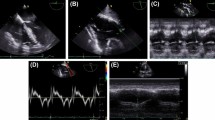
Abstract
Purpose of Review
This review aims to highlight the perioperative echocardiographic evaluation of right ventricular (RV) function with strengths and limitations of commonly used and evolving techniques. It explains the value of transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE) and describes the perioperative changes of RV function echocardiographers should be aware of.
Recent Findings
RV dysfunction is an entity with strong influence on outcome. However, its definition and assessment in the perioperative interval are not well-defined. Moreover, values assessed by TTE and TEE are not interchangeable; while some parameters seem to correlate well, others do not. Myocardial strain analysis and three-dimensional echocardiography may overcome the limitations of conventional echocardiographic measures and provide further insight into perioperative cardiac mechanics.
Summary
Echocardiography has become an essential part of modern anesthesiology in patients with RV dysfunction. It offers the opportunity to evaluate not only global but also regional RV function and distinguish alterations of RV contraction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Echocardiography is an essential part of modern anesthesia in patients undergoing the entire spectrum of cardiac surgery [1••], catheter-based cardiac procedures [2], and in high-risk non-cardiac surgery [3]. Although the right ventricle (RV) and its contribution to cardiac physiology and patient outcome has been neglected for a long time, the importance of RV function has been recognized in recent years in surgery [4] and non-surgery patients [5]. Perioperative RV dysfunction or failure has a wide range of etiologies and is a complex and dynamic entity [4]. It is a predictor of morbidity and mortality in various non-cardiac [6] and cardiac surgery procedures [7, 8], where it seems to be a strong predictor of mortality independent from the estimated surgical risk [8•].
Beyond cardiac magnetic resonance tomography (cMRT), as gold standard of cardiac imaging, echocardiography and catheter-based measurements offer the opportunity to monitor RV function intraoperatively. However, only echocardiography allows the operator to evaluate RV morphology and global function as well as regional differences and pathologies of associated structures. The RV has particular anatomical and functional characteristics that need to be addressed during echocardiographic evaluation and decision-making [9,10,11]. Moreover, the surgical or interventional procedure can have influence on the perioperative course of RV function [4, 9, 12] making assessment challenging.
There are several conventional echocardiographic parameters commonly used for evaluation of RV function, with specific strengths and limitations in the perioperative period. Furthermore, there are upcoming techniques for echocardiographic assessment of RV function with promising features.
This review aims to describe the current state of the art echocardiographic assessment of perioperative RV function and give further insight into evolving techniques in this field of research.
Anatomical and Pathophysiological Background
A profound understanding of RV anatomy and pathophysiology helps to understand diagnostic and management options.
RV Anatomy
The RV has a singular and complex form [9]. It seems triangular from a lateral view and crescent-shaped in a cross-sectional view. The RV has a thin free wall that is formed of transverse fibers, with sparse subendocardial longitudinal fibers [13]. The interventricular septum forms its medial border containing oblique helical fibers that cross each other at 60° [13]. Appearing smaller than the left ventricle (LV), the RV has a larger volume than the LV [9, 13], and its mass is only about 1/6 of the mass of the LV [14]. The RV can be subdivided into three parts: the inflow tract, the trabeculated apical portion, and the outflow tract [9, 14].
RV Pathophysiology
The main function of the RV is to receive systemic venous blood and eject it into the pulmonary circulation. Under normal circumstances, the RV is coupled to a low pressure, highly distensible arterial system [14, 15]. The RV is linked in series with the LV. In the absence of shunts and valvular regurgitation, the stroke volume of the RV is equivalent to the stroke volume of the LV. However, because of the greater end-diastolic volume of the RV, the right ventricular ejection fraction (RV EF) is lower than the left ventricular ejection fraction (LV EF) [9, 14].
Numerous mechanisms are essential for RV contraction. Perhaps the most important mechanism is the bellows-like inward motion of the RV free wall. Other important mechanisms contain the contraction of the longitudinal fibers initiating the shortening of the long axis and movement of the tricuspid annulus toward the apex [14]. The typical contraction of the RV is sequential, starting with the inflow tract (sinus), proceeding with the heavily trabeculated apical portion, and ending with the contraction of the outflow tract (infundibulum), delayed by 25–50 ms [9, 13, 14].
Although the interventricular septum (IVS) does not exclusively reflect RV function, it is of great importance for RV geometry and function. Approximately 30 to 40% of RV stroke volume is dependent on LV contraction by bulging of the ventricular septum into the RV cavity [16], explaining the great importance of IVS for RV function in systole and diastole [17].
Definition of RV Dysfunction and Failure
Although there is no generally accepted definition of RV failure, it can be defined as a combination of systemic arterial hypotension with increased right ventricular filling pressures [18]. Another common definition focuses on the functional approach and defines RV failure as the inability of the RV to provide adequate blood flow through the pulmonary circulation at normal central venous pressure [19].
RV dysfunction definitions vary in the literature. According to the current guidelines of the American Society of Echocardiography (ASE) and the European Association of Cardiovascular Imaging (EACVI), RV dysfunction is present when the echocardiographic parameters used to quantify RV function are less than the lower or greater than the higher reference value of the normal range (mean ± 2 × standard deviation), pointing out that available validated data are insufficient for the classification of abnormal categories into mild, moderate, and severe [10, 11]. Therefore, based on the current guidelines, only a dichotomous approach to RV impairment is feasible.
Echocardiography
Evaluation of RV function needs multiple acoustic windows, and the report should present an assessment based on both qualitative and quantitative parameters [10, 11]. Qualitative evaluation of RV function may be performed by experienced operators [20, 21], but, may have random results [11]. The guidelines on RV function assessment focus on transthoracic echocardiography (TTE), while guidelines on transesophageal echocardiography (TEE) mainly neglect the evaluation of RV function [3, 22]. Moreover, parameters assessed by TTE and TEE are not interchangeable, and normal values of RV function assessed by TEE are not well-established [23].
RV Systolic Function
Conventional Echocardiographic Parameters
Several RV parameters are recommended by current guidelines (Table 1), each of these with its own strengths and limitations, particularly regarding the perioperative interval (Table 2).
Linear Measurements
Quantitation of RV dimensions is essential and reduces interobserver variability compared with visual assessment alone [10]. These parameters are simple, easy to obtain, fast, and well-validated [10]. However, evaluation by two-dimensional echocardiography is challenging because of the complex geometry of the RV, having a high intra- and interobserver variability depending on the assessed view [10]. RV dimensions are best estimated from a RV-focused four-chamber view, with a diameter of > 41 mm at base and > 35 mm at mid-level indicating RV dilatation [10]. RV wall thickness, performed in end-diastole, below the tricuspid annulus is an easy to perform single-site measurement, with values of > 5 mm suggesting RV free wall hypertrophy.
Tricuspid Annular Plane Systolic Excursion (TAPSE)
TAPSE is a measure of longitudinal movement of the lateral tricuspid annulus to the apex during systole. Although it is a regional measure, it correlates with global systolic RV function [11, 25] and patient outcome in different clinical conditions [4]. TAPSE < 17 mm is highly suggestive of RV systolic dysfunction [10]. It is an easy to obtain, but angle-dependent parameter of RV longitudinal function with excellent temporal resolution measured by M-mode echocardiography [10, 11]. Therefore, alignment of the M-mode cursor with the longitudinal motion of the lateral tricuspid annulus should be achieved. This is simply performed in a four-chamber view by TTE [10] and needs an alternative approach by using TEE [1], since adequate alignment in TEE mid-esophageal four-chamber view is not feasible. Possible solutions addressing this issue are the use of alternative views with superior alignment or the use of alternative technologies [1]. Alternative views with superior alignment exist for visualization of the RV [22]. Particularly, the modified deep transgastric view focusing the right ventricle and the transgastric RV inflow view has been evaluated in the literature, however, showing underestimation of TAPSE by TEE using these alternative views in comparison to TAPSE by TTE using an apical four-chamber view [26, 27]. Therefore, alternative and less angle-dependent techniques seem more promising. Measurement of TAPSE by anatomic M-mode which evaluates two-dimensional pixel samples along a freely positioned cursor line presented increased precision [26, 28]. A further approach is to measure TAPSE with speckle-tracking echocardiography, which is mainly angle-independent [28, 29]. First investigations showed promising and unbiased results; however, there seems to be doubt about precision and reliability [29, 30].
Moreover, TAPSE may not be an optimal parameter in the perioperative setting of cardiac surgery [31]. Several trials reported about reduced TAPSE after cardiac surgery, explaining this condition with pericardiotomy [32] or surgery and extracorporeal circulation [33], while global RV function was preserved. Therefore, the accurate assessment of perioperative RV function in cardiac surgery patients with this parameter is highly questionable.
Tricuspid Annular Systolic Velocity (RV S′)
RV S′ has a lot of alikeness with TAPSE and showed also to correlate with global RV function and patient outcome [10, 11, 25]. An RV S′ velocity less than 9.5 cm/s indicates RV systolic dysfunction measured by TTE in awake spontaneous breathing patients [10]. It seems to be less dependent on loading conditions [10, 11]. But it has not been studied perioperatively by TEE as intensely as TAPSE, possibly because of the angle dependence of this tissue Doppler–derived parameter [1, 34]. Alternative views with more accurate alignment and the use of novel technologies were reported for addressing this issue [34]. Yet, similar to the existing literature on TAPSE, velocities measured by tissue Doppler imaging in alternative views, assessed by TEE, underestimated RV S′ in comparison to RV S′, assessed by TTE from the apical four-chamber view [27, 35].
A novel speckle-tracking-based method for measuring RV S′ in the mid-esophageal four-chamber view was described by Mauermann et al. [29], and showed promising results in 24 out of 25 evaluated patients.
Right Ventricular Fractional Area Change (RV FAC)
RV FAC is a measure of global RV systolic function [10, 11]. It is obtained in a four-chamber view and describes the change of RV area in relation to diastolic RV area. It is of great importance to assess the entire RV in the image plane, including the apex during the complete contraction. RV FAC of < 35% suggests RV systolic dysfunction [10].
RV FAC correlates well with RV ejection fraction (RV EF) assessed by cMRI [36], and with echocardiographic measured three-dimensional RV EF [37]. Assessed either by TTE or by TEE, RV FAC is associated with postoperative morbidity and mortality [8, 38]. Data comparing TTE and TEE measures of RV FAC are sparse. Skinner et al. [39] showed that TTE and TEE measures of RV FAC did not differ significantly in spontaneously breathing patients. Roberts and colleagues found a fair agreement between TEE and TTE measurement of RV FAC in a heterogenous cardiac surgery population [40].
Right Ventricular Myocardial Performance Index (RV MPI)
RV MPI is a non-geometric measure of global RV function, calculated by the summation of the isovolumetric contraction and relaxation times divided by the ejection time [41]. Evaluated from the same heartbeat, the isovolumic contraction time, the isovolumic relaxation time, and ejection time intervals should be measured either by pulsed-wave (PW) Doppler or tissue Doppler (TDI) [10, 11]. In conditions with elevated right atrial pressure, which shortens isovolumetric relaxation time, RV MPI may be incorrectly reduced. RV MPI >0.43 assessed by PW-Doppler or >0.54 evaluated by TDI implies RV dysfunction [10].
RV MPI measured by TTE showed to be a predictor of adverse events, mainly in patients with pulmonary hypertension [41, 42]. Preoperative RV MPI evaluated by TEE was independently associated with mortality and morbidity after left-sided heart valve surgery [38].
Michaux et al. [43] compared RV MPI measurements in awake spontaneously breathing patients scheduled for coronary artery bypass surgery, preoperatively by TTE and intraoperatively after induction of anesthesia by TEE. RV MPI values were significantly lower in anesthetized patients under positive pressure ventilation. Moreover, variability was fourfold higher in TEE measures. Therefore, Michaux and colleagues questioned RV MPI as an adequate parameter for RV function assessment in anesthetized patients under positive pressure ventilation [43]. Roberts at al. found only slight agreement for RV MPI between TEE and TTE evaluation in anesthetized patients [40].
Novel Echocardiographic Parameters
Three-dimensional Right Ventricular Ejection Fraction (3D-RV EF)
According to the current ASE/EACVI guidelines, evaluation of three-dimensional RV volumetry and ejection fraction (3D-RV EF) should complement two-dimensional measures of RV function, when feasible [10]. Three-dimensional echocardiography allows the evaluation of RV shape, size, and function without the limitations of two-dimensional assessment. It is independent of geometric assumptions; includes the evaluation of the inflow, outflow, and apical segments; and correlates well with cMRI [10]. 3D-RV EF depends on an adequate image quality and requires an offline analysis as software is often not included on the echo machines [10]. TTE [44] and TEE [22] guidelines for performing three-dimensional echocardiography of the right heart were published. TEE may provide high-resolution and superior quality images of the right heart compared to TTE [22]. 3D-RV EF correlates well with cardiac magnetic resonance–derived RV EF, as the gold standard for evaluation of RV function [45], and is feasible intraoperatively [46, 47]. However, the predictive value in cardiac and non-cardiac surgery patients was not tested in adequately powered prospective studies [12, 20, 48]. Nonetheless, 3D-RV EF assessed by TEE is increasingly used as the reference method in the intraoperative setting [20, 49, 50]. The current ASE/EACVI guidelines state that 3D-RV EF < 45%, assessed by TTE in awake spontaneous breathing patients, implies dysfunction [10].
RV Strain Analysis
Myocardial strain analysis with speckle-tracking echocardiography (STE) is an upcoming technology to assess perioperative right ventricular function [51••]. Strain is a dimensionless parameter describing myocardial deformation, the fractional change in the length of myocardial segments. Deformation may appear in longitudinal, circumferential, or radial dimension. Strain can have positive or negative values, which reflect lengthening or shortening, respectively. It has the advantage to be less dependent on loading conditions than other echocardiographic parameters of RV function assessment, has a small interobserver variability, is relatively angle-independent, and does not rely on geometric assumptions [52]. It is, however, influenced by image quality, artefacts as reverberation or attenuation, and is vendor-dependent [52]. In the context of the RV, predominantly right ventricular global longitudinal strain (RV GLS), which incorporates RV free wall segments and the interventricular septum segments, or right ventricular longitudinal free wall strain (RV FWS), which exclusively includes RV free wall segments, is used in clinical practice [52].
Two-dimensional RV strain (2D-RV strain) correlates well with cMRI-derived RV EF and seems to have higher diagnostic accuracy for detection of RV dysfunction compared to conventional parameters [36, 53]. To standardize deformation imaging, the ASE and EACVI published a consensus document, which includes recommendations for two-dimensional RV strain assessment [54]. The current ASE/EACVI guideline on chamber quantification states that 2D-RV strain parameters are useful for estimating RV global and regional systolic function and have an established prognostic value in various cardiac conditions [10]. 2D-RV free wall strain greater than - 20 is expected to be abnormal in awake, spontaneous breathing patients evaluated by TTE [10].
Perioperative TTE–derived 2D-RV FWS is reduced after cardiac surgery with [24, 55] and without extracorporal circulation [56]; however, it remains unchanged after transcatheter aortic valve repair (TAVR) [24]. It predicts RV failure after left ventricular assist device (LVAD) surgery [57, 58] and correlates with postoperative mortality in cardiac surgery [8, 59], as well as in patients undergoing TAVR [60].
Less is known for intraoperative 2D-RV strain assessed by TEE since most of the knowledge on RV strain analysis has been obtained by TTE in awake patients. Intraoperative 2D-RV strain analysis is feasible [61] and impaired 2D-RV GLS seems to be associated with postoperative atrial fibrillation [62] and prolonged postoperative vasoactive support [63]. Surprisingly, intraoperative 2D-RV strain assessed by TEE poorly predicts RV failure after LVAD implantation [64, 65].
There is inconsistent data, however, on the perioperative course of 2D-RV strain in cardiac surgery [49, 50, 66, 67]. This variability might be explained by differences in evaluated surgical procedures, severity of the conditions, time of echocardiographic assessment, and the use of vasoactive treatment during echocardiography. Various applied techniques for myocardial protection during cardiopulmonary bypass and different surgical practice itself may additionally influence postoperative RV function, and therefore strain, respectively. Furthermore, assessment of the complex RV with two-dimensional strain analysis might be biased, mainly by out-of-plane movement of the analyzed speckles [68] and foreshortened views [69]. For all mentioned above, there are no generally accepted normal TEE values for intraoperative 2D-RV strain. More trials, standardization, and probably alternative techniques, overcoming the limitations of two-dimensional approach, seem to be necessary to draw further conclusions.
Three-dimensionally derived RV FWS (3D-RV FWS) overcomes these limitations by 3D full-volume assessment that incorporates the entire RV and is independent of geometric assumptions [48, 52]. Intraoperative 3D-RV FWS is feasible and promising; however, existing data is sparse [51••].
In a retrospective case series, Keller et al. [70] analyzed one-time intraoperative 3D-RV FWS, compared this method to other techniques of intraoperative RV FWS assessment, and found good agreement between analyzed methods. In another retrospective trial, Keller and colleagues showed differences in perioperative changes of 3D-RV FWS between mitral valve surgery, off-pump CABG (OPCAB), and in percutaneous mitral valve repair (PMVR) [71•]. Regarding our own published data [72], 3D-RV FWS seems not to differ significantly in awake, spontaneously breathing patients and in patients under general anesthesia and positive pressure ventilation with stable hemodynamics. It remained unchanged after sternotomy but deteriorated after on-pump coronary artery bypass surgery in our group of patients. More data is needed to draw reliable conclusions.
RV Diastolic Function
Various acute and chronic conditions are associated with RV diastolic dysfunction (RVDD), including both pressure and volume overload conditions in LV dysfunction, heart valve disease, pulmonary embolism, lung disease, ischemic heart disease, cardiomyopathies, and different systemic diseases affecting the heart and the lung [11]. Assessment of RVDD is of clinical value as it serves as an early marker of RV dysfunction which commonly develops before obvious systolic RV dysfunction and before RV dilatation appears [11]. Presence of RVDD is associated with less exercise capacity and is an independent predictor of mortality in patients with heart failure and pulmonary hypertension [11].
RVDD seems to be of prognostic relevance also in the perioperative setting [58, 73, 74].
For evaluation of RVDD, current ASE/EACVI guidelines recommend assessment of tricuspid valve inflow (Et, At) and lateral tricuspid annular tissue Doppler velocity (et´). Traditionally perioperative RVDD was recognized in patients with severe LV dysfunction [58, 73]. Recently, Sumin et al. [74•] found up to 46% of patients scheduled for non-emergency CABG surgery with preserved LV and RV systolic function to have RVDD using the recommended approach [11]. Analyzing their data, the decreased Et/At ratio was the best echocardiographic marker predicting postoperative heart failure development after CABG surgery in their patient population.
The best monitoring tool for perioperative RVDD assessment needs to be established [75]. Recently, strain-based parameters have been described for assessment of left ventricular diastolic dysfunction [76], but the authors of this review are unaware of studies on RVDD based on strain analysis. Unquestionably, more data is needed to draw conclusions on RVDD in the perioperative setting.
TTE Versus TEE Assessment
Both, TTE and TEE, are crucial parts of perioperative RV function assessment [1, 10]. TEE offers superior image quality and is the imaging mode of choice in the intraoperative setting complementing invasive hemodynamic measures. But it may be not adequate in awake patients in the direct postoperative period. In contrast, validated data on reference values largely relies on guidelines derived from TTE studies on the awake patient [10, 11], but image quality after cardiac surgery can be challenging. Image plane and angles of assessment have an effect on the values of RV measures [10, 11], and even slight modifications showed statistically significant differences in measured values, particularly in the context of RV function assessment [77].
For TEE evaluation, the best perioperative measure is yet to be established, since the parameters of RV function assessment are not interchangeable between TTE and TEE [12]. Particularly, the regional longitudinal parameters TAPSE and RV S′ seem to be underestimated by TEE compared to those by TTE evaluation [27, 40]. But also, RV MPI as global measure of RV function showed at most moderate correlation [40, 43] and a high inter- and intraobserver variability [43]. Therefore, echocardiographers should be cautious in extrapolating these measures validated by TTE studies to their TEE examinations. Global measures of RV function as RV FAC [39, 40], 3D-RV EF [46], 2D-RV GLS [61, 78], and 3D-RV FWS [72] assessed by TEE seem to correlate better with TTE assessment and may provide better agreement with validated reference values.
Perioperative Course
Cardiac surgery causes functional and geometric changes of the RV that are not entirely understood and often depend on the performed procedures. Existing data suggest postoperative reduced longitudinal RV function while global RV function is preserved [32, 33]. Moreover, data from 2D strain-based analyses suggests that longitudinal RV function decreases, whereas transverse/circumferential function might improve [49, 55].
Moreover, administration of anesthetics and the change from spontaneous respiration to intermittent positive pressure ventilation may affect “normal” echocardiographic values [79, 80]. Therefore, normal values assessed by TTE in spontaneous breathing patients are not interchangeable with values assessed by TEE in anesthetized and ventilated patients. Further research is required addressing this issue.
Comparison to Other Techniques of RV Function Assessment
Cardiac magnetic resonance tomography (cMRT) remains the gold standard of cardiac imaging. But it is not practical in the perioperative setting with rapid changes of hemodynamic conditions.
The use of pulmonary artery catheter (PAC) has been the gold standard of cardiac output measurement for the last decades. Although its use in critical ill patients has been questioned heavily in the last years, PAC remains an effective means of monitoring RV performance in the perioperative setting [81]. It is still a frequently used monitoring tool in cardiac surgery [82]. Particularly in complex and high-risk cases (e.g., LVAD implantation, heart transplantation, patient with severely depressed LV or RV function), PAC is of great value in perioperative decision-making [83]. But values of cardiac output measurement seem not to be interchangeable between PAC and echocardiography, even when 3D echocardiography is used [84, 85]. However, there are also reports describing a good correlation between both methods [86]. PAC complements information on RV function by providing pulmonary artery pressure monitoring and oxygen saturation [87, 88]. This is particularly important in difficult and complex situations and procedures [89, 90].
Future Directions
Global echocardiographic parameters of RV function assessment [40, 46] and strain-based parameters [61, 78] appear to have better agreement between TTE and TEE, and seem to describe RV function more accurately in the perioperative interval [50, 59]. Future studies should focus on exploring these parameters and their influence on patient outcome. Two-dimensional RV strain analysis is a promising technique needing standardization and validation in the context of cardiac and non-cardiac surgery [91, 92]. Having the opportunity to distinguish longitudinal, radial, and circumferential contraction [49, 55] RV strain analysis allow to differentiate perioperative changes of RV function in different procedures. Although in its infancy, perioperative three-dimensional RV strain analysis offers many features to overcome the problems of two-dimensional strain assessment and may help in decision-making [71, 72].
In high-risk patients and complex surgical procedures (e.g., LVAD implantation, heart transplantation, LCOS), combination of echocardiography and PAC on detection of RV failure should be explored [82].
Identifying RVDD seems to be of value in the perioperative setting. However, more data, either assessed by echocardiography, by PAC, or even both techniques, is needed to draw further conclusions [75, 88].
The number of parameters or even technologies for RV function assessment has increased significantly, and evaluation has become more complex and time-consuming. In the context of a dynamic intraoperative setting with several demanding tasks, artificial intelligence might help the anesthesiologist in decision-making in time-critical situations [93, 94].
Conclusion
Both TTE and TEE are essential for perioperative assessment of RV function. For a complete evaluation of the complex structured RV, various parameters and views are required. RV contraction seems to change in the perioperative course, particularly in cardiac surgery. Novel echocardiographic techniques have found a reduced motion of longitudinal fibers and an increased deformation of transversal fibers after cardiac surgery. Myocardial strain analysis and three-dimensional echocardiography may provide further insight into these alterations of systolic and diastolic RV function in the perioperative period.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Nicoara A, Skubas N, Ad N, Finley A, Hahn RT, Mahmood F, et al. Guidelines for the use of transesophageal echocardiography to assist with surgical decision-making in the operating room: a surgery-based approach: From the American Society of Echocardiography in Collaboration with the Society of Cardiovascular Anesthesiologists and the Society of Thoracic Surgeons. J Am Soc Echocardiogr. 2020;33(6):692–734. https://doi.org/10.1016/j.echo.2020.03.002Recent ASE/EACVI guideline highlighting the importance of TEE in intraoperative decision-making.
Hahn RT, Little SH, Monaghan MJ, Kodali SK, Williams M, Leon MB, et al. Recommendations for comprehensive intraprocedural echocardiographic imaging during TAVR. JACC Cardiovasc Imaging. 2015;8(3):261–87. https://doi.org/10.1016/j.jcmg.2014.12.014.
Flachskampf FA, Badano L, Daniel WG, Feneck RO, Fox KF, Fraser AG, et al. Recommendations for transoesophageal echocardiography: update 2010. Eur J Echocardiogr. 2010;11(7):557–76. https://doi.org/10.1093/ejechocard/jeq057.
Subramani S, Sharma A, Arora L, Hanada S, Krishnan S, Ramakrishna H. Perioperative right ventricular dysfunction: analysis of outcomes. Journal of Cardiothoracic and Vascular Anesthesia. 2021;(in press). https://doi.org/10.1053/j.jvca.2021.01.032.
Amsallem M, Mercier O, Kobayashi Y, Moneghetti K, Haddad F. Forgotten no more: a focused update on the right ventricle in cardiovascular disease. JACC Heart Fail. 2018;6(11):891–903. https://doi.org/10.1016/j.jchf.2018.05.022.
Chou J, Ma M, Gylys M, Seong J, Salvatierra N, Kim R, et al. Preexisting right ventricular dysfunction is associated with higher postoperative cardiac complications and longer hospital stay in high-risk patients undergoing nonemergent major vascular surgery. Journal of Cardiothoracic and Vascular Anesthesia. 2019;33(5):1279–86. https://doi.org/10.1053/j.jvca.2018.10.011.
Bootsma IT, de Lange F, Koopmans M, Haenen J, Boonstra PW, Symersky T, et al. Right ventricular function after cardiac surgery is a strong independent predictor for long-term mortality. J Cardiothorac Vasc Anesth. 2017;31(5):1656–62. https://doi.org/10.1053/j.jvca.2017.02.008.
Peyrou J, Chauvel C, Pathak A, Simon M, Dehant P, Abergel E. Preoperative right ventricular dysfunction is a strong predictor of 3 years survival after cardiac surgery. Clin Res Cardiol. 2017;106(9):734–42. https://doi.org/10.1007/s00392-017-1117-yProspective trial including 400 cardiac surgery patients showing that echocardiography-assessed RV dysfunction predicts postoperative mortality independent of EuroScore.
Haddad F, Couture P, Tousignant C, Denault AY. The right ventricle in cardiac surgery, a perioperative perspective: I. Anatomy, physiology, and assessment. Anesth Analg. 2009;108(2):407–21. https://doi.org/10.1213/ane.0b013e31818f8623.
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16(3):233–70. https://doi.org/10.1093/ehjci/jev014.
Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, Chandrasekaran K, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr. 2010;23(7):685–713; quiz 86-8. https://doi.org/10.1016/j.echo.2010.05.010.
Zochios V, Protopapas AD, Parhar K. Markers of right ventricular dysfunction in adult cardiac surgical patients. Journal of Cardiothoracic and Vascular Anesthesia. 2017;31(5):1570–4. https://doi.org/10.1053/j.jvca.2017.06.019.
Buckberg G, Hoffman JI. Right ventricular architecture responsible for mechanical performance: unifying role of ventricular septum. J Thorac Cardiovasc Surg. 2014;148(6):3166–71 e1-4. https://doi.org/10.1016/j.jtcvs.2014.05.044.
Dell’Italia LJ. The right ventricle: anatomy, physiology, and clinical importance. Curr Probl Cardiol. 1991;16(10):653–720. https://doi.org/10.1016/0146-2806(91)90009-y.
Haddad F, Hunt SA, Rosenthal DN, Murphy DJ. Right ventricular function in cardiovascular disease, part i. Circulation. 2008;117(11):1436–48. https://doi.org/10.1161/CIRCULATIONAHA.107.653576.
Yamaguchi S, Harasawa H, Li KS, Zhu D, Santamore WP. Comparative significance in systolic ventricular interaction. Cardiovascular Research. 1991;25(9):774–83. https://doi.org/10.1093/cvr/25.9.774.
Santamore WP, Dell’Italia LJ. Ventricular interdependence: significant left ventricular contributions to right ventricular systolic function. Prog Cardiovasc Dis. 1998;40(4):289–308. https://doi.org/10.1016/s0033-0620(98)80049-2.
Kinch JW, Ryan TJ. Right ventricular infarction. New England Journal of Medicine. 1994;330(17):1211–7. https://doi.org/10.1056/nejm199404283301707.
Greyson CR. Pathophysiology of right ventricular failure. Critical Care Medicine. 2008;36(1):S57–65. https://doi.org/10.1097/01.Ccm.0000296265.52518.70.
Magunia H, Schmid E, Hilberath JN, Haberle L, Grasshoff C, Schlensak C, et al. 2D echocardiographic evaluation of right ventricular function correlates with 3D volumetric models in cardiac surgery patients. J Cardiothorac Vasc Anesth. 2017;31(2):595–601. https://doi.org/10.1053/j.jvca.2016.11.020.
Orde S, Slama M, Yastrebov K, McLean A, Huang S, College of Intensive Care Medicine of A, et al. Subjective right ventricle assessment by echo qualified intensive care specialists: assessing agreement with objective measures. Crit Care. 2019;23(1):70. https://doi.org/10.1186/s13054-019-2375-z.
Hahn RT, Abraham T, Adams MS, Bruce CJ, Glas KE, Lang RM, et al. Guidelines for performing a comprehensive transesophageal echocardiographic examination: recommendations from the American Society of Echocardiography and the Society of Cardiovascular Anesthesiologists. J Am Soc Echocardiogr. 2013;26(9):921–64. https://doi.org/10.1016/j.echo.2013.07.009.
Gebhardt BR, Asher S, Maslow A. The limitations of using transthoracic echocardiographic-derived normative values for grading intraoperative transesophageal echocardiography examinations of the right ventricle: are they really interchangeable? J Cardiothorac Vasc Anesth. 2020;34(5):1260–2. https://doi.org/10.1053/j.jvca.2020.02.024.
Kempny A, Diller GP, Kaleschke G, Orwat S, Funke A, Schmidt R, et al. Impact of transcatheter aortic valve implantation or surgical aortic valve replacement on right ventricular function. Heart. 2012;98(17):1299–304. https://doi.org/10.1136/heartjnl-2011-301203.
Tamborini G, Marsan NA, Gripari P, Maffessanti F, Brusoni D, Muratori M, et al. Reference values for right ventricular volumes and ejection fraction with real-time three-dimensional echocardiography: evaluation in a large series of normal subjects. J Am Soc Echocardiogr. 2010;23(2):109–15. https://doi.org/10.1016/j.echo.2009.11.026.
Flo Forner A, Hasheminejad E, Sabate S, Ackermann MA, Turton EW, Ender J. Agreement of tricuspid annular systolic excursion measurement between transthoracic and transesophageal echocardiography in the perioperative setting. Int J Cardiovasc Imaging. 2017;33(9):1385–94. https://doi.org/10.1007/s10554-017-1128-9.
Mauermann E, Vandenheuvel M, Francois K, Bouchez S, Wouters P. Right ventricular systolic assessment by transesophageal versus transthoracic echocardiography: displacement, velocity, and myocardial deformation. J Cardiothorac Vasc Anesth. 2020;34(8):2152–61. https://doi.org/10.1053/j.jvca.2020.03.009.
Korshin A, Gronlykke L, Nilsson JC, Moller-Sorensen H, Ihlemann N, Kjoller M, et al. The feasibility of tricuspid annular plane systolic excursion performed by transesophageal echocardiography throughout heart surgery and its interchangeability with transthoracic echocardiography. Int J Cardiovasc Imaging. 2018;34(7):1017–28. https://doi.org/10.1007/s10554-018-1306-4.
Mauermann E, Vandenheuvel M, Francois K, Bouchez S, Wouters P. A Novel speckle-tracking based method for quantifying tricuspid annular velocities in TEE. J Cardiothorac Vasc Anesth. 2019;33(10):2636–44. https://doi.org/10.1053/j.jvca.2019.05.036.
Shen T, Picard MH, Hua L, Burns SM, Andrawes MN. Assessment of tricuspid annular motion by speckle tracking in anesthetized patients using transesophageal echocardiography. Anesth Analg. 2018;126(1):62–7. https://doi.org/10.1213/ane.0000000000002614.
Maus TM. TAPSE: A red herring after cardiac surgery. J Cardiothorac Vasc Anesth. 2018;32(2):779–81. https://doi.org/10.1053/j.jvca.2017.11.013.
Raina A, Vaidya A, Gertz ZM, Susan C, Forfia PR. Marked changes in right ventricular contractile pattern after cardiothoracic surgery: implications for post-surgical assessment of right ventricular function. J Heart Lung Transplant. 2013;32(8):777–83. https://doi.org/10.1016/j.healun.2013.05.004.
Tamborini G, Muratori M, Brusoni D, Celeste F, Maffessanti F, Caiani EG, et al. Is right ventricular systolic function reduced after cardiac surgery? A two- and three-dimensional echocardiographic study. Eur J Echocardiogr. 2009;10(5):630–4. https://doi.org/10.1093/ejechocard/jep015.
Vandenheuvel M, Bouchez S, Wouters P, Mauermann E. Assessing right ventricular function in the perioperative setting, part I: echo-based measurements. Anesthesiol Clin. 2019;37(4):675–95. https://doi.org/10.1016/j.anclin.2019.08.011.
Tousignant CP, Bowry R, Levesque S, Denault AY. Regional differences in color tissue Doppler-derived measures of longitudinal right ventricular function using transesophageal and transthoracic echocardiography. J Cardiothorac Vasc Anesth. 2008;22(3):400–5. https://doi.org/10.1053/j.jvca.2007.07.014.
Focardi M, Cameli M, Carbone SF, Massoni A, De Vito R, Lisi M, et al. Traditional and innovative echocardiographic parameters for the analysis of right ventricular performance in comparison with cardiac magnetic resonance. Eur Heart J Cardiovasc Imaging. 2015;16(1):47–52. https://doi.org/10.1093/ehjci/jeu156.
Imada T, Kamibayashi T, Ota C, Carl Shibata S, Iritakenishi T, Sawa Y, et al. Intraoperative right ventricular fractional area change is a good indicator of right ventricular contractility: a retrospective comparison using two- and three-dimensional echocardiography. J Cardiothorac Vasc Anesth. 2015;29(4):831–5. https://doi.org/10.1053/j.jvca.2014.12.005.
Haddad F, Denault AY, Couture P, Cartier R, Pellerin M, Levesque S, et al. Right ventricular myocardial performance index predicts perioperative mortality or circulatory failure in high-risk valvular surgery. J Am Soc Echocardiogr. 2007;20(9):1065–72. https://doi.org/10.1016/j.echo.2007.02.017.
Skinner H, Kamaruddin H, Mathew T. Tricuspid annular plane systolic excursion: comparing transthoracic to transesophageal echocardiography. J Cardiothorac Vasc Anesth. 2017;31(2):590–4. https://doi.org/10.1053/j.jvca.2016.09.001.
Roberts SM, Klick J, Fischl A, King TS, Cios TJ. A comparison of transesophageal to transthoracic echocardiographic measures of right ventricular function. J Cardiothorac Vasc Anesth. 2020;34(5):1252–9. https://doi.org/10.1053/j.jvca.2019.11.039.
Tei C, Ling LH, Hodge DO, Bailey KR, Oh JK, Rodeheffer RJ, et al. New index of combined systolic and diastolic myocardial performance: a simple and reproducible measure of cardiac function--a study in normals and dilated cardiomyopathy. J Cardiol. 1995;26(6):357–66.
Tei C, Dujardin KS, Hodge DO, Bailey KR, McGoon MD, Tajik AJ, et al. Doppler echocardiographic index for assessment of global right ventricular function. J Am Soc Echocardiogr. 1996;9(6):838–47. https://doi.org/10.1016/s0894-7317(96)90476-9.
Michaux I, Seeberger M, Schuman R, Skarvan K, Filipovic M. Feasibility of measuring myocardial performance index of the right ventricle in anesthetized patients. J Cardiothorac Vasc Anesth. 2010;24(2):270–4. https://doi.org/10.1053/j.jvca.2009.09.010.
Lang RM, Badano LP, Tsang W, Adams DH, Agricola E, Buck T, et al. EAE/ASE recommendations for image acquisition and display using three-dimensional echocardiography. European Heart Journal - Cardiovascular Imaging. 2012;13(1):1–46. https://doi.org/10.1093/ehjci/jer316.
Pickett CA, Cheezum MK, Kassop D, Villines TC, Hulten EA. Accuracy of cardiac CT, radionucleotide and invasive ventriculography, two- and three-dimensional echocardiography, and SPECT for left and right ventricular ejection fraction compared with cardiac MRI: a meta-analysis. Eur Heart J Cardiovasc Imaging. 2015;16(8):848–52. https://doi.org/10.1093/ehjci/jeu313.
Fusini L, Tamborini G, Gripari P, Maffessanti F, Mazzanti V, Muratori M, et al. Feasibility of intraoperative three-dimensional transesophageal echocardiography in the evaluation of right ventricular volumes and function in patients undergoing cardiac surgery. J Am Soc Echocardiogr. 2011;24(8):868–77. https://doi.org/10.1016/j.echo.2011.05.002.
Karhausen J, Dudaryk R, Phillips-Bute B, Rivera JD, de Lange F, Milano CA, et al. Three-dimensional transesophageal echocardiography for perioperative right ventricular assessment. Ann Thorac Surg. 2012;94(2):468–74. https://doi.org/10.1016/j.athoracsur.2012.03.073.
Muraru D, Spadotto V, Cecchetto A, Romeo G, Aruta P, Ermacora D, et al. New speckle-tracking algorithm for right ventricular volume analysis from three-dimensional echocardiographic data sets: validation with cardiac magnetic resonance and comparison with the previous analysis tool. Eur Heart J Cardiovasc Imaging. 2016;17(11):1279–89. https://doi.org/10.1093/ehjci/jev309.
Donauer M, Schneider J, Jander N, Beyersdorf F, Keyl C. Perioperative changes of right ventricular function in cardiac surgical patients assessed by myocardial deformation analysis and 3-dimensional echocardiography. J Cardiothorac Vasc Anesth. 2020;34(3):708–18. https://doi.org/10.1053/j.jvca.2019.08.026Prospective trial evaluating two-dimensional RV strain demonstrating that after on-pump CABG surgery, longitudinal RV function is reduced while circumferential RV function is increased.
Gronlykke L, Korshin A, Holmgaard F, Kjoller SM, Gustafsson F, Nilsson JC, et al. Severe loss of right ventricular longitudinal contraction occurs after cardiopulmonary bypass in patients with preserved right ventricular output. Int J Cardiovasc Imaging. 2019;35(9):1661–70. https://doi.org/10.1007/s10554-019-01616-7.
Benson MJ, Silverton N, Morrissey C, Zimmerman J. Strain imaging: an everyday tool for the perioperative echocardiographer. J Cardiothorac Vasc Anesth. 2020;34(10):2707–17. https://doi.org/10.1053/j.jvca.2019.11.035Current review article summarizing the evidence on myocardial strain analysis in the perioperative setting.
Mor-Avi V, Lang RM, Badano LP, Belohlavek M, Cardim NM, Derumeaux G, et al. Current and evolving echocardiographic techniques for the quantitative evaluation of cardiac mechanics: ASE/EAE consensus statement on methodology and indications endorsed by the Japanese Society of Echocardiography. J Am Soc Echocardiogr. 2011;24(3):277–313. https://doi.org/10.1016/j.echo.2011.01.015.
Lu KJ, Chen JX, Profitis K, Kearney LG, DeSilva D, Smith G, et al. Right ventricular global longitudinal strain is an independent predictor of right ventricular function: a multimodality study of cardiac magnetic resonance imaging, real time three-dimensional echocardiography and speckle tracking echocardiography. Echocardiography. 2015;32(6):966–74. https://doi.org/10.1111/echo.12783.
Badano LP, Kolias TJ, Muraru D, Abraham TP, Aurigemma G, Edvardsen T, et al. Standardization of left atrial, right ventricular, and right atrial deformation imaging using two-dimensional speckle tracking echocardiography: a consensus document of the EACVI/ASE/Industry Task Force to standardize deformation imaging. Eur Heart J Cardiovasc Imaging. 2018;19(6):591–600. https://doi.org/10.1093/ehjci/jey042.
Moya Mur JL, Garcia Martin A, Garcia Lledo A, Lazaro Rivera C, Rincon Diaz LM, Miguelena Hycka J, et al. Geometrical and functional cardiac changes after cardiac surgery: a phisiopatological explanation based on speckle tracking. Int J Cardiovasc Imaging. 2018;34(12):1905–15. https://doi.org/10.1007/s10554-018-1424-z.
Khani M, Hosseintash M, Foroughi M, Naderian M, Khaheshi I. Assessment of the effect of off-pump coronary artery bypass (OPCAB) surgery on right ventricle function using strain and strain rate imaging. Cardiovasc Diagn Ther. 2016;6(2):138–43. https://doi.org/10.21037/cdt.2016.02.03.
Grant AD, Smedira NG, Starling RC, Marwick TH. Independent and incremental role of quantitative right ventricular evaluation for the prediction of right ventricular failure after left ventricular assist device implantation. J Am Coll Cardiol. 2012;60(6):521–8. https://doi.org/10.1016/j.jacc.2012.02.073.
Kato TS, Jiang J, Schulze PC, Jorde U, Uriel N, Kitada S, et al. Serial echocardiography using tissue Doppler and speckle tracking imaging to monitor right ventricular failure before and after left ventricular assist device surgery. JACC Heart Fail. 2013;1(3):216–22. https://doi.org/10.1016/j.jchf.2013.02.005.
Ternacle J, Berry M, Cognet T, Kloeckner M, Damy T, Monin JL, et al. Prognostic value of right ventricular two-dimensional global strain in patients referred for cardiac surgery. J Am Soc Echocardiogr. 2013;26(7):721–6. https://doi.org/10.1016/j.echo.2013.03.021.
Medvedofsky D, Koifman E, Jarrett H, Miyoshi T, Rogers T, Ben-Dor I, et al. Association of right ventricular longitudinal strain with mortality in patients undergoing transcatheter aortic valve replacement. Journal of the American Society of Echocardiography. 2020;33(4):452–60. https://doi.org/10.1016/j.echo.2019.11.014.
Tousignant C, Desmet M, Bowry R, Harrington AM, Cruz JD, Mazer CD. Speckle tracking for the intraoperative assessment of right ventricular function: a feasibility study. J Cardiothorac Vasc Anesth. 2010;24(2):275–9. https://doi.org/10.1053/j.jvca.2009.10.022.
Ting PC, Chou AH, Chien-Chia Wu V, Tsai FC, Chu JJ, Chen CY, et al. Relationship between right ventricular function and atrial fibrillation after cardiac surgery. J Cardiothorac Vasc Anesth. 2017;31(5):1663–71. https://doi.org/10.1053/j.jvca.2017.05.018.
Ting PC, Wu VC, Liao CC, Chou AH, Tsai FC, Lin PJ, et al. Preoperative right ventricular dysfunction indicates high vasoactive support needed after cardiac surgery. J Cardiothorac Vasc Anesth. 2019;33(3):686–93. https://doi.org/10.1053/j.jvca.2018.07.048.
Alfirevic A, Makarova N, Kelava M, Sale S, Soltesz E, Duncan AE. Predicting right ventricular failure after LVAD implantation: role of tricuspid valve annulus displacement. J Cardiothorac Vasc Anesth. 2020;34(5):1204–10. https://doi.org/10.1053/j.jvca.2019.08.045.
Silverton NA, Patel R, Zimmerman J, Ma J, Stoddard G, Selzman CH, et al. Intraoperative transesophageal echocardiography and right ventricular failure after left ventricular assist device implantation. J Cardiothorac Vasc Anesth. 2018;32(5):2096–103. https://doi.org/10.1053/j.jvca.2018.02.023.
Korshin A, Gronlykke L, Holmgaard F, Kjoller SM, Gustafsson F, Nilsson JC, et al. Right ventricular transverse displacement increases following cardiac surgery: possibly compensating loss in tricuspid annular plane systolic excursion (TAPSE). J Clin Monit Comput. 2020;34(6):1139–48. https://doi.org/10.1007/s10877-020-00466-2 Prospective study evaluating two-dimensional RV strain in on-pump CABG surgery showing postoperative reduced longitudinal RV function and increased transversal function.
Singh A, Huang X, Dai L, Wyler D, Alfirevic A, Blackstone EH, et al. Right ventricular function is reduced during cardiac surgery independent of procedural characteristics, reoperative status, or pericardiotomy. J Thorac Cardiovasc Surg. 2020;159(4):1430–8 e4. https://doi.org/10.1016/j.jtcvs.2019.04.035.
Wu VC-C, Takeuchi M, Otani K, Haruki N, Yoshitani H, Tamura M, et al. Effect of through-plane and twisting motion on left ventricular strain calculation: direct comparison between two-dimensional and three-dimensional speckle-tracking echocardiography. Journal of the American Society of Echocardiography. 2013;26(11):1274–81.e4. https://doi.org/10.1016/j.echo.2013.07.006.
Jacobs LD, Salgo IS, Goonewardena S, Weinert L, Coon P, Bardo D, et al. Rapid online quantification of left ventricular volume from real-time three-dimensional echocardiographic data. European Heart Journal. 2006;27(4):460–8. https://doi.org/10.1093/eurheartj/ehi666.
Keller M, Lang T, Schilling A, Nowak-Machen M, Rosenberger P, Magunia H. Novel mesh-derived right ventricular free wall longitudinal strain analysis by intraoperative three-dimensional transoesophageal speckle-tracking echocardiography: a comparison with conventional parameters. Int J Cardiovasc Imaging. 2019;35(12):2177–88. https://doi.org/10.1007/s10554-019-01669-8.
Keller M, Heller T, Lang T, Patzelt J, Schreieck J, Schlensak C, et al. Acute changes of global and longitudinal right ventricular function: an exploratory analysis in patients undergoing open-chest mitral valve surgery, percutaneous mitral valve repair and off-pump coronary artery bypass grafting. Cardiovasc Ultrasound. 2020;18(1):32. https://doi.org/10.1186/s12947-020-00218-xRetrospective analysis presenting different alterations of RV contraction following different procedures.
Labus J, Winata J, Schmidt T, Nicolai J, Tomko M, Sveric K, et al. Perioperative course of three-dimensional-derived right ventricular strain in coronary artery bypass surgery: a prospective, observational, pilot trial. J Cardiothorac Vasc Anesth. 2021;35(6):1628–37. https://doi.org/10.1053/j.jvca.2021.01.026.
Jin Y, Wang H, Wang Z, Jiang H, Tao D, Wu H. The evaluation of preoperative right ventricular diastolic dysfunction on coronary artery disease patients with left ventricular dysfunction. Echocardiography. 2014;31(10):1259–64. https://doi.org/10.1111/echo.12652.
Sumin AN, Korok EV, Sergeeva TJ. Preexisting right ventricular diastolic dysfunction and postoperative cardiac complications in patients undergoing nonemergency coronary artery bypass surgery. J Cardiothorac Vasc Anesth. 2021;35(3):799–806. https://doi.org/10.1053/j.jvca.2020.09.100Retrospective analysis detecting RVDD as a predictor of postoperative heart failure.
Chen T, Maslow AD. Right ventricular diastolic dysfunction: the missing link. Journal of Cardiothoracic and Vascular Anesthesia. 2021;35(3):807–10. https://doi.org/10.1053/j.jvca.2020.10.052.
Ebrahimi F, Kohanchi D, Gharedaghi MH, Petrossian V. Intraoperative assessment of left-ventricular diastolic function by two-dimensional speckle tracking echocardiography: relationship between pulmonary capillary wedge pressure and peak longitudinal strain rate during isovolumetric relaxation in patients undergoing coronary artery bypass graft surgery. J Cardiothorac Vasc Anesth. 2019;33(4):1014–21. https://doi.org/10.1053/j.jvca.2018.06.011.
Genovese D, Mor-Avi V, Palermo C, Muraru D, Volpato V, Kruse E, et al. Comparison between four-chamber and right ventricular–focused views for the quantitative evaluation of right ventricular size and function. Journal of the American Society of Echocardiography. 2019;32(4):484–94. https://doi.org/10.1016/j.echo.2018.11.014.
Kurt M, Tanboga IH, Isik T, Kaya A, Ekinci M, Bilen E, et al. Comparison of transthoracic and transesophageal 2-dimensional speckle tracking echocardiography. J Cardiothorac Vasc Anesth. 2012;26(1):26–31. https://doi.org/10.1053/j.jvca.2011.05.014.
Bolliger D, Seeberger MD, Kasper J, Bernheim A, Schumann RM, Skarvan K, et al. Different effects of sevoflurane, desflurane, and isoflurane on early and late left ventricular diastolic function in young healthy adults. Br J Anaesth. 2010;104(5):547–54. https://doi.org/10.1093/bja/aeq066.
Filipovic M, Wang J, Michaux I, Hunziker P, Skarvan K, Seeberger MD. Effects of halothane, sevoflurane and propofol on left ventricular diastolic function in humans during spontaneous and mechanical ventilation. Br J Anaesth. 2005;94(2):186–92. https://doi.org/10.1093/bja/aei028.
Rozental O, Thalappillil R, White RS, Tam CW. To swan or not to swan: indications, alternatives, and future directions. J Cardiothorac Vasc Anesth. 2021;35(2):600–15. https://doi.org/10.1053/j.jvca.2020.07.067.
Judge O, Ji F, Fleming N, Liu H. Current use of the pulmonary artery catheter in cardiac surgery: a survey study. J Cardiothorac Vasc Anesth. 2015;29(1):69–75. https://doi.org/10.1053/j.jvca.2014.07.016.
Habicher M, Zajonz T, Heringlake M, Böning A, Treskatsch S, Schirmer U, et al. S3 guidelines on intensive medical care of cardiac surgery patients: hemodynamic monitoring and cardiovascular system-an update. Anaesthesist. 2018;67(5):375–9. https://doi.org/10.1007/s00101-018-0433-6.
De Simone R, Wolf I, Mottl-Link S, Bottiger BW, Rauch H, Meinzer HP, et al. Intraoperative assessment of right ventricular volume and function. Eur J Cardiothorac Surg. 2005;27(6):988–93. https://doi.org/10.1016/j.ejcts.2005.01.022.
Milam AJ, Ghoddoussi F, Lucaj J, Narreddy S, Kumar N, Reddy V, et al. Comparing the mutual interchangeability of ECOM, FloTrac/Vigileo, 3D-TEE, and ITD-PAC cardiac output measuring systems in coronary artery bypass grafting. J Cardiothorac Vasc Anesth. 2021;35(2):514–29. https://doi.org/10.1053/j.jvca.2020.03.048.
Yano K, Toyama Y, Iida T, Hayashi K, Takahashi K, Kanda H. Comparison of right ventricular function between three-dimensional transesophageal echocardiography and pulmonary artery catheter. J Cardiothorac Vasc Anesth. 2021;35(6):1663–9. https://doi.org/10.1053/j.jvca.2020.11.012.
Raymond M, Gronlykke L, Couture EJ, Desjardins G, Cogan J, Cloutier J, et al. Perioperative right ventricular pressure monitoring in cardiac surgery. J Cardiothorac Vasc Anesth. 2019;33(4):1090–104. https://doi.org/10.1053/j.jvca.2018.08.198.
Grønlykke L, Couture EJ, Haddad F, Amsallem M, Ravn HB, Raymond M, et al. Preliminary experience using diastolic right ventricular pressure gradient monitoring in cardiac surgery. Journal of Cardiothoracic and Vascular Anesthesia. 2020;34(8):2116–25. https://doi.org/10.1053/j.jvca.2019.12.042.
Cronin B, O’Brien EO, Gu W, Banks D, Maus T. Intraoperative 3-dimensional echocardiography-derived right ventricular volumetric analysis in chronic thromboembolic pulmonary hypertension patients before and after pulmonary thromboendarterectomy. J Cardiothorac Vasc Anesth. 2019;33(6):1498–503. https://doi.org/10.1053/j.jvca.2018.09.038.
Kanemaru E, Yoshitani K, Kato S, Fujii T, Tsukinaga A, Ohnishi Y. Comparison of right ventricular function between patients with and without pulmonary hypertension owing to left-sided heart disease: assessment based on right ventricular pressure-volume curves. J Cardiothorac Vasc Anesth. 2020;34(1):143–50. https://doi.org/10.1053/j.jvca.2019.05.025.
Abuelkasem E, Wang DW, Subramaniam K. Pro: myocardial deformation imaging should be used perioperatively for assessment of cardiac function. J Cardiothorac Vasc Anesth. 2019;33(11):3196–200. https://doi.org/10.1053/j.jvca.2019.05.009.
Cios TJ, Roberts SM, Klick JC. Con: strain imaging should not be a routine part of the intraoperative TEE exam during cardiac surgery. J Cardiothorac Vasc Anesth. 2019;33(11):3201–3. https://doi.org/10.1053/j.jvca.2019.02.041.
Genovese D, Rashedi N, Weinert L, Narang A, Addetia K, Patel AR, et al. Machine learning–based three-dimensional echocardiographic quantification of right ventricular size and function: validation against cardiac magnetic resonance. Journal of the American Society of Echocardiography. 2019;32(8):969–77. https://doi.org/10.1016/j.echo.2019.04.001.
Chen X, Owen CA, Huang EC, Maggard BD, Latif RK, Clifford SP, et al. Artificial intelligence in echocardiography for anesthesiologists. J Cardiothorac Vasc Anesth. 2021;35(1):251–61. https://doi.org/10.1053/j.jvca.2020.08.048.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare there are no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Cardiovascular Anesthesia
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Labus, J., Uhlig, C. Role of Echocardiography for the Perioperative Assessment of the Right Ventricle. Curr Anesthesiol Rep 11, 306–317 (2021). https://doi.org/10.1007/s40140-021-00474-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40140-021-00474-1