Abstract
Purpose
To evaluate the prevalence of dental caries and dental developmental defects (DDD) in childhood cancer survivors (CCS) and identify disease and treatment-related risk factors.
Methods
CCS aged up to 21 years, diagnosed with a malignancy before the age of 10 years and in remission for at least one year were included. Data were collected from patients’ medical records and through a clinical examination where presence of dental caries and prevalence of DDD were recorded. Fisher’s exact test was used to assess possible correlations and multivariate regression analysis to determine risk factors for defect development.
Results
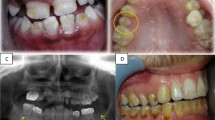
Seventy CCS with a mean chronological age of 11.2 years at the time of examination, mean age at cancer diagnosis of 4.17 years, and a mean post-treatment follow-up time of 5.48 years were included. Mean DMFT/dmft was 1.31, with 29% of survivors presenting with at least one carious lesion. Younger patients on the day of examination and patients treated with higher radiation dose, showed significantly higher prevalence of dental caries. The prevalence of DDD was 59%, with demarcated opacities being the defect most commonly observed (40%). Age at dental examination, diagnosis, age at diagnosis, and time that have elapsed since the end of treatment were the factors significantly affecting its prevalence. Regression analysis revealed that age at examination was the only factor significantly associated with the presence of coronal defects.
Conclusion
A great number of CCS presented with at least one carious lesion or a DDD, with the prevalence being significantly associated with various disease-specific characteristics, but age at dental examination the only significant predictor.
Similar content being viewed by others
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
13 May 2023
A Correction to this paper has been published: https://doi.org/10.1007/s40368-023-00802-5
References
Alberth M, Kovalecz G, Nemes J, et al. Oral health of long-term childhood cancer survivors. Pediatr Blood Cancer. 2004;43:88–90. https://doi.org/10.1002/pbc.20023.
Avsar A, Elli M, Derka O, et al. Long-term effects of chemotherapy on caries formation, dental development, and salivary factors in childhood cancer survivors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:781–9. https://doi.org/10.1016/j.tripleo.2007.02.029.
Blaauwbroek R, Groenier KH, Kamps WA, Meyboom-de Jong B, Postma A. Late effects in adult survivors of childhood cancer: the need for life-long follow-up. Ann Oncol. 2007;18:1898–902. https://doi.org/10.1093/annonc/mdm336.
Bosse KR, Majzner RG, Mackall CL, Maris JM. Immune-based approaches for the treatment of pediatric malignancies. Annual Review of Cancer Biol. 2020;4:353–70. https://doi.org/10.1146/annurev-cancerbio-030419-033436.
Busenhart DM, Erb J, Rigakos G, et al. Adverse effects of chemotherapy on the teeth and surrounding tissues of children with cancer: a systematic review with meta-analysis. Oral Oncol. 2018;83:64–72. https://doi.org/10.1016/j.oraloncology.2018.06.001.
Çetiner D, Çetiner S, Uraz A, Alpaslan GH, Alpaslan C, Memikoğlu TUT, KaradeniZ C. Oral and dental alterations and growth disruption following chemotherapy in long-term survivors of childhood malignancies. Support Care Cancer. 2019;27:1891–9. https://doi.org/10.1007/s00520-018-4454-0.
Collett WK, Thonard JC. The effect of fractional radiation on dentinogenesis in the rat. J Dent Res. 1965;44:84–90. https://doi.org/10.1177/00220345650440013501.
Dahllöf G. Oral and dental late effects after pediatric stem cell transplantation. Biol Blood Marrow Transplant. 2008;14:81–3. https://doi.org/10.1016/j.bbmt.2007.11.007.
Duggal MS, Curzon MEJ, Bailey CC, et al. Dental parameters in the long-term survivors of childhood cancer compared with siblings. Oral Oncol. 1997;33:348–53. https://doi.org/10.1016/s1368-8375(97)89103-8.
Effinger K, Migliorati C, Hudson M, et al. Oral and dental late effects in survivors of childhood cancer: a Children’s oncology report group. Support Care Cancer. 2014;22:2009–19. https://doi.org/10.1007/s00520-014-2260-x.
Ekstrand K, Gimenez T, Ferreira FR, Mendes FM, Braga MM. The international caries detection and assessment system—ICDAS: a systematic review. Caries Res. 2018;52(5):406–19. https://doi.org/10.1159/000486429.
Gawade P, Hudson M, Kaste S, et al. A systematic review of dental late effects in survivors of childhood cancer. Pediatr Blood Cancer. 2014;61:407–16. https://doi.org/10.1002/pbc.24842.
Halperson E, Matalon V, Goldstein G, SaiegSpilberg S, Herzog K, Fux-Noy A, Shmueli A, Ram D, Moskovitz M. The prevalence of dental developmental anomalies among childhood cancer survivors according to types of anticancer treatment. Sci Rep. 2022;12(1):4485. https://doi.org/10.1038/s41598-022-08266-1.
Hölttä P, Alaluusua S, Saarinen-Pihkala UM, et al. Long-term adverse effects on dentition in children with poor-risk neuroblastoma treated with high-dose chemotherapy and autologous stem cell transplantation with or without total body irradiation. Bone Marrow Transplant. 2002;29:121–7. https://doi.org/10.1038/sj.bmt.1703330.
Jaffe N, Toth BB, Hoar RE, et al. Dental and maxillofacial abnormalities in long-term survivors of childhood cancer: effects of treatment with chemotherapy and radiation to the head and neck. Pediatrics. 1984;73:816–23.
Jeong KH, Kim D, Song YM, Sung J, Kim YH. Epidemiology and genetics of hypodontia and microdontia: a study of twin families. Angle Orthod. 2015;85:980–5. https://doi.org/10.2319/052814-376.1.
Jodłowska A, Postek-Stefańska L. Duration and dose of chemotherapy and dental development. Dent Med Probl. 2022;59(1):45–58. https://doi.org/10.17219/dmp/138914.
Kang CM, Hahn SM, Kim HS, et al. Clinical risk factors influencing dental developmental disturbances in childhood cancer survivors. Cancer Res Treat. 2018;50(3):926–35. https://doi.org/10.4143/crt.2017.296.
Kaste SC, Hopkins KP, Bowman LC. Dental abnormalities in long-term survivors of head and neck rhabdomyosarcoma. Med Pediatr Oncol. 1995;25:96–101. https://doi.org/10.1002/mpo.2950250209.
Kaste SC, Hopkins KP, Jones D, Crom D, Greenwald CA, Santana VM. Dental abnormalities in children treated for acute lymphoblastic leukemia. Leukemia. 1997;11:792–6. https://doi.org/10.1038/sj.leu.2400670.
Kaste SC, Hopkins KP, Bowman LC, et al. Dental abnormalities in children treated for neuroblastoma. Med Pediatr Oncol. 1998;30:22–7. https://doi.org/10.1002/(sici)1096-911x(199801)30:1%3c22::aid-mpo8%3e3.0.co;2-2.
Maguire A, Welbury RR. Long-term effects of antineoplastic chemotherapy and radiotherapy on dental development. Dent Updete. 1996;23:188–94.
Minicucci EM, Lopes LF, Crocci AJ. Dental abnormalities in children after chemotherapy treatment for acute lymphoid leukemia. Leuk Res. 2003;27:45–50. https://doi.org/10.1016/s0145-2126(02)00080-2.
Nunn JH, Welbury RR, Gordon PH, et al. Dental caries and dental anomalies in children treated by chemotherapy for malignant disease: a study in the north of England. Int J Paed Dent. 1991;1:131–5. https://doi.org/10.1111/j.1365-263x.1991.tb00331.x.
Oliveira AFB, Chaves AMB, Rosenblatt A. The influence of enamel defects on the development of early childhood caries in a population with low socioeconomic status: a longitudinal study. Caries Res. 2006;40:296–302. https://doi.org/10.1159/000093188.
Pajari U, Lanning M. Developmental defects of teeth in survivors of childhood ALL are related to the therapy and age at diagnosis. Med Pediatr Oncol. 1995;24:310–4. https://doi.org/10.1002/mpo.2950240508.
Pajari U, Lanning M, Larmas M. Prevalence and location of enamel opacities in children after antineoplastic therapy. Commun Dent Oral Epidemiol. 1988;16:222–6. https://doi.org/10.1111/j.1600-0528.1988.tb01759.x.
Pedersen LB, Clausen N, Schrøder H, Schmidt M, Poulsen S. Microdontia and hypodontia of premolars and permanent molars in childhood cancer survivors after chemotherapy. Int J Paediatr Dent. 2012;22:239–43. https://doi.org/10.1111/j.1365-263X.2011.01199.
Proc P, Szczepańska J, Herud A, Zubowska M, Fendler W, Młynarski W. Dental caries among childhood cancer survivors. Medicine. 2019;98(6): e14279. https://doi.org/10.1097/MD.0000000000014279.
Singh N, Scully C, Joyston-Bechal S. Oral complications of cancer therapies: prevention and management. Clin Oncol. 1996;8:15–24. https://doi.org/10.1016/s0936-6555(05)80034-2.
Sonis AL, Tarbell N, Valachovic RW, et al. Dentofacial development in long-term survivors of acute lymphoblastic leukemia. A comparison of three treatment modalities. Cancer. 1990;66:2645–52.
Stene T, Koppang HS. Autoradiographic investigation of proliferative responses in rat incisor pulp after vincristine administration. Scand J Dent Res. 1980;88(2):96–103. https://doi.org/10.1111/j.1600-0722.1980.tb01200.x.
van der Pas-van Voskuilen IGM, Veerkamp JSJ, Raber-Durlacher JE, et al. Long-term adverse effects of hematopoietic stem cell transplantation on dental development in children. Supp Care Cancer. 2009;17:1169–75. https://doi.org/10.1007/s00520-008-0567-1.
Ward E, DeSantis C, Robbins A, Kohler B, Jemal A. Childhood and adolescent cancer statistics, 2014. CA Cancer J Clin. 2014;64:83–103. https://doi.org/10.3322/caac.21219.
Wellbury RR, Craft AW, Murray JJ, et al. Dental health of survivors of malignant disease. Arch Dis Child. 1984;59:1186–7. https://doi.org/10.1136/adc.59.12.1186.
WMA. WMA declaration of Helsinki: Ethical principles for medical research involving human subjects. World Medical Association Fortaleza, Brazil amendment (2013). Available at https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involvinghuman-subjects/. Accessed 1 Jun 2022.
Willberg P, Kanellopoulos A, Ruud E, Hjermstad MJ, Fossa SD, Herlofson BB. Dental abnormalities after chemotherapy in long-term survivors of childhood acute lymphoblastic leukemia 7–40 years after diagnosis. Supp Care Cancer. 2016;24:1497–506. https://doi.org/10.1007/s00520-015-2940-1.
Acknowledgements
None
Funding
The authors received no funding for the current work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. KK conceived the idea. Material preparation and data collection was performed by KS. AK assisted sample selection and AP led the writing. The first draft of the manuscript was written KS and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval
The study was performed in accordance with the Declaration of Helsinki (WMA 2013) ethical standards and the research protocol was submitted and approved by the Ethics Committee of the '[removed for blind peer review]’ (N363, approved on 22/6/2018).
Consent to participate
Informed consent to participate was obtained from all participants and/or their legal guardians.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Seremidi, K., Kavvadia, K., Kattamis, A. et al. Dental caries and dental developmental defects as adverse effects of antineoplastic treatment in childhood cancer survivors. Eur Arch Paediatr Dent 24, 357–365 (2023). https://doi.org/10.1007/s40368-023-00789-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40368-023-00789-z