Abstract
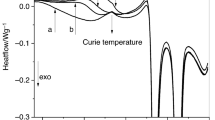
The crystallization kinetics of Cu43Zr48Al9 and (Cu43Zr48Al9)98Y2 bulk metallic glasses in non-isothermal and isothermal conditions was studied by differential scanning calorimetry. In the non-isothermal and isothermal modes, the average activation energy of (Cu43Zr48Al9)98Y2 is larger than that of Cu43Zr48Al9, meaning the higher stability against crystallization of (Cu43Zr48Al9)98Y2. In addition, the average activation energies for Cu43Zr48Al9 and (Cu43Zr48Al9)98Y2 calculated using Arrhenius equation in isothermal mode are larger than the values calculated by Kissinger–Akahira–Sunose method in non-isothermal mode, indicating that the energy barrier is higher in isothermal mode. The Johnson–Mehl–Avrami model was used to analyze the crystallization kinetics in the non-isothermal and isothermal modes. The Avrami exponent n for Cu43Zr48Al9 is above 2.5, indicating that the crystallization is mainly determined by a diffusion-controlled three-dimensional growth with an increasing nucleation rate, while the Avrami exponent n for (Cu43Zr48Al9)98Y2 is in the range of 1.5–2.5 in the non-isothermal mode, implying that the crystallization is mainly governed by diffusion-controlled three-dimensional growth with decreasing nucleation rate. Finally, the Avrami exponents n for Cu43Zr48Al9 and (Cu43Zr48Al9)98Y2 are different in the non-isothermal and isothermal conditions, which imply different nucleation and growth behaviors during the crystallization processes.












Similar content being viewed by others
References
W.L. Johnson, MRS Bull. 24, 42 (1999)
M. Telford, Mater. Today 7, 36 (2004)
J.C. Huang, T.H. Chuang, Mater. Chem. Phys. 57, 195 (1999)
A. Inoue, Acta Mater. 48, 279 (2000)
J. Cui, J.S. Li, J. Wang, H.C. Kou, J.C. Qiao, S. Gravier, J.J. Blandin, J. Non-Cryst. Solids 404, 7 (2014)
A. Inoue, W. Zhang, Mater. Trans. 43, 2921 (2002)
F.G. Coury, W.J. Botta, C. Bolfarini, C.S. Kiminami, M.J. Kaufman, J. Non-Cryst. Solids 406, 79 (2014)
B. Li, J.S. Li, X.H. Fan, J. Chen, Rare Met. Mater. Eng. 43, 1558 (2014). (in Chinese)
Y. Zhang, J. Chen, G.L. Chen, X.J. Liu, Appl. Phys. Lett. 89, 131904 (2006)
H.W. Xu, Y.Y. Du, Y. Deng, Trans. Nonferrous Met. Soc. China 22, 842 (2012). (in Chinese)
D.H. Xu, D. Gang, W.L. Johnson, Phys. Rev. Lett. 92, 245504 (2004)
K.G. Prashanth, S. Scudino, K.B. Surreddi, M. Sakaliyska, B.S. Murty, J. Eckert, Mater. Sci. Eng., A 279, 513 (2009)
H.E. Kissinger, Anal. Chem. 29, 1702 (1957)
Y.H. Li, C. Yang, L.M. Kang, H.D. Zhao, S.G. Qu, X.Q. Li, W.W. Zhang, Y.Y. Li, J. Non-Cryst. Solids 432, 440 (2016)
J.C. Qiao, J.M. Pelletier, J. Non-Cryst. Solids 357, 2590 (2011)
L. Deng, B.W. Zhou, H.S. Yang, X. Jiang, B.Y. Jiang, X.G. Zhang, J. Alloys Compd. 632, 429 (2015)
X.X. Hu, J.C. Qiao, J.M. Pelletier, Y. Yao, J. Non-Cryst. Solids 432, 254 (2016)
A.A. Soliman, S. Al-Heniti, A. Al-Hajry, M. Al-Assiri, G. Al-Barakati, Thermochim. Acta 413, 57 (2004)
Q.P. Cao, J.W. Liu, J.F. Li, Y.H. Zhou, X.D. Wang, J.Z. Jiang, J. Non-Cryst. Solids 357, 1182 (2011)
X.F. Wang, D. Wang, B. Zhu, Y.J. Li, F.S. Han, J. Non-Cryst. Solids 385, 111 (2014)
K.N. Lad, R.T. Savalia, A. Pratap, G.K. Dey, S. Banerjee, Thermochim. Acta 473, 74 (2008)
W. Lu, B. Yan, W.H. Huang, J. Non-Cryst. Solids 351, 3320 (2005)
S. Venkataraman, E. Rozhkova, J. Eckert, L. Schulta, D.J. Sordelet, Intermetallics 13, 833 (2005)
L.K. Zhang, Z.H. Chen, Q. Zheng, D. Chen, Phys. B 411, 149 (2013)
Z.F. Yao, J.C. Qiao, C. Zhang, J.M. Pelletier, Y. Yao, J. Non-Cryst. Solids 415, 42 (2015)
Y.J. Yang, D.W. Xing, J. Shen, J.F. Sun, S.D. Wei, H.J. He, D.G. McCartney, J. Alloys Compd. 415, 106 (2006)
S.W. Wei, B.Z. Ding, T.Q. Lei, Z.Q. Hu, Mater. Lett. 37, 263 (1998)
M. Avrami, J. Chem. Phys. 8, 212 (1940)
M. Avrami, J. Chem. Phys. 9, 177 (1941)
J. Wang, H.C. Kou, J.S. Li, X.F. Gu, H. Zhong, H. Chang, L. Zhou, J. Phys. Chem. Solids 70, 1448 (2009)
C. Peng, Z.H. Chen, X.Y. Zhao, A.L. Zhang, L.K. Zhang, D. Chen, J. Non-Cryst. Solids 405, 7 (2014)
K.G. Raval, K.N. Lad, A. Pratap, A.M. Awasthi, S. Bhardwaj, Thermochim. Acta 425, 47 (2005)
P. Gong, K.F. Yao, S.F. Zhao, J. Therm. Anal. Calorim. 121, 697 (2015)
Acknowledgements
This work was supported financially by the Special Research Project for the Education Department of Shaanxi Province (No. 14JK1351) and the President Fund of Xi’an Technological University (No. 0852-302021407).
Author information
Authors and Affiliations
Corresponding author
Additional information
Available online at http://link.springer.com/journal/40195
Rights and permissions
About this article
Cite this article
Yang, K., Fan, XH., Li, B. et al. Non-isothermal Crystallization Kinetics and Isothermal Crystallization Kinetics in Supercooled Liquid Region of Cu–Zr–Al–Y Bulk Metallic Glass. Acta Metall. Sin. (Engl. Lett.) 31, 290–298 (2018). https://doi.org/10.1007/s40195-017-0625-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40195-017-0625-0