Abstract
Aims
Biochar affects the progress of plant diseases caused by soilborne pathogens, frequently featuring U-shaped biochar dose/disease response curves. This study tested this phenomenon in common bean (Phaseolus vulgaris L.) with several biochars.
Methods
Four biochars prepared from two feedstocks (eucalyptus wood and greenhouse wastes) each at 350 and 600 °C were tested on bean seedling growth and infection caused by Rhizoctonia solani at concentrations of 0–3 % by weight. Biochar direct toxicity to R. solani was quantified in vitro.
Results
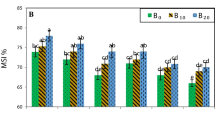
In general, lower concentrations (≤1 %) of biochar suppressed damping-off, whereas higher concentrations (3 %) were ineffective at disease protection. Plant growth in the absence of the pathogen was generally improved at all doses by the four biochars. Maximum growth response (G-Rmax) generally occurred at higher biochar doses than maximum disease reduction (D-Rmax). Direct toxicity to the pathogen could not explain disease reduction.
Conclusion
Inverted U-shaped biochar dose/plant growth and biochar dose/disease reduction curves are emerging as common patterns in biochar/crop/pathogen systems. Frequently, the inflection between growth promotion and suppression occurs at different doses than the inflection between disease suppression and promotion. We term this the “Shifted Rmax-Effect”. As there is no simple rule-of-thumb for crop/soil/biochar/dose/pathogen combinations, the possible effects of biochar on plant pathogens should not be overlooked.








Similar content being viewed by others
Abbreviations
- AUMPC:
-
Area under mortality progress curve
- CFU:
-
Colony forming units
- EC50:
-
Effective concentration for 50 % growth inhibition
- EUC:
-
Eucalyptus wood chips
- GC/MS:
-
Gas chromatography - mass spectrometry
- GHW:
-
Greenhouse pepper plant wastes
- HTT:
-
Highest Treatment Temperature
- RT:
-
Retention time
References
Ahrens WH, Cox DJ, Budhwar G (1990) Use of the arcsine and square root transformations for subjectively determined percentage data. Weed Sci 38:452–458
Baronti S, Alberti G, Delle Vedove G, Di Gennaro F, Fellet G, Genesio L, Miglietta F, Peressotti A, Vaccari FP (2010) The biochar option to improve plant yields: first results from some field and pot experiments in Italy. Ital J Agron 5:3–12
Bell DK, Sumner DR (1987) Survival of Rhizoctonia solani and other soilborne basidiomycetes in fallow soil. Plant Dis 71:911–915
Bennett WF (1993) Nutrient deficiencies and toxicities in crop plants. American Phytopathological Society Press, St. Paul
Bonanomi G, Antignani V, Pane C, Scala E (2007) Suppression of soilborne fungal diseases with organic amendments. J Plant Pathol 89:311–324
Brady N, Weil R (1999) The nature and properties of soil Prentice-Hall Inc. Upper Saddle River, New Jersey
Calabrese EJ, Blain RB (2009) Hormesis and plant biology. Environ Pollut 157:42–48
Cedergreen N, Felby C, Porter JR, Streibig JC (2009) Chemical stress can increase crop yield. Field Crop Res 114:54–57
Coleman R, Yang V, Woodward B, Lebow P, Clausen C (2010) Efficacy of fatty acid chemistry: candidate mold and decay fungicides. In: Proceedings One Hundred Sixth Annual Meeting of the American Wood Protection Association, Hyatt Regency Riverfront, Savannah, Georgia, May 23
Elad Y, David DR, Meller Harel Y, Borenshtein M, Ben Kalifa H, Silber A, Graber ER (2010) Induction of systemic resistance in plants by biochar, a soil-applied carbon sequestering agent. Phytopathology 100:913–921
Elad Y, Cytryn E, Meller Harel Y, Lew B, Graber ER (2011) The biochar effect: plant resistance to biotic stresses. Phytopathol Mediterr 50:335–349
Elmer WH, Pignatello JJ (2011) Effect of biochar amendments on mycorrhizal associations and Fusarium crown and root rot of asparagus in replant soils. Plant Dis 95:960–966
Finney DJ (1952) Probit analysis. Cambridge University Press, New York
Gaskin JW, Speir RA, Harris K, Das KC, Lee RD, Morris LA, Fisher DS (2010) Effect of peanut hull and pine chip biochar on soil nutrients, corn nutrient status, and yield. Agron J 102:623–633
Graber ER, Elad Y (2013) Biochar impact on plant resistance to disease. In: Ladygina N, Rineau F (eds) Biochar and soil biota. CRC Press (Taylor and Francis Group), Boca Raton
Graber ER, Tsechansky L, Khanukov J, Oka Y (2011) Sorption, volatilization, and efficacy of the fumigant 1, 3-dichloropropene in a biochar-amended soil. Soil Sci Soc Am J 75:1365–1373
Graber ER, Tsechansky L, Gerstl Z, Lew B (2012) High surface area biochar negatively impacts herbicide efficacy. Plant Soil 353:95–106
Graber ER, Frenkel O, Jaiswal AK, Elad Y (2014a) How may biochar influence severity of diseases caused by soilborne pathogens? Carbon Manag 5:169–183
Graber ER, Tsechansky L, Lew B, Cohen E (2014b) Reducing capacity of water extracts of biochars and their solubilization of soil Mn and Fe. Eur J Soil Sci 65:162–172
Herr LJ (1987) Populations of Rhizoctonia solani in soil under crops in rotation with sugar beet. Ann Appl Biol 110:17–24
Jaiswal AK, Elad Y, Graber ER, Frenkel O (2014) Rhizoctonia solani suppression and plant growth promotion in cucumber as affected by biochar pyrolysis temperature, feedstock and concentration. Soil Biol Biochem 69:110–118
Kloss S, Zehetner F, Dellantonio A, Hamid R, Ottner F, Liedtke V, Schwanninger M, Gerzabek MH, Soja G (2012) Characterization of slow pyrolysis biochars: effects of feedstocks and pyrolysis temperature on biochar properties. J Environ Qual 41:990–1000
Ko WH, Hora FK (1971) A selective medium for the quantitative determination of Rhizoctonia solani in soil. Phytopathology 61:707–710
Lehmann J (2007) A handful of carbon. Nature 447:143–144
Liu DL, An M, Johnson IR, Lovett JV (2003) Mathematical modeling of allelopathy. III. A model for curve-fitting allelochemical dose responses. Nonlinearity Biol Toxicol Med 1:37–50
Massad TJ, Dyer LA, Vega GC (2012) Costs of defense and a test of the carbon-nutrient balance and growth-differentiation balance hypotheses for two co-occurring classes of plant defense. Plos One 7. doi: e47554 10.1371/journal.pone.0047554
Matsubara Y, Hasegawa N, Fukui H (2002) Incidence of Fusarium root rot in asparagus seedlings infected with arbuscular mycorrhizal fungus as affected by several soil amendments. J Jpn Soc Hortic Sci 71:370–374
Mehari ZH, Meller Harel Y, Rav-David D, Graber ER, Elad Y (2013) The nature of systemic resistance induced in tomato (Solanum lycopersicum) by biochar soil treatments. IOBC/WPRS Bullet 89:227–230
Meller Harel Y, Elad Y, Rav-David D, Borenstein M, Shulchani R, Lew B, Graber ER (2012) Biochar mediates systemic response of strawberry to foliar fungal pathogens. Plant Soil 357:245–257
Nerome M, Toyota K, Islam T, Nishijima T, Matsuoka T, Sato K, Yamaguchi Y (2005) Suppression of bacterial wilt of tomato by incorporation of municipal biowaste charcoal into soil. Soil Microorg 59:9–14
Patrick Z, Toussoun T (1965) Plant residues and organic amendments in relation to biological control. In: Baker KF, Snyder WC (eds) Ecology of soil-borne plant pathogens: prelude to biological control. University of California Press, Berkeley
Pierik R, Tholen D, Poorter H, Visser EJW, Voesenek L (2006) The Janus face of ethylene: growth inhibition and stimulation. Trends Plant Sci 11:176–183
Rajkovich S, Enders A, Hanley K, Hyland C, Zimmerman AR, Lehmann J (2012) Corn growth and nitrogen nutrition after additions of biochars with varying properties to a temperate soil. Biol Fertil Soils 48:271–284
Rondon MA, Lehmann J, Ramirez J, Hurtado M (2007) Biological nitrogen fixation by common beans (Phaseolus vulgaris L.) increases with bio-char additions. Biol Fertil Soils 43:669–708
Schroeder KL, Paulitz TC (2008) Effect of inoculum density and soil tillage on the development and severity of Rhizoctonia root rot. Phytopathology 98:304–314
Spokas KA (2010) Review of the stability of biochar in soils: predictability of O:C molar ratios. Carbon Manage 1:289–303
Spokas KA, Cantrell KB, Novak JM, Archer DW, Ippolito JA, Collins HP, Boateng AA, Lima IM, Lamb MC, McAloon AJ, Lentz RD, Nichols KA (2012) Biochar: a synthesis of its agronomic impact beyond carbon sequestration. J Environ Qual 41:973–989
Sun Y, Gao B, Yao Y, Fang J, Zhang M, Zhou Y, Chen H, Yang L (2014) Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem Eng J 240:574–578
Van Zwieten L, Kimber S, Morris S, Chan KY, Downie A, Rust J, Joseph S, Cowie A (2010) Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 327:235–246
Velini ED, Alves E, Godoy MC, Meschede DK, Souza RT, Duke SO (2008) Glyphosate applied at low doses can stimulate plant growth. Pest Manag Sci 64:489–496
Viger M, Hancock RD, Miglietta F, Taylor G (2014) More plant growth but less plant defence? First global gene expression data for plants grown in soil amended with biochar. GCB Bioenergy. In Press DOI: 10.1111/gcbb.12182
Ye SF, Yu JQ, Peng YH, Zheng JH, Zou LY (2004) Incidence of Fusarium wilt in Cucumis sativus L. is promoted by cinnamic acid, an autotoxin in root exudates. Plant Soil 263:143–150
Zwart DC, Kim SH (2012) Biochar amendment increases resistance to stem lesions caused by Phytophthora spp. in tree seedlings. Hortscience 47:1736–1740
Acknowledgments
We thank Indira Paudel, Dalia Rav David, Prof. Dani Shtienberg, Dr. Yephet Ben-Yephet, Menahem Borenshtein, Ludmilla Tsechansky, and Ran Shulhani for their help in the experiments. We would also like to thank three anonymous reviewers for their valuable advice. The research was funded by The Chief Scientist of the Ministry of Agriculture and Rural Development, Israel. Project no: 1321653. Contribution from the Agricultural Research Organization, The Volcani Center, Bet Dagan, Israel, No: 546/14
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Johannes Lehmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 42 kb)
Rights and permissions
About this article
Cite this article
Jaiswal, A.K., Frenkel, O., Elad, Y. et al. Non-monotonic influence of biochar dose on bean seedling growth and susceptibility to Rhizoctonia solani: the “Shifted Rmax-Effect”. Plant Soil 395, 125–140 (2015). https://doi.org/10.1007/s11104-014-2331-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-014-2331-2