Abstract
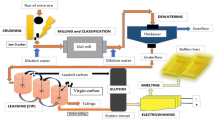
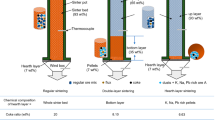
The low-grade nickel laterite ore used in this study was successfully upgraded through selective reduction by industrialized rotary kilns followed by magnetic separation. The results indicated that both iron metallization extent and nickel metallization extent showed obvious linear correlation. In addition, rational roasting temperature and prolonged grinding time could enhance the overall fineness level of reduced laterite nickel ore. The moderate roasting temperature at a range of 1100–1150 °C should be suitable enough to a conversion of total nickel to metallic nickel and partly inhibited the conversion of total iron. Ultimately, the obtained ferronickel alloy with a nickel content of 7.32% and an iron content of 84.15% was prepared successfully from the low-grade nickel laterite ore containing a nickel content of 1.92%. Nickel and iron recoveries were 96.72 and 89.08% respectively, which achieved the highly efficient recovery and utilization of iron and nickel from low-grade laterite nickel ore.














Similar content being viewed by others
References
Yan LY, Wang AJ (2016) Analysis of global nickel supply and demand patterns. China Min Mag 25:1–5
Li JH, Li YY, Zheng S (2015) Research review of laterite nickel ore metallurgy. Nonferrous Metals Science and Engineering 6:35–40
Yu L (2013) Development situation of Russian sulfide nickel ore resources. World Nonferrous Metal 3:60–62
Wang ZH, Chu MS, Liu ZG, Wang H, Zhao W, Gao LH (2017) Preparing ferro-nickel alloy from low-grade laterite nickel ore based on metallized reduction-magnetic separation. Metals 7(313):1–2
Rao MJ, Li GH, Jiang T, Luo J, Zhang YB, Fan XH (2013) Carbothermic reduction of nickeliferous laterite ores for nickel pig iron production in China: a review. JOM 65(11):1573–1580
Quast K, Connor JN, Skinner W, Robinson DJ, Addai-Mensah J (2015) Preconcentration strategies in the processing of nickel laterite ores part 1: literature review. Miner Eng 79:261–268
Elliott R, Pickles CA, Peacey J (2017) Ferronickel particle formation during the carbothermic reduction of a limonitic laterite ore. Miner Eng 100:166–176
Li B, Wang H, Wei YG, Hu JH (2011b) Physicochemical and mechanical properties of nickel laterite ore powder. Adv Mater Res 183-185:1741–1745
Li Y, Li S, Han Y (2011a) Deep reduction/magnetic separation of laterite for concentration of Ni and Fe. J Northeast Univ (Natural Science) 32:740–744
Mudd GM (2010) Global trends and environmental issues in nickel mining: sulfides versus laterites. Ore Geol Rev 38:9–26
Zhu DQ, Cui Y, Vining K, Hapugoda S, Douglas J, Pan J, Zheng GL (2012) Upgrading low nickel content laterite ores using selective reduction followed by magnetic separation. Int J Miner Process 106-109:1–7
Tang X, Liu R, Yao L, Ji Z, Zhang Y, Li S (2014) Ferronickel enrichment by fine particle reduction and magnetic separation from nickel laterite ore. Int J Miner Metall Mater 21:955–961
Pickles CA (2004) Microwave heating behaviour of nickeliferous limonitic laterite ores. Miner Eng 17:775–784
Lee HY, Kim SG, Oh JK (2005) Electrochemical leaching of nickel from low-grade laterites. Hydrometallurgy 77:263–268
Loveday BK (2008) The use of oxygen in high pressure acid leaching of nickel laterites. Miner Eng 21:533–538
Moskalyk RR, Alfantazi AM (2002) Nickel laterite processing and electrowinning practice. Miner Eng 15:593–605
Swamy YV, Kar BB, Mohanty JK (2003) Physico-chemical characterization and sulphatization roasting of low-grade nickeliferous laterites. Hydrometallurgy 69:89–98
Guo XY, Li D, Kyung HP, Tian QH, Wu Z (2009) Leaching behavior of metals from a limonitic nickel laterite using a sulfation-roasting-leaching process. Hydrometallurgy 99:144–150
Pickles CA, Forster J, Elliott R (2014) Thermodynamic analysis of the carbothermic reduction roasting of a nickeliferous limonitic laterite ore. Miner Eng 65:33–40
Jungah K, Gjergj D, Hideaki T, Katsunori O, Seiji M, Toyohisa F (2010) Calcination of low-grade laterite for concentration of Ni by magnetic separation. Miner Eng 23:282–288
Zhai YC, Mu WN, Liu Y, Xu Q (2010) A green process for recovering nickel from nickeliferous laterite ores. Trans Nonferrous Met Soc Chin 20:65–70
Harris CT, Peacey JG, Pickles CA (2013) Selective sulphidation and flotation of nickel from a nickeliferous laterite ore. Miner Eng 54:21–31
Li G, Shi T, Rao M, Jiang T, Zhang Y (2012) Beneficiation of nickeliferous laterite by reduction roasting in the presence of sodium sulfate. Miner Eng 32:19–26
Jiang M, Sun T, Liu Z, Kou J, Liu N, Zhang S (2013) Mechanism of sodium sulfate in promoting selective reduction of nickel laterite ore during reduction roasting process. Int J Miner Process 123:32–38
Pickles CA, Elliott R (2015) Thermodynamic analysis of selective reduction of nickeliferous limonitic laterite ore by carbon monoxide. Miner Process Ext Metall 124(4):208–216
Landers M, Grafe M, Gilkes R, Saunders M, Wells M (2011) Nickel distribution and speciation in rapidly dehydroxylated goethite in oxide-type lateritic nickel ores: XAS and TEM spectroscopic (EELS and EFTEM) investigation Aust. J Earth Sci 58:745–765
Lu J, Liu SJ, Du WG, Pan F, Yang S (2013) The effect of sodium sulphate on the hydrogen reduction process of nickel laterite ore. Miner Eng 49:153–154
Liu SH, Li QL, Song JW (2018) Study on the grinding kinetics of copper tailing powder. Powder Technol 330:107–109
Funding
The authors received financial support from the National Natural Science Foundation of China (51704061) and China Postdoctoral Science Foundation (2016 M601321) and Fundamental Research Funds of the Central Universities of China (N162503003) and Northeastern University Postdoctoral Science Foundation (20170306).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Gao, L., Liu, Z., Pan, Y. et al. Separation and Recovery of Iron and Nickel from Low-Grade Laterite Nickel Ore Using Reduction Roasting at Rotary Kiln Followed by Magnetic Separation Technique. Mining, Metallurgy & Exploration 36, 375–384 (2019). https://doi.org/10.1007/s42461-018-0012-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42461-018-0012-z