Abstract
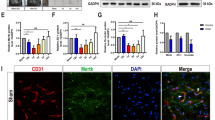
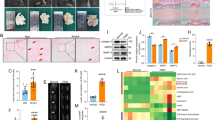
Neurogenic intermittent claudication (NIC), a classic symptom of lumbar spinal stenosis (LSS), is associated with neuronal apoptosis. To explore the novel therapeutic target of NIC treatment, we constructed the rat model of NIC by cauda equina compression (CEC) method and collected dorsal root ganglion (DRG) tissues, a region responsible for sensory and motor function, for mRNA sequencing. Bioinformatic analysis of mRNA sequencing indicated that upregulated metallothionein 2A (MT2A), an apoptosis-regulating gene belonging to the metallothionein family, might participate in NIC progression. Activated p38 MAPK mediated motor dysfunction following LSS and it was also found in DRG tissues of rats with NIC. Therefore, we supposed that MT2A might affect NIC progression by regulating p38 MAPK pathway. Then the rat model of NIC was used to explore the exact role of MT2A. Rats at day 7 post-CEC exhibited poorer motor function and had two-fold MT2A expression in DRG tissues compared with rats with sham operation. Co-localization analysis showed that MT2A was highly expressed in neurons, but not in microglia or astrocytes. Subsequently, neurons isolated from DRG tissues of rats were exposed to hypoxia condition (3% O2, 92% N2, 5% CO2) to induce cell damage. Gain of MT2A function in neurons was performed by lentivirus-mediated overexpression. MT2A overexpression inhibited apoptosis by inactivating p38 MAPK in hypoxia-exposed neurons. Our findings indicated that high MT2A expression was related to NIC progression, and MT2A overexpression protected against NIC through inhibiting activated p38 MAPK-mediated neuronal apoptosis in DRG tissues.






Similar content being viewed by others
Data availability
The data that support the findings of the current study are available from the corresponding author upon reasonable request.
References
Williamson E, Boniface G, Marian IR, Dutton SJ, Garrett A, Morris A, et al. The Clinical effectiveness of a physiotherapy delivered physical and psychological group intervention for older adults with neurogenic claudication: the BOOST randomized controlled trial. J Gerontol A Biol Sci Med Sci. 2022;77(8):1654–64. https://doi.org/10.1093/gerona/glac063.
Jensen RK, Harhangi BS, Huygen F, Koes B. Lumbar spinal stenosis. BMJ. 2021;373:n1581. https://doi.org/10.1136/bmj.n1581.
Cao YL, Duan Y, Zhu LX, Zhan YN, Min SX, Jin AM. TGF-β1, in association with the increased expression of connective tissue growth factor, induce the hypertrophy of the ligamentum flavum through the p38 MAPK pathway. Int J Mol Med. 2016;38(2):391–8. https://doi.org/10.3892/ijmm.2016.2631.
Walter KL, O’Toole JE. Lumbar Spinal Stenosis. JAMA. 2022;328(3):310. https://doi.org/10.1001/jama.2022.6137.
Ishimoto Y, Yoshimura N, Muraki S, Yamada H, Nagata K, Hashizume H, et al. Associations between radiographic lumbar spinal stenosis and clinical symptoms in the general population: the wakayama spine study. Osteoarthr Cartil. 2013;21(6):783–8. https://doi.org/10.1016/j.joca.2013.02.656.
Ma B, Shi J, Jia L, Yuan W, Wu J, Fu Z, et al. Over-expression of PUMA correlates with the apoptosis of spinal cord cells in rat neuropathic intermittent claudication model. PLoS ONE. 2013;8(5):e56580. https://doi.org/10.1371/journal.pone.0056580.
Deer TR, Kim CK, Bowman RG 2nd, Ranson MT, Yee BS. Study of percutaneous lumbar decompression and treatment algorithm for patients suffering from neurogenic claudication. Pain Physician. 2012;15(6):451–60.
Jensen RK, Lauridsen HH, Andresen ADK, Mieritz RM, Schiottz-Christensen B, Vach W. Diagnostic screening for lumbar spinal stenosis. Clin Epidemiol. 2020;12:891–905. https://doi.org/10.2147/CLEP.S263646.
Bussieres A, Cancelliere C, Ammendolia C, Comer CM, Zoubi FA, Chatillon CE, et al. Non-surgical interventions for lumbar spinal stenosis leading to neurogenic claudication: a clinical practice guideline. J Pain. 2021;22(9):1015–39. https://doi.org/10.1016/j.jpain.2021.03.147.
Chuang HC, Tsai KL, Tsai KJ, Tu TY, Shyong YJ, Jou IM, et al. Oxidative stress mediates age-related hypertrophy of ligamentum flavum by inducing inflammation, fibrosis and apoptosis through activating Akt and MAPK pathways. Aging (Albany NY). 2020;12(23):24168–83. https://doi.org/10.18632/aging.104105.
Yang Y, Xu J, Su Q, Wu Y, Li Q, Ma Z, et al. Lysophosphatidic acid induced apoptosis, DNA damage, and oxidative stress in spinal cord neurons by upregulating LPA4/LPA6 receptors. Mediat Inflamm. 2022;2022:1818758. https://doi.org/10.1155/2022/1818758.
Kobayashi S, Uchida K, Yayama T, Takeno K, Miyazaki T, Shimada S, et al. Motor neuron involvement in experimental lumbar nerve root compression : a light and electron microscopic study. Spine. 2007;32(6):627–34. https://doi.org/10.1097/01.brs.0000257559.84494.15.
Park SH, Hong JY, Kim WK, Shin JS, Lee J, Ha IH, et al. Effects of SHINBARO2 on rat models of lumbar spinal stenosis. Mediat Inflamm. 2019;2019:7651470. https://doi.org/10.1155/2019/7651470.
Ito T, Ohtori S, Inoue G, Koshi T, Doya H, Ozawa T, et al. Glial phosphorylated p38 MAP kinase mediates pain in a rat model of lumbar disc herniation and induces motor dysfunction in a rat model of lumbar spinal canal stenosis. Spine. 2007;32(2):159–67. https://doi.org/10.1097/01.brs.0000251437.10545.e9.
Shunmugavel A, Martin MM, Khan M, Copay AG, Subach BR, Schuler TC, et al. Simvastatin ameliorates cauda equina compression injury in a rat model of lumbar spinal stenosis. J Neuroimmune Pharmacol. 2013;8(1):274–86. https://doi.org/10.1007/s11481-012-9419-3.
Sung HC, Chang KS, Chen ST, Hsu SY, Lin YH, Hou CP, et al. Metallothionein 2A with antioxidant and antitumor activity is upregulated by caffeic acid phenethyl ester in human bladder carcinoma cells. Antioxidants (Basel). 2022. https://doi.org/10.3390/antiox11081509.
Kwak SY, Jang WI, Park S, Cho SS, Lee SB, Kim MJ, et al. Metallothionein 2 activation by pravastatin reinforces epithelial integrity and ameliorates radiation-induced enteropathy. EBioMedicine. 2021;73:103641. https://doi.org/10.1016/j.ebiom.2021.103641.
Lores-Padin A, Mavrakis E, Fernandez B, Garcia M, Gonzalez-Iglesias H, Pereiro R, et al. Gold nanoclusters as elemental label for the sequential quantification of apolipoprotein E and metallothionein 2A in individual human cells of the retinal pigment epithelium using single cell-ICP-MS. Anal Chim Acta. 2022;1203:339701. https://doi.org/10.1016/j.aca.2022.339701.
Voinsky I, Zoabi Y, Shomron N, Harel M, Cassuto H, Tam J, et al. Blood RNA Sequencing Indicates upregulated BATF2 and LY6E and downregulated ISG15 and MT2A expression in children with autism spectrum disorder. Int J Mol Sci. 2022;23(17):9843. https://doi.org/10.3390/ijms23179843.
Liu X, Quan J, Shen Z, Zhang Z, Chen Z, Li L, et al. Metallothionein 2A (MT2A) controls cell proliferation and liver metastasis by controlling the MST1/LATS2/YAP1 signaling pathway in colorectal cancer. Cancer Cell Int. 2022;22(1):205. https://doi.org/10.1186/s12935-022-02623-w.
Wahyudi LD, Yu SH, Cho MK. The effect of curcumin on the cadmium-induced mitochondrial apoptosis pathway by metallothionein 2A regulation. Life Sci. 2022;310:121076. https://doi.org/10.1016/j.lfs.2022.121076.
Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12(1):1–21. https://doi.org/10.1089/neu.1995.12.1.
Takenobu Y, Katsube N, Marsala M, Kondo K. Model of neuropathic intermittent claudication in the rat: methodology and application. J Neurosci Methods. 2001;104(2):191–8. https://doi.org/10.1016/s0165-0270(00)00342-3.
Zanos TP, Silverman HA, Levy T, Tsaava T, Battinelli E, Lorraine PW, et al. Identification of cytokine-specific sensory neural signals by decoding murine vagus nerve activity. Proc Natl Acad Sci U S A. 2018;115(21):E4843-e4852. https://doi.org/10.1073/pnas.1719083115.
Sui X, Kong N, Ye L, Han W, Zhou J, Zhang Q, et al. p38 and JNK MAPK pathways control the balance of apoptosis and autophagy in response to chemotherapeutic agents. Cancer Lett. 2014;344(2):174–9. https://doi.org/10.1016/j.canlet.2013.11.019.
Takahashi H, Aoki Y, Saito J, Nakajima A, Sonobe M, Akatsu Y, et al. Time course of changes in serum oxidative stress markers to predict outcomes for surgical treatment of lumbar degenerative disorders. Oxid Med Cell Longev. 2020;2020:5649767. https://doi.org/10.1155/2020/5649767.
Zhai G, Liang W, Xu Y. High expression of lysophosphatidic acid induces nerve injury in LSS patients via AKT mediated NF-κB p65 pathway. Front Pharmacol. 2021;12:641435. https://doi.org/10.3389/fphar.2021.641435.
Lee J, Choi H, Park C, Jeon S, Yune T. Jmjd3 mediates neuropathic pain by inducing macrophage infiltration and activation in lumbar spinal stenosis animal model. Int J Mol Sci. 2021. https://doi.org/10.3390/ijms222413426.
Fukutake T. Intermittent claudication secondary to spine and/or spinal cord diseases. Brain Nerve. 2021;73(6):671–83. https://doi.org/10.11477/mf.1416201818.
Deer T, Sayed D, Michels J, Josephson Y, Li S, Calodney AK. A review of lumbar spinal stenosis with intermittent neurogenic claudication: disease and diagnosis. Pain Med. 2019;20(Suppl 2):S32-s44.
Desai A, Ball PA, Bekelis K, Lurie J, Mirza SK, Tosteson TD, et al. SPORT: Does incidental durotomy affect longterm outcomes in cases of spinal stenosis? Neurosurgery. 2015. https://doi.org/10.1227/01.neu.0000462078.58454.f4. (discussion S63).
Gao QY, Wei FL, Li T, Zhu KL, Du MR, Heng W, et al. Oblique lateral interbody fusion vs minimally invasive transforaminal lumbar interbody fusion for lumbar spinal stenosis: a retrospective cohort study. Front Med (Lausanne). 2022. https://doi.org/10.3389/fmed.2022.829426.
Ahn Y, Oh HK, Kim H, Lee SH, Lee HN. Percutaneous endoscopic lumbar foraminotomy: an advanced surgical technique and clinical outcomes. Neurosurgery. 2014;75(2):124–33. https://doi.org/10.1227/neu.0000000000000361. (discussion 132-123).
Berenpas F, Weerdesteyn V, Geurts AC, van Alfen N. Long-term use of implanted peroneal functional electrical stimulation for stroke-affected gait: the effects on muscle and motor nerve. J Neuroeng Rehabil. 2019;16(1):86. https://doi.org/10.1186/s12984-019-0556-2.
Kobayashi S. Pathophysiology, diagnosis and treatment of intermittent claudication in patients with lumbar canal stenosis. World J Orthop. 2014;5(2):134–45. https://doi.org/10.5312/wjo.v5.i2.134.
Gitelman A, Hishmeh S, Morelli BN, Joseph SA Jr, Casden A, Kuflik P, et al. Cauda equina syndrome: a comprehensive review. Am J Orthop (Belle Mead NJ). 2008;37(11):556–62.
Liu Y, Yang R, Kong Q, Wang Y, Zhang B, Sun J, et al. Metabolomic changes in rat model of cauda equina injury. World Neurosurg. 2017;102:449–58. https://doi.org/10.1016/j.wneu.2017.03.072.
Khan M, Shunmugavel A, Dhammu TS, Matsuda F, Singh AK, Singh I. Oral administration of cytosolic PLA2 inhibitor arachidonyl trifluoromethyl ketone ameliorates cauda equina compression injury in rats. J Neuroinflamm. 2015;12:94. https://doi.org/10.1186/s12974-015-0311-y.
Kaur H, Xu N, Doycheva DM, Malaguit J, Tang J, Zhang JH. Recombinant Slit2 attenuates neuronal apoptosis via the Robo1-srGAP1 pathway in a rat model of neonatal HIE. Neuropharmacology. 2019;158:107727. https://doi.org/10.1016/j.neuropharm.2019.107727.
Chio JCT, Wang J, Badner A, Hong J, Surendran V, Fehlings MG. The effects of human immunoglobulin G on enhancing tissue protection and neurobehavioral recovery after traumatic cervical spinal cord injury are mediated through the neurovascular unit. J Neuroinflamm. 2019;16(1):141. https://doi.org/10.1186/s12974-019-1518-0.
Kim H, Hong JY, Jeon WJ, Lee J, Lee YJ, Ha IH. Melittin regulates iron homeostasis and mediates macrophage polarization in rats with lumbar spinal stenosis. Biomed Pharmacother. 2022;156:113776. https://doi.org/10.1016/j.biopha.2022.113776.
Kim H, Hong JY, Jeon WJ, Lee J, Ha IH. Evaluation of the effects of differences in silicone hardness on rat model of lumbar spinal stenosis. PLoS ONE. 2021;16(5):e0251464. https://doi.org/10.1371/journal.pone.0251464.
Hsu YC, Chuang HC, Tsai KL, Tu TY, Shyong YJ, Kuo CH, et al. Administration of N-acetylcysteine to regress the fibrogenic and proinflammatory effects of oxidative stress in hypertrophic ligamentum flavum cells. Oxid Med Cell Longev. 2022;2022:1380353. https://doi.org/10.1155/2022/1380353.
Tan J, Shi J, Shi G, Liu Y, Liu X, Wang C, et al. Changes in compressed neurons from dogs with acute and severe cauda equina constrictions following intrathecal injection of brain-derived neurotrophic factor-conjugated polymer nanoparticles. Neural Regen Res. 2013;8(3):233–43. https://doi.org/10.3969/j.issn.1673-5374.2013.03.005.
Mehlenbacher MR, Elsiesy R, Lakha R, Villones RLE, Orman M, Vizcarra CL, et al. Metal binding and interdomain thermodynamics of mammalian metallothionein-3: enthalpically favoured Cu(+) supplants entropically favoured Zn(2+) to form Cu(4) (+) clusters under physiological conditions. Chem Sci. 2022;13(18):5289–304. https://doi.org/10.1039/d2sc00676f.
Na H, Li X, Zhang X, Xu Y, Sun Y, Cui J, et al. lncRNA STEAP3-AS1 modulates cell cycle progression via affecting CDKN1C expression through steap3 in colon cancer. Mol Ther Nucleic Acids. 2020;21:480–91. https://doi.org/10.1016/j.omtn.2020.06.011.
Wang XL, Schnoor M, Yin LM. Metallothionein-2: An emerging target in inflammatory diseases and cancers. Pharmacol Ther. 2023;244:108374. https://doi.org/10.1016/j.pharmthera.2023.108374.
Huang X, Deng J, Xu T, Xin W, Zhang Y, Ruan X. Downregulation of metallothionein-2 contributes to oxaliplatin-induced neuropathic pain. J Neuroinflamm. 2021;18(1):91. https://doi.org/10.1186/s12974-021-02139-6.
Li J, Lei Y, Zhao Y. Metallothionein-2A protects cardiomyocytes from hypoxia/reper-fusion through inhibiting p38. Cell Biochem Biophys. 2023;81(1):69–75. https://doi.org/10.1007/s12013-022-01118-9.
Subhramanyam CS, Wang C, Hu Q, Dheen ST. Microglia-mediated neuroinflammation in neurodegenerative diseases. Semin Cell Dev Biol. 2019;94:112–20. https://doi.org/10.1016/j.semcdb.2019.05.004.
Chen NN, Wei F, Wang L, Cui S, Wan Y, Liu S. Tumor necrosis factor alpha induces neural stem cell apoptosis through activating p38 MAPK pathway. Neurochem Res. 2016;41(11):3052–62. https://doi.org/10.1007/s11064-016-2024-8.
Acknowledgements
This research was supported by the National Natural Science Foundation of China (82071365).
Author information
Authors and Affiliations
Contributions
C.G. W. and Z.C. W.: conceptualization, interpretation of results, and writing—original draft. Y. Z., X.J. D., and J.H. X.: investigation and data collection. J.W. Z, W. X., Z.R. W., and W. L.: data analysis and visualization. B. M.: conceptualization and writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Animal experiments were approved by the Animal Welfare Ethics Committee of Tongji Hospital (Approval number: 2021-DW-(101)). All operations were performed in accordance with the US National Research Council's Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, C., Wang, Z., Zi, Y. et al. Compensatory upregulation of MT2A alleviates neurogenic intermittent claudication through inhibiting activated p38 MAPK-mediated neuronal apoptosis. Human Cell 37, 675–688 (2024). https://doi.org/10.1007/s13577-024-01043-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-024-01043-4