu et al. 2016), Botrytis cinerea (Kolton et al. 2017; Mehari et al. 2015) or Fusarium oxysporum (Akhter et al. 2015, 2016; Jaiswal et al. 2020).
There is not much literature on the use of biochar against PPNs, but the available studies have proposed different potential mechanisms for nematode control, such as changes in the biodiversity of soil nematode populations, induction of plant defenses or direct toxicity (reviewed in Poveda et al. 2021). For example, the application of wheat straw biochar resulted in an increased abundance of fungivore nematodes and decreased abundance of PPNs from the genus Hischmanniella, Pratylenchus, Psilenchus or Tylenchus, among others (Zhang et al. 2013). Huang et al. (2015) observed a reduced infection of Meloidogyne graminicola in rice plants after the application of oak wood biochar, linked to the induction of plant defenses (local H2O2 accumulation and a transcriptional enhancement of ethylene-related genes). An enhanced resistance of carrot plants against Pratylenchus penetrans was also reported, probably due to the induction of plant defenses, although changes in soil pH or direct toxicity could not be ruled out (George et al. 2016). Direct toxicity has also been reported by Rahayu and Sari (2017), where a direct application of biochar from burned log wood in an in vitro assay presented a mortality rate of the nematode Pratylenchus coffeae similar to that of chemical nematicides and reduced its population in the roots of coffee seedlings (Rahayu and Sari 2017).
In the particular case of tomato plants, biochar from several plant sources has been proven effective in reducing Meloidogyne incognita damage through a reduction of the number of galls, egg masses, eggs, infective juveniles or final populations (Arshad et al. 2020, 2021; Eche and Okafor 2020; Ibrahim et al. 2019). However, biochar from by-products/wastes of vitivinicultural origin, as grape pomace, has not yet been evaluated for pathogen control, including PPNs. In this respect, the grape production industry is of great economic importance, not only in southern European countries, but all over the world, with an annual production of 77.8 million tons in 2018, of which 57% were wine grapes (Kokkinomagoulos and Kandylis 2020). The wine sector is continuously expanding and produces a large number of by-products and wastes than can be transformed into valuable products such as biochar, revalorizing them, reducing the waste management problem and contributing to the circular economy. Hence, we used grape pomace, a residue from the wine industry, as the raw material from which two different biochars were produced. A characterization of the two biochars produced at 350 and 700 °C was performed, and their main element composition, major functional organic groups and structure were analysed, as well as the cyto- and phytotoxicity of their aqueous extracts in biological assays. Finally, we explored its potential as a control method for RKN management.
2 Materials and methods
2.1 Biochar production
Two different biochars were used in the study. They were prepared from wine industry residues (grape pomace, GP) from the 2017 harvest, kindly provided by Vinos Coloman, S.A.T. (Ciudad Real, Spain). The grape pomace used was mainly a mixture of grape varieties “Garnacha” and “Tempranillo”. The material was ground in a blender and pyrolysed in a rotatory quartz furnace (Carbolite CB HTR 11/150P8, Carbolite Gero) at 350 °C or 700 °C for 2 h. The working temperature was reached at a 10 °C min−1 heating rate and N2 was continuously passed at a 1 NL min−1 flow rate. The resulting chars were identified as BC350 and BC700 and, after washing (see below), BC350W and BC700W.
2.2 Biochar characterisation
Elemental analysis: the raw material and both biochars (BC350 and BC700) were analysed with a LECO CHNS-932 analyser (LECO) to determine C, nitrogen (N), sulfur (S), and H concentration. Surface area (SDA) and micropore volume (Vmicro) were determined by CO2 adsorption at 0 °C in an automated gas adsorption analyser (Tristar 3020, Micromeritics Instrument Corp.). Samples were prepared for the adsorption analysis by degasifying under vacuum for 7 h at 150 °C (VacPrep 061, Micromeritics Instrument Corp.). SDA and Vmicro were calculated with the Dubinin–Astakhov equation (Dubinin and Astakhov 1971). The amount of surface oxygen groups (SOG) was analysed through a temperature programmed desorption under N2 flow (1 mLN min–1) (TPD–N2) in a vertical quartz reactor. Each sample was heated to 900 °C with a heating rate of 10 °C min–1 and the amount of CO and CO2 released was continuously measured with an Ultramat 23 NDIR analyser (Siemens AG). The higher heating value (HHV) was determined with a calorimetric bomb (IKA C2000, IKA®-Werke GmbH and CO. KG), according to technical specification UNE-EN 5400. Three independent biological replicates were used.
Inductively coupled plasma mass spectrometry (ICP-MS) analysis: a multi-elemental analysis was performed by inductively coupled plasma-mass spectrometry (ICP-MS Nex-ION 300XX, PerkinElmer). Dry GP, biochar (BC350, BC350W, BC700 and BC700W) and dried plant samples (shoots and roots separately) were ground to a fine powder. One hundred mg of each sample (except 50 mg for roots) were digested under acidic oxidative conditions (2 mL digestion mixture with HNO3:H2O2:H2O, 0.6:0.4:1 (v:v) using concentrated reagents) using an autoclave at 121 °C and 1.5 atm for 30 min (Ortega-Villasante et al. 2007). The digestions were filtered through a 0.45 μm cellulose filter and diluted in milli-Q water to 5 mL. Six and four independent biological replicates were used for biochar and plant measurements, respectively.
Synchrotron radiation-based Fourier transform infrared microspectroscopy (SR-FTIR) analysis: pellets for SR-FTIR analysis were prepared by mixing 198 mg of KBr (IR-grade, Sigma-Aldrich, ref. no. 221864) and 2 mg (1% m/m) of dehydrated GP, each biochar sample, and their corresponding aqueous extracts. The aqueous extracts were previously lyophilised with a LyoMicron lyophiliser (Coolvacuum Technologies, S.L.). The mixture was thoroughly ground in an agate mortar to form a fine homogenous powder, which was placed into a Manual Hydraulic FTIR Press GS15011 (Specac). Using a 10 mm diameter vessel, a translucent disc-shaped tablet was obtained using a pressure of 3500 kg for 5 min. Subsequently, each pellet was placed on a transparent CaF2 window and the SR-FTIR spectra were obtained at the BL01-MIRAS beamline of ALBA Synchrotron (Barcelona, Spain), using a Hyperion 3000 microscope (Bruker) coupled to a spectrophotometer (VERTEX 70 spectrometer and 50 µm HgCdTe (MCT) detector). The aperture size was 10 µm × 10 µm, and the microscope objective magnification was 36×. For each KBr pellet sample, between 25 and 45 points was analysed, and each obtained spectrum corresponds to the accumulation of 128 scans with a resolution of 4 cm−1 in a wavenumber range of 4000–900 cm−1. Spectra were corrected and normalized using a baseline correction algorithm and vector normalization procedure, respectively. Second derivative data and principal component analysis (PCA) were performed using OPUS 7.5 (Bruker) and Unscrambler® X 10.3 (CAMO Software) software (Barón-Sola et al. 2021).
Scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDX): the surface morphology of each biochar and the GP was analysed in more than three different zones by scanning electron microscopy. Samples were gold coated with a sputter coater (Q150T S, Quorum Tech.) and scanned with a scanning electron microscope (S-3000N, Hitachi High-Tech) operating at 20 kV. Local elemental composition was analysed by X-ray energy-dispersive spectroscopy (EDX) (QUANTAX EDS model XFlash 6–30, Bruker Corp.). Several types of crystals were found and further analysed by EDX. These mineral-like particles were classified into 4 main types based on their predominant elements (excluding C and O): crystals composed mostly of Ca (type 1); K and Ca (type 2); P, K, Ca and Mg (type 3) or Si, K and Al (type 4). For each type of crystalline structure, a different number of particles were analysed by EDX: type 1, n = 23; type 2, n = 6; type 3, n = 18; type 4, n = 3. Images were captured at different zooming magnifications.
2.3 Preparation of biochar aqueous extracts
Both biochars were immersed in distilled water at the concentration of 3% (w/v) for 24 h in constant agitation, filtered through filter paper (FILTER-LAB®, ref. no. 1305) and dried for 24–48 h at room temperature. Washed biochars were identified as BC350W and BC700W. The aqueous extracts obtained during the filtration process were collected, stored at − 20 °C and identified as BC350AE and BC700AE. The aqueous extracts were centrifuged for 5 min at 12,000×g, the supernatant was filtered through a 0.2 µm syringe filter and used to prepare six dilutions (1/2, 1/5, 1/10, 1/50, 1/100 and 1/1000). The pH values of these dilutions were measured with a pH meter BASIC 20 (Crison Instruments S.A.).
2.4 Plant material and amplification of nematode populations
Solanum lycopersicum L. cv. Marmande (Semillas Batlle S.A, ref no. 015901) was used for all the experiments. For the in vitro assays, seeds were surface sterilized for 30 min with a sterilization solution (20% commercial bleach (35 g of active chlorine per L) and 1 μg μL−1 Triton X-100) and rinsed thoroughly with sterile distilled water.
Meloidogyne javanica (Treub 1885) Chitwood 1949 population was amplified in vitro on cucumber roots (Cucumis sativus L. var. Prior) and egg mass hatching to obtain infective second-stage juvenile (J2) nematodes for the in vitro infection assays was performed according to Diaz-Manzano et al. (2016). The M. javanica population for the nematicidal activity assay and soil experiments was maintained in S. lycopersicum L. cv. Marmande plants in pots at 25 °C. Egg masses of M. javanica were handpicked from infected tomato roots and incubated in tap water at 25 °C for 24–96 h to obtain J2s from the hatched eggs.
2.5 Phytotoxicity assays with biochar aqueous extracts
Square plates (120 mm) containing a thin layer of 1% plant agar in distilled water (Duchefa Biochemie, ref. no. P1001) and 3 layers of sterile filter paper (0.17 mm thickness, FILTER-LAB®, ref. no. 1305) were used to sow 15 sterile tomato seeds per plate with sterile tweezers. Sterilization of tomato seedlings was performed as described above. Four mL from each dilution and the original aqueous extracts (1×; BC350AE and BC700AE) or distilled water (control) were added to the plates. Plates were sealed with one layer of Parafilm® and covered with aluminium foil. After 2 days of stratification at 4 °C, the plates were transferred to a dark growth chamber at 26 °C in a vertical position. The percentage of seed germination and root length were measured after 3 and 7 days, respectively. Plants were scanned and the root length was measured using the image processing package Fiji, based on ImageJ (US National Institutes of Health, Bethesda, MD) (Schindelin et al. 2012). For root length, a total of 100 plants per treatment from at least 3 independent biological replicates were measured. For the germination assays, a total of ≥ 60 plants from 4 independent biological replicates per treatment were assayed.
2.6 Cytotoxicity assays with biochar aqueous extracts
The human hepatocarcinoma cell line (HuH7) was obtained from American Type Culture Collection. HuH7 cells were cultured in DMEM (Lonza) supplemented with 10% heat inactivated fetal bovine serum (FBS), 2 mM l-glutamine, 1% antibiotic (penicillin–streptomycin) and incubated in a humidified incubator at 37 °C and 5% CO2. When cells reached about 80% confluency, they were treated with BC350AE or BC700AE at different concentrations (50%, 20%, 10%, 2%, and 1%) for 24 h. The 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay, to evaluate cell viability, was performed following Mosmann (1983) with some modifications. Basically, after the different incubations were finished, the conditioned medium was removed and cells were washed with PBS and incubated with 0.5 mg mL−1 MTT dye (Sigma, ref. no. CT01-5) in DMEM medium without phenol red for 30 min at 37 °C. Blue formazan crystals were solubilised with dimethylsulfoxide (DMSO) and a colorimetric evaluation was performed using a spectrophotometer NanoDrop 2000 (Bioteck S.p.A.) at 555 and 690 nm (this last one to subtract background) . Three biological replicates per treatment were used.
2.7 Nematicidal activity assay with biochar aqueous extracts
Potential nematicidal activity of the aqueous extracts was tested as described in Andrés et al. (2018). Three consecutive aqueous extracts were obtained from washing BC350 and BC700 three consecutive times with distilled water (3% w/v) as described above, and were named BC350AE, BC350AE-1, BC350AE-2, BC700AE, BC700AE-1, and BC700AE-2. These extracts were tested in vitro on M. javanica J2s. The pH was adjusted to 6.4 with 0.2 M acetic acid for half of each of the extracts.
For the nematode treatments, a nematode inoculum consisting of 500 J2s in tap water was concentrated through a 25 μm filter and the nematodes were suspended in either 500 μL of tap water (control) or each of the biochar aqueous extracts. The J2s suspension was divided into 4 aliquots (100 μL) used as replicates of approximately 100 J2s each and placed in a 96-well plate (BD Falcon) for every treatment. The plates were covered to prevent evaporation and maintained in the dark at 25 °C. The dead J2s were counted under a binocular microscope after 72 h. The nematicidal activity was expressed as percent of dead J2s corrected according to the Schneider-Orelli’s formula (Püntener 1981). Four independent biological replicates were analysed.
2.8 M. javanica in vitro infection assays with biochar aqueous extracts
Tomato seeds were sown in square plates and incubated as mentioned above for the phytotoxicity assays. Four dilutions from each aqueous extract were used: 1/2, 1/5, 1/10 and 1/50 for BC350AE and 1/5, 1/10, 1/50 and 1/100 for BC700AE. Three days after germination in the dark at 26 °C, plants with similar root length were inoculated with 10–20 J2s per root tip, covered with a thin layer of 0.8% modified Gamborg B5 medium (Gamborg et al. 1968) to favor nematode penetration, and maintained at identical conditions for 3 days. The number of galls per plant was measured 3 days post inoculation. Twenty-four independent biological replicates per treatment were used (a total of ≥ 240 plants per treatment).
2.9 Analysis of nematode infectivity/reproduction and plant physiological parameters in tomato plants grown in biochar-treated soils
Tomato seeds were germinated for 3 weeks in vermiculite at 25 ± 2 °C, 60%RH, under long-day photoperiod (16 h light/8 h dark). The biochar treatment was prepared by mixing BC350W at a concentration of 0.75% (w/w) with a substrate consisting of sandy loam soil. Pots containing 1000 g of the moistened substrate (either soil without biochar as control or with 0.75% BC350W) were inoculated with 2000 J2s of M. javanica each and incubated for 5 days in a growth chamber at 25 °C. After this incubation period, 3-week-old tomato seedlings maintained in vermiculite were transplanted into the pots, grown under the same conditions and fertilised with 50 mL of a 0.3% solution of 20–20–20 (N-P-K) every 10 days. Seven weeks after inoculation, roots were processed according to Andrés et al. (2018). The number of egg masses was counted under a stereomicroscope after erioglaucine staining (Atamian et al. 2012), and nematode infectivity was determined as the number of egg masses per plant. Eggs were extracted from the entire root system (Hussey and Barker 1973) and the reproduction of the nematode was expressed as the number of eggs per plant. Eggs were counted as the average of three aliquots of 100 µL per plant under a stereomicroscope. The multiplication rate of the nematode was estimated by dividing the number of eggs per plant by the J2s inoculum. Physiological parameters (number of leaves, number of tomatoes, average tomato weight, plant fresh biomass and relative water content, and RWC) were assessed in the same plants mentioned above. Twenty biological replicates were used for average tomato weight and RWC, and 15 for total fresh biomass, number of leaves and number of tomatoes. A minimum of 15 biological replicates were used for infection and reproduction parameters.
Fresh biomass was measured immediately after removing the plant from the pot. The RWC was determined based on the method of Barrs and Weatherley (1962). Basically, small leaflet pieces, excluding midrib and margin, were cut and weighted (fresh weight; FW). Then, the leaflets were immersed in distilled water and incubated at 4 °C for 24 h. The adhering water of the pieces was blotted dry in a piece of paper, and turgid weight (TW) was measured. The leaflets were left in an oven at 70 °C for 24 h before dry weight (DW) was measured. RWC was calculated according to this formula: RWC (%) = ((FW − DW)/(TW − DW)) * 100. More than 10 independent biological replicates were assessed.
For the treatments in Additional file 1: Figs. S4–S6, tomato seedlings were also grown in vermiculite for 3 weeks under the same conditions explained before and transplanted and grown in pots with 800 g of sandy soil and the different concentrations of BC700 or BC350 either washed or unwashed, as indicated in the figure legends. They were maintained in a growth chamber at 26 ± 2 °C, 60%RH, under long-day photoperiod (16 h light/8 h dark) for 3 weeks and fertilised with 150 mL of a 0.3% solution of 20–20–20 (N-P-K) every 15 days. Parameters shown in Additional file 1: Fig. S6 were measured 6 weeks after germination.
2.10 Data analysis
Significant differences between samples were tested using the IBM® SPSS® Statistics for Windows version 24.0 software (IBM Corp 2016). The number of tomatoes per plant was transformed with log(x + 1) to adjust the values as closely as possible to a normal distribution. The data was subjected to different statistical analyses according to the dataset’s characteristics. The ICP-MS data was subjected to One-Way ANOVA + Duncan’s Multiple Range Test. Cell viability data was subjected to One-Way ANOVA + Tukey’s Range Test. Seed germination, root length, number of leaves per plant, number of tomatoes per plant, average tomato weight, RWC and total fresh biomass were subjected to the Mann–Whitney U test. Egg masses per plant, eggs per plant and eggs per J2 were subjected to the Student’s t-test. Confidence intervals were established with a significance of 5% (p < 0.05) for each test.
3 Results and discussion
The results presented are firstly centered in the detailed characterization of two biochars produced at 350 and 700 °C from grape pomace, an abundant residue from the wine industry. Other characteristics as its cyto- and phytotoxicity in biological assays were also performed, and the main objective was to study its potential as a method for RKN management.
3.1 Grape pomace and biochar characterization
Biochar yield depends on pyrolysis temperature, heating rate and residence time, and it decreases as the pyrolysis temperature increases (Gheorghe et al. 2009). Accordingly, our data indicates that the yield of the biochar from GP obtained at 700 °C (BC700) was lower than that obtained at 350 °C (BC350), i.e. 28.4% and 45.7%, respectively (Table 1). Jin et al. (2020) reported similar yields from GP biochar produced at 300 °C and 700 °C (55% and 31.8%, respectively). In addition, the physicochemical properties of the GP differed from both biochars, as expected, due to the decomposition of components in the raw material with increasing temperature. The SDA did not suffer pronounced changes as a result of pyrolysis, i.e. when compared to GP, it decreased around 12% at 350 °C, whereas it increased by 11% at 700 °C. However, Vmicro decreased by 50% at 350 °C but increased by 17% at 700 °C (Table 1). Changes in the SDA occur via the organic volatilization and the restructure of aromatic clusters. Hence, gases are released from the carbon matrix when a particle is heated enough to undergo chemical reorganization, resulting in the generation of many small-sized pores (Gale et al. 1995), which seem to be enhanced in the biochar pyrolysed at 700 °C, but not at 350 °C (Table 1). The HHV is directly related to the potential of the material for energy production, and it was approximately 27.5 MJ kg−1 for both biochars, 1.34 times higher than for GP (20.4 MJ kg−1; Table 1). Interestingly, although the pyrolysis temperatures were contrastingly different, both biochars showed similar HHV values. In the same way, GP torrefied at 275–300 °C showed HHVs of 27.1 and 25.3 MJ kg−1, respectively (Pala et al. 2014; Tamelová et al. 2021). Gupta et al. (2019) also showed no differences in HHV when comparing pyrolysis temperatures similar to those in this study (400 °C and 700 °C for teak sawdust). Therefore, both biochars are equivalent in terms of their energy production capacity.
We also measured the amount of CO and CO2 released during a temperature-programmed desorption, which provides information about the amount of oxygen-containing surface groups (SOG) present in the sample (Table 1). In this sense, GP showed a high content of SOG, particularly CO2 evolving groups, reflecting its acidic nature. However, the thermal treatment led to a progressive loss of CO and CO2 with increasing temperature, highly notable for CO2 evolving groups, thus modifying the surface character of the biochars and making them more alkaline. Accordingly, aqueous extracts from both biochars showed an alkaline pH that increased with rising pyrolysis temperatures (pH above 8 for BC350AE and pH above 12 for BC700AE; Additional file 1: Table S1). In this respect, Windeatt et al. (2014) reported pH levels between 6.1 and 11.6 for biochars obtained from eight different feedstocks, most of them being above 9.5. A significant correlation between biochar pH and the sum of potassium (K), calcium (Ca), magnesium (Mg), and sodium (Na) concentrations has been described (Enders et al. 2012). Therefore, the high pH values obtained in the biochar aqueous extracts could also be attributed to a high alkali matter content of the biochars, as there was a significant increase in K, Ca and Mg in BC350 and BC700 (Table 2) compared to the raw material. In this respect, alkaline biochars have the potential to increase soil pH (Biederman and Harpole 2013; Streubel et al. 2011), thus both generated biochars might have potential for the amendment of acidic soils.
When analysing the content of major elements in biochars (Table 2), total C content was 1.3 and 1.5-fold higher for BC350 and BC700, respectively, compared to the raw material. Similar values of C content for GP biochar have been previously reported (Pala et al. 2014; Tamelová et al. 2021; Trakal et al. 2017). In general, C content increases with increasing temperature, which could be attributed to an increase in the rate of carbonization and the development of aromatic carbon structures. With respect to H and S content, both decreased sharply with temperature (Table 2). In this respect, the H/C ratio is used to measure the degree of aromaticity and it generally decreases with the increasing temperature (Ahmad et al. 2013; Shafizadeh 1985) due to H loss and C increase, which is in line with our results. Overall, the tendencies observed for C, H, N and S are in accordance with other reports on GP biochar (Hoffmann et al. 2019; Manolikaki et al. 2016; Pala et al. 2014; Tamelová et al. 2021; Trakal et al. 2017). In order to check whether these elements might solubilize from the biochar matrix of both BC700 and BC350, they were washed with distilled water for 24 h to obtain their respective aqueous extracts, and then dried and named BC350W and BC700W. No significant differences were observed for C, H, N and S concentration when comparing washed and unwashed biochars (Table 2). However, significant differences (p < 0.05) for other elements determined by ICP-MS (Table 2) were found in both biochar samples when compared to GP except for iron (Fe) in BC350W and silicon (Si) in BC350W and BC700, with values similar to those of the raw material. K and Ca were the dominant elements in all the examined biochar samples, followed by Mg and phosphorus (P), which is consistent with other studies on GP biochar (Jin et al. 2020; Manolikaki et al. 2016). Interestingly, the concentration of this group of elements was in most cases higher in the biochar samples than in GP, except for K and Si content in BC350W and BC700W, respectively, which were lower (1.3 and twofold decreases, respectively). Therefore, much of the mineral content in the feedstock seems to have been concentrated in the biochar due to the loss of C, H and O during pyrolysis.
The washing process caused significant changes in K, Mg, P, aluminium (Al), Si and Mg content in BC700W with respect to BC700. Nevertheless, the content of all the analysed elements was higher in BC700W than in GP, except for Si (Table 2). In BC350W, the washing process reduced the concentration of all the analysed elements respect to the unwashed biochar (BC350). These results strongly suggested that most of the elements in the biochar are present in a water-soluble form, particularly in BC350, as a significant concentration decrease was detected after washing the samples (Table 2). In this regard, Atalay et al. (2022) used pyrolysis products of pumpkin wastes as a source of K to crystallize struvite-K, a water-soluble mineral with potential as a slow-release fertilizer, where 80% of the K from the pyrolysis products could be recovered as struvite-K crystals. Therefore, biochar obtained from GP might have potential as a slow-release fertilizer, gradually releasing the elements it contains as the soil is irrigated, though it is important to note that the presence of these elements in the soil does not necessarily imply their bioavailability. The fertilization capacity of the generated biochars was not the main focus of this study, however future research could help elucidate this putative use.
Functional organic groups in the raw GP and washed/unwashed biochars at both pyrolysis temperatures (350 °C and 700 °C) were characterized by SR-FTIR, where we observed meaningful differences in corrected adjusted average second derivative spectra (Fig. 1; Additional file 1: Fig. S1). PCA data revealed that GP clearly segregated from the charred material at 350 °C and 700 °C before and after washing. Washed (Fig. 1) and unwashed (Additional file 1: Fig. S1) biochars grouped closely in most regions. Those biochars showed a clear decrease of O and H containing functional groups as compared to GP (Fig. 1; Additional file 1: Fig. S1), following the pattern observed in biochars elaborated from different plant raw materials (Askeland et al. 2019) and in accordance with the data obtained from the elemental analysis (Table 2). In the region corresponding to aliphatic compounds, from 3100 to 2800 cm−1, we observed several peaks such as 2923 (assigned to νasCH2, asymmetric vibrating cis-alkene group) and 2850 cm−1 (νsCH2, symmetric stretching) of PC-1, which appeared in GP and may correspond to unsaturated fatty acids. Other peaks found in GP were 3062 and 2944 cm−1, corresponding to C-H stretching vibrations from fatty acids (aromatic νCH and νasC–H, respectively) of PC-2 (Jin et al. 2020; Johnston 2017; Lago et al. 2021). Several of those peaks were only detected in the biochar produced at 350 °C, albeit with lower intensity than GP, and remained after washing (Fig. 1; Additional file 1: Fig. S2). This occurred with the different types of fatty acids normally present during wine fermentation, some of them proposed as potential markers of alcoholic fermentation (Csutoras et al. 2022), and therefore expected to be mainly present in GP, but to a lesser extent in the pyrolysed material. GP µSR-FTIR spectra also showed a clear peak of the C=O stretching vibration band at 1741 cm−1, which may correspond to esterified fatty acids (i.e. glycerolipids and storage triacyl glycerols), virtually absent in both washed/unwashed biochars (Fig. 1; Additional file 1: Figs. S1–S3). Therefore, there was a loss of carboxylic functional groups during pyrolysis, partially contributing to the alkalinisation of the biochars produced at both charring temperatures, as previously described for GP biochar (Jin et al. 2020) and in accordance with the alkaline nature of the obtained biochar aqueous extracts (Additional file 1: Table S1). In the 1700–1300 cm−1 region we observed smaller differences between samples, with several peaks of higher intensity in GP at 1596, 1511, 1486 and 1459 attributed to N–H, C–N amide, N=N, and –NH3 stretching vibration, respectively, at PC-1. Another peaks of N–H and –NH2 stretching vibrations (1502 and 1417 cm−1, respectively) contributed to PC-2 differences of GP compared to 350 °C and 700 °C biochars (Fig. 1). Most of these functional groups correspond to proteins and nucleic acids, which were eventually modified by pyrolysis and almost disappeared from the material pyrolysed at 700 °C, as observed with other plant-derived biochars elaborated at similar temperatures (Xu et al. 2021). In the carbohydrates and lignin region (1300–900 cm−1) GP also segregated from both biochars, with clear peaks at 1186, 1089 and 1066 cm−1 in the PC-1 loading corresponding to phenolic and C–O–C stretching vibrations, characteristic of saccharides and lignin (Jin et al. 2020; Johnston 2017). At 700 °C, most residual C functional groups substantially decreased, resembling the typical FTIR spectra of graphite (Keiluweit et al. 2010). This was clearly observed in the unwashed biochar samples shown in Additional file 1: Fig. S3, where the secondary derivative of BC700 had a wavy pattern in the 1700–900 cm−1 region, where characteristic peaks of amine, carbonyl and carboxyl groups were essentially absent. Hence, our SR-FTIR data suggested that most volatile functional groups had been lost during pyrolysis, while BC350 showed nonetheless an intermediate alteration pattern among GP and BC700, in line with the results shown by Jin et al. (2020), Rajapaksha et al. (2015) and Wu et al. (2012). Such loss of volatile functional groups can explain the significant reduction of the H and N concentration shown in Table 2, whereas the elemental analysis revealed the accumulation of non-volatile alkaline metals such as K, Ca and Mg (Table 2), both probably contributing to the biochar alkalinization during pyrolysis, as formerly discussed.
SR-FTIR spectroscopy of grape pomace (GP) and washed biochars. Biochars were prepared at 350 °C and 700 °C and washed with distilled water at a concentration of 3% (w/v) for 24 h (BC350W and BC700W). Full 2nd derivative average spectra (wavenumber range between 3100 and 900 cm−1) along with the first two main principal components (PC-1 and PC-2) for different wavenumber ranges, which explain the largest contributions of signal sifts between samples. Numbers in red indicate the wavenumber values of the most representative peaks. For each KBr pellet sample, between 25 and 45 points were analysed, and each obtained spectrum corresponds to the accumulation of 128 scans with a resolution of 4 cm−1 in a wavenumber range of 4000–900 cm−1
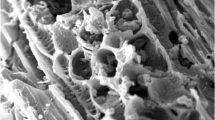
We also characterized the biochar through SEM, and the images were used to visually display the variation in the structure of biochars produced at different temperatures and after the washing process. It revealed a general morphology consistent of particles of varied sizes with a prominent porous structure, which seems to be accentuated after pyrolysis as compared to the raw materials (Fig. 2). Micropores are abundant in both biochars, and probably more abundant in BC700/BC700W than in BC350/BC350W in accordance with the data presented in Table 1. In addition, micropores are interspersed with macropores (internal width > 0.05 µm; Downie et al. 2009) of very irregular size and shape (Fig. 2). Microorganisms, usually of sizes bigger than 0.5 µm, could easily colonize those macropores and take advantage of the potential protection, nutrient and water availability conferred by them (Jaafar et al. 2015; Pietikäinen et al. 2000; Saito and Marumoto 2002; Thies and Rilling 2009; Warnock et al. 2007). The pyrolysis temperature and the washing process appear to have an impact on particle size. As the pyrolysis temperature increased from 350 °C to 700 °C, the overall particle size seemed to decrease (Fig. 2a, g). While BC350W particles resemble those of BC350 (Fig. 2a, d, respectively), the particles of BC700W appeared significantly smaller than those of its unwashed counterpart (Fig. 2j, g, respectively), implying either the contact with water, mechanical agitation or both, causing particle disruption. These biochar particles showed in general sharp angular edges with uneven pores of various diameters. Some of them presented a tubular pore structure (Fig. 2, red arrowheads), with slit-shaped or elongated pores (Fig. 2f, l), presumably derived from conservated xylem elements from the plant (Wang et al. 2006). Some particles showed a more disorganized pore placement (Fig. 2c, e, h, k) and other particles contained both areas of organized even pores and areas of disorganized pore placement (Fig. 2i). Interestingly, many pores contained crystalline structures of different morphology (globular, tabular, etc.) attached to their walls. Those structures varied in size, with the largest observed between 15 and 17 µm in diameter (e.g. in Fig. 2b), and were abundant in those areas with a more disorganized pore placement (Fig. 2b, c, g, h, i, k). Accordingly, it has been reported that inorganic components present in the feedstock undergo changes during the carbonization process: devolatilization, vaporization, and incorporation of metals into the silicate structures, etc. During the last stages of combustion, these inorganic compounds can transform from amorphous phases to crystalline forms (Wornat et al. 1995). Different types of minerals can coexist in the same biochar particles (Amonette and Joseph 2009), as observed in Fig. 2c, h. Those mineral-like particles were present in both biochars but were more abundant in BC700, where most of the biochar particles have almost all their pores filled with crystals (Fig. 2g). When comparing the washed biochars with their unwashed counterparts, crystals look slightly less abundant (Fig. 2c, e, h, k). This might be indicative of the solubility of the mineral structures, mentioned above with the ICP-MS data, as the concentration of most of the elements decreased in the washed biochars, particularly in BC350W, respect to the unwashed ones (BC350, BC700; Table 2). The presence of this soluble inorganic matter suggested a positive environment for the growth of different microorganisms as well as plant roots, either on the surface of the biochar particles or inside their macropores. After an EDX analysis, these mineral-like particles were classified into 4 main types based on their predominant elements: crystals composed mostly of Ca (type 1); K and Ca (type 2); P, K, Ca and Mg (type 3) or Si, K and Al (type 4).
SEM images of GP and biochars at different magnifications. a BC350 at 50-fold magnification. b, c BC350 at 500-fold magnification. d BC350W at 50-fold magnification. e, f BC350W at 500-fold magnification. g BC700 at 50-fold magnification. h, i BC700 at 500-fold magnification. j BC700W at 50-fold magnification. k, l BC700W at 500-fold magnification. m, n GP at 50 and 500-fold magnification, respectively. o GP at 650-fold magnification. Red arrowheads indicate areas with tubular pore structure. Yellow arrowheads indicate examples of crystalline structures inside the biochars’ pores
Figure 3 shows biochar pores with crystalline particles (Fig. 3a, d) and the average percentages of the predominant elements of each particle type when excluding C and O from the total percentage. Type 1 crystals (Fig. 3b) are composed of an average percentage of 86% Ca, followed by 7% K and 4% P, and show an irregular morphology with overlapping plates or rosettes. Type 2 crystals (Fig. 3c) are composed of an average percentage of 45% K and 43% Ca, and its shape resembles an aggregated compact mineral with an irregular shape. Type 3 crystals are composed on average of 34% P, 31% K, 17% Ca and 16% Mg and have a distinctive smooth surface and globular shape (Fig. 3e). Type 4 crystals are composed of an average percentage of 49% Si, 27% K and 17% Al and are morphologically similar to the previous particles, but with an irregular surface (Fig. 3f). These findings appear to be in agreement with the reports inferring the putative mineral types that may be found in biochar, including quartz (SiO2), calcite (CaCO3), sylvite (KCl), anhydrite (CaSO4), hydroxyapatite (Ca10(PO4)6(OH)2), Ca phosphates, amorphous silica, and oxides and hydroxides of Ca, Mg, Fe, Al, titanium (Ti), manganese (Mn) or zinc (Zn) (Amonette and Joseph 2009).
SEM–EDX images of biochar particles filled with crystals. a, d Pores filled with mineral-like particles were detected. Graphical representation of the average percentage of the predominant elements in each type of crystal (excluding C and O) and a representative EDX spectra with an image of the corresponding particle, marked with an arrowhead in the same image (b type 1; c type 2; e type 3; f type 4). Red arrowheads, type 1 particles. Yellow arrowhead, type 2 particle. Blue arrowheads, type 3 particles. Green arrowheads, type 4 particles. Means ± standard errors (n ≥ 3)
3.2 Effect of biochar aqueous extracts on seedling germination and growth
Different effects of biochar aqueous extracts have been previously observed in plants, both beneficial and inhibitory (Bian et al. 2019; Marra et al. 2018, respectively). In order to check whether biochar aqueous extracts from GP may exert effects on plant growth, we checked germination and seedling growth after their in planta application. There were no significant differences in seed germination with the undiluted extract or any of the various dilutions of BC350 and BC700 aqueous extracts (BC350AE, Fig. 4a and BC700AE, Fig. 4b). However, root length was significantly shorter in the undiluted aqueous extract (1×) and in the 1/2 and 1/5 dilutions of BC350AE and BC700AE (Fig. 4c, d). Furthermore, significant growth inhibition was also observed at higher dilutions (1/10 and 1/50) of BC700AE (Fig. 4d), but not at equivalent dilutions of BC350AE. Therefore, our results indicated that both biochars’ aqueous extracts showed some degree of toxicity, although it was remarkably higher in BC700AE than in BC350AE. Similar inhibitory effects were reported for Lepidium sativum and Brassica rapa root growth after treatment with olive mill waste biochar aqueous extracts (Marra et al. 2018), or with extracts of six biochars from varied feedstocks subjected to different thermochemical processes and temperatures, that produced significant inhibition of corn shoot growth, but no effect on seed germination (Rogovska et al. 2012).
Effect of different dilutions of biochar aqueous extracts (BC350AE and BC700AE) on tomato seed germination, seedling growth and animal cell viability. a, b Germination and c, d root length of 7-day-old tomato seedlings grown in different dilutions of biochar aqueous extracts (a, c BC350AE; b, d BC700AE). pH values are displayed above each dilution. e Cell viability (MTT) assay indicates the percentage of viable hepatocytes after their exposure to BC350AE or BC700AE diluted at different percentages, as indicated in the X axis, with cell culture medium. Means ± standard errors (a, b n ≥ 60; c, d n = 100; e n = 3). Statistically significant differences with respect to the distilled water used as a control according to Mann–Whitney U test (a–d) and with respect to distilled water at that same dilution (50%) with culture media according to One Way ANOVA, Post hoc test: Tukey Test (e). ***p < 0.001; **p < 0.01
Interestingly, the aqueous extracts were highly alkaline, with an average pH of 12.3 for 1× BC700AE and 8.4 for 1× BC350AE (Additional file 1: Table S1), although the pH gradually decreased with higher dilutions, correlating with their lower growth-inhibitory effects. This high pH was probably caused by high levels of alkaline metals such as K, Ca or Mg in both biochars (Table 2), which may also contribute to a germination and seedling growth inhibition due to osmotic stress, as previously reported (Joseph et al. 2021). On the other hand, the toxic effects of the aqueous extracts cannot be entirely attributed to pH or salinity, since the 1/100 dilution of BC700AE showed no significant reduction in root length despite its pH of 9.9, higher than the pH of every BC350AE dilution that showed inhibitory effects (1/2, 1/5; Fig. 4c, d; Additional file 1: Table S1). It is therefore feasible that water-soluble compounds could be also responsible for the observed toxic effects, in addition to the alkaline pH. In this sense, polycyclic aromatic hydrocarbons (PAHs) have been found in biochar aqueous extracts that caused corn seedling growth inhibition (Rogovska et al. 2012). SR-FTIR spectra of lyophilized biochar aqueous extracts (BC350AE and BC700AE) grouped well separated from the raw material and their washed biochars (GP, BC350W, BC700W) in the 1700–900 cm−1 region, with peaks corresponding to aromatic phenols and ketones, phenolic ethers, carboxylic esters or carbonyls (i.e. 1563, 1598, 1509 or 1025 cm−1; Additional file 1: Fig. S2) or graphite (1300–1000 cm−1; Additional file 1: Fig. S3), which may correspond to charred saccharide- or lignin-derived materials (Jin et al. 2020; Johnston 2017). Eventually, substances carrying these diverse functional groups may be toxic for plant growth. In this respect, Ruzickova et al. (2021) described water-soluble organic compounds as a possible cause of biochar phytotoxicity from different certified biochars. In contrast, the 1/1000 dilution of BC700AE showed a total opposite effect, as the root length was significantly increased respect to the control (Fig. 4d). Similarly, aqueous extracts obtained from wheat straw biochar significantly enhanced yield, leaf soluble sugar content and shoot and root biomass of Chinese cabbage plants (Bian et al. 2019; Lou et al. 2016). Further focused research could be performed to elucidate this possible growth promotion at certain GP biochar-derived aqueous extract dilutions.
3.3 Cytotoxicity of the aqueous extracts on animal cells
We believe it is important to assess the potential toxicity of the biochar aqueous extracts that could be produced by irrigation of biochar-treated soils. Hence, we treated hepatocytes with the aqueous extracts of both biochars (BC350AE, BC700AE) and determined cell viability trough a MTT assay. Cells were treated for 24 h by replacing the culture medium with increasing concentrations of BC350AE, BC700AE and distilled water as control (Fig. 4e). The 100% cell culture medium treatment was used as a positive control for cell viability. The 50% culture medium in distilled water showed a significant loss of viability respect to the 100% culture media, most probably due to differences in osmolarity. Nevertheless, in the 50% treatment, cell viability in BC350AE and distilled water only differed by 6.6% (being 55.1% for water and 48.5% for BC350AE), but there was a significant reduction in BC700AE (only 15.4% cell viability; p < 0.05), when compared to both BC350AE and distilled water. Therefore, BC350AE did not cause relevant toxicity to animal cells, as cell viability was similar in both BC350AE and water. However, some compounds present in BC700AE may have cytotoxic effects at high concentrations, as shown by the strong reduction in cell viability.
It is important to mention that there is no scientific literature on the effect of biochar aqueous extracts on animal cells, though there are studies on the direct effect of solid biochar. Low cytotoxicity of biochar produced from Alternanthera philoxeroides was reported when tested on human bronchial epithelial cells (Du et al. 2021). Human skin keratinocyte cells exposed to a ferromanganese-bearing sludge-derived biochar also showed insignificant cellular toxicity (Hung et al. 2022). However, biochar cytotoxicity at high doses has been reported in some cases (Dong et al. 2018, 2019). Our results suggest that mammalian cells should not be affected by putative soluble compounds released due to soil irrigation after BC350 application.
3.4 Nematicidal activity of biochar aqueous extracts on M. javanica juveniles
Phenolic compounds present in grapevine tissues have been shown to reduce the survival of RKNs (Wallis 2022), and treatment with some biochars has been effective for nematode control (Huang et al. 2015; George et al. 2016; Rahayu and Sari 2017; Rahman et al. 2014; Zhang et al. 2013). Therefore, we checked whether the biochar aqueous extracts and the biochar itself may exert any effect on nematode performance. In the first place, we tested whether the biochar aqueous extracts showed any direct negative effect against PPNs by assessing the in vitro mortality of J2s of a globally widespread RKN species, M. javanica (Escobar et al. 2015). Both biochars were washed three consecutive times (see Sect. 2) to obtain three different extracts (BC350AE, BC350AE-1, BC350AE-2; BC700AE, BC700AE-1, BC700AE-2). The J2s were incubated for 72 h in each of those extracts. None of the treatments showed any nematicidal activity (defined as J2 mortality > 80%) except for BC700AE, with a mortality rate of 100% (Fig. 5a). As RKNs are attracted to and aggregate at low pH (Wang et al. 2009), we wanted to rule out any possible negative effect directly caused by the high pH of BC700AE (pH 12.26). Therefore, we conducted the same nematicidal bioassay adjusting the pH of the extracts to 6.4 with acetic acid. In contrast to the 100% mortality rate of BC700AE (pH 12.26), the same extract with its pH adjusted to 6.4 showed a very low mortality (3%). Additionally, a KCl/KOH solution with a similar pH to that of BC700AE (12.3) showed a nematicidal activity of 97.9%, close to the 100% obtained with BC700AE. We can, therefore, conclude that the main cause of the nematicidal activity observed in BC700AE was most probably its high alkaline nature, and that no direct effects of the aqueous extracts on second-stage larvae (J2) were observed. Similar results have been reported in the literature, e.g. oak wood biochar exudates did not show a direct negative effect on the viability of M. graminicola juveniles after a 72 h incubation (Huang et al. 2015). Likewise, biochar exudates from olive mil wastes pyrolysed at 500 °C, 800 °C and 1000 °C were not effective against M. incognita (Marra et al. 2018).
Effect of biochar aqueous extracts on nematode mortality and infection parameters of M. javanica in tomato seedlings. a Percentage of mortality of second-stage juveniles (J2s) of M. javanica assessed after incubation with extracts obtained from washing BC350 and BC700 three consecutive times at a concentration of 3% (w/v) in distilled water, with its original pH or adjusted to 6.4, as indicated in the columns. The percentage of mortality of a KCl/KOH solution with alkaline pH (12.3; positive control) is also indicated as a reference for the effect of pH on nematode mortality. b, c Number of galls per plant 3 days post inoculation in tomato seedlings (6 days after germination) grown in different dilutions of aqueous extracts (b BC350AE; c BC700AE). Control, distilled water. Means ± standard errors (a n = 4; b, c n = 24). No significant differences in any of the treatments as compared to control were encountered
Although no evident direct toxicity was observed on the mortality of M. javanica J2s, we tested whether the aqueous extracts could have an inhibitory effect on the infection during the plant-nematode interaction. We performed infection tests with M. javanica in tomato seedlings grown in different dilutions of the aqueous extracts (Fig. 5b, c). Some of the chosen dilutions had previously shown to be phytotoxic to tomato seedling growth (Fig. 4c: 1/2, 1/5 for BC350AE; Fig. 4d: 1/5, 1/10, 1/50 for BC700AE) while some were non-phytotoxic (Fig. 4c: 1/10, 1/50 for BC350AE; Fig. 4d: 1/100 for BC700AE). No significant differences in the number of galls per plant were found for any of the treatments, suggesting no evident effect on the plant-nematode interaction during penetration, migration or establishment of M. javanica. Similarly, Huang et al. (2015) showed no differences in M. graminicola penetration or development in rice plants when incubated in biochar exudates.
3.5 Effect of BC350W in soil on plant growth and nematode control
We have shown in the previous sections a growth-inhibitory effect on tomato seedlings when treated with aqueous extracts from BC700, even at high dilutions (Fig. 4d). Accordingly, when different concentrations of BC700 (3%, 1.5% and 0.75% (w/w) were added to the soil, 3-week-old transplanted tomato plants did not survive because of their toxic effect (Additional file 1: Fig. S4). Due to the high phytotoxicity at low concentrations of both BC700 directly applied to the soil and its aqueous extract (BC700AE) (Additional file 1: Fig. S4; Fig. 4), as well as its proven cytotoxicity at high doses (Fig. 4e), we centered our study from now on in BC350.
When different concentrations of BC350 (3%, 1.5% and 0.75% (w/w)) were added to the soil, we observed some growth-inhibitory effects on tomato plants 3 weeks after transplantation. Plants were smaller and both roots and shoots showed inhibited growth compared to the control (Additional file 1: Fig. S5), although a less pronounced effect than with BC700 (Additional file 1: Fig. S4). This toxicity may result from phytotoxic compounds found in the biochar, as we confirmed the presence of phenolics, ketones, phenolic ethers, carboxylic esters, and carbonyl groups in their aqueous extracts (Additional file 1: Figs. S2, S3) that might cause some toxicity, as observed in seedling growth (Fig. 4c). Additionally, the highly alkaline nature of the water-soluble extracts (Table 2, Additional file 1: Fig. S1) and the presence of soluble compounds (Table 2) could also contribute to plant growth disturbances. In line with this, Kloss et al. (2012) and Yargicoglu and Reddy (2014) reported the presence of heavy metals and polycyclic aromatic hydrocarbons (PAHs) in biochars, produced during pyrolysis. PAHs content increases with pyrolysis temperature, but biochars produced at temperatures under 500 °C shows lower total concentrations of PAHs, suggesting they may be suitable for agronomic use (Rogovska et al. 2012). This is consistent with the lower toxicity observed for the biochar produced at 350 °C as compared to the one generated at 700 °C (Additional file 1: Figs. S4, S5). In line with this, when several biochars obtained by gasification at 1200 °C from four different feedstocks were compared, grape marc biochar resulted in the lowest PAHs content, (∼ 5 mg kg−1), compared to conifer wood, poplar wood or wheat straw biochars (∼ 26 to 33 mg kg−1; Visioli et al 2016).
Considering these results, we decided to wash BC350 with distilled water (see Sect. 2), and the washed biochar (BC350W) was applied at different concentrations (3%, 1.5% and 0.75% (w/w)) to the soil. Tomato plants did not show significant differences compared to control plants in terms of seed germination, number of leaves, fresh biomass and shoot length at 0.75% and 1.5% concentrations, but the biomass was significantly reduced at 3% (p < 0.05) when grown for 3 weeks in those conditions (Additional file 1: Fig. S6). Yet, the highest concentration tested (3%) showed some plant toxicity, while those at 1.5% and 0.75% showed no effect in all parameters related to plant growth (Additional file 1: Fig. S6). As biochar has the potential to increase the water retention capacity of the soil (Basso et al. 2013) and could, therefore, increase the water retention capacity of plants (Akhtar et al. 2014), we measured the RWC of the leaves. In all three biochar concentrations the RWC was higher than in the control plants, indicating that the biochar treatment offers some advantages on the plant water status, which might be pertinent for plant growth under drought conditions. Given that there was an enhanced RWC and no signs of phytotoxicity at the two lowest concentrations of BC350W in soil, all further experiments were carried out with a biochar concentration of 0.75% (w/w). It is important to note that although no phytotoxic effects were observed at 1.5% and 0.75% at the conditions stablished in our experiments, no effect on growth promotion was observed either, something previously reported for other cases (Schnell et al. 2012). A growth-promoting effect might be expected due to the higher content of soluble elements in the biochar (such as K, P or Mg, among others) as compared to the unprocessed GP, (Table 2). However, the fertilization regime used (see Sect. 2), might have buffered this putative growth promotion effect of biochar, or nutrient availability to the plant roots was not adequate.
Yet, there is strong evidence that biochar application can promote defense against pathogens in a wide variety of crops (Poveda et al. 2021). In tomato plants, biochar application has been proven to be effective against pathogens such as Alternaria solani (Rasool et al. 2021), R. solanacearum (Gu et al. 2017; Lu et al. 2016), B. cinerea (Kolton et al. 2017; Mehari et al. 2015) or F. oxysporum (Akhter et al. 2015, 2016; Jaiswal et al. 2020). In these cases, biochar application was able to reduce the incidence or severity of the infection by 29–70%, due to changes in microbial population densities (Kolton et al. 2017; Lu et al. 2016), growth or mobility inhibition (Gu et al. 2017; Rasool et al. 2021), alteration of root exudates (Akhter et al. 2015) or induction of priming or systemic resistance (Jaiswal et al. 2020; Mehari et al. 2015). In this respect, PPNs are a major threat for the agriculture and there is yet scarce information on the ability of biochar to control them (reviewed in Andrés and González-Coloma 2022; reviewed in Poveda et al. 2021). For example, there are a few studies (in rice, grapevine, carrot or tomato) that involve the control of RKNs through the application of biochar produced from different sources such as wheat straw, poultry litter, rice and spelt husk, shea nut shell or wood (Arshad et al. 2020, 2021; Gao et al. 2018; George et al. 2016; Huang et al. 2015; Ibrahim et al. 2019; Rahayu and Sari 2017; Rahman et al. 2014; Zhang et al. 2013). The ones carried out on tomato plants showed infection reductions due to the induction of plant resistance (Arshad et al. 2021) or changes in soil nematode population densities (Ibrahim et al. 2019). However, there are no studies on PPN control involving biochar produced from grape pomace. Therefore, we tested the potential of washed biochar (BC350W) to promote tolerance/resistance to nematodes when applied at a 0.75% (w/w) rate in soil.
Seven weeks after inoculation with the RKN M. javanica, the time required for the nematode to complete its life cycle, the average tomato weight, the RWC of the leaves and the total fresh biomass of the plant (Fig. 6a–c, respectively) were assessed, showing no significant differences between treatments. In contrast, significant differences were observed for the number of leaves (Fig. 6d) and the number of tomatoes per plant (Fig. 6e). In the inoculated plants grown with biochar, tomato production was significantly enhanced by 46% in comparison to the inoculated plants grown in control soil (Fig. 6e). However, the average tomato weight was highly variable in each treatment and therefore there were no significant differences among them. It may suggest different fructification times among treatments, but no direct relation with biochar was observed. In line with this, it has been reported that the application of Douglas fir biochar caused a significant decrease in the flowering time in two different S. lycopersicum cultivars and one Solanum pimpinellifolium cultivar (French and Iyer-Pascuzzi 2018) while patula pine biochar caused earlier fruit emergence in S. lycopersicum (Gallo-Saravia et al. 2018). Analysis of the nutrient content of biochar treated and untreated plants, either inoculated or non-inoculated, did not result in significant differences in any of the tested elements among treatments, except for Si. A significant decrease in the content of Si was observed in the aboveground parts of the non-inoculated plants exposed to BC350W and in the inoculated control, when compared to the non-inoculated control (Table 3). Therefore, even if the concentration of most of the analysed elements was higher in both biochar amendments than in GP (Table 2), a proportion of them is likely present in a water-soluble form (as their concentration decreased in the washed biochars; Table 2), but it does not seem to be directly related to an enhanced nutritional status in the plant tissues 10 weeks after germination. Possible causes include limited ratios of absorption or availability, or a relatively short experiment or that the fertilization regime used provided enough nutrients to sustain an appropriate growth (see Sect. 2). In contrast, there is an extensive literature on the improvement of tomato height, biomass, yield or quality parameters after biochar application (Eizenberg et al. 2017; El-Bassi et al. 2021; French and Iyer-Pascuzzi 2018; Graber et al. 2010; Guo et al. 2021; He et al. 2021; Suo et al. 2021; Velli et al. 2021).
Effect of washed biochar (BC350W) on plant biometric parameters and M. javanica reproduction parameters on infected tomato plants. a Average weight of each tomato, b relative water content (RCW), c total fresh biomass, d number of leaves per plant, e number of tomatoes per plant, f number of egg masses per plant, g eggs per plant and h multiplication rate (MR). Means ± standard errors (n ≥ 15). BC350W, washed biochar; control, sandy loam soil with no biochar. Statistically significant differences with respect to the control according to the Mann–Whitney U test (a–e) or Student’s t-test (f–h) **p < 0.01; *p < 0.05
The effects of biochar treatments on nematode infectivity were evaluated in in vivo assays on tomato plants. Interestingly, the number of egg masses per plant was reduced by 42% in the plants exposed to BC350W (Fig. 6f) as compared to non-treated plants, suggesting that biochar application reduced by almost half the nematode’s ability to penetrate and/or establish within the roots. In addition, the number of eggs per plant and the multiplication rate of the nematode (number of eggs per plant/J2 inoculum) (Fig. 6g, h) were also significantly reduced (39%) by the biochar treatment. These parameters indicate that the biochar obtained from the pyrolysis of GP at 350 °C is able to enhance the tolerance/resistance of tomato plants to RKNs in pots in controlled conditions. Further research will be needed to determine whether this effect is also maintained in field conditions. Additionally, this opens the possibility of this biochar being effective against other pathogens, including different types of PPNs.
Therefore, biochar application represents a potential strategy to combat PPNs, which can be achieved through different mechanisms (Poveda et al. 2021). Changes in soil biodiversity is one of these reported mechanisms, as biochar application can decrease the populations of PPNs, including M. javanica, by increasing the abundance of fungivore nematodes and other plant beneficial microorganisms (Rahman et al. 2014; Zhang et al. 2013). Direct toxicity of biochar constitutes another mechanism, as described for biochar produced from burned wood logs and applied at a 0.5–4% rate, which resulted in a 38–75% mortality of P. coffeae due to nematicidal compounds derived from the biochar (Rahayu and Sari 2017). However, we confirmed that a direct nematicidal effect of the GP biochar analysed in this study is very unlikely (Fig. 5a), but the induction of plant defenses might be feasible due to the reduction in infection and reproduction parameters measured in controlled conditions (Fig. 6). In this respect, a 95% reduction of P. penetrans infection on carrot plants and an 87% reduction in the number of nematodes in the soil fraction have been reported following the application of spelt husk biochar, possibly caused by an induction of a system-wide defense response (George et al. 2016). In a similar way, a combination of rice husk biochar and biocontrol agents (Trichoderma harzianum and Bacillus subtilis) triggered the expression of defense-related genes (PR-1b and JERF3) in tomato plants, enhancing its biomass and reducing M. incognita infection (Arshad et al. 2021). Moreover, the application of 1.2% (w/v) oak wood biochar resulted in induced resistance against M. graminicola probably through a local accumulation of H2O2 and a transcriptional enhancement of ethylene-related genes, which influenced plant defense genes (Huang et al. 2015). Further research will help elucidate whether some of these mechanisms explain the nematode-resistance response observed following 0.75% BC350W treatment in tomato.
Interestingly, our results did not show an enhancement in plant growth except for tomato production 7 weeks after inoculation with M. javanica. We observed, however, a suppression of nematode infectivity following the application of BC350W at low doses (0.75%), which seems to be in agreement with the proposed “Shifted Rmax-Effect” (Jaiswal et al. 2015). In this way, disease suppression and growth enhancement would have inverted U-shape curves relative to dose, and both curves may be shifted relative to each other. As a result, maximum disease suppression occurs normally at low doses where no enhancement of plant growth is expected, while the maximum plant growth response typically occurs at higher biochar doses (Jaiswal et al. 2018).
4 Conclusions
Recycling of GP waste in southern European countries could constitute a relevant contribution to circular economy as grapes and wine production are economically significant in this area. Wine production generates a large amount of waste that may be transformed into valuable products such as biochar, reducing the waste management problem. In this work we describe the main characteristics of two different biochars (obtained after the pyrolysis of GP at 350 °C and 700 °C) and their corresponding aqueous extracts, including their elemental content, differences in the main functional organic groups by SR-FTIR, as well as their cyto- and phytotoxicity. SEM images of the biochar surface revealed abundant micro and macropores filled with mineral-like particles of different composition that may provide a positive environment for the growth of microorganisms. In addition, we observed that M. javanica reproduction in tomato plants was significantly reduced in BC350W-treated compared to untreated control plants (grown in pots under controlled conditions). To our knowledge, this is the first time that an effect on PPN control using GP-derived biochar is described, although the mechanism of action, as well as their efficacy under field conditions, should be investigated in future research. Therefore, the use of GP biochar could be a promising strategy for the control of PPNs that could perhaps be extended to other pathogens or pests in the near future.
Availability of data and materials
The datasets generated and/or analysed during the current study are available from the corresponding authors on reasonable request.
References
Adebajo SO, Oluwatobi F, Akintokun PO, Ojo AE, Akintokun AK, Gbodope IS (2022) Impacts of rice-husk biochar on soil microbial biomass and agronomic performances of tomato (Solanum lycopersicum L.). Sci Rep 12:1787. https://doi.org/10.1038/s41598-022-05757-z
Ahmad M, Lee SS, Rajapaksha AU, Vithanage M, Zhang M, Cho JS, Lee SE, Ok YS (2013) Trichloroethylene adsorption by pine needle biochars produced at various pyrolysis temperatures. Bioresour Technol 143:615–622. https://doi.org/10.1016/J.BIORTECH.2013.06.033
Akhtar SS, Li G, Andersen MN, Liu F (2014) Biochar enhances yield and quality of tomato under reduced irrigation. Agr Water Manag 138:37–44. https://doi.org/10.1016/j.aGPat.2014.02.016
Akhter A, Hage-Ahmed K, Soja G, Steinkellner S (2015) Compost and biochar alter mycorrhization, tomato root exudation, and development of Fusarium oxysporum f. sp. lycopersici. Front Plant Sci 6:529. https://doi.org/10.3389/fpls.2015.00529
Akhter A, Hage-Ahmed K, Soja G, Steinkellner S (2016) Potential of Fusarium wilt-inducing chlamydospores, in vitro behaviour in root exudates and physiology of tomato in biochar and compost amended soil. Plant Soil 406:425–440. https://doi.org/10.1007/s11104-016-2948-4
Ali N, Khan S, Yao H, Wang J (2019) Biochars reduced the bioaccessibility and (bio)uptake of organochlorine pesticides and changed the microbial community dynamics in agricultural soils. Chemosphere 224:805–815. https://doi.org/10.1016/j.chemosphere.2019.02.163
Amonette J, Joseph S (2009) Characteristics of biochar: micro-chemical properties. In: Lehmann J, Joseph J (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 35–52
Andrés MF, González-Coloma A (2022) Agro-industrial by-products and wastes as source of biopesticides. In: Dumas B, Prigent-Combaret C (eds) Recent advances and prospective in the biocontrol of plant diseases. ITSE, London
Andrés MF, González-Coloma A, Muñoz R, de la Peña F, Julio LF, Burillo J (2018) Nematicidal potential of hydrolates from the semi industrial vapor-pressure extraction of Spanish aromatic plants. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-017-9429-z
Arshad U, Naveed M, Javed N, Gogi MD, Ali MA (2020) Biochar application from different feedstocks enhances plant growth and resistance against Meloidogyne incognita in tomato. Int J Agric Biol 24(4):961–968. https://doi.org/10.17957/IJAB/15.1522
Arshad U, Azeem F, Mustafa G, Bakhsh A, Toktay H, McGiffen M, Nawaz MA, Naveed M, Ali MA (2021) Combined application of biochar and biocontrol agents enhances plant growth and activates resistance against Meloidogyne incognita in tomato. Gesunde Pflanz 73(4):591–601. https://doi.org/10.1007/s10343-021-00580-4
Askeland M, Clarke B, Paz-Ferreiro J (2019) Comparative characterization of biochars produced at three selected pyrolysis temperatures from common woody and herbaceous waste streams. PeerJ 7:e6784. https://doi.org/10.7717/peerj.6784
Atalay S, Sargin I, Arslan G (2022) Crystallization of struvite-K from pumpkin wastes. J Sci Food Agric 102(2):523–530. https://doi.org/10.1002/jsfa.11380
Atamian HS, Roberts PA, Kaloshian I (2012) High and low throughput screens with root-knot nematodes Meloidogyne spp. J vis Exp 12(61):3629. https://doi.org/10.3791/3629
Barón-Sola A, Toledo-Basantes M, Arana-Gandía M, Martínez F, Ortega-Villasante C, Dučić T, Yousef I, Hernández LE (2021) Synchrotron radiation-Fourier transformed infrared microspectroscopy (μSR-FTIR) reveals multiple metabolism alterations in microalgae induced by cadmium and mercury. J Hazard Mater 419:126502. https://doi.org/10.1016/j.jhazmat.2021.126502
Barrs HD, Weatherley PE (1962) A re-examination of the relative turgidity technique for estimating water deficits in leaves. Aust J Biol Sci 15:413–428
Basso AS, Miguez FE, Laird DA, Horton R, Westgate M (2013) Assessing potential of biochar for increasing water-holding capacity of sandy soils. GCB Bioenergy 5(2):132–143. https://doi.org/10.1111/gcbb.12026
Bhatia SK, Palai AK, Kumar A, Bhatia RK, Patel AK, Thakur VK, Yang Y (2021) Trends in renewable energy production employing biomass-based biochar. Bioresour Technol 340:125644. https://doi.org/10.1016/j.biortech.2021.125644
Bian R, Joseph S, Shi W, Li L, Taherymoosavi S, Pan G (2019) Biochar DOM for plant promotion but not residual biochar for metal immobilization depended on pyrolysis temperature. Sci Total Environ 662:571–580. https://doi.org/10.1016/j.scitotenv.2019.01.224
Biederman LA, Harpole WS (2013) Biochar and its effects on plant productivity and nutrient cycling: a meta-analysis. GCB Bioenergy 5(2):202–214. https://doi.org/10.1111/gcbb.12037
Blanco-Canqui H (2017) Biochar and soil physical properties. Soil Sci Soc Am J 81:687–711. https://doi.org/10.2136/sssaj2017.01.0017
Chandra S, Bhattacharya J (2019) Influence of temperature and duration of pyrolysis on the property heterogeneity of rice straw biochar and optimization of pyrolysis conditions for its application in soils. J Clean Prod 215:1123–1139. https://doi.org/10.1016/j.jclepro.2019.01.079
Cheng H, Jones DL, Hill P, Bastami MS, Tu C (2017) Influence of biochar produced from different pyrolysis temperature on nutrient retention and leaching. Arch Agron Soil Sci 64:850–859. https://doi.org/10.1080/03650340.2017.1384545
Chitwood DJ (2003) Research on plant-parasitic nematode biology conducted by the United States department of agriculture-agricultural research service. Pest Manag Sci 59(6–7):748–753. https://doi.org/10.1002/ps.684
Csutoras C, Bakos-Barczi N, Burkus B (2022) Medium chain fatty acids and fatty acid esters as potential markers of alcoholic fermentation of white wines. Acta Aliment 51(1):33–42. https://doi.org/10.1556/066.2021.00129
Cui Q, Xia J, Yang H, Liu J, Shao P (2021) Biochar and effective microorganisms promote Sesbania cannabina growth and soil quality in the coastal saline-alkali soil of the Yellow River Delta. China. Sci Total Environ 756:143801. https://doi.org/10.1016/j.scitotenv.2020.143801
Díaz-Manzano FE, Olmo R, Cabrera J, Barcala M, Escobar C, Fenoll C (2016) Long-term in vitro system for maintenance and amplification of root-knot nematodes in Cucumis sativus roots. Front Plant Sci 7:124. https://doi.org/10.3389/fpls.2016.00124
Directive 2009/128/EC of the European Parliament and of the Council of 21 October 2009 establishing a framework for community action to achieve the sustainable use of pesticides. OJ L 309, pp 71–86. https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32009L0128. Accessed 24 Nov 2009
Dong CD, Tsai ML, Chen CW, Hung CM (2018) Remediation and cytotoxicity study of polycyclic aromatic hydrocarbon-contaminated marine sediments using synthesized iron oxide–carbon composite. Environ Sci Pollut Res Int 25(6):5243–5253. https://doi.org/10.1007/S11356-017-9354-1/FIGURES/11
Dong CD, Lung SCC, Chen CW, Lee JS, Chen YC, Wang WCV, Chen CJ, Hung CM, Lin CH (2019) Assessment of the pulmonary toxic potential of nano-tobacco stem-pyrolyzed biochars. Environ Sci Nano 6:1527–1535. https://doi.org/10.1039/C8EN00968F
Downie A, Crosky A, Munroe P (2009) Physical properties of biochar. In: Lehmann J, Joseph J (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 13–33
Du YD, Zhang XQ, Shu L, Feng Y, Lv C, Liu HQ, Xu F, Wang Q, Zhao CC, Kong Q (2021) Safety evaluation and ibuprofen removal via an Alternanthera philoxeroides-based biochar. Environ Sci Pollut Res Int 28(30):40568–40586. https://doi.org/10.1007/S11356-020-09714-Z
Dubinin MM, Astakhov VA (1971) Development of the concepts of volume filling of micropores in the adsorption of gases and vapors by microporous adsorbents. Russ Chem Bull 20:8–12. https://doi.org/10.1007/BF00849307
Eche CO, Okafor OE (2020) Control potential of some indigenous biochars against Meloidogyne incognita [(Kofoid and White) Chitwood] in tomato (Solanum lycopersicum L.). J Entomol Nematol 12(2):32–38. https://doi.org/10.5897/JEN2020.0251
Eizenberg H, Plakhine D, Ziadne H, Tsechansky L, Graber ER (2017) Non-chemical control of root parasitic weeds with biochar. Front Plant Sci 8:939. https://doi.org/10.3389/fpls.2017.00939
El-Bassi L, Azzaz AA, Jellali S, Akrout H, Marks EAN, Ghimbeu CM, Jeguirim M (2021) Application of olive mill waste-based biochars in agriculture: impact on soil properties, enzymatic activities and tomato growth. Sci Total Environ 755(Pt 1):142531. https://doi.org/10.1016/J.SCITOTENV.2020.142531
Enders A, Hanley K, Whitman T, Joseph S, Lehmann J (2012) Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresour Technol 114:644–653. https://doi.org/10.1016/j.biortech.2012.03.022
Escobar C, Barcala M, Cabrera J, Fenoll C (2015) Overview of root-knot nematodes and giant cells. In: Escobar C, Fenoll C (eds) Advances in botanical research. Academic Press Inc., London, pp 1–32. https://doi.org/10.1016/bs.abr.2015.01.001
Fahad S, Hussain S, Saud S, Hassan S, Tanveer M, Ihsan MZ, Shah AN, Ullah A, Nasrullah KF, Ullah S, Alharby H, Nasim W, Wu C, Huang J (2016) A combined application of biochar and phosphorus alleviates heat-induced adversities on physiological, agronomical and quality attributes of rice. Plant Physiol Biochem 103:191–198. https://doi.org/10.1016/j.plaphy.2016.03.001
French E, Iyer-Pascuzzi AS (2018) A role for the gibberellin pathway in biochar-mediated growth promotion. Sci Rep 8(1):1–10. https://doi.org/10.1038/s41598-018-23677-9
Gai X, Wang H, Liu J, Zhai L, Liu S, Ren T, Liu H (2014) Effects of feedstock and pyrolysis temperature on biochar adsorption of ammonium and nitrate. PLoS ONE 9:e113888. https://doi.org/10.1371/journal.pone.0113888
Gale TK, Fletcher TH, Bartholomew CH (1995) Effects of pyrolysis conditions on internal surface areas and densities of coal chars prepared at high heating rates in reactive and nonreactive atmospheres. Energy Fuels 9(3):513–524. https://doi.org/10.1021/ef00051a017
Gallo-saravia M, Lugo-sierra L, Barrera-zapata R (2018) Evaluación de biochar como alternativa de sustrato en cultivos de tomate. Scientia Et Technica 23(2):297–303. https://doi.org/10.22517/23447214.17691
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50(1):151–158. https://doi.org/10.1016/0014-4827(68)90403-5
Gao S, Doll DA, Stanghellini MS, Westerdahl BB, Wang D, Hanso NBD (2018) Deep injection and the potential of biochar to reduce fumigant emissions and effects on nematode control. J Environ Manag 223:469–477. https://doi.org/10.1016/j.jenvman.2018.06.031
George C, Kohler J, Rillig MC (2016) Biochars reduce infection rates of the root-lesion nematode Pratylenchus penetrans and associated biomass loss in carrot. Soil Biol Biochem 95:11–18. https://doi.org/10.1016/j.soilbio.2015.12.003
Gheorghe C, Marculescu C, Badea A, Dinca C, Apostol T (2009) Effect of pyrolysis conditions on bio-char production from biomass. In: 3rd WSEAS Int Conf on renewable energy sources, pp 239–41
Ginebra M, Muñoz C, Calvelo-Pereira R, Doussoulin M, Zagal E (2022) Biochar impacts on soil chemical properties, greenhouse gas emissions and forage productivity: a field experiment. Sci Total Environ 806(Pt 2):150465. https://doi.org/10.1016/j.scitotenv.2021.150465
Graber ER, Harel YM, Kolton M, Cytryn E, Silber A, David DR, Tsechansky L, Borenshtein M, Elad Y (2010) Biochar impact on development and productivity of pepper and tomato grown in fertigated soilless media. Plant Soil 337(1):481–496. https://doi.org/10.1007/S11104-010-0544-6/TABLES/6
Gu Y, Hou Y, Huang D, Hao Z, Wang X, Wei Z, Jousset A, Tan S, Xu D, Shen O, Xu Y, Fridman VP (2017) Application of biochar reduces Ralstonia solanacearum infection via effects on pathogen chemotaxis, swarming motility, and root exudate adsorption. Plant Soil 415:269–281. https://doi.org/10.1007/s11104-016-3159-8
Guo L, Yu H, Kharbach M, Zhang W, Wang J, Niu W (2021) Biochar improves soil-tomato plant, tomato production, and economic benefits under reduced nitrogen application in Northwestern China. Plants 10(4):759. https://doi.org/10.3390/PLANTS10040759
Gupta GK, Gupta PK, Mondal MK (2019) Experimental process parameters optimization and in-depth product characterizations for teak sawdust pyrolysis. Waste Manag 87:499–511. https://doi.org/10.1016/J.WASMAN.2019.02.035
Hansen V, Müller-Stöver D, Imparato V, Krogh PH, Jensen LS, Dolmer A, Hauggaard-Nielsen H (2017) The effects of straw or straw-derived gasification biochar applications on soil quality and crop productivity: a farm case study. J Environ Manag 186(Pt 1):88–95. https://doi.org/10.1016/j.jenvman.2016.10.041
He X, Xie H, Gao D, Khashi U, Rahman M, Zhou X, Wu F (2021) Biochar and intercropping with potato-onion enhanced the growth and yield advantages of tomato by regulating the soil properties, nutrient uptake, and soil microbial community. Front Microbiol 12:695447. https://doi.org/10.3389/FMICB.2021.695447
Hoffmann V, Jung D, Zimmermann J, Rodriguez Correa C, Elleuch A, Halouani K, Kruse A (2019) Conductive carbon materials from the hydrothermal carbonization of vineyard residues for the application in electrochemical double-layer capacitors (EDLCs) and direct carbon fuel cells (DCFCs). Materials 12(10):1703. https://doi.org/10.3390/MA12101703
Huang WK, Ji HL, Gheysen G, Debode J, Kyndt T (2015) Biochar-amended potting medium reduces the susceptibility of rice to root-knot nematode infections. BMC Plant Biol 15(1):267. https://doi.org/10.1186/s12870-015-0654-7
Huang XF, Li SQ, Li SY, Ye GY, Lu LJ, Zhang L, Yang LY, Qian X, Liu J (2019) The effects of biochar and dredged sediments on soil structure and fertility promote the growth, photosynthetic and rhizosphere microbial diversity of Phragmites communis (Cav.) Trin. ex Steud. Sci Total Environ 697:134073. https://doi.org/10.1016/j.scitotenv.2019.134073
Hung CM, Chen CW, Huang CP, Tsai ML, Wu CH, Lin YL, Cheng YR, Dong CD (2022) Efficacy and cytotoxicity of engineered ferromanganese-bearing sludge-derived biochar for percarbonate-induced phthalate ester degradation. J Hazard Mater 422:126922. https://doi.org/10.1016/J.JHAZMAT.2021.126922
Hussey RS, Barker KR (1973) A comparison of methods of collecting inocula of Meloidogyne spp., including a new technique. Plant Dis 57:1025–1028
IBM Corp (2016) IBM SPSS statistics for windows, version 24.0. IBM Corp, Armonk
Ibrahim F, Quainoo AK, Kankam F (2019) Effect of shea nut shell biochar on root knot nematodes (Meloidogyne spp.) of tomato (Solanum lycopersicum L.). Annu Res Rev Biol 30:1–7. https://doi.org/10.9734/ARRB/2018/
Jaafar NM, Clode PL, Abbott LK (2015) Soil microbial responses to biochars varying in particle size, surface and pore properties. Pedosphere 25(5):770–780. https://doi.org/10.1016/S1002-0160(15)30058-8
Jaiswal AK, Frenkel O, Elad Y, Lew B, Graber ER (2015) Non-monotonic influence of biochar dose on bean seedling growth and susceptibility to Rhizoctonia solani: the “shifted Rmax-effect.” Plant Soil 395:125–140. https://doi.org/10.1007/s11104-014-2331-2
Jaiswal AK, Elad Y, Cytryn E, Graber ER, Frenkel O (2018) Activating biochar by manipulating the bacterial and fungal microbiome through pre-conditioning. New Phytol 219(1):363–377. https://doi.org/10.1111/nph.15042
Jaiswal AK, Alkan N, Elad Y, Sela N, Philosoph AM, Graber ER, Frenkel O (2020) Molecular insights into biochar-mediated plant growth promotion and systemic resistance in tomato against Fusarium crown and root rot disease. Sci Rep 10(1):1–15. https://doi.org/10.1038/S41598-020-70882-6
Jin Q, Wang Z, Feng Y, Kim YT, Stewart AC, O’Keefe SF, Neilson AP, He Z, Huang H (2020) Grape pomace and its secondary waste management: biochar production for a broad range of lead (Pb) removal from water. Environ Res 186:109442. https://doi.org/10.1016/J.ENVRES.2020.109442
Johnston CT (2017) Biochar analysis by Fourier-transform infra-red spectroscopy. In: Singh B, Camps-Arbestain M, Lehmann J (eds) Biochar: a guide to analytical methods. CRC Press, Boca Raton, pp 199–213
Joseph S, Cowie AL, Van Zwieten L, Bolan N, Budai A, Buss W, Cayuela ML, Graber ER, Ippolito JA, Kuzyakov Y, Luo Y, Ok YS, Palansooriya KN, Shepherd J, Stephens S, Weng ZH, Lehmann J (2021) How biochar works, and when it doesn’t: a review of mechanisms controlling soil and plant responses to biochar. GCB Bioenergy 13(11):1731–1764. https://doi.org/10.1111/gcbb.12885
Kamau S, Karanja NK, Ayuke FO, Lehmann J (2019) Short-term influence of biochar and fertilizer-biochar blends on soil nutrients, fauna and maize growth. Biol Fertil Soils 55:661–673. https://doi.org/10.1007/s00374-019-01381-8
Kavitha B, Reddy PVL, Kim B, Lee SS, Pandey SK, Kim KH (2018) Benefits and limitations of biochar amendment in agricultural soils: a review. J Environ Manag 227:146–154. https://doi.org/10.1016/j.jenvman.2018.08.082
Keiluweit M, Nico PS, Johnson MG, Kleber M (2010) Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ Sci Technol 44(4):1247–1253. https://doi.org/10.1021/es9031419
Kim HS, Kim KR, Yang JE, Ok YS, Owens G, Nehls T, Wessolek G, Kim KH (2016) Effect of biochar on reclaimed tidal land soil properties and maize (Zea mays L.) response. Chemosphere 142:153–159. https://doi.org/10.1016/j.chemosphere.2015.06.041
Kloss S, Zehetner F, Dellantonio A, Hamid R, Ottner F, Liedtke V, Schwanninger M, Gerzabek MH, Soja G (2012) Characterization of slow pyrolysis biochars: effects of feedstocks and pyrolysis temperature on biochar properties. J Environ Qual 41:990–1000. https://doi.org/10.2134/jeq2011.0070
Kokkinomagoulos E, Kandylis P (2020) Sustainable exploitation of by-products of vitivinicultural origin in winemaking. Proceedings 67(1):5. https://doi.org/10.3390/ASEC2020-07521
Kolton M, Graber ER, Tsehansky L, Elad Y, Cytryn E (2017) Biochar-stimulated plant performance is strongly linked to microbial diversity and metabolic potential in the rhizosphere. New Phytol 213:1393–1404. https://doi.org/10.1111/nph.14253
Lago BC, Silva CA, Melo LCA, Morais EG (2021) Predicting biochar cation exchange capacity using Fourier transform infrared spectroscopy combined with partial least square regression. Sci Total Environ 794:148762. https://doi.org/10.1016/j.scitotenv.2021.148762
Lehmann J, Joseph S (2009) Biochar for environmental management: an introduction. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 1–12
Liu B, Cai Z, Zhang Y, Liu G, Luo X, Zheng H (2019) Comparison of efficacies of peanut shell biochar and biochar-based compost on two leafy vegetable productivity in an infertile land. Chemosphere 224:151–161. https://doi.org/10.1016/j.chemosphere.2019.02.100
Lou Y, Joseph S, Li L, Graber ER, Liu X, Pan G (2016) Water extract from straw biochar used for plant growth promotion: an initial test. BioRes 11(1):249–266
Lu Y, Rao S, Huang F, Cai Y, Wang G, Cai K (2016) Effects of biochar amendment on tomato bacterial wilt resistance and soil microbial amount and activity. Int J Agron. https://doi.org/10.1155/2016/2938282
Manolikaki II, Mangolis A, Diamadopoulos E (2016) The impact of biochars prepared from agricultural residues on phosphorus release and availability in two fertile soils. J Environ Manag 181:536–543. https://doi.org/10.1016/J.JENVMAN.2016.07.012
Marra R, Vinale F, Cesarano G, Lombardi N, d’Errico G, Crasto A, Mazzei P, Piccolo A, Incerti G, Woo SL, Scala F, Bonanomi G (2018) Biochars from olive mill waste have contrasting effects on plants, fungi and phytoparasitic nematodes. PLoS ONE 13(6):e0198728. https://doi.org/10.1371/journal.pone.0198728
Martínez-Gómez Á, Poveda J, Escobar C (2022) Overview of the use of biochar from main cereals to stimulate plant growth. Front Plant Sci 13:912264. https://doi.org/10.3389/fpls.2022.912264
Mehari ZH, Elad Y, Rav-David D, Graber ER, Harel YM (2015) Induced systemic resistance in tomato (Solanum lycopersicum) against Botrytis cinerea by biochar amendment involves jasmonic acid signaling. Plant Soil 395:31–44. https://doi.org/10.1007/s11104-015-2445-1
Mehmood S, Ahmed W, Ikram M, Imtiaz M, Mahmood S, Tu S, Chen D (2020) Chitosan modified biochar increases soybean (Glycine max L.) resistance to salt-stress by augmenting root morphology, antioxidant defense mechanisms and the expression of stress-responsive genes. Plants 9(9):1173. https://doi.org/10.3390/plants9091173
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Meth 65:55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Ortega-Villasante C, Hernández LE, Rellán-Álvarez R, Del Campo FF, Carpena-Ruiz RO (2007) Rapid alteration of cellular redox homeostasis upon exposure to cadmium and mercury in alfalfa seedlings. New Phytol 176:96–107. https://doi.org/10.1111/j.1469-8137.2007.02162.x
Pala M, Kantarli IC, Buyukisik HB, Yanik J (2014) Hydrothermal carbonization and torrefaction of grape pomace: a comparative evaluation. Bioresour Technol 161:255–262. https://doi.org/10.1016/J.BIORTECH.2014.03.052
Pietikäinen J, Kiikkilä O, Fritze H (2000) Charcoal as a habitat for microbes and its effect on the microbial community of the underlying humus. Oikos 89:231–242
Poveda J, Martínez-Gómez Á, Fenoll C, Escobar C (2021) The use of biochar for plant pathogen control. Phytopathology 111(9):1490–1499. https://doi.org/10.1094/PHYTO-06-20-0248-RVW
Püntener W (1981) Manual for field trials in plant protection, 2nd edn. Agricultural Division, Ciba-Geigy Limited, Basel
Rahayu DS, Sari NP (2017) Development of Pratylenchus coffeae in biochar applied soil, coffee roots and its effect on plant growth. Pelita Perkebunan 33(1):24–32. https://doi.org/10.22302/iccri.jur.pelitaperkebunan.v33i1.251
Rahman L, Whitelaw-Weckert MA, Orchard B (2014) Impact of organic soil amendments, including poultry litter biochar, on nematodes in a Riverina, NSW Vineyard. Soil Res 52(6):604–619. https://doi.org/10.1071/SR14041
Rajapaksha AU, Vithanage M, Ahmad M, Seo DC, Cho JS, Lee SE, Lee SS, Ok YS (2015) Enhanced sulfamethazine removal by steam-activated invasive plant-derived biochar. J Hazard Mater 290:43–50. https://doi.org/10.1016/j.jhazmat.2015.02.046
Ramzani PMA, Shan L, Anjum S, Khan WU, Ronggui H, Iqbal M, Virk ZA, Kausar S (2017) Improved quinoa growth, physiological response, and seed nutritional quality in three soils having different stresses by the application of acidified biochar and compost. Plant Physiol Bioch 116:127–138. https://doi.org/10.1016/j.plaphy.2017.05.003
Rasool M, Akhter A, Soja G, Haider MS (2021) Role of biochar, compost and plant growth promoting rhizobacteria in the management of tomato early blight disease. Sci Rep 11(1):6092. https://doi.org/10.1038/s41598-021-85633-4
Rogovska N, Laird D, Cruse RM, Trabue S, Heaton E (2012) Germination tests for assessing biochar quality. J Environ Qual 41(4):1014–1022. https://doi.org/10.2134/jeq2011.0103
Ruzickova J, Koval S, Raclavska H, Kucbel M, Svedova B, Raclavsky K, Juchelkova D, Scala F (2021) A comprehensive assessment of potential hazard caused by organic compounds in biochar for agricultural use. J Hazard Mater 403:123644. https://doi.org/10.1016/j.jhazmat.2020.123644
Saito M, Marumoto T (2002) Inoculation with arbuscular mycorrhizal fungi: the status quo in Japan and the future prospects. Plant Soil 244:273–279. https://doi.org/10.1023/A:1020287900415
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9(7):676–682. https://doi.org/10.1038/nmeth.2019
Schnell RW, Vietor DM, Provin TL, Munster CL, Capareda S (2012) Capacity of biochar application to maintain energy crop productivity: soil chemistry, sorghum growth, and runoff water quality effects. J Environ Qual 41(4):1044–1051. https://doi.org/10.2134/jeq2011.0077
Shafizadeh F (1985) Pyrolytic reactions and products of biomass. In: Overend RP, Milne TA, Mudge LK (eds) Fundamentals of thermochemical biomass conversion. Springer, Dordrecht, pp 183–217. https://doi.org/10.1007/978-94-009-4932-4_11
She D, Sun X, Gamareldawla AHD, Nazar EA, Hu W, Edith K, Yu S (2018) Benefits of soil biochar amendments to tomato growth under saline water irrigation. Sci Rep 8(1):14743. https://doi.org/10.1038/s41598-018-33040-7
Song D, Xi X, Zheng Q, Liang G, Zhou W, Wang X (2019) Soil nutrient and microbial activity responses to two years after maize straw biochar application in a calcareous soil. Ecotoxicol Environ Saf 180:348–356. https://doi.org/10.1016/j.ecoenv.2019.04.073
Streubel JD, Collins HP, Garcia-Perez M, Tarara J, Granatstein D, Kruger CE (2011) Influence of contrasting biochar types on five soils at increasing rates of application. Soil Sci Soc Am J 75:1402–1413. https://doi.org/10.2136/sssaj2010.0325
Suo F, You X, Yin S, Wu H, Zhang C, Yu X, Sun R, Li Y (2021) Preparation and characterization of biochar derived from co-pyrolysis of Enteromorpha prolifera and corn straw and its potential as a soil amendment. Sci Total Environ 798:149167. https://doi.org/10.1016/j.scitotenv.2021.149167
Tamelová B, Malaťák J, Velebil J, Gendek A, Aniszewska M (2021) Energy utilization of torrefied residue from wine production. Materials 14(7):1610. https://doi.org/10.3390/ma14071610
Thies JE, Rilling MC (2009) Characteristics of biochar: biological properties. In: Lehmann J, Joseph J (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 85–105
Trakal L, Raya-Moreno I, Mitchell K, Beesley L (2017) Stabilization of metal(loid)s in two contaminated agricultural soils: comparing biochar to its non-pyrolysed source material. Chemosphere 181:150–159. https://doi.org/10.1016/j.chemosphere.2017.04.064
van Dijk M, Morley T, Rau ML, Saghai Y (2021) A meta-analysis of projected global food demand and population at risk of hunger for the period 2010–2050. Nat Food 2:494–501. https://doi.org/10.1038/s43016-021-00322-9
Velli P, Manolikaki I, Diamadopoulos E (2021) Effect of biochar produced from sewage sludge on tomato (Solanum lycopersicum L.) growth, soil chemical properties and heavy metal concentrations. J Environ Manag 297:113325. https://doi.org/10.1016/j.jenvman.2021.113325
Verdejo-Lucas S, Cortada L, Sorribas F, Ornat C (2009) Selection of virulent populations of Meloidogyne javanica by repeated cultivation of Mi resistance gene tomato rootstocks under field condition. Plant Pathol 58:990–998. https://doi.org/10.1111/j.1365-3059.2009.02089.x
Visioli G, Conti FD, Menta C, Bandiera M, Malcevschi A, Jones DL, Vamerali T (2016) Assessing biochar ecotoxicology for soil amendment by root phytotoxicity bioassays. Environ Monit Assess 188(3):166. https://doi.org/10.1007/s10661-016-5173-y
Wallis CM (2022) Microplate bioassay to examine the effects of grapevine-isolated stilbenoids on survival of root knot nematodes. BMC Res Notes 15:220. https://doi.org/10.1186/s13104-022-06106-z
Wang D, Hao S, Wang Q, Xue J (2006) Anatomy of the Late Devonian sphenopsid Rotafolia songziensis, with a discussion of stelar architecture of the sphenophyllales. Int J Plant Sci 167(2):373–383. https://doi.org/10.1086/499115
Wang C, Bruening G, Williamson VM (2009) Determination of preferred pH for root-knot nematode aggregation using pluronic F-127 gel. J Chem Ecol 35(10):1242–1251. https://doi.org/10.1007/s10886-009-9703-8
Warnock DD, Lehmann J, Kuyper TW, Rillig MC (2007) Mycorrhizal responses to biochar in soil—concepts and mechanisms. Plant Soil 300:9–20. https://doi.org/10.1007/s11104-007-9391-5
Windeatt JH, Ross AB, Williams PT, Forster PM, Nahil MA, Singh S (2014) Characteristics of biochars from crop residues: potential for carbon sequestration and soil amendment. J Environ Manag 146:189–197. https://doi.org/10.1016/j.jenvman.2014.08.003
Wornat MJ, Hurt RH, Yang NYC, Headley TJ (1995) Structural and compositional transformations of biomass chars during combustion. Combust Flame 100:131–143. https://doi.org/10.1016/0010-2180(94)00055-W
Wu W, Yang M, Feng Q, McGrouther K, Wang H, Lu H, Chen Y (2012) Chemical characterization of rice straw-derived biochar for soil amendment. Biomass Bioenergy 47:268–276. https://doi.org/10.1016/j.biombioe.2012.09.034
Xi J, Li H, Xi J, Tan S, Zheng J, Tan Z (2020) Effect of returning biochar from different pyrolysis temperatures and atmospheres on the growth of leaf-used lettuce. Environ Sci Pollut Res Int 27(28):35802–35813. https://doi.org/10.1007/s11356-020-09840-8
Xu M, Gao P, Yang Z, Su L, Wu J, Yang G, Zhang X, Ma J, Peng H, Xiao Y (2019) Biochar impacts on phosphorus cycling in rice ecosystem. Chemosphere 225:311–319. https://doi.org/10.1016/j.chemosphere.2019.03.069
Xu S, Chen J, Peng H, Leng S, Li H, Qu W, Hu Y, Li H, Jiang S, Zhou W, Leng L (2021) Effect of biomass type and pyrolysis temperature on nitrogen in biochar, and the comparison with hydrochar. Fuel 291:120128. https://doi.org/10.1016/j.fuel.2021.120128
Yakout SM (2017) Physicochemical characteristics of biochar produced from rice straw at different pyrolysis temperature for soil amendment and removal of organics. Proc Natl Acad Sci 87:207–214. https://doi.org/10.1007/s40010-017-0343-z
Yargicoglu EN, Reddy KR (2014) Evaluation of PAH and metal contents of different biochars for use in climate change mitigation systems. In: Proceedings of international conference on sustainable infrastructure (ICSI), pp 114–125. https://doi.org/10.1061/9780784478745.011
You X, Yin S, Suo F, Xu Z, Chu D, Kong Q, Zhang C, Li Y, Liu L (2021) Biochar and fertilizer improved the growth and quality of the ice plant (Mesembryanthemum crystallinum L.) shoots in a coastal soil of Yellow River Delta, China. Sci Total Environ 775:144893. https://doi.org/10.1016/j.scitotenv.2020.144893
Zhang XK, Li Q, Liang WJ, Zhang M, Bao XL, Xie ZB (2013) Soil nematode response to biochar addition in a Chinese wheat field. Pedosphere 23(1):98–103. https://doi.org/10.1016/S1002-0160(12)60084-8
Zhao F, Zou G, Shan Y, Ding Z, Dai M, He Z (2019) Coconut shell derived biochar to enhance water spinach (Ipomoea aquatica Forsk) growth and decrease nitrogen loss under tropical conditions. Sci Rep 9(1):20291. https://doi.org/10.1038/s41598-019-56663-w
Zheng H, Wang X, Chen L, Wang Z, Xia Y, Zhang Y, Wang H, Luo X, Xing B (2018) Enhanced growth of halophyte plants in biochar-amended coastal soil: roles of nutrient availability and rhizosphere microbial modulation. Plant Cell Environ 41(3):517–532. https://doi.org/10.1111/pce.12944
Acknowledgements
We thank Bodega S.A.T Coloman (Pedro Muñoz, Ciudad Real, Spain) for their support and kindly donation of the grape pomace used for the study. We also thank the support received from UCLM intramural funds and to EU FEDER for new equipment and maintenance that complemented all the grants. We also thank Fundación Tatiana Pérez de Guzmán el Bueno, as AM-G was recipient of a PhD grant. The FTIR experiments were performed at MIRAS beamline at ALBA Synchrotron with the collaboration of ALBA staff and supported by travel grants for experiment no. 2017092364. We thank Javier Cabrera for his support and help from the begining of this project. We also thank for the useful technical help to Esperanza Salvador and Isidoro Poveda from the SEM-EDX laboratory, SIDI, UAM, Madrid, Spain.
Funding
This work was supported by the Spanish Government (PID2019-105924RB-I00 MCIN/AEI/10.13039/501100011033; RED2018-102407-T) to CE, and (AGL2014-53771-R; AGL2017-87591-R) to LEH, and by the Castilla-La Mancha Government (SBPLY/17/180501/000287 and SBPLY/21/180501/000033) to CE. AM-G was recipient of a PhD grant from Fundación Tatiana Pérez de Guzmán el Bueno. EU FEDER funds complemented all the grants.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. AM-G performed most of the experiments and participated actively in the writing of the manuscript. FED-M assisted in most of the experiments performed. MFA and AG-C participated in the reproduction assays of M. javanica and the nematicidal assays. OG-T performed the cytotoxicity assays. AB and IY participated in the SR-FTIR analysis. IM and ED produced the biochar and participated in the analysis of its physicochemical properties and elemental analysis. LEH participated in the experimental design, SR-FTIR, ICP-MS analysis, and in the critical writing and reading of the manuscript. CE designed and supervised the experiments and contributed to the writing and critical reading of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no competing interests to disclose, financial or otherwise.
Additional information
Handling Editor: Hailong Wang.
Supplementary Information
Additional file 1: Table S1.
pH values of different dilutions of the aqueous extracts (BC350AE and BC700AE) obtained from washing BC350 and BC700 (3% (w/v) with distilled water), respectively, for 24 h. Figure S1. SR-FTIR spectroscopy of grape pomace (GP) and biochars prepared at 350 °C and 700 °C (BC350, BC700, respectively). Full 2nd derivative spectra (wavenumber range between 3100 and 900 cm−1) along with the first two main principal components (PC-1 and PC-2) for different wavenumber ranges, which explain the largest contributions of signal shifts between samples. Numbers in red indicate the wavenumber values of the most representative peaks. Figure S2. SR-FTIR spectroscopy of grape pomace (GP), washed (BC350W) and unwashed (BC350) biochar prepared at 350 °C, and BC350 aqueous extract (BC350AE). Full 2nd derivative spectra (wavenumber range between 3100 and 900 cm−1) along with the first two main principal components (PC-1 and PC-2) for different wavenumber ranges, which explain the largest contributions of signal shifts between samples. Numbers in red indicate the wavenumber values of the most representative peaks. Figure S3. SR-FTIR spectroscopy of grape pomace (GP), washed (BC700W) and unwashed (BC700) biochar prepared at 700 °C, and BC700 aqueous extract (BC700AE). Full 2nd derivative spectra (wavenumber range between 3100 and 900 cm−1) and along with the first two main principal components (PC-1 and PC-2) for different wavenumber ranges, which explain the largest contributions of signal shifts between samples. Numbers in red indicate the wavenumber values of the most representative peaks. Figure S4. Effect of different concentrations (3%, 1.5% and 0.75%) of the unwashed biochar obtained at 700 °C (BC700) on 3-week old tomato plants 24 h and 6 days after its transplantation into biochar-amended sandy soils. Figure S5. Effect of different concentrations (3%, 1.5% and 0.75%) of the unwashed biochar obtained at 350 °C (BC350) on roots and shoots of tomato plants 3 weeks after its transplantation into biochar-amended sandy soils. Figure S6. Effect of different concentrations of BC350W on the percentage of seed germination (a), number of leaves (b), shoot length (c), relative water content (RCW) (d) and total fresh biomass (f) of 6-week-old tomato plants. Means ± standard errors (n ≥ 8). Significant differences with respect to the control according to Mann–Whitney U test. ***p < 0.001; **p < 0.01.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martínez-Gómez, Á., Andrés, M.F., Barón-Sola, Á. et al. Biochar from grape pomace, a waste of vitivinicultural origin, is effective for root-knot nematode control. Biochar 5, 30 (2023). https://doi.org/10.1007/s42773-023-00228-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42773-023-00228-8