Abstract
Hypocreomycetidae is a highly diverse group with species from various habitats. This subclass has been reported as pathogenic, endophytic, parasitic, saprobic, fungicolous, lichenicolous, algicolous, coprophilous and insect fungi from aquatic and terrestrial habitats. In this study, we focused on freshwater fungi of Hypocreomycetidae which resulted 41 fresh collections from China and Thailand. Based on morphological and phylogenetic analyses, we identified 26 species that belong to two orders (Hypocreales and Microascales) and six families (Bionectriaceae, Halosphaeriaceae, Microascaceae, Nectriaceae, Sarocladiaceae and Stachybotryaceae). Ten new species are introduced and 13 new habitats and geographic records are reported. Mariannaea superimposita, Stachybotrys chartarum and S. chlorohalonatus are recollected from freshwater habitats in China. Based on phylogenetic analysis of combined LSU, ITS, SSU, rpb2 and tef1-α sequences data, Emericellopsis is transferred to Hypocreales genera incertae sedis; Pseudoacremonium is transferred to Bionectriaceae; Sedecimiella is placed in Nectriaceae; Nautosphaeria and Tubakiella are excluded from Halosphaeriaceae and placed in Microascales genera incertae sedis; and Faurelina is excluded from Hypocreomycetidae. Varicosporella is placed under Atractium as a synonym of Atractium. In addition, phylogenetic analysis and divergence time estimates showed that Ascocodina, Campylospora, Cornuvesica and Xenodactylariaceae form distinct lineages in Hypocreomycetidae and they evolved in the family/order time frame. Hence, a new order (Xenodactylariales) and three new families (Ascocodinaceae, Campylosporaceae and Cornuvesicaceae) are introduced based on phylogenetic analysis, divergence time estimations and morphological characters. Ancestral character state analysis is performed for different habitats of Hypocreomycetidae including freshwater, marine and terrestrial taxa. The result indicates that marine and freshwater fungi evolved independently from terrestrial ancestors. The results further support those early diverging clades of this subclass, mostly comprising terrestrial taxa and freshwater and marine taxa have been secondarily derived, while the crown clade (Nectriaceae) is represented in all three habitats. The evolution of various morphological adaptations towards their habitual changes are also discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hypocreomycetidae (Sordariomycetes) is an ecologically and morphologically diverse group. It comprises plant or human pathogens, endophytes, parasites, saprobes, fungicolous, lichenicolous, algicolous, coprophilous and insect fungi from various habitats, including freshwater, marine and terrestrial habitats (Maharachchikumbura et al. 2015; Hyde et al. 2017, 2020a). The members of Hypocreomycetidae have light-colored perithecia, nonamyloid or amyloid ascal rings, or those which lack apical rings and most taxa lack true paraphyses (Zhang et al. 2006). Hypocreomycetidae was established by Eriksson and Winka (1997) based on morphology and a single gene (SSU) phylogenetic analysis. Eriksson (2006) showed that Hypocreomycetidae formed a monophyletic clade within Sordariomycetes and four orders viz. Coronophorales, Halosphaeriales, Hypocreales and Microascales were accepted in Hypocreomycetidae. Coronophorales, Halosphaeriales and Hypocreales are monophyletic, while Microascales is paraphyletic. Zhang et al. (2006) updated the phylogenetic tree for Sordariomycetes using LSU, SSU, tef1-α and rpb2 sequence data. Their analysis concurred with Eriksson (2006), except Melanospora, which formed a sister clade with Coronophorales and can be recognized as a distinct order. Thus, a new order Melanosporales was introduced to accommodate Melanospora (Zhang et al. 2006). Subsequently, Tang et al. (2007) accepted five orders in Hypocreomycetidae. In contrast, Hibbett et al. (2007) accepted only four orders in the subclass whereas Halosphaeriales was placed under Microascales based on phylogenetic analysis. Lumbsch and Huhndorf (2010) accepted four orders and 18 families in Hypocreomycetidae.
Boonyuen et al. (2011) and Réblová et al. (2011) introduced Savoryellales and Glomerellales in Hypocreomycetidae, respectively. Maharachchikumbura et al. (2015) re-evaluated the classification of Sordariomycetes based on LSU, SSU, tef1-α and rpb2 sequence data. A new order, Falcocladiales, was added to Hypocreomycetidae; thus, seven orders and 30 families were accepted in this subclass (Maharachchikumbura et al. 2015). Consequently, Jones et al. (2015) introduced a new order Torpedosporales and Maharachchikumbura et al. (2016) placed Pleurotheciales in Hypocreomycetidae. Yang et al. (2016) introduced Fuscosporellales in the subclass. Hongsanan et al. (2017) and Hyde et al. (2017) showed that Conioscyphales, Fuscosporales, Pleurotheciales and Savoryellales formed a monophyletic clade, sister to Hypocreomycetidae. Therefore, these four orders were transferred to a newly introduced subclass Savoryellomycetidae. This was confirmed by Dayarathne et al. (2019) based on phylogenetic analysis and divergent time estimates. In addition, Parasympodiellales was assigned in Hypocreomycetidae and Melanosporales was placed under Coronophorales (Hernández-Restrepo et al. 2017; Hongsanan et al. 2017). Seven orders Coronophorales, Falcocladiales, Glomerellales, Hypocreales, Parasympodiellales, Torpedosporales and Microascales were accepted in Hypocreomycetidae based on both phylogenetic analysis and divergence time estimates (Hongsanan et al. 2017; Hyde et al. 2017, 2020a; Hernández-Restrepo et al. 2017). The recent treatment of Hypocreomycetidae was provided by Wijayawardene et al. (2022), who accepted eight orders (including Cancellidiales) and 38 families. Xiao et al. (2023) introduced a new family Polycephalomycetaceae in Hypocreales. Eight orders and 39 families are currently accepted in the Hypocreomycetidae (Wijayawardene et al. 2022). However, few orders and families remain polyphyletic; thus, this subclass needs to be revised (Hyde et al. 2020a; Huang et al. 2021).
The evolution study of Hypocreomycetidae is mainly focused on Halosphaeriaceae as this family comprises most marine species with a few species from freshwater and terrestrial habitats (Sakayaroj et al. 2011; Jones et al. 2017, 2019). Several studies have discussed the evolution of morphological characters and the origin of Halosphaeriaceae (Sakayaroj et al. 2011; Jones et al. 2017, 2019). However, there is no specific studies on the evolution of Hypocreomycetidae especially for freshwater fungi.
Several studies have recommended the multiple origins of freshwater fungi and the evolution of freshwater ascomycetes from terrestrial habitats (Shearer 1993; Vijaykrishna et al. 2006; Hyde et al. 2021). Belliveau et al. (2005) investigated the evolution of aquatic hyphomycetes based on molecular data and their results demonstrated multiple origins of aquatic hyphomycetes. However, their study did not obtain any firm conclusions concerning their ancestors. It stated that the sexual and asexual morphs have concurrent adaptations to freshwater habitats as several sexual morphs of freshwater hyphomycetes have been reported on tree branches decaying in the water. However, the evolution of aquatic hyphomycetes, either terrestrial asexual or sexual morphs, is undetermined, and this needs further studies based on molecular and morphological data (Webster and Descals 1979; Webster 1992). Vijaykrishna et al. (2006) initially investigated the origin of freshwater ascomycetes based on the molecular clock, and their results stated that freshwater ascomycetes originated from terrestrial fungi with multiple and independent evolution. Freshwater ascomycetes have unique adaptations to survive in freshwater habitats; an example is the freshwater ascomycetes decompose lignocellulose in woody litter, softening the wood, which is thought to be a better adaptation for degrading wood in water-logged conditions. Another example is the unique morphological characters of freshwater ascomycetes, such as the asci with massive apical rings which help eject ascospores into the air and underwater to dispersal in freshwater. The appendages of ascospores can help the species to attached to the substrates in the running water (Ho and Hyde 2000; Hyde and Goh 2003; Vijaykrishna et al. 2006). However, these adaptions have also been found in terrestrial fungi, indicating that freshwater ascomycetes share a common ancestor with terrestrial ascomycetes (Shearer et al. 2009). Recently, Hyde et al. (2021) investigated the evolution of freshwater Diaporthomycetidae based on phylogenetic analysis and divergence time estimates; their result indicated that freshwater Diaporthomycetidae have evolved from terrestrial fungi and has evolved on several occasions.
Studies concluded that the fungi originated from aquatic habitats and then migrated to terrestrial habitats (Vijaykrishna et al. 2006; Beakes and Sekimoto 2009; Beakes et al. 2012; Jones et al. 2014). Vijaykrishna et al. (2006) made a conclusion based on the previous studies (e.g. Shearer 1993; Wong et al. 1998; Hyde and Wong 2000; Cai et al. 2003) that fungi may occur as pathogens, saprobes or endophytes on plants, then become adapted to the aquatic environment, when these plants invaded water. Some studies stated that fungi originated from the sea and then migrated to terrestrial habitats (Beakes and Sekimoto 2009; Jones et al. 2011, 2014). There is still controversial, and it would be interesting if further studies could focus on this. Hypocreomycetidae is widely distributed worldwide and has been reported in freshwater, terrestrial and marine environments. One-hundred and fifty-six Hypocreomycetidae species have been reported from freshwater habitats and are distributed in five orders viz. Coronophorales, Glomerellales, Hypocreales, Microascales and Torpedosporales (Calabon et al. 2021, 2022). Thus, studying the evolution of Hypocreomycetidae is important for understanding the possible transition and evolution of aquatic and terrestrial ascomycetes.
In this study, we aim to (1) investigate freshwater fungi in Hypocreomycetidae with fresh collections based on morphological and multi-gene phylogenetic analyses; (2) establish the divergence time of orders and families in Hypocreomycetidae based on molecular clock analyses and (3) explore the evolution of Hypocreomycetidae based on ancestral state analysis.
Materials and methods
Isolation and morphological examination
Samples (submerged woods) were collected from freshwater habitats (lakes and streams) in China and Thailand. The samples were brought to the laboratory in plastic bags. Sample incubation, observation and morphological studies were done following the methods outlined by Luo et al. (2018). Fungal species were isolated using single spore isolation following the method described in Senanayake et al. (2020). Germinating ascospores and conidia were transferred to fresh potato dextrose agar (PDA) media and incubated at room temperature for 2–4 weeks, and cultures were grown for 1–2 months. The cultures obtained from Thailand are deposited at the Mae Fah Luang University Culture Collection (MFLUCC) and the cultures obtained from China are deposited at the Kunming Institute of Botany Culture Collection (KUNCC). Herbarium specimens from China and Thailand were prepared following the methods provided by Luo et al. (2018), and the herbariums were deposited at Mae Fah Luang University (MFLU) and Herbarium of Cryptogams Kunming Institute of Botany Academia Sinica (Herb. KUN-HKAS) respectively. Facesoffungi and Index Fungorum numbers are registered as outlined in Jayasiri et al. (2015) and Index Fungorum (2023). The descriptions of the species are added to GMS database (Chaiwan et al. 2021).
DNA extraction, PCR amplification and sequencing
Genomic DNA was extracted from fungal mycelium or directly from the ascomatal tissue thalli of fungi as outlined by Wanasinghe et al. (2018). EZ gene™ Fungal gDNA Kit (GD2416) was used to extract genomic DNA following the manufacturer’s instructions. The primers are summarized in Table 1. The amplification reactions were performed in 25 μL of PCR mixtures containing 12.5 μL of 2 × Power Taq PCR Master Mix (a premix and ready-to-use solution, including 0.1 Units/μL Taq DNA Polymerase, 500 μm dNTP Mixture each (dATP, dCTP, dGTP, dTTP), 20 mM Tris–HCl pH 8.3, 100 Mm KCl, 3 mM MgCl2, stabilizer and enhancer), 1 μL of each primer, 1 μL DNA template and 9.5 μL nuclease-free water. PCR amplification was confirmed on 1% agarose electrophoresis gels stained with ethidium bromide. Purification and sequencing of PCR products were carried out using the above-mentioned PCR primers at Kunming Tsingke Biological Engineering Technology and Services Co., Ltd. (Kunming, P.R. China).
Sequence alignment and phylogenetic analyses
Sequences featuring a high degree of similarity were determined from a BLAST search for each gene to identify the closest matches with taxa in Sordariomycetes. The sequences were assembled using BioEdit and aligned with MAFFT v.7 online program (http://mafft.cbrc.jp/alignment/server/) (Katoh and Standleym 2013; Katoh et al. 2019) and final improvements were made when necessary, using BioEdit v7.2.3 (Hall 1999).
Maximum likelihood (ML) analysis was performed using RAxML-HPC v.8 (Stamatakis 2006; Stamatakis et al. 2008) on the XSEDE Teragrid of the CIPRES Science Gateway online flatform (Miller et al. 2010) with rapid bootstrap analysis, followed by 1000 bootstrap replicates. The final tree was selected amongst suboptimal trees from each run by comparing likelihood scores under the GTRGAMMA substitution model.
MP bootstrap analyses were performed with PAUP v4.0b10 (Swofford 2002). The analysis was performed using the heuristic search option with 1000 random taxa addition and tree bisection and reconnection (TBR) as the branch-swapping algorithm. All characters were unordered and of equal weight, and gaps were treated as missing data. Maxtrees were unlimited, branches of zero length were collapsed and all multiple, equally parsimonious trees were saved. Clade stability was assessed using a bootstrap (BT) analysis with 1000 replicates, each with 10 replicates of the random stepwise addition of taxa (Hillis and Bull 1993).
Bayesian analysis was performed by MrBayes v3.1.2 (Ronquist et al. 2012). The model of evolution was estimated by MrModeltest 2.2 (Nylander 2004). Posterior probabilities (Rannala and Yang 1996) were performed by Markov Chain Monte Carlo Sampling (MCMC) in MrBayes v. 3.1.2. Six simultaneous Markov Chains were run for 1 billion generations, and trees were sampled every 1000 generation (resulting in 100,000 trees). The first 20,000 trees representing the burn-in phase of the analyses were discarded and the remaining 80,000 (post-burning) trees used for calculating posterior probabilities (PP) in the majority rule consensus tree.
Several recent studies have discussed the criteria to define a species (Boekhout et al. 2021; Chethana et al. 2021; Maharachchikumbura et al. 2021; Voigt et al. 2021). Maharachchikumbura et al. (2021) detailed the integrative approaches including morphological species concept (MSC), biological species concept (BSC), and Evolutionary/phylogenetic species concept (PSC) with chemotaxonomy and divergence time estimation for species delimitation in Ascomycota. In this study, the new species and families were established based on morphological characters, phylogenetic analysis and divergence time estimates. In addition, we also performed base pair comparison for the newly described taxa following Jeewon and Hyde (2016).
Calibration, divergence time and evolutionary rate estimations
This study used one secondary and a fossil calibration for the divergence time estimates. Paleoophiocordyceps coccophagus, a fossil of Hypocreales, which is similar to the asexual morph of Hirsutella and Hymenostilbe (Sung et al. 2007) that are synonyms of Ophiocordyceps (Ophiocordycipitaceae, Hypocreales; Quandt et al. 2014) was used to calibrate the Ophiocordyceps crown, using an exponential distribution (offset = 100, mean = 27.5, with 97.5% CI of 200 MYA) (Sung et al. 2008; Samarakoon et al. 2016). The crown age of Sordariomycetes with a normal distribution (mean = 250, SD = 40, with 97.5% CI = 338 MYA) was selected as a secondary calibration point (Hyde et al. 2017).
Divergence time estimates were carried out by BEAST v 1.8.4 (Drummond et al. 2012). Aligned sequence data were partitioned separately for LSU, ITS, SSU, rpb2 and tef1-α dataset, and loaded to prepare an XML file constructed in BEAUti v1.8.0. The substitution models were selected based on jModeltest2.1.1; GTR + I + G for ITS and LSU, TIM3 + I + G for SSU, TrN + I + G for tef1-α and TIM2 + I + G for rpb2. However, the models TIM2, TIM3 and TrN were unavailable in BEAUti 1.8.4; thus, TN93 was selected by setting the ‘‘All Equal’’ for the base frequencies. An uncorrelated relaxed clock model (Drummond and Rambaut 2007) with a lognormal distribution of rates for each gene estimate was used for the analyses. We used a Yule tree prior, which assumes a constant speciation rate per lineage, and a randomly generated starting tree (Hyde et al. 2017). The analysis was run for 300 million generations and parameters were sampled every 30,000 generations. The effective sample sizes were checked in Tracer v.1.6 (Rambaut et al. 2013), and the acceptable values were greater than 200. Maximum clade creditability (MCC) trees were annotated using TreeAnnotator v1.8.0 and then visualized in FigTree v.1.4.2 (Rambaut 2014).
Ancestral character state analyses
Ancestral character state analysis (Thiyagaraja et al. 2020, 2021) was carried out to reconstruct the evolutionary relationship of habitual changes in Hypocrealmycetidae. The following states were established: freshwater, marine and terrestrial fungi. The platform Reconstruct Ancestral State in Phylogenies (RASP 3.2.1) was used to construct ancestral character analysis, using the Bayesian Binary MCMC based on the divergence time estimate tree (Yu et al. 2015, 2019). This approach was performed and visualized in RASP 3.2.1 using settings: 10 chains, a sampling frequency of 100, a temperature of 0.1, state frequencies fixed (JC), and among-site rate variation equal. The trees were edited using Microsoft PowerPoint 2013 and converted to jpeg files using Adobe Photoshop CS6 Extended 10.0 (Adobe Systems. U.S.A.).
Results
Phylogenetic analysis
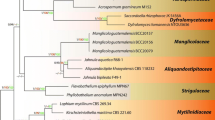
The multi-gene dataset (LSU, ITS, SSU, rpb2 and tef1-α) was used to reveal the relationships of orders and families in Hypocreomycetidae (Fig. 1). The alignment comprised 527 taxa from six subclasses (Diaporthomycetidae, Hypocreomycetidae, Lulworthiomycetidae, Savoryellomycetidae, Sordariomycetidae and Xylariomycetidae) of Sordariomycetes with four Dothideomycetes taxa as outgroup. The dataset comprised 4577 characters including gaps (LSU: 902 bp, ITS: 639 bp, SSU: 1010 bp, rpb2: 1072 bp, tef1-α: 954 bp). The statistical analyses resulted in largely the same topology with high support for most branches in the ML and BI analyses and with similar overall topologies of order and family level relationships in agreement with previous work based on ML and BI analyses (Hongsanan et al. 2017; Hyde et al. 2020a). Therefore, the best scoring RAxML tree is shown in Fig. 1, with a likelihood value of − 189,870.011043.
Phylogenetic tree based on RAxML analyses of combined LSU, SSU, ITS, tef1-α and rpb2 sequence data. Maximum likelihood bootstrap ≥ 75% (MLBS) and Bayesian posterior probabilities ≥ 0.95 (PP) are indicated at the nodes. The freshwater species are in red and newly obtained strains are in bold. The tree is rooted with Aureobasidium microstictum, Dothidea insculpta, D. sambuci and Pseudoseptoria collariana
The present phylogenetic analysis comprises 41 families including three new families (Ascocodinaceae, Campylosporaceae and Cornuvesicaceae). Most freshwater taxa in Hypocreomycetidae are included in the phylogenetic analysis except those species which lack sequence data in GenBank. Phylogenetic analysis showed that freshwater taxa are distributed in 10 families viz. Bionectriaceae, Campylosporaceae, Halosphaeriaceae, Juncigenaceae, Microascaceae, Nectriaceae, Reticulascaceae, Sarocladiaceae, Stachybotryaceae and Triadelphiaceae (Fig. 1). Our fresh collections clustered within two orders (Hypocreales and Microascales) and six families (Nectriaceae, Bionectriaceae, Stachybotryaceae, Sarocladiaceae, Halosphaeriaceae and Microascaceae) which represent 26 species including10 new species, 13 new records and 3 new collections. The acquired phylogenetic results are discussed where applicable in the notes below.
Molecular clock analysis
In recent years, divergence time estimates have been used in fungal taxonomic studies, especially in ranking higher taxa. Hyde et al. (2017) and Liu et al. (2017a) have recommended the range to recognize subclasses, orders, and families in the classes Sordariomycetes and Dothideomycetes. In Sordariomycetes, the stem ages of orders and families are recommended within 150–250 MYA and 50–150 MYA, respectively. According to the divergence time estimates (Fig. 2), the crown and stem ages of Sordariomycetes are 302 MYA and 323 MYA (Fig. 2), which are quite similar to Hyde et al. (2020a). The crown and stem ages of Hypocreomycetidae are 290 MYA and 302 MYA, respectively. The stem ages of all the orders in this subclass are listed in Table 2, including a new order Xenodactylariales. The phylogenetic analysis and divergence time estimates support all orders.
Ancestral character state reconstruction of terrestrial, freshwater and marine habitats in Hypocreomycetidae and maximum clade credibility (MCC) tree with divergence time estimates for Hypocreomycetidae. Multistate coding datasets analyzed with Bayesian Binary MCMC approaches. Pie charts at terminals show the representative character and the internal nodes represent the marginal probabilities for each alternative ancestral area. The divergence time are shown at the nodes (MYA)
The stem ages of the orders are compared with previous studies (Hyde et al. 2017, 2020a). In our study, the stem ages of Falcocladiales, Glomerellales, Hypocreales and Microascales are similar to the previous studies (Table 2). However, the divergence time of Coronophorales and Torpedosporales are older than Hyde et al. (2017, 2020a) (Table. 1). The different fossils, models and substitution rate variation can cause different results of divergence times (Beimforde et al. 2014). Our study uses one fossil data (Paleoophiocordyceps coccophagus and a secondary calibration (crown age of Sordariomycetes). In contrast, Hyde et al. (2017) used one fossil datum (Paleoophiocordyceps coccophagus) and two secondary calibrations (the divergence time of Sordariomycetes and Leotiomycetes and crown age of Sordariomycetes) and Hyde et al. (2020a) used two secondary calibrations (crown ages of Sordariomycetes and Dothideomycetes). In addition, Hyde et al. (2017) mentioned that the taxon sampling and number of base pair differences between fungal groups could affect the crown node age, and the crown node age can become older by including more taxa in the data set. Our analysis is focused on the subclass Hypocreomycetidae and the taxa were mostly selected from Hypocreomycetidae. In comparison, Hyde et al. (2017, 2020a) focused on the class Sordariomycetes. Thus, the divergence time of Coronophorales and Torpedosporales were different from that of Hyde et al. (2017, 2020a).
Ancestral character analysis
Savoryellomycetidae, Diaporthomycetidae, Sordariomycetidae, Lulworthiomycetidae and Xylariomycetidae show a terrestrial ancestor. Hypocreomycetidae also shares terrestrial as the common ancestor, whereas marine and freshwater taxa have independently evolved. Within the Hypocreales clade, the Stachybotryaceae clade comprised mostly freshwater taxa and clustered together with mostly terrestrial Incertae sedis species with terrestrial ancestor. Gliomastix represents marine, terrestrial and newly sequenced freshwater fungi which were recovered in the Bionectriaceae clade with several terrestrial and marine fungal genera. Clonostachys and its sexual genus Bionectria mainly comprised terrestrial fungi except for two newly sequenced species which were reported from freshwater habitats. All these genera represent Bionectriaceae which formed a sister clade to the mostly marine fungal genera Emericellopsis with a marine ancestor which had high support. Calcarisporiaceae, Clavicipitaceae, Cocoonihabitaceae, Cordycipitaceae, Flammocladiellaceae, Hypocreaceae, Niessliaceae, Ophiocordycipitaceae and Polycephalomycetaceae show terrestrial ancestor and marine and freshwater taxa show independent evolution. The Myrotheciomycetaceae clade mostly comprises terrestrial fungi with terrestrial ancestors.
Cai et al. (2010) reported the first freshwater Mariannaea species and expected a large discovery of freshwater taxa (Cai et al. 2010; Hu et al. 2017). Mariannaea humicola and Mariannaea punicea were recorded from soil (Lombard et al. 2015; Hu et al. 2017), while all other species, including our newly sequenced strains reported from freshwater habitats. Thelonectria are mostly saprobes in terrestrial habitats (Salgado-Salazar et al. 2016), while T. discophora (= Nectria discophora) was reported from aquatic (Shearer and Webster 1991) and terrestrial habitats (Zeng and Zhuang 2013). Our newly sequenced strains clustered together with terrestrial species of Thelonectria with freshwater ancestor and the node was not statistically supported. Throughout the Nectriaceae clade, several genera such as Acremonium, Atractium, Fusicolla, Rodentomyces and Sedecimiella comprised freshwater, marine or terrestrial species, while several clades represent all three habitats such as Chaetopsina, Cosmospora, Neocosmospora and Volutella. The basal clade of Nectriaceae are mostly marine habitats which also shows terrestrial ancestor, and the freshwater and terrestrial habitats show independent evolution. The Ophiocordycipitaceae clade mainly comprised terrestrial fungi and several Tolypocladium species reported from marine habitats which together formed a clade with exclusively terrestrial Niessliaceae clade. Clavicipitaceae to Hypocreaceae clades comprised marine and terrestrial taxa with the ancestor of a terrestrial habitat. Halosphaeriaceae, one of the most prominent marine ascomycetous families, assigned within Microascales has a marine ancestor (Raghukumar 2017), which was also confirmed in our study.
Taxonomy
Sordariomycetes, Hypocreomycetidae
The recent treatment of Hypocreomycetidae was provided by Hyde et al. (2020a) and Wijayawardene et al. (2022), and are followed in the present study. We provided an updated phylogenetic tree for Hypocreomycetidae which includes 41 families and seven orders. Based on phylogenetic analysis and divergence time estimates, a new order (Xenodactylariales) and three new families (Ascocodinaceae, Campylosporaceae, and Cornuvesicaceae) are introduced. Furthermore, the placements of several orders, families and genera are discussed and revised based on phylogenetic analysis. Emericellopsis and Pseudoacremonium are transferred to Hypocreales genera incertae sedis and Bionectriaceae, respectively; Sedecimiella is placed in Nectriaceae; Nautosphaeria and Tubakiella are excluded from Halosphaeriaceae and placed in Microascales genera incertae sedis; Varicosporella is placed under Atractium with synonymy of A. aquatica instead of V. aquatica. Faurelina is excluded from Hypocreomycetidae.
In this study, 41 fresh collections were made from freshwater habitats. Based on phylogenetic analysis and morphological characters, ten new species are introduced, and 13 new habitats and geographic records and 3 new collections are reported. Detailed descriptions and illustrations are provided.
Glomerellales Chadef. ex Réblová, W. Gams & Seifert, Stud. Mycol. 68: 170 (2011).
Glomerellales was established by Réblová et al. (2011) with three families viz. Australiascaceae, Glomerellaceae and Reticulascaceae based on multi-locus phylogenetic analysis. Two additional families Plectosphaerellaceae and Malaysiascaceae were subsequently added to this order by Maharachchikumbura et al. (2016) and Tibpromma et al. (2018). Currently, five families are accepted in Glomerellales (Hyde et al. 2020a). Members of Glomerellales have been reported as endophytes, pathogens, and saprobes from both terrestrial and aquatic habitats (Harter 1916; Rong and Gams 2000; Marsault and Peterson 2017; Cannon et al. 2012a; Jayawardena et al. 2016; Marin-Felix et al. 2017; Zhou et al. 2017; Hyde et al. 2020a). Freshwater species of the order are distributed in three genera (Bertia, Cylindrotrichum and Kylindria) in Reticulascaceae (Luo et al. 2019).
In our phylogenetic analysis, Glomerellales formed a monophyletic clade basal to Hypocreomycetidae; Plectosphaerellaceae is basal to Glomerellales; Australiascaceae, Glomerellaceae and Malaysiascaceae clustered together as a monophyletic clade and sister to Reticulascaceae (Fig. 1) with the stem ages of Australiascaceae (48 MYA), Malaysiascaceae (35 MYA), and Glomerellaceae (35 MYA) which fall within the genus status (Fig. 2). Hyde et al. (2021) mentioned the revision of Malaysiascaceae as the stem age of this family accord within genus level and our analysis also agree with Hyde et al. (2020a). Thus, the status of these three families may need further study. In addition, the genus Ascocodinaea formed a distinct lineage within Glomerellales (Fig. 1), and the divergence time estimates showed that the stem age of Ascocodinaea (96.8 MYA, Fig. 2) falls within the family level.
Ascocodinaceae D.F. Bao, K.D. Hyde & Z.L. Luo, fam. nov.
Index Fungorum number: IF 900276; Facesoffungi number: FoF 13919.
Etymology: Named after the type genus, Ascocodinaea.
Type genus: Ascocodina C.D. Viljoen, M.J. Wingf. & K. Jacobs.
Fungicolous. Sterile setae and conidiophores abundant, arising from the host surface among perithecia. Sexual morph: Perithecia forming directly on the hymenial surface, gregarious, superficial to semi-immersed, gray to black, translucent brown in 3% KOH, ovoidal, with an acute apex, collapsing deeply by lateral pinching when dry, with stiff, erect, acute, unbranched, septate, black setae arising as modified cells of the surface of the upper half of the perithecium, thick-walled. Perithecial wall translucent brown by transmitted light, thin-walled cells of textura epidermoidea at the surface. perithecial apex formed of cells enlarged and arranged in files. Ostiolar canal periphysate; periphyses continuous with the paraphyses. Paraphyses abundant among, and overreaching, mature asci, infrequently branched, septate, slightly enlarged at the tip. Asci cylindrical, 8-spored; apex with a thin ring pierced by a pore. Ascospores uniseriate with overlapping ends, ellipsoidal to fusiform, slightly curved, multi-septate, central cells translucent brown and end cells hyaline, smooth-walled. Asexual morph: Conidiophores macronematous, mononematous, stiff, erect, unbranched, black, morphologically indistinguishable from the sterile setae, each bearing a single, terminal, integrated conidiogenous cell. Conidiogenous cells monophialidic, enteroblastic, proliferating percurrently or sympodially; tip not flared, with slight periclinal thickening at the conidiogenous locus. Conidia broadly ellipsoidal, cylindrical, or inequilateral, often slightly curved, 0–1-septate, hyaline, lacking a visible basal abscission scar, smooth-walled, held in a drop of hyaline slime at the tip of each conidiophore (Samuels et al. 1997).
Notes: Ascocodinaea was introduced by Samuels et al. (1997) to accommodate two fungicolous species, Ascocodinaea polyporicola and A. stereicola (type). Previously, the placement of Ascocodinaea was not well-resolved. Ascocodinaea was originally placed in Lasiosphaeriaceae (Sordariales), based on similar morphological characters with Lasiosphaeriaceae, such as the dark, setose, pseudoparenchymatic ascomata and dematiaceous phialidic asexual morphs (Samuels et al. 1997). Réblová et al. (1999) transferred Ascocodinaea to Chaetosphaeriaceae based on characters of asci, ascospores and perithecial and the dictyochaeta-like asexual morph. Huhndorf et al. (2004) provided a phylogenetic analysis for Sordariales based on LSU sequence data and the result showed that the placement of Ascocodinaea is far from Lasiosphaeriaceae and had close affinities to Glomerella (Collectotrichum). Thus, Ascocodinaea was excluded from Lasiosphaeriaceae and treated as genus incertae sedis in Hypocreomycetidae (Lumbsch and Huhndorf 2007; Wijayawardene et al. 2012). Maharachchikumbura et al. (2015) placed Ascocodinaea in Glomerellales genera incertae sedis, which was accepted by later studies (Maharachchikumbura et al. 2016; Wijayawardene et al. 2017; Hyde et al. 2020a).
Only LSU sequence is available for A. stereicola; in our LSU phylogenetic analysis Ascocodinaea stereicola clustered as a distinct clade and sister to Plectosphaerellacea. In our multi-locus phylogenetic analysis, Ascocodinaea stereicola formed a distinct lineage within Glomerellales (Fig. 1). Currently, only A. polyporicola and A. stereicola are accepted to the genus, and sequence data of A. polyporicola were not available in the GenBank. However, A. polyporicola differs from A. stereicola in having larger ascospores, conidia and conidiophores, finer and much more intricately branched paraphyses (Samuels et al. 1997).
Morphologically, Ascocodinaea differs from Australiascaceae in having uniseriate ascospores with translucent brown central cells and hyaline end cells and broadly ellipsoidal, cylindrical, or inequilateral, 0–1-septate conidia. While, ascospores of Australiascaceae are biseriate, hyaline and conidia are ellipsoid to cylindrical-ellipsoidal, septate, aggregated in slime or in chains. Ascocodinaea can be easily distinguished from Glomerellaceae by the uniseriate, multi-septate, ascospores with translucent brown central cells and hyaline end cells. However, ascospores of Glomerellaceae are uni- to biseriate, hyaline and aseptate, in addition, conidia of Glomerellaceae sometimes have a filiform appendage, the base is rounded to truncate, sometimes with a prominent hilum, a character were not found in Ascocodinaea (Maharachchikumbura et al. 2016; Hyde et al. 2020a). Ascocodinaea differs from Reticulascaceae in having unbranched conidiophores and broadly ellipsoidal or inequilateral, 0–1-septate conidia, whereases, conidiophores of Reticulascaceae are branched or unbranched and conidia are pyriform to cylindrical, 1- or multi-septate. In addition, conidiogenous cells of Ascocodinaea have proliferating percurrent growth; this was not observed in Reticulascaceae (Réblová et al. 2011). Malaysiascaceae differs from Australiascaceae in having bi-seriate, hyaline ascospores that become 1-septate and pale brown after discharge. Ascocodinaea is different from Plectosphaerellaceae in having asci with a thin ring pierced by a pore and uniseriate with overlapping ends ascospores with translucent brown central cells and hyaline end cells. However, the asci of Plectosphaerellaceae lack an apical ring, and ascospores are irregularly arranged, hyaline or pale brown (Maharachchikumbura et al. 2016; Giraldo and Crous 2019).
Divergence time estimates showed that the stem age of Ascocodinaea is around 96.8 MYA which falls within the family range (Hyde et al. 2017). Therefore, a new family Ascocodinaceae is introduced to accommodate Ascocodinaea based on morphological and phylogenetic analyses and divergence time estimates.
Hypocreales Lindau, in Engler & Prantl, Nat. Pflanzenfam., Teil. I (Leipzig) 1(1): 343 (1897).
The most recent treatment of Hypocreales was provided by Hyde et al. (2020a) and Wijayawardene et al. (2022). Hyde et al. (2020a) listed 14 families under Hypocreales, while, Wijayawardene et al. (2022) accepted 15 families in the order where Cylindriaceae was additionally added. Earlier, Hyde et al. (2020a) placed Cylindriaceae in Xylariomycetidae. This was later confirmed by the analysis of Samarakoon et al. (2022). Hence, Cylindriaceae should be excluded from Hypocreales and placed in Xylariomycetidae. Xiao et al. (2023) recently introduced a new family Polycephalomycetaceae to Hypocreales. Perera et al. (2023) provided an updated phylogenetic analysis of combined gene analysis of ITS, LSU, rpb2, tef1-α and tub2 for Hypocreales and accepted 17 families including three new families (Ijuhyaceae, Stromatonectriaceae and Xanthonectriaceae). Based on our phylogenetic analysis, the placements of a few genera, such as, Emericellopsis, Pseudoacremonium and Sedecimiella need further revisions. In our study, Pseudoacremonium and Sedecimiella are transferred to Bionectriaceae and Nectriaceae, respectively. Emericellopsis was excluded from Myrotheciomycetaceae and placed in Hypocreales genera incertae sedis. In our fresh collections, 39 freshwater strains were placed in Bionectriaceae, Nectriaceae, Sarocladiaceae and Stachybotryaceae within Hypocreales.
Bionectriaceae Samuels & Rossman, Stud. Mycol. 42: 15 (1999).
Bionectriaceae was introduced by Rossman et al. (1999) to accommodate 26 genera. The classification of Bionectriaceae has been revised and refined by several studies based on phylogenetic analyses (Rossman et al. 2001; Maharachchikumbura et al. 2015, 2016; Wijayawardene et al. 2018). The recent treatment of Bionectriaceae was provided by Wijayawardene et al. (2022) with the acceptance of 47 genera. In our phylogenetic analyses, Bionectriaceae clustered with Tilachlidiaceae. Tilachlidiaceae was introduced by Lombard et al. (2015) and three genera Psychronectria, Septofusidium and Tilachlidium, are accepted in the family. Our phylogenetic analysis suggests that Tilachlidiaceae may need to be revised and placed under Bionectriaceae (Fig. 1). In addition, our phylogenetic analysis showed that two Septofusidium species (S. berolinense and S. herbarum) and Pseudoacremonium sacchari clustered within Bionectriaceae (Fig. 1). Septofusidium was previously placed within Tilachlidiaceae (Lombard et al. 2015; Hyde et al. 2020a), recently, Perera et al. (2023) transferred it to Bionectriaceae based on phylogenetic analysis. However, the taxonomy of Septofusidium needs further studies, as Septofusidium is polyphyletic (Perera et al. 2023) and the type species lacks sequence data. Pseudoacremonium was placed in Hypocreales genera incertae sedis (Crous et al. 2014; Hyde et al. 2020a). Based on our phylogenetic analysis, Pseudoacremonium is transferred to Bionectriaceae.
In addition, our three fresh collections made from freshwater habitats and the phylogenetic analysis placed them in Clonostachys and Gliomastix within Bionectriaceae. The three fresh collections are identified as Clonostachys rosea, Gliomastix masseei and a new species Clonostachys aquatica based on phylogenetic analysis and morphological characters. Clonostachys rosea, and Gliomastix masseei were reported from freshwater habitats for the first time.
Clonostachys Corda, Pracht-Fl. Eur. Schimmelbild.: 31 (1839).
Clonostachys has a worldwide distribution and is commonly found in tropical and subtropical regions (Schroers 2001; Domsch et al. 2007). Species in the genus are saprobes, endophytes, plant pathogens and mycoparasites from various habitats including soil (Schroers 2001; Toledo et al. 2006; Zhang et al. 2008; Moreira et al. 2016). Clonostachys was linked to Bionectria by Rossman et al. (2013). Based on One Fungus–One Name concept, Rossman et al. (2013) synonymized Bionectria under Clonostachys by giving priority to older and frequently used name Clonostachys. The asexual morph of Clonostachys is characterized by penicillate, frequently sporodochial and, in many cases, dimorphic conidiophores (Schroers 2001). The sexual morph of Clonostachys is characterized by solitary to gregarious, subglobose or globose to ovoid, white, yellow, pale orange, tan, or brown perithecia with KOH- and LA- perithecial walls and narrowly clavate to clavate asci containing eight ascospores (Schroers 2001). There are more than 100 records of Clonostachys listed in Index Fungorum (2023), of which 65 species are commonly accepted (Rossman 2014; Lombard et al. 2015; Dao et al. 2016; Prasher and Chauhan 2017; Lechat and Fournier 2018, 2019, 2020a; Zeng and Zhuang 2022). In this study, a new species Clonostachys aquatica is introduced with detailed description and illustration and C. rosea was collected from freshwater habitat for the first time.
Clonostachys aquatica D.F. Bao, Z.L. Luo & K.D. Hyde sp. nov.
Index Fungorum number: IF 900263, Facesoffungi number: FoF 13920; Fig. 3
Etymology: Referring the fungus was collected from aquatic habitat.
Holotype: KUN-HKAS 125804.
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies on natural substrate effuse, superficial, gregarious, velvety, white, shining. Mycelium superficial to semi-immersed, composed of hyaline, branched, smooth, septate hyphae. Conidiophores 75–105 × 3–5 µm (x̄ = 90.2 × 4.1 μm, n = 10), borne on aerial mycelium, macronematous, mononematous, penicillate, cylindrical, branched, septate, hyaline, smooth and thin-walled, bearing short branches in the upper part. Phialides 7–14 × 2–4 µm (x̄ = 10.7 × 3.0 μm, n = 25), in whorls of 2–4, hyaline, subulate to subcylindrical, swollen at the apex, smooth-walled. Conidia (3–)4–5(–6) × 2–3 µm (x̄ = 4.6 × 2.9 μm, n = 50), arranged in false heads on conidiogenous cells tips, solitary, ellipsoidal to obovoidal, rounded at both ends, hyaline, aseptate, smooth- and thin-walled.
Culture characteristics: Conidia germinating on PDA within 12 h. Colonies growing on PDA reaching 3 cm in 14 days at room temperature, surface effused, mycelium sparse, white.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Xingyun Lake, 12 July 2021, L.L. Li, L487 (KUN-HKAS 125804, holotype), ex-type culture, KUNCC 22–12454 = CGMCC 3.24271.
GenBank numbers: ITS = OP876724, LSU = OP875077.
Notes: In our phylogenetic analysis, Clonostachys aquatica clustered sister to C. rossmaniae with 94%ML/98%MP/1.00PP support (Fig. 5). Morphologically, Clonostachys aquatica is similar to C. rossmaniae in having penicillate, branched hyaline conidiophores and one-celled, ellipsoidal to ovoidal, hyaline and similar size of conidia. However, C. aquatica is different from C. rossmaniae in having subulate to subcylindrical phialides which are swollen at the apex, while, phialides of C. rossmaniae are almost cylindrical, terminate, flask-shaped and the apex are not swollen (Schroers 2001). We also compared the base pair differences of ITS nucleotides between C. aquatica and C. rossmaniae and found 1.75% differences. Therefore, Clonostachys aquatica is introduced as a new species based on both morphological and phylogenetic analyses, as recommended by Chethana et al. (2021).
Clonostachys rosea (Link) Schroers, Samuels, Seifert & W. Gams, Mycologia 91(2): 369 (1999).
Index Fungorum number: IF 485209; Facesoffungi number: FoF 13921; Fig. 4
Saprobic on submerged decaying wood. Sexual morph: see Schroers et al. (1999). Asexual morph: Hyphomycetous. Colonies appearing as white patches on the host. Conidiophores 61–100 × 3–5 µm (x̄ = 80.7 × 4.4 μm, n = 20), arising from stroma, mononematous, monomorphic, hyaline, smooth-walled, penicillate, stipitate. Penicilli solitary to gregarious, not sporodochial, bi- or quater-verticillate; branches of the penicillus divergent, each branch terminating in metulae and adpressed phialides. Phialides 7–11 × 2–4 µm (x̄ = 9.5 × 2.6 μm, n = 15), in whorls of 2–6, narrowly flask-shaped, slightly tapering toward the apex, with visible periclinal thickening, collarettes inconspicuous, hyaline, smooth-walled. Intercalary phialides not observed. Conidia 5–6 × 2–3 µm (x̄ = 5.3 × 2.7 μm, n = 30), broadly ellipsoidal to oval, rarely minutely curved, ends broadly rounded, straight, aseptate, bi-guttulate, hilum laterally displaced, almost median or invisible, hyaline, smooth-walled.
Culture characteristics: Conidia germinating on PDA within 12 h. Colonies growing on PDA reaching 3.5 cm in 7 days at room temperature, surface effused, smooth, margin entire, initially white, becoming pale yellowish orange, reverse yellowish, orange at center.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Xihu Lake, 8 May 2021, S.P. Huang, L415 (KUN-HKAS 125782), living culture, KUNCC 22–12453.
GenBank numbers: ITS = OP876720.
Notes: Clonostachys rosea was initially introduced as Gliocladium roseum by Bainier (1907). Schroers et al. (1999) found Clonostachys rosea quite different from other Gliocladium species in morphology, ecology, and DNA sequence data. Therefore, Gliocladium roseum was placed within Clonostachys and synonymized G. roseum under Clonostachys rosea. Clonostachys rosea has a cosmopolitan distribution and have been found in soil, insects, nematodes, win and as endophytes in different plants (Krauss et al. 2002; Verdejo-Lucas et al. 2002; Toledo et al. 2006; Mamarabadi et al. 2008; Viccini et al. 2009; Costa et al. 2012; Muvea et al. 2014; Sun et al. 2015a, b).
In our phylogenetic analysis, the new collection KUNCC 22–12453 clustered with Clonostachys rosea (Fig. 5). Our new isolate fits well with the description of C. rosea (Schroers et al. 1999). Therefore, we identified our new isolate as C. rosea, which has been reported from freshwater habitats for the first time.
Phylogenetic tree based on RAxML analyses of a combined ITS and tub2 dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Fusarium acutatum (CBS 402.97) and Nectria cinnabarina (CBS 189.87). The combined gene analysis comprises 80 strains with 1123 characters after aligned including gaps (ITS: 497 bp, tub2: 626 bp), of which 469 were parsimony-informative, 124 were parsimony-uninformative, and 530 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -12,821.753182. The matrix had 653 distinct alignment patterns, with 26.55% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.211507, C = 0.281104, G = 0.249523, T = 0.257866; substitution rates AC = 1.093935, AG = 3.149063, AT = 1.106154, CG = 0.657912, CT = 3.704654, GT = 1.000000; gamma distribution shape parameter α = 0.311435. Bootstrap support values for RAxML (blue) and MP (red) greater than 60% and Bayesian posterior probabilities (black) greater than 0.90 are given at each node
Gliomastix Guég., Bull. Soc. mycol. Fr. 21: 240 (1905)
Gliomastix was introduced by Guéguen (1905), characterized by darkly pigmented phialoconidia. It is typified by Gliomastix murorum which was previously named as G. chartarum (Hughes 1958; Gams 1971). The placement of Gliomastix is controversial and debated by many authors (Gams 1971; Matsushima 1975; Domsch et al. 2007; Lechat et al. 2010; Kiyuna et al. 2011; Summerbell et al. 2011). Gams (1971) placed Gliomastix in a section of Acremonium. Matsushima (1975) placed Acremonium masseei and A. polychromum in Gliomastix and Lechat et al. (2010) linked G. fusigera with the sexual morph of Hydropisphaera bambusicola. Kiyuna et al. (2011) and Summerbell et al. (2011) revised and compiled the taxonomy of Gliomastix. Kiyuna et al. (2011) agreed with Gams’s concept and accepted Gliomastix as a section of Acremonium. Furthermore, A. felinum was synonymized under Gliomastix felina and a new species A. tumulicola also was introduced. However, Summerbell et al. (2011) did not follow the Gams’s concept and recognized Gliomastix as a distinct genus. Their phylogenetic analysis supported Gliomastix differs from previous morphological concepts by excluding several distantly related species e.g., Acremonium cerealis and A. inflatum. Maharachchikumbura et al. (2015) and Hyde et al. (2020a) followed the treatment of Summerbell et al. (2011) treated Gliomastix as a distinct genus in Bionectriaceae. Our phylogenetic analysis also showed that Gliomastix formed a monophyletic clade within Bionectriaceae (Fig. 1), which agrees with Summerbell et al. (2011).
In this study, we collected a Gliomastix taxon which is identified as G. masseei based on both phylogenetic analysis and morphological characters. The new isolate was first time reported from freshwater habitats as well as from China.
Gliomastix masseei (Sacc.) Matsush., Icon. microfung. Matsush. lect. (Kobe): 76 (1975).
Index Fungorum number: IF 314510; Facesoffungi number: FoF 13922; Fig. 6
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Hyphomycetous. Colonies effuse, partly immersed, dark brown to black, velvety, hairy. Mycelium mostly immersed, partly superficial, consisting of septate, branched, smooth-walled, hyaline hyphae. Conidiophores 28–40 × 2–3 µm (x̄ = 34 × 2.7 μm, n = 20), micronematous, to semi-macronematous, cylindrical, hyaline to subhyaline, tractate and darker at the apex, aseptate, smooth, thin-walled. Conidiogenous cells polyblastic, terminal, determinate, subhyaline to pale brown, smooth. Conidia 6–8 × 3–4 µm (x̄ = 7.1 × 3.5 μm, n = 30), catenate, in branched chains, ellipsoid-fusiform, narrow and truncate at both ends, hyaline when young, brown to dark brown at maturity, darker at both ends, smooth, thick-walled, aseptate.
Culture characteristics: Conidia germinating on PDA within 24 h. Colonies growing on PDA reaching 5.0 cm in 7 days at room temperature, circular, with velvety to cotton, dense, greyish aerial mycelium, initially white, with light grey immersed hyphae, forming dark, grey, concentric rings.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Erhai Lake, 1 April 2021, S.P. Huang, L240 (KUN-HKAS 125790), living culture, KUNCC 22–12524.
GenBank numbers: ITS = OP876719, LSU = OP875073.
Notes: In our phylogenetic analysis, the newly obtained strain clustered with other strains of Gliomastix masseei (Figs. 1, 7). The morphology of our strain fits well with the description of G. masseei (Kiyuna et al. 2011). Thus, the new isolate is identified as G. masseei. Gliomastix masseei has been reported from India, Italy and Japan. Our new isolate was collected from China which expands the geographical distribution of G. masseei (Kiyuna et al. 2011; Summerbell et al. 2011).
Phylogenetic tree based on RAxML analyses of a combined ITS and LSU dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Bionectria byssicola (CBS 364.78) The combined gene analysis including 23 strains with 1526 characters after aligned including gaps (ITS: 626 bp. LSU: 900 bp), of which 151 were parsimony-informative, 82 were parsimony-uninformative and 1293 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -3922.297293. The matrix had 297 distinct alignment patterns, with 21.90% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.242056, C = 0.254477, G = 0.285780, T = 0.217687; substitution rates AC = 1.319516, AG = 1.218603, AT = 1.905682, CG = 0.505055, CT = 5.019153, GT = 1.000000; gamma distribution shape parameter α = 0.049053. Bootstrap support values for RAxML (blue) and MP (red) greater than 75% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Septofusidium W. Gams, Cephalosporium-artige Schimmelpilze (Stuttgart): 147 (1971)
Septofusidium was introduced by Gams (1971) with S. elegantulum as the type. Currently, five species viz. Septofusidium berolinense, S. elegantulum, S. herbarum, S. stevensiae, S. variabile are included in the genus (Gams 1971; Samson 1974; Samuels 1989; Tan and Shivas 2022). The taxa have been reported as parasitic on foliicolous fungi (Gams 1971; Samson 1974; Samuels 1989). Lombard et al. (2015) initially provided phylogenetic analysis for Septofusidium and their result showed that Septofusidium berolinense and S. herbarum clustered with two strains of Tilachlidium brachiatum. They therefore introduced a new family Tilachlidiaceae to accommodate Septofusidium and Tilachlidium. However, all the families of Hypocreales were not considered in their phylogenetic analysis. Perera et al. (2023) showed that Septofusidium berolinense and S. herbarum clustered within Bionectriaceae. Hence, they transferred Septofusidium to Bionectriaceae based on phylogenetic analysis. We obtained a similar result with Perera et al. (2023). However, the taxonomy of this genus needs further study, as Septofusidium is polyphyletic and sequence data of the type species Septofusidium elegantulum is lacking in GenBank.
Pseudoacremonium Crous, in Crous et al., Persoonia 32: 241 (2014)
Pseudoacremonium was introduced by Crous et al. (2014) and comprises a single species P. sacchari and is placed in Hypocreales genera incertae sedis (Crous et al. 2014; Hyde et al. 2020a). In our phylogenetic analysis, P. sacchari clustered as a sister taxon to Septofusidium berolinense within Bionectriaceae (Fig. 1). Thus, we transfer Pseudoacremonium to Bionectriaceae.
Nectriaceae Tul. & C. Tul. [as 'Nectriei'], Select. fung. carpol. (Paris) 3: 3 (1865)
Nectriaceae a highly diverse group with a worldwide distribution and it has higher diversity in warm temperate and tropical regions (Rossman et al. 1999; Rossman 2000; Chaverri et al. 2011; Schroers et al. 2011; Hyde et al. 2014; Lombard et al. 2015). Several authors have studied and revised the taxonomy of Nectriaceae (Petch 1938; Munk 1957; Dennis 1960; Kreisel 1969; Rossman et al. 1999; Lumbsch and Huhndorf 2010; Lombard et al. 2015; Maharachchikumbura et al. 2016). A recent treatment of Nectriaceae was provided by Wijayawardene et al. (2022), they accepted 70 genera in the family. Nectriaceae comprises mostly freshwater taxa of Hypocreomycetidae. In this study, we focused on the freshwater taxa in Nectriaceae, 25 fresh collections were made from freshwater habitats in China and Thailand. Phylogenetic analyses placed them in 11 genera (Aquanectria, Atractium, Cosmospora, Chaetopsina, Gliocladiopsis, Mariannaea, Neonectria, Neocosmospora, Paracremonium, Thelonectria and Volutella). Eight new species; Atractium fusiforme, Cosmospora cylindricospora, Mariannaea suae, M. yunnanensis, Neocosmospora aquatica, Neonectria aquatica, Thelonectria aquatica and Thelonectria cylindricospora, six new geographical and habitat records (Aquanectria penicillioides, Gliocladiopsis tenuis, Mariannaea dimorpha, Neocosmospora brevis, Paracremonium binnewijzendii and Volutella ciliata) are introduced with detailed description and illustration based on phylogenetic analysis and morphological characters.
Aquanectria L. Lombard & Crous, in Lombard, van der Merwe, Groenewald & Crous, Stud. Mycol. 80: 207 (2015)
Aquanectria was introduced by Lombard et al. (2015) to accommodate Flagellospora penicillioides (A. penicillioides) and Heliscus submersus (A. submersa) based on phylogenetic analysis. Subsequently, five additional species were introduced to Aquanectria. Currently, seven species are accepted in the genus (Gordillo and Decock 2019). Aquanectria is known to form both sexual and asexual morphs. The sexual morph is characterized by perithecial, superficial, ovate to subglobose, brown-orange to orange-red ascomata with papillate ostiolar region, 8-spored, cylindrical to clavate asci and ellipsoid to fusiform, hyaline, 1-septate ascospores; the asexual morphs of this genus are hyphomycetes that are characterized by erect, solitary, septate, hyaline, branched conidiophores with verticillate penicillus with 1–4 phialides, cylindrical, collarette phialides and filiform, curved to slightly sigmoid, aseptate to 1-septate, hyaline conidia (Lombard et al. 2015; Huang et al. 2018; Gordillo and Decock 2019).
Aquanectria species have been reported from China, Colombia, Ecuador, French Guiana, Singapore, the UK, and the USA (Lombard et al. 2015; Huang et al. 2018; Gordillo and Decock 2019). This study describes a new geographical and habitat record Aquanectria penicillioides based on morphological characteristics and phylogenetic evidence.
Aquanectria penicillioides (Ingold) L. Lombard & Crous, in Lombard, van der Merwe, Groenewald & Crous, Stud. Mycol. 80: 207 (2015).
Index Fungorum number: IF 810950; Facesoffungi number: FoF 05440; Fig. 8
Aquanectria penicillioides (KUN-HKAS125807) a–c Appearance of ascomata on the host. d Section through ascoma. e, f Section of peridium in 3% KOH (arrowed in e and f, turning red dark to purple in 3% KOH). g section of peridium. h Paraphyses. i–l Asci. m Ascal apical ring. n–t Ascospores. Scale bars: d–f = 50 μm, g = 30 μm, h–l = 20 μm, g, m–r = 10 μm
Saprobic on submerged decaying wood. Sexual morph: Ascomata 165–220 × 141–185 µm (x̄ = 192 × 162.6 μm, n = 8), perithecial, solitary or cespitose, superficial, pyriform or globose to subglobose, slightly papillate, yellow to orange, paler at the apex, KOH + , turning dark red to purple in 3% KOH. Peridium 22–35 µm (x̄ = 27.6 μm, n = 15), comprise two layers, outer layer comprises of orange textura angularis to textura globulosa cells; inner layer of hyaline to pale yellow textura prismatica cells. Paraphyses 6–13 µm (x̄ = 60.8 × 7.3 μm, n = 15), hyaline to subhyaline, septate, branched, smooth-walled. Asci 56–66 × 6–8 µm (x̄ = 60.8 × 7.3 μm, n = 30), unitunicate, 8-spored, cylindrical, with an apical ring. Ascospores 9–12 × 3–4 µm (x̄ = 10.6 × 3.5 μm, n = 30), 1–2-seriate, clavate to fusiform, gradually narrowing towards the ends, uniseptate, slightly constricted at septum, hyaline, guttulate, smooth-walled. Asexual morph: Undetermined.
Material examined: China, Yunnan Province, on submerged decaying wood in a stream, 12 May 2021, D.F. Bao, H360 (KUN-HKAS 125807).
GenBank numbers: ITS = OP876699, LSU = OP872569, tub2 = OQ025188, his3 = OQ064510, tef1-α = OQ064517.
Notes: Aquanectria penicillioides, the type species of Aquanectria, was originally introduced as Flagellospora penicillioides (Ingold 1942; Ranzoni 1956). Lombard et al. (2015) showed that F. penicillioides clustered with Heliscus submersus in a well-supported clade and sister to Gliocladiopsis. Therefore, a new genus Aquanectria was introduced to accommodate F. penicillioides and H. submersus, with the synonymization of F. penicillioides under Aquanectria penicillioides (Lombard et al. 2015).
Aquanectria penicillioides is characterized by perithecial, superficial, ovate to subglobose, brown-orange to orange-red ascomata, with a papillate ostiolar region, cylindrical to clavate, 8-spored asci and ellipsoid to fusiform, hyaline, 1-septate ascospores. The new isolate fits well with the original description of A. penicillioides (Ranzon 1956). In our phylogenetic analysis, the new isolate KUN-HKAS 125807 clustered with A. penicillioides (CBS 257.54) with 100%ML /100%MP /1PP support (Fig. 9). Thus, we identified the new isolate as A. penicillioides. This species has been reported from freshwater habitats in the USA (Ingold 1942; Ranzoni 1956; Lombard et al. 2015). In this study, the new isolate was collected from freshwater habitat in China, it is a new record for China.
Phylogenetic tree based on RAxML analyses of a combined his3, ITS, tef1-α and tub2 dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Dematiocladium celtidis (CBS 115994). The combined gene analysis included 25 strains with 2269 characters after aligned including gaps (his3: 539 bp ITS: 548 bp, tef1-α: 546 bp, tub2: 636 bp), of which 496 were parsimony-informative, 228 were parsimony-uninformative and 1545 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -10,069.552607. The matrix had 848 distinct alignment patterns, with 15.22% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.220957, C = 0.321481, G = 0.230158, T = 0.227403; substitution rates AC = 1.910524, AG = 3.670736, AT = 1.710679, CG = 0.917283, CT = 6.457579, GT = 1.000000; gamma distribution shape parameter α = 0.221306. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Atractium Link, Mag. Gesell. naturf. Freunde, Berlin 3(1–2): 10 (1809)
≡ Varicosporella Lechat & J. Fourn.
Sexual morphs: Ascomata on submerged wood, superficial, nonstromatic, obpyriform, pale orange, turing or not turning red or purple in 3% KOH, turning pale yellow in lactic acid. Predium composed of angular to flattened cells. Hamathecium composed of fugacious moniliform paraphyses. Asci 8-spored, cylindric-clavate with a J- flat- tened apical apparatus. Ascospores uniseriate, ellipsoid to fusiform, 1-septate, slightly constricted at septum, hyaline to pale-yellow brown, ornamented with short sinuous ribs (Lechat and Fournier 2015). Asexual morphs: See Gräfenhan et al. (2011).
Notes: Atractium was established with A. stilbaster as the type species by Link (1809). Link (1825) reinterpreted the generic concept of Atractium to include the pale or colourful synnematous taxa with slimy conidial masses, usually with falcate, septate conidia. The genus was previously listed as a synonym of Fusarium (Wollenweber and Reinking 1935). However, Atractium has synnematous conidiophores which is different from Fusarium. Hence, Gräfenhan et al. (2011) proposed them as two distinct genera. In addition, the epitype of A. stilbaste (the type of Atractium) was designated by Gräfenhan et al. (2011), and they accepted three species (A. crissum, A. holubovae and A. stilbaster) in Atractium. Currently, 26 names of Atractium are listed in the Index Fungorum (2023), of which only three species are accepted in the genus while the placement of the other 23 species remains uncertain (Gräfenhan et al. 2011). Atractium species are commonly associated with water (Gräfenhan et al. 2011) and species in the genus have been found from Canada, Germany, and the Philippines (Seifert 1985; Seifert et al. 1995; Sivichai et al. 2002; Fryar et al. 2004).
Atractium is only known by its asexual morphs and is characterized by synnematous conidiophores branching once or twice monochasial, 2-level verticillate, monoverticillate or irregularly biverticillate; monophialidic, hyaline, subulate conidiogenous cells and septate, clavate, obovoid or gently curved, rarely ellipsoidal, conidia with a rounded apical cell, and somewhat conical basal cell, lacking a differentiated foot, some species produce chlamydospores (Gräfenhan et al. 2011). In this study, a sexual species Varicosporella aquatica is transferred to Atractium and a new species Atractium fusiformis is introduced based on morphological and phylogenetic analyses. The morphological description of sexual morph of this genus is also provided.
Atractium aquatica (Lechat & J. Fourn) D.F. Bao, K.D. Hyde & Z.L. Luo, comb. nov.
≡ Varicosporella aquatica Lechat & J. Fourn., Ascomycete.org 7(1): 2 (2015).
Index Fungorum number: IF 900476; Facesoffungi number: FoF 13923;
Notes: Varicosporella was introduced by Lechat and Fournier (2015) with a single species V. aquatica. Phylogenetic analysis of Lechat and Fournier (2015) showed that V. aquatica grouped within Nectriaceae in a basal branch distant from extant genera. However, they did not include all the genera of Nectriaceae in their phylogenetic analysis. In our phylogenetic analysis, V. aquatica clustered with Atractium species within Nectriaceae (Fig. 11). Thus, we transferred V. aquatica to Atractium, and synonymized it under A. aquatica based on phylogenetic analysis.
Atractium aquatica is characterized by superficial, astromatic, obpyriform, orange ascomata, unitunicate cylindrical asci with a discoid refractive inamyloid apical apparatus, and ellipsoid with narrowly to broadly rounded ends, two-celled, pale brown ascospores ornamented with short sinuous ridges with fusarium-like asexual morph (Lechat and Fournier 2015). The morphology of this species fits well with Nectriaceae. Our phylogenetic analysis showed that A. aquatica clustered as a sister taxon to A. fusiformis. Atractium aquatica is similar to A. fusiformis in having pyriform, orange ascomata, uniseptate, cylindrical asci with an apical ring and two-celled, pale brown ascospores. However, A. aquatica differs from A. fusiformis in having ellipsoid with rounded ends ascospores and the ascospores wall are roughened by short, sinuous, brown, thick ribs, sometimes anastomosed. While ascospores of A. fusiformis are ellipsoid to fusiform, gradually narrowing towards the ends and ascospores walls are smooth when young, becoming verrucose with age. In addition, asci (140–165 × 17–20 vs. 65–80 × 7–10 µm) and ascospores (21–24 × 8.5–10 (–11) vs. 10–14 × 5–6 µm) of A. aquatica are comparatively larger than those of A. fusiformis.
Atractium fusiformis D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900264; Facesoffungi number: FoF 13924; Fig. 10
Atractium fusiformis (KUN-HKAS 125815, holotype) a–b Appearance of ascomata on the host. c Vertical section of ascoma. d Section of peridium in 3% KOH. e Peridium. f Paraphyses. g–k Asci. l Apical ring of ascus. m, o–t Ascospores. n Germinating ascopore. u Surface view of culture on PDA. v Reverse view of culture on PDA. Scale bars: c–d = 50 μm, e = 30 μm, f–k = 20 μm, l–n = 5 μm
Etymology: Refers the fusiform ascospores of this fungus.
Holotype: KUN-HKAS: 125,815.
Saprobic on submerged decaying wood. Sexual morph: Ascomata 100–150 × 85–160 µm (x̄ = 125.6 × 123.5 μm, n = 10), perithecial, solitary or cespitose, superficial or with a minute stroma, globose to subglobose or pyriform, slightly papillate, yellow to orange, KOH + , turning to purple in the 3% KOH, surface smooth to slightly roughened. Peridium 19–25 µm (x̄ = 22.2 μm, n = 10) comprise orange to hyaline ells of textura angular to textura prismatica cells. Paraphyses 2–5 (x̄ = 3.5 μm, n = 15), hyaline to subhyaline, septate, branched, smooth-walled. Asci 65–80 × 7–10 µm (x̄ = 72.4 × 8.5 μm, n = 10), unitunicate, 8-spored, cylindrical, with an apical ring. Ascospores 10–14 × 5–6 µm (x̄ = 11.8 × 5.4 μm, n = 30), uniseriate, ellipsoid to fusiform, gradually narrowing towards the ends, uniseptate, slightly constricted at septum, hyaline when young, pale brown at maturity, guttulate, smooth-walled when young, becoming verrucose with age. Asexual morph: Undetermined.
Culture characteristics: Ascospore germinated on PDA media within 12 h. Colony reached 2.5–3 cm at room temperature for one week, circular, flat, with fluffy, dense, white mycelium, edge entire, reverse pale yellowish.
Material examined: CHINA, Yunnan Province, Yiliang County, on submerged decaying wood in a small stream, 16 May 2021, H.W. Shen, L525 (KUN-HKAS 125815, holotype), ex-type culture, KUNCC 22–12523 = CGMCC 3.24269; ibid, H387 (KUN-HKAS 125814, paratype), ex-paratype culture, KUNCC 22–12522. CHINA, Yunnan Province, Shizong County, on submerged decaying wood in a small stream, 12 May 2021, D.F. Bao, L731 (KUN-HKAS 125809, paratype), ex-paratype culture, KUNCC 22–12521. CHINA, Yunnan Province, Dali City, on submerged decaying wood in a Cibihu lake, 2 May 2021, S.P. Huang, L702 (KUN-HKAS 125789, paratype), ex-paratype culture, KUNCC 22–12452.
GenBank numbers: KUNCC 22–12523: ITS = OP876725, LSU = OP875078, tub2 = OQ025192. KUNCC 22–12522: ITS = OP876711, LSU = OP875067. KUKUNCC 22–12521: ITS = OP876729, LSU = OP875082, tub2 = OQ025196. KUNCC 22–12452: ITS = OP876727, LSU = OP875080, tub2 = OQ025195.
Notes: In this study, we introduce a new sexual species Atractium fusiformis in Atractium. Morphologically, A. fusiformis fits well with the generic concepts of Nectriaceae in having orange, orange-red, perithecial, globose to subglobose, pyriform ascomata, unitunicate, cylindrical or ellipsoidal asci with apical ring and uniseriate, hyaline to yellow, fusiform or ellipsoidal, septate ascospores (Hyde et al. 2020a). In the phylogenetic analysis, four newly obtained strains of Atractium fusiformis (KUNCC 22–12522, KUNCC 22–12523, KUNCC 22–12521 and KUNCC 22–12452) clustered together and sister to A. aquatica with low support (Fig. 11). However, A. fusiformis can be distinguished from A. aquatica in having ellipsoid with rounded ends ascospores and much larger asci and ascospores (Lechat and Fournier 2015). Thus, we introduce our new isolate as a new species based on both phylogeny and morphology.
Phylogenetic tree based on RAxML analyses of a combined ITS, LSU and tub2 dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Neocosmospora haematococca (CBS 119600 and CBS 101573). The combined gene analysis included 74 strains with 2037 characters after aligned, including gaps (ITS: 605 bp, LSU: 827 bp, tub2: 605 bp), of which 660 were parsimony-informative, 161 were parsimony-uninformative and 1216 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -22,438.740286. The matrix had 986 distinct alignment patterns, with 10.17% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.233899, C = 0.265503, G = 0.269577, T = 0.231020; substitution rates AC = 1.264773, AG = 2.524299, AT = 1.254123, CG = 0.974153, CT = 4.563937, GT = 1.000000; gamma distribution shape parameter α = 0.220117. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Cosmospora Rabenh., Hedwigia 2: 59 (1862)
Cosmospora was established by Rabenhorst (1862) with C. coccinea as the type. The generic concept of Cosmospora was previously relatively broad, encompassing a great deal of asexual morph variability and the sexual morph usually with a small, orange or reddish KOH + and thin-walled perithecia, cylindrical asci with or without an apical ring, and 8-spored, uniseriate, 1-septate ascospores. Gräfenhan et al. (2011) strictly refined the generic concept of Cosmospora to include only the species growing on polypores and xylariaceous fungi and having acremonium-like or verticillium-like asexual states. They accepted eight species in Cosmospora, 12 species were subsequently introduced to the genus. Currently, 20 species are accepted in the genus (Gräfenhan et al. 2011; Herrera et al. 2015; Zeng and Zhuang 2016; Luo et al. 2019; Lechat and Fournier 2021).
Species in Cosmospora are characterized by superficial, solitary to gregarious, orange-red to bright red, pyriform perithecia, cylindrical to narrowly clavate asci and ellipsoidal, multi-septate, verrucose or tuberculate, yellow–brown ascospores; ellipsoidal, oblong, clavate or allantoid and aseptate conidia (Gräfenhan et al. 2011; Herrera et al. 2015; Zeng and Zhuang 2016). This study introduces a new species Cosmospora cylindricospora based on phylogenetic and morphological analyses.
Cosmospora cylindricospora D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900265; Facesoffungi number: FoF 13925; Fig. 12
Etymology: Referring to the cylindrical conidia of this fungus.
Holotype: KUN-HKAS 125785.
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies effuse, white, shining, velvety, hairy. Mycelium partly superficial, composed of branched, septate, brown to dark brown, smooth hyphae. Conidiophores 58–90 × 4–6 μm (x̅ = 74.2 × 5 μm, n = 15) macronematous, mononematous, solitary, unbranched, straight or slightly flexuous, subhyaline to pale brown, paler towards the apex, 3–6-septate, smooth. Conidiogenous cells polyblastic, integrated, determinate, terminal, hyaline, denticulate, hyaline, smooth-walled. Conidia 92–124 × 4–5 μm (x̅ = 108 × 4.5 μm, n = 30), acrogenous, solitary, cylindrical, straight or slightly curved, tapering towards the apex, rounded at the apex, truncate at the base, hyaline, aseptate or sometimes with several pseudoseptate, with small guttules, smooth-walled.
Culture characteristics: Colonies on PDA reaching 4–4.5 cm diam. after three weeks at room temperature. Colony medium dense, circular, flattened to raised, surface slightly rough with hyphal tufts, edge entire, velvety to fluffy; from above, white to white yellowish at the margin, light green to yellowish green at the centre; from below, radiating outwards colony, white to cream at the margin, dark green at the middle, dark yellowish green at the centre.
Material examined: CHINA, Yunnan Province, Cangshan Mountain, on submerged decaying wood in a stream, 9 July 2021, J. He, L594 (KUN-HKAS 125785, holotype), ex-type culture, KUNCC 22–12662 = CGMCC 3.24270.
GenBank numbers: ITS = OP876700, LSU = OP872570, tub2 = OQ025193, rpb2 = OQ077584.
Notes: Cosmospora cylindricospora matches the generic concept of Cosmospora in having hyaline and aseptate conidia. However, Cosmospora cylindricospora is distinct from other species of the genus by its cylindrical, straight or slightly curved and longer conidia. Cosmospora cylindricospora shares similar morphological characteristics such as simple, unbranched conidiophores and hyaline, unicellular, smooth conidia with C. khandalensis and C. lavitskiae. However, C. cylindricospora differs from C. khandalensis and C. lavitskiae in having polyblastic conidiogenous cells and cylindrical, straight or slightly curved conidia with rounded apex and truncated base. Whereas conidiogenous cells of C. khandalensis and C. lavitskiae are monophialidic and conidia are ovoid to ellipsoidal or reniform (Sukapure and Thirumalachar 1966; Zhdanova 1966). In addition, our phylogenetic analysis showed that C. cylindricospora formed a distinct lineage within the genus (Fig. 13). Thus, Cosmospora cylindricospora is introduced as a new species and it is the second Cosmospora species reported from freshwater habitats in China.
Phylogenetic tree based on RAxML analyses of a combined ITS, LSU and tub2 dataset. Tree topology of the RAxML and MP analyses are similar. The tree is rooted to Cosmosporella cavisperma (CBS 172.31) and C. olivacea (KUMCC 17–0321). The combined gene of ITS, LSU and tub2 analysis included 19 strains with 1977 characters after aligned including gaps (ITS: 591 bp, LSU: 901 bp, tub2: 585 bp), of which 173 were parsimony-informative, 110 were parsimony-uninformative and 1694 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -5471.730276. The matrix had 348 distinct alignment patterns, with 17.48% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.225094, C = 0.272713, G = 0.272067, T = 0.230126; substitution rates AC = 1.175555, AG = 3.331353, AT = 1.155285, CG = 1.079199, CT = 7.475654, GT = 1.000000; gamma distribution shape parameter α = 0.020000. Bootstrap support values for RAxML (blue) and MP (red) greater than 60% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Chaetopsina Rambelli, Atti Accad. Sci. Ist. Bologna, Cl. Sci. Fis. Rendiconti 3: 5 (1956).
Chaetopsina, a hyphomycetous genus, was introduced by Rambelli (1956) with C. fulva as the type species. Since then, many numbers of hyphomycetes species have been introduced in Chaetopsina (Rambelli and Lunghini 1976; Sutton and Hodges 1976; 1979; Morgan-Jones 1979; Crane and Schoknecht 1982; Kirk 1985; Samuels 1985; Wingfield 1987; Holubova-Jechova 1990; Merli 1992; Zucconi and Rambelli 1993). Samuels (1985) described four sexual species of Nectria sensu lato having Chaetopsina asexual morphs. These species were later placed in a newly introduced genus Chaetopsinectria by Luo and Zhuang (2010a). Rossman et al. (2016) recommended the generic name Chaetopsina instead of Chaetopsinectria based on its priority, widespread use, and a greater number of names and this was accepted by later studies (Lechat and Fournier 2019, 2020b).
The sexual morph of Chaetopsina is characterized by perithecial, solitary, superficial, non-stromatic, obpyriform, red, becoming dark red in KOH ascomata with an acute apex, 8-spored, clavate asci with a simple apex or an apical ring and ellipsoid to fusiform, 1-septate, hyaline, smooth to striate ascospores. The asexual morphs are characterized by setiform, tapering towards acutely rounded apex, base bulbous, mostly flexuous, yellow–brown, turning red-brown in KOH, fertile in mid region, unbranched, verruculose conidiophores and the fertile region consisting of irregularly branched dense aggregated conidiogenous cells, conidiogenous cells are hyaline, mono- to polyphialidic, ampulliform to lageniform and conidia are subcylindrical, aseptate hyaline, smooth, with bluntly rounded ends and base rarely with flattened hilum (Rambelli 1956; Luo and Zhuang 2010a).
Currently, 27 species are included in Chaetopsina of which only 12 species have sequence data in GenBank. Chaetopsina species are widespread in tropical and neotropical areas and have been found on leaves, bark, dead palm leaves or ascomycetous stromata (Sutton and Hodges 1976; Rambelli and Lunghini 1976; Crane and Schoknecht 1982; Kirk 1985; Wingfield 1987; Merli 1992; Zucconi and Rambelli 1993; Lechat and Fournier 2019, 2020b). In this study, Chaetopsina penicillate was collected from freshwater habitats in China, and it is the first species of this genus reported from freshwater habitats.
Chaetopsina penicillata Samuels, Mycotaxon 22(1): 24 (1985).
Index Fungorum number: IF 105142; Facesoffungi number: FoF 13926; Fig. 14
Saprobic on decaying wood submerged in a lake. Sexual morph: Undetermined. Asexual morph: Colonies effuse, hairy, brown to dark brown, with a white to buff slimy conidial mass at the tip of conidiophores, shining. Mycelium partly immersed, composed brown, septate, branched smooth hyphae. Conidiophores 130–160 × 7–10 µm (x̄ = 146.9 × 8.4 μm, n = 25), macronematous, mononematous, reddish brown, paler towards the apex, septate, smooth-walled, arising from stromatic cells, each composed of a well-defined stipe and a complex penicillate head consisting of series of penicillate branches and terminating with conidiogenous cells and a head of slimy conidia. Conidiogenous cells 11–17 × 2–5 µm (x̄ = 14.1 × 3.3 μm, n = 30), polyblastic, hyaline to pale brown, parallel, obovoid, tapering towards apex, smooth-walled. Conidia 20–25 × 7–9 µm (x̄ = 22.8 × 8.3 μm, n = 43), solitary, slimy, straight, fusiform to clavate, gradually narrowing towards the ends, aseptate, hyaline, smooth-walled, guttulate.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Cibihu Lake, 20 July 2021, S.P. Huang, L701 (KUN-HKAS 102466), living culture, KUNCC 22–12664.
GenBank number: ITS = OP985128, LSU = OP985134.
Notes: In our phylogenetic analysis, the new collection KUNCC 22–12664 clustered with Chaetopsina penicillata with 100%ML/ 100%MP /1 PP support (Fig. 15). Morphologically, our new collection fits well with the original description of C. penicillata except the size of conidiophores. The conidiophores of our new collection are much shorter than the holotype (130–160 vs. 350–370 µm) (Samuels 1985). We identified our new collection as C. penicillata based on both morphological characters and phylogenetic analysis. Chaetopsina penicillata was introduced by Samuels (1985) from terrestrial habitats in Ecuador, Jamaica and New Zealand. This study provides the first report of C. penicillata from freshwater habitats in China.
Phylogenetic tree based on RAxML analyses of a combined ITS, LSU, rpb2, cmd, acl and his3. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Calostilbe striispora (CBS 133491). The combined gene analysis included 44 strains with 4142 characters after aligned including gaps (ITS: 613 bp, LSU: 877 bp, rpb2: 861 bp, cmd: 474 bp, acl: 959 bp, his3: 358 bp), of which 952 were parsimony-informative, 461 were parsimony-uninformative and 2671 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -31,301.685669. The matrix had 1548 distinct alignment patterns, with 51.88% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.236102, C = 0.265619, G = 0.272345, T = 0.225934; substitution rates AC = 1.173873, AG = 2.751368, AT = 1.373236, CG = 0.927145, CT = 5.682462, GT = 1.000000; gamma distribution shape parameter α = 0.214794. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Gliocladiopsis S.B. Saksena, Mycologia 46(5): 662 (1954)
Gliocladiopsis was established by Saksena (1954) with G. sagariensis as type species. The type species has been synonymized several times based on its morphological characteristics (Agnihothrudu 1959; Barron 1968; Crous and Wingfield 1993). Gliocladiopsis has been initially synonymized under Cylindrocarpon or Cylindrocladium (Agnihothrudu 1959; Barron 1968). Crous and Wingfield (1993) resurrected Gliocladiopsis based on its distinct characteristics of dense, penicillate conidiophores, and synonymized G. sagariensis under G. tenuis. Later, G. irregularis and G. sumatrensis were added to this genus (Crous and Peerally 1996; Crous et al. 1997). Lombard and Crous (2012) re-evaluated the taxonomy of Gliocladiopsis based on both phylogeny and morphology and the results showed that G. sagariensis and G. tenuis are distinct species, thus, G. sagariensis is reinstated as the type species for the genus; in addition, five species were introduced in the genus. Consequently, 10 new species had since then been added in the genus (Liu and Cai 2013; Parkinson et al. 2017; Hyde et al. 2018; Zhai et al. 2019; Gordillo and Decock 2019; Perera et al. 2020). Currently, 19 species are included in the genus (Crous and Peerally 1996; Crous et al. 1997; Lombard and Crous 2012; Liu and Cai 2013; Parkinson et al. 2017; Hyde et al. 2018; Zhai et al. 2019; Gordillo and Decock 2019; Perera et al. 2020).
So far, only one sexual species (G. pseudotenuis) has been reported in Gliocladiopsis. Gliocladiopsis pseudotenuis was iniality introduced by Schoch et al. (2000) as Glionectria tenuis and it was incorrectly linked to the asexual morph of Gliocladiopsis tenuis. However, Phylogenetic analysis and morphological observations showed that it is a distinct species wihtin Gliocladiopsis. Therefore, Lombard and Crous (2012) provided a new name Gliocladiopsis pseudotenuis for Glionectria tenuis. The sexual morph of Gliocladiopsis is characterized by superficial, obovoid to broadly obpyriform ascomata that are turning red-brown in 3% KOH + with a dark red stromatic base, unitunicate, 8-spored, cylindrical, sessile asci with flattened apex and a refractive apical apparatus and uniseriate, hyaline, ellipsoidal, smooth, 1-septate ascospores that are becoming brown and verruculose with age; The asexual morphs are characterized by hyaline, penicillate conidiophores, which consist of a simple-septate stipe bearing 2–4 successive whorls of branches subtending whorls of phialides and hyaline, cylindrical, 0–1-septate conidia accumulating in whitish to pale yellowish mucoid drops (Lombard and Crous 2012; Gordillo and Decock 2019).
Morphologies of Gliocladiopsis species are quite similar which make species identification very difficult. Thus, phylogeny is important to identify the Gliocladiopsis species. Of the 19 species, 14 have the sequence in the GenBank. Therefore, sequence data of the other five species are required to understand the taxonomy of the genus better. Gliocladiopsis species have been isolated from soil, diseased plants root and asymptomatic rhizomes (Booth 1966; Crous et al. 1997; Dann et al. 2012; Parkinson et al. 2017; Li et al. 2008). Only two species G. aquaticus and G. guangdongensis were isolated from freshwater habitats. This study collected Gliocladiopsis tenuis from freshwater habitats in China for the first time.
Gliocladiopsis tenuis (Bugnic.) Crous & M.J. Wingf., Mycol. Res. 97(4): 446 (1993).
Index Fungorum number: IF 359449; Facesoffungi number: FoF 06876; Fig. 16
Saprobic on submerged decaying wood in a lake. Sexual morph: Undetermined. Asexual morph: appearing as white masses on the substrate, becoming yellowish with age. Conidiophores 75–110 × 4–6 μm (x̄ = 93 × 5.2 μm, n = 16), penicillate, macronematous, mononematous, consisting of a stipe bearing a penicillate arrangement of fertile branches, hyaline, septate, branched, without stipe extensions and terminal vesicles. Conidiogenous cells apparatus with several series of hyaline, branched, smooth-walled branches. Phialides 9–14 × 3–4 μm (x̄ = 11.4 × 3.4 μm, n = 44), doliiform to cymbiform to cylindrical, arranged in terminal whorls of 3–6 per branch, with minute collarettes, central phialide frequently extending above the rest. Conidia 15–20 × 2–3 μm (x̄ = 17.1 × 2.3 μm, n = 45), in chains, cylindrical with rounded ends, aseptate to 1-septate, hyaline, smooth-walled, straight.
Culture characteristics: Colonies on PDA reaching 3.5–4.0 cm diameter after 3 weeks at room temperature; colony from above, dense, circular, with edge entire, fluffy to floccose, with white tufts and black droplets, dark brown to black at the margin; reverse reddish-brown to dark brown.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Chenghai Lake, 28 May 2021, H.W. Shen, L603 (KUN-HKAS 125800), living culture, KUNCC 22–12663.
GenBank numbers: ITS = OP876701, tub2 = OQ025194, his3 = OQ064512, tef1-α = OQ064522.
Notes: In the phylogenetic analysis, the new isolate KUNCC 22–12663 clustered with the ex-type of G. tenuis (IMI 68205) and four extant strains of G. tenuis with 100% ML/ 100% MP/ 1 PP support (Fig. 17). Morphologically, the new isolate fits well with the original description of G. tenuis (Crous and Wingfield 1993), thus, we identified the new isolate as G. tenuis based on phylogenetic analysis and morphological characters.
Phylogenetic tree based on RAxML analyses of a combined his3, ITS, tef1-α, and tub2 dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Dematiocladium celtidis (CBS 115994). The combined gene analysis included 45 strains with 2221 characters after aligned including gaps (his3: 515 bp, ITS: 539 bp, tef1-α: 538 bp, tub2: 629 bp), of which 249 were parsimony-informative, 231 were parsimony-uninformative and 1741 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -7798.259460. The matrix had 565 distinct alignment patterns, with 6.78% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.218239, C = 0.323881, G = 0.229746, T = 0.228134; substitution rates AC = 1.311343, AG = 2.782041, AT = 1.439742, CG = 0.500710, CT = 5.688029, GT = 1.000000; gamma distribution shape parameter α = 0.183224. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Gliocladiopsis tenuis has been reported from terrestrial habitats in India, Indonesia, South-East Asia, Thailand and Vietnam (Crous and Wingfield 1993; Crous and Peerally 1996; Perera et al. 2020). In this study, the new collection was isolated from freshwater habitats for the first time and introduced as a new geographical record for China.
Mariannaea G. Arnaud ex Samson, Stud. Mycol. 6: 74 (1974)
Mariannaea was formally established by Samson (1974) to accommodate M. camptospora, M. elegans and the variety M. elegans var. punicea and typified by M. elegans. The genus is characterized by branched, septate conidiophores with hyaline, flask-shaped phialides, 1–2-celled, hyaline conidia mostly forming imbricate chains or slimy heads (Samson 1974; Samson and Bigg 1988; Samuels and Seifert 1991). The sexual morph of Mariannaea has been linked to Nectria (Samuels and Seifert 1991) and Cosmospora (Gräfenhan et al. 2011). However, Cosmospora and Nectria have been shown polyphyletic within Nectriaceae, while Mariannaea formed a monophyletic clade within Nectriaceae. Therefore, Gräfenhan et al. (2011) retained Mariannaea as a distinct genus in Nectriaceae.
The classification and species concept of Mariannaea were reviewed by Hu et al. (2017), they designated an epi-type for M. elegans (the type species of Mariannaea) and accepted 15 species in the genus. Five new species were further introduced in the genus (Crous et al. 2019; Hyde et al. 2020b; Boonmee et al. 2021; Watanabe and Hirose 2021; Yang et al. 2021). Currently, 20 species are accepted in the genus (Index Fungorum 2023). This study collected five fresh collections from freshwater habitats in Yunnan Province. Two new species M. yunnanense and M. suae, as well as two new records are introduced based on phylogenetic analysis and morphological characters.
Mariannaea dimorpha Z.Q. Zeng & W.Y. Zhuang, Mycol. Progr. 13(4): 969 (2014).
Index Fungorum number: IF 570079; Facesoffungi number: FoF 13927; Fig. 18
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies on natural substrate superficial, scattered or gregarious, effuse, white, hairy, with glistening conidia. Mycelium composed of septate, branched, smooth, hyaline to brown hyphae. Conidiophores 230–450 × 12–14 µm (x̄ = 338.7 × 13.1 μm, n = 10), macronematous, mononematous, cylindrical, erect, straight or slightly flexouse, septate, hyaline when young, becoming orange brown at base and gradually paler towards apex, hyaline at the apex, smooth at the base, gradually verruculose towards the apex, branched, branching 1–2 levels verticillate, bearing short branches in the upper part with a terminal whorl of 5–7 phialides. Phialides 19–29 × 3–5 µm (x̄ = 23.9 × 3.8 μm, n = 20), flask-like, hyaline to subhyaline, slightly swollen at base, tapering at apex, smooth when young, verruculose at maturity. Conidia 10–14 × 3–4 µm (x̄ = 12.3 × 3.5 μm, n = 30), oblong to fusiform, acuminate at apex, pointed at base, straight, (0)-1-septate, slightly constricted at septum, hyaline, smooth-walled.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Nanpan River, 12 July 2018, X. He, S1725 (KUN-HKAS 125803), living culture, KUNCC 22–12458.
GenBank numbers: ITS = OP876732, LSU = OP875086.
Notes: Mariannaea dimorpha was introduced by Zeng and Zhuang (2014) with its sexual and asexual morphs. It was isolated on rotten bark from China. Phylogenetic analysis showed that our new isolate (KUNCC 22–12458) clustered with the holotype of M. dimorpha (Fig. 19). There are no differences in ITS region between these two species, we therefore identified our species as M. dimorpha. Our new isolate was collected from China and first reported from freshwater habitat.
Phylogenetic tree based on RAxML analyses of a combined ITS and LSU dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Stachybotrys chartarum (CBS 129.13). The combined gene analysis included 56 strains with 1429 characters after aligned including gaps (ITS: 583 bp and LSU: 846 bp), of which 230 were parsimony-informative, 89 were parsimony-uninformative and 1110 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -9021.203912. The matrix had 692 distinct alignment patterns, with 32.42% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.242101, C = 0.249327, G = 0.274549, T = 0.234024; substitution rates AC = 1.654970, AG = 2.830985, AT = 1.713621, CG = 0.691184, CT = 6.463431, GT = 1.000000; gamma distribution shape parameter α = 0.165519. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Morphologically, our new isolate is similar to the holotype of M. dimorpha in having septate, hyaline conidiophores which branching 1–2 levels verticillate, phialidic conidiogenous cells and fusiform straight, (0)-1-septate, hyaline, smooth-walled conidia (Zeng and Zhuang 2014). However, the conidiophores of our isolate are smooth at the base and gradually verruculose towards the apex and hyaline when young, becoming orange brown at the base and gradually became paler towards apex and the phialides are smooth when young, verruculose at maturity. However, these characters were not found from the holotype (Zeng and Zhuang 2014).
Mariannaea yunnanensis D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900266; Facesoffungi number: FoF 13928; Fig. 20
Etymology: Referring to Yunnan Province, China, where the fungus was collected.
Holotype: KUN-HKAS 125786.
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies on natural substrate superficial, effuse, white, hairy. Mycelium immersed, subhyaline to pale brown, composed of branched, septate hyphae. Conidiophores 220–330 × 6–10 µm (x̄ = 274.8 × 7.8 μm, n = 11), macronematous, mononematous, cylindrical, erect, straight or slightly flexouse, septate, smooth-walled, hyaline to subhyaline, branched, branching verticillately at 2–3 level verticillate, bearing short branches in the upper part with a terminal whorl of 3–6 phialides, and 1–2 lower nodes of 3–4 phialides. Phialides 12–23 × 2–4 µm (x̄ = 17.6 × 3.1 μm, n = 30), flask-like, hyaline, slightly swollen at base, tapering at apex, smooth-walled. Conidia 6–8 × 3–4 µm (x̄ = 7.1 × 3.6 μm, n = 30), ellipsoidal to fusiform, acuminate at apex, pointed at base, aseptate, hyaline, smooth and thin-walled.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in a stream, 12 May 2021, D.F. Bao, B213 (KUN-HKAS 125786, holotype), ex-type culture, KUNCC 22–12455= CGMCC 3.24273; ibid, B215 (KUN-HKAS 125788, paratype), ex-paratype culture, KUNCC 22–12456 = CGMCC 3.24272.
GenBank numbers: KUNCC 22–12455: ITS = OP876702, LSU = OP875058, tub2 = OQ025185. KUNCC 22–12456 ITS = OP876703, LSU = OP875059, tub2 = OQ025186.
Notes: In our phylogenetic analysis, two new isolates of Mariannaea yunnanensis (KUNCC 22–12455 and KUNCC 22–12456) formed a distinct lineage within Mariannaea which closed to M. aquaticola, M. humicola, M. pinicola and M. samuelsii (Fig. 19). Mariannaea yunnanensis resembles M. aquaticola, M. humicola, M. pinicola and M. samuelsii in having macronematous, mononematous, cylindrical, erect, septate, hyaline conidiophores which bearing short branches on the upper part, phialidic, hyaline, smooth-walled conidiogenous cells and ellipsoidal to fusiform, aseptate conidia. However, the conidiophores of M. aquaticola has branches with whorls of 3–4 phialides, while, branch whorls of M. yunnanensis have 3–6 phialides (Cai et al. 2010). Mariannaea yunnanensis is different from M. humicola in having longer conidiophores (220–330 vs. 80–100 µm) and larger conidia (6–8 × 3–4 vs. 4–6 × 2–3 µm) and the conidiophores with a terminal whorl of 3–6 phialides. Whereas conidiophores of M. humicola with a terminal whorl of 1–5 phialides (Lombard et al. 2015). Mariannaea yunnanensis can be distinguished from M. pinicola by its fewer terminal or intercalary whorls phialides (3–6 vs. 9) (Samuels and Seifert 1991). The conidiophores of Mariannaea yunnanensis are much longer than those of M. samuelsii (220–330 vs.100–200 μm). The conidiophores terminal whorl has 3–6 phialides and 1–2 lower nodes of 3–4 phialides in M. yunnanensis, while, terminal whorl has (2–)3–5 phialides and 1–2 lower nodes of 1–3 phialides in M. samuelsii (Gräfenhan et al. 2011).
Mariannaea suae D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900267; Facesoffungi number: FoF 13929; Fig. 21
Etymology: “suae” (Lat.) in memory of the Chinese mycologist Prof. Hong-Yan Su (4 April 1967–3 May 2022).
Holotype: KUN-HKAS 125787.
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies on natural substrate superficial, scattered, effuse, white, hairy. Mycelium composed of branched, septate, hyaline to medium brown hyphae, smooth. Conidiophores 254–679 × 5–24 µm (x̄ = 466 × 13 μm, n = 10), macronematous, mononematous, single, cylindrical, erect, straight or slightly flexuous, septate, hyaline, smooth-walled, bearing short branches in the upper part with a terminal whorl of more than 10 phialides, branching one level verticillate. Phialides 16–23 × 1–2 µm (x̄ = 19.6 × 1.8 μm, n = 25), flask-like, cylindrical, hyaline, tapering towards apex, smooth-walled. Conidia 6–8 × 3–5 µm (x̄ = 6.8 × 4 μm, n = 30), ellipsoidal to oval, rounded at both ends, straight, aseptate, hyaline, guttulate, smooth-walled.
Material examined: CHINA, Yunnan Province, Cangshan Mountain on submerged decaying wood in a stream, 9 July 2021, J. He, B217 (KUN-HKAS 125787, holotype), ex-type culture, KUNCC 22–12525 = CGMCC 3.24274.
GenBank numbers: ITS = OP876704, LSU = OP875060.
Notes: Mariannaea suae is introduced here based on phylogenetic analysis and its distinct morphology. Mariannaea suae is unique in the genus by its conidiophores which bear short branches in the upper part with a terminal whorl of more than ten phialides, branching one level verticillate. While, conidiophores of other species less than ten phialides in the terminal whorl, and usually branching 1–3 levels verticillate (Cai et al. 2010; Gräfenhan et al. 2011; Zeng and Zhuang 2014; Lombard et al. 2015; Hu et al. 2017; Yang et al. 2021). In our phylogenetic analysis, Mariannaea suae formed a distinct lineage within Mariannaea (Fig. 19). Therefore, we introduce our new isolate as a new species based on phylogenetic and morphological analyses.
Mariannaea superimposita (Matsush.) Samuels [as 'superimpositus'], Mycologia 81(3): 353 (1989).
Index Fungorum number: IF 136166; Facesoffungi number: FoF 05444; Fig. 22
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies on natural substrate superficial, scattered, effuse, white, hairy. Mycelium composed of septate, branched, smooth, hyaline to medium brown hyphae. Conidiophores 250–460 × 6–14 µm (x̄ = 355.7 × 9.8 μm, n = 10), macronematous, mononematous, simple, cylindrical, erect, straight or slightly flexouse, septate, hyaline when young, becoming orange brown at base and gradually paler towards apex, hyaline at apex, smooth-walled, branching 1–2 levels verticillate, bearing short branches in the upper part with 3–4 phialides in verticils. Phialides 21–31 × 2–4 µm (x̄ = 26 × 3.2 μm, n = 30), flask-like, hyaline, cylindrical, slightly swollen at base, tapering at apex, smooth-walled. Conidia 10–14 × 3–5 µm (x̄ = 12.1 × 3.6 μm, n = 30), ellipsoidal to fusiform, acuminate at apex, pointed at base, straight, or slightly curved, (0)-1-septate, slightly constricted at the septum, hyaline, smooth-walled.
Material examined: CHINA, Yunnan Province, Shizong County, on submerged decaying wood in a stream, 12 May 2021, D.F. Bao, H355 (KUN-HKAS 125808), living culture, KUNCC 22–12457.
GenBank numbers: ITS = OP876707, LSU = OP875063, tub2 = OQ025187.
Notes: Matsushima (1975) initially introduced Mariannaea superimposita as Penicillifer superimpositus. It was later transferred to Mariannaea by Samuels (1989) and phylogenetic analysis also confirmed the placement of the species within Mariannaea (Nonaka et al. 2015). In the phylogenetic analysis (Fig. 19), our new isolate (KUNCC 22–12457) clustered with Mariannaea superimposita (CBS 113472 and CBS 124559). Morphologically, the new isolate is quite similar to M. superimposita (Matsushima 1975); we, therefore, identified our new isolate as M. superimposita.
Mariannaea superimposita has been reported from China, Japan and Venezuela (Matsushima 1975; Luo et al. 2019). In this study, the new isolate was recollected from freshwater habitats in China.
Neocosmospora E.F. Sm., Bull. U.S. Department of Agriculture 17: 45 (1899).
Neocosmospora was established by Smith (1899). Species in the genus are saprobes, endophytes, and plant and animal pathogens and are commonly found in soil, plant debris, living plant material, air and water (Sandoval-Denis et al. 2019; Guarnaccia et al. 2021). Neocosmospora is characterised by orange to red-brown, smooth-walled to coarsely warted perithecia, producing globose to ellipsoidal, 0–1-septate, distinctly ornamented (striate, cerebriform to spinulose), yellow golden brown ascospores; while asexual morphs produce distinctive very long and narrow, acremonium-like aerial monophialides (Sandoval-Denis et al. 2019).
Currently, 129 records of Neocosmospora are listed in Index Fungorum (2023). However, only N. haematococca has been reported from freshwater habitats. In this study, two species were collected from freshwater habitats. Based on multi-locus phylogenetic analysis and morphological characters, the species were identified as N. aquatica sp. nov. and N. brevis.
Neocosmospora aquatica D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900268; Facesoffungi number: FoF 13930; Fig. 23
Etymology: Epithet refers to the collection from an aquatic habitat.
Holotype: KUN-HKAS 125810.
Saprobic on submerged decaying wood from freshwater habitats. Sexual morph: Undetermined. Asexual morph: Colonies on natural substrate superficial, effuse, gregarious, velvety, white. Mycelium superficial to semi-immersed, composed of hyaline, branched, smooth hyphae. Conidiophores 40–80 × 2–4 µm (x̄ = 59.8 × 3.3 μm, n = 20), borne on aerial mycelium, semi-macronematous to macronematous, mononematous, cylindrical, unbranched or irregularly laterally branched, aseptate, hyaline, smooth-walled, bearing terminal single phialides. Conidiogenous cells monophialidic, integrated, terminal, determinate, hyaline, subulate to subcylindrical, with a discrete flared collarette, smooth. Conidia 6–7 × 2–4 µm (x̄ = 6.5 × 3 μm, n = 32), arranged in false heads on phialide tips, ellipsoidal to obovoidal, hyaline, smooth- and thin-walled, aseptate.
Material examined: CHINA, Yunnan Province, Yiliang County, on submerged decaying wood in a stream, 16 May 2021, H.W. Shen, H391 (KUN-HKAS 125810, holotype), ex-type culture, KUNCC 22–12459 = CGMCC 3.24275; ibid, H411 (KUN-HKAS 125811, paratype), ex-paratype culture, KUNCC 22–12460 = CGMCC 3.24276.
GenBank numbers: KUNCC 22–12459: ITS = OP876713, LSU = OP875069, tef1-α = OQ064518. KUNCC 22–12460: ITS = OP876716, LSU = OP875072, tef1-α = OQ064520.
Notes: In our phylogenetic analysis, Neocosmospora aquatica clustered as a sister taxon to N. parceramosa (Fig. 24). Neocosmospora aquatica resembles N. parceramosa in having simple, hyaline smooth-walled conidiophores, monophialidics, subulate to subcylindrical conidiogenous cells, with a discrete flared collarette and ellipsoidal, subcylindrical to clavate, hyaline, conidia that clustering in false heads at the tip of monophialides. However, Neocosmospora aquatica differs from N. parceramosa in having aseptate conidiophores and conidia. While, N. parceramosa has sepate conidiophores and conidia (Sandoval-Denis et al. 2019). In addition, nucleotide comparison between N. aquatica and N. parceramosa for ITS and tef1-α gene regions revealed 1.8% and 1.2% differences respectively. Thus, the new isolate is introduced as a new species (Chethana et al. 2021; Pem et al. 2021).
Phylogenetic tree based on RAxML analyses of a combined ITS, LSU, rpb2 and tef1-α, dataset. Tree topology of the RAxML and MP analyses are similar. The tree is rooted to Geejayessia atrofusca (NRRL 22316) and G. cicatricum (CBS 125552). The combined gene analysis including 147 strains with 3495 characters after aligned including gaps (ITS: 492 bp, LSU: 483 bp, rpb2: 1823 bp, tef1-α: 697 bp), of which 917 were parsimony-informative, 349 were parsimony-uninformative and 2229 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -56,493.984935. The matrix had 1584 distinct alignment patterns, with 18.80% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.238608, C = 0.274649, G = 0.260503, T = 0.226239; substitution rates AC = 1.514461, AG = 4.749211, AT = 1.456537, CG = 0.930417, CT = 9.220959, GT = 1.000000; gamma distribution shape parameter α = 0.258278. Bootstrap support values for RAxML (blue) and MP (red) greater than 60% and Bayesian posterior probabilities (black) greater than 0.90 are given at each node
Neocosmospora brevis Sand.-Den. & Crous, in Sandoval-Denis, Lombard & Crous, Persoonia 43: 119 (2019).
Index Fungorum number: IF 831176; Facesoffungi number: FoF 13931; Fig. 25
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies on natural substrate superficial, effuse, gregarious, scattered, velvety, white, with a white slimy conidial mass on the conidiophores. Mycelium superficial to semi-immersed, composed of branched, hyaline, smooth hyphae. Conidiophores 100–140 × 2–4 µm (x̄ = 122.5 × 3 μm, n = 32), borne on aerial mycelium, semi-macronematous to macronematous, mononematous, cylindrical, unbranched or rarely branched, septate, hyaline to pale brown, smooth-walled, bearing terminal single phialides. Conidiogenous cells phialidic, integrated, terminal, determinate, hyaline, 6–12 × 3–4 µm (x̄ = 9.1 × 3.4 μm, n = 30), cylindrical to subcylindrical, with a discrete flared collarettes, smooth. Conidia arranged in false heads on phialide tips, oval, ellipsoidal, clavate, with round ends, hyaline, smooth- and thin-walled, aseptate.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Chenghai Lake, 28 May 2021, S. Nuan, H314 (KUN-HKAS 125799), living culture, KUNCC 22–12461.
GenBank numbers: ITS = OP876705, LSU = OP875061, tef1-α = OQ064516.
Notes: Neocosmospora brevis was described by Sandoval-Denis et al. (2019), which was collected from soil–water polluted with diethyleneglycerol and ethylenglycerol, Citrus sinensis and a human eye in Belgium, Italy and the USA (Sandoval-Denis et al. 2019). In this study, a new isolate (KUNCC 22–12461) was identified as Neocosmospora brevis based on both phylogenetic and morphological analysis which was collected on submerged decaying wood from freshwater habitats in China. It is a new record for China.
In our phylogenetic analysis, the new isolate clustered with Neocosmospora brevis with significant support (Fig. 24). The nucleoide comparision of ITS and tef1-α between our new isolate (KUNCC 22–12461) and Neocosmospora brevis (CBS 130326) revealed 1 bp and 3 bp differences, respectively. Our new isolate is morphologically similar to the holotype of N. brevis in having cylindrical, unbranched or rarely branched, septate conidiophores, phialidic conidiogenous cells and oval, ellipsoidal straight or slightly curved, hyaline, smooth- and thin-walled microconidia, the macroconidia were not observed in the new isolate. However, our new isolate (KUNCC 22–12461) has longer conidiophores than the holotype (100–140 vs. 36.5–59 μm) and aseptate conidia, while the holotype has 0–1(–2)-septate, which may due to their different hosts and habitats. The holotype of N. brevis was collected from soil–water with diethyleneglycerol and ethylenglycerol (Sandoval-Denis et al. 2019). However, our new isolate is a saprobe collected on submerged wood from freshwater habitats.
Neonectria Wollenw., Annls mycol. 15(1/2): 52 (1917).
Neonectria, is a cosmopolitan genus commonly distributed in tropical and temperate regions. The species occur as saprobes, pathogens and some species are isolated as soil inhabitants (Brayford 1993; Chaverri et al. 2011). Species of Neonectria sensu lato are characterised by subglobose to broadly obpyriform, smooth to roughened, red perithecia that are becoming dark red in 3% KOH, and with an acute to constricted apex that is sometimes knobby, the perithecial wall is ca. 50 μm thick and generally composed of two regions, sometimes with an outer region that forms textura epidermoidea, that may or may not be covered with another region of cells; and the ascospores are hyaline, generally bicellular, rarely multi-cellular, and smooth or finely ornamented (Rossman et al. 1999; Chaverri et al. 2011).
Neonectria was linked to the asexual genus Cylindrocarpon. Neonectria and Cylindrocarpon was informally classified into several groups based on the combined morphology of asexual morph and sexual morph (Neonectria into five informal groups and Cylindrocarpon into four groups, Booth 1959, 1966; Brayford and Samuels 1993; Samuels and Brayford 1994). Phylogenetic analysis revealed that Neonectria and Cylindrocarpon are phylogenetically congeneric (Mantiri et al. 2001; Brayford et al. 2004; Seifert et al. 2003). Halleen et al. (2004) introduced a new asexual morph genus Campylocarpon to accommodate C. fasciculare, which is the first formal segregation from Cylindrocarpon. A taxonomic revision of Neonectria sensu lato was provided by Chaverri et al. (2011) based on multi-locus phylogenetic analysis, morphological characters and ecological data. Their phylogenetic analysis showed that five distinct highly supported clades that correspond to some extent with the informal Neonectria and Cylindrocarpon groups. Hence, three genera llyonectria, Rugonectria and Thelonectria were introduced to accommodate three Neonectria sensu lato informal groups. Currently, 55 Neonectria species epithets are listed in the Index Fungorum (2023). However, only N. lugdunensis has been reported from freshwater habitats. This study introduces a new freshwater species Neonectria aquatica based on phylogenetic analysis and morphological characters.
Neonectria aquatica D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900269; Facesoffungi number: FoF 13932; Fig. 26
Etymology: Referring to the aquatic habitat of this fungus.
Holotype: KUN-HKAS 125779.
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies appearing as white patches on the host, velvety, white, shinning. Mycelium superficial to semi-immersed, consisting of branched, hyaline, septate, smooth hyphae. Conidiophores borne on aerial mycelium, short, mononematous, cylindrical, loosely branched with branches terminating in several phialidies, hyaline, septate, smooth and thin-walled. Conidiogenous cells 17–31 × 2–4 µm (x̄ = 23.9 × 3.2 μm, n = 30), monophialidic, integrated, terminal, determinate, hyaline, cylindrical to subcylindrical, smooth-walled. Conidia 27–31 × 6–8 µm (x̄ = 29 × 7 μm, n = 40), acrogenous, solitary, cylindrical with round ends, uniseptate, straight or slightly curved, hyaline, slightly swollen at the apex, smooth- and thin-walled.
Material examined: CHINA, Yunnan Province, Cangshang Mountain, on submerged decaying wood in a stream, 1 March 2019, Z.Q. Zhang, S-3086 (KUN-HKAS 125779, holotype), ex-type culture, KUNCC 22–12462.
GenBank numbers: ITS = OP876733, LSU = OP875087, tub2 = OQ025197.
Notes: In our phylogenetic analysis, Neonectria aquatica formed a basal clade to N. lugdunensis and N. obtusispora within Neonectria (Fig. 27). Neonectria aquatica is similar to N. lugdunensis and N. obtusispora in having short, mononematous, cylindrical conidiophores loosely branched with branches terminating in several phialidies and cylindrical, straight or slightly curved, hyaline, smooth-walled conidia (Luo and Zhuang 2010b). However, Neonectria aquatica can be distinguished from N. lugdunensis and N. obtusispora by having uniseptate, cylindrical conidia with round ends. While, N. lugdunensis has 0–1 septate conidia that are rounded at the distal end, with a lateral hilum (Luo and Zhuang 2010b) and conidia of N. obtusispora are 1–3 septate with rounded apex and somewhat pointed basal cell (Booth 1966). Therefore, we introduce our collection as a new species based on phylogenetic analysis and morphological characters following the guidelines of Chethana et al. (2021).
Phylogenetic tree based on RAxML analyses of a combined ITS, LSU, tef1-α and tub2 dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Fusarium incarnatum (CBS 161.25) and F. equiseti (NL19-25,004). The combined gene analysis including 62 strains with 2872 characters after aligned including gaps (ITS: 567 bp, LSU: 831 bp, tef1-α: 819 bp, tub2: 655 bp), of which 865 were parsimony-informative, 137 were parsimony-uninformative and 1870 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -18,747.693262. The matrix had 1202 distinct alignment patterns, with 20.40% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.229010, C = 0.278072, G = 0.258275, T = 0.234642; substitution rates AC = 1.435040, AG = 2.735484, AT = 1.610328, CG = 0.862374, CT = 5.534221, GT = 1.000000; gamma distribution shape parameter α = 0.217640. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Paracremonium L. Lombard & Crous, in Lombard, van der Merwe, Groenewald & Crous, Stud. Mycol. 80: 233 (2015).
Paracremonium is an acremonium-like genus, was established by Lombard et al. (2015) with P. inflatum as the type species. Eight species were further introduced in the genus (Lynch et al.2016; Crous et al. 2017, 2021a; Zhang et al. 2017, 2021a; Al-Bedak et al. 2019; Ming et al. 2021). Paracremonium is only known by the aseual morphs and characterized by hyaline, septate, branched hyphae which sometimes forming sterile coils from which conidiophores arise (Lombard et al. 2015). Species of Paracremonium are widely distributed and have been reported from Canada, California, China, India, Netherlands and Egypt (Lynch et al. 2016; Crous et al. 2017, 2021a; Al-Bedak et al. 2019; Zhang et al. 2021a; Ming et al. 2021). In this study, a new geographic record Paracremonium binnewijzendii is described.
Paracremonium binnewijzendii Houbraken, van der Kleij & L. Lombard, in Crous et al., Persoonia 39: 321 (2017).
Index Fungorum number: IF 823317; acesoffungi number: FoF 13933; Fig. 28
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: colonies appearing as white patches on the host, effuse, superficial, gregarious, velvety, white. Mycelium superficial to semi-immersed, composed of hyaline, septate, branched, smooth, hyphae. Conidiophores borne on aerial mycelium, macronematous, mononematous, branched, hyaline, septate, smooth-walled, bearing branches on conidiophores with phialides in verticils, branching 1–2 levels verticillate, Conidiogenous cells 30–45 × 2–3 µm (x̄ = 38.9 × 2.7 μm, n = 20), phialidic, integrated, terminal, determinate, hyaline, cylindrical to subcylindrical, with a discrete flared collarettes, smooth-walled. Conidia 6–7 × 4–5 µm (x̄ = 6.5 × 4.2 μm, n = 30), arranged in false heads on conidiogenous cells tips, solitary, globose to subglobose, ellipsoidal, rounded at both ends, hyaline, aseptate, smooth- and thick-walled.
Culture characteristics: Colonies on PDA, 1.5–2 cm diam after one week at room temperature, margin regular, smooth surface, entire edge, cottony or woolly, whitish, well-defined edges with no pigmentation of the agar, reverse brown to dark yellow ate centre, pale yellow at the edge.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Chenghai Lake, 28 May 2021, L.L. Li, H372 (KUN-HKAS 125796), living culture, KUNCC 22–12526. CHINA, Yunnan Province, Cangshan Mountain, on submerged decaying wood in a stream, 1 March 2019, Z.Q. Zhang, S3093 (KUN-HKAS 125780), living culture, KUNCC 22–12463.
GenBank number: KUNCC 22–12526: ITS = OP876709, LSU = OP875065, tub2 = OQ025189. KUNCC 22–12463: ITS = OP876734, LSU = OP875088, tub2 = OQ025198.
Notes: In our phylogenetic analysis, our new isolate clustered with two strains of Paracremonium binnewijzendii with 100% ML/ 100% MP/ 1.00 PP support (Fig. 29). Paracremonium binnewijzendii was described by Crous et al. (2017), and is characterized by subcylindrical, hyaline, smooth, phialides with an inconspicuous collarette conidiogenous cells and aseptate, ellipsoidal, smooth conidia. Morphologically, our species is almost identical to the holotype of P. binnewijzendii, except the conidiophores of our new isolate that bear branches on conidiophores with phialides in verticils, branching 1–2 levels verticillate. Their morphological differences are probably due to the different growing environments and hosts. The holotype of P. binnewijzendii was collected in soil from stream embankment in the Netherlands. While, our new isolated was collected on submerged decaying wood from freshwater habitats in China, and it is a new record for China.
Phylogenetic tree based on RAxML analyses of a combined ITS, LSU and tub2 dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Nalanthamala vermoesenii (CBS 230.48 and CBS 110893). The combined gene analysis included 23 strains with 2013 characters after aligned including gaps (ITS: 544 bp, LSU: 864 bp, tub2: 605 bp) of which 319 were parsimony-informative, 67 were parsimony-uninformative and 1627 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of—5662.553723. The matrix had 402 distinct alignment patterns, with 18.25% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.231117, C = 0.267958, G = 0.280534, T = 0.220391; substitution rates AC = 1.209101, AG = 2.644357, AT = 1.498111, CG = 0.870374, CT = 7.193584, GT = 1.000000; gamma distribution shape parameter α = 0.160525. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Sedecimiella K.L. Pang, Alias & E.B.G. Jones, in Pang, Alias, Chiang, Vrijmoed & Jones, Bot. Mar. 53(6): 495 (2010).
Sedecimiella was introduced with a single species S. taiwanensis by Pang et al. (2010). Sedecimiella taiwanensis is a mangrove-based marine fungus collected from Taiwan, China. Sedecimiella taiwanensis is placed in Hypocreales genera incertae sedis (Pang et al. 2010; Hyde et al. 2020a) and was characterized by orange to dark brown, pyriform with globose to subglobose venter, coriaceous, ostiolate ascomata, two-layered peridium, 16-spored, unitunicate, cylindrical asci with short pedicellate lacking an apical pore and globose, one-celled, hyaline ascospores. In our phylogenetic analysis, Sedecimiella taiwanensis clustered with two Acremonium species (A. minutisporum and A. vitellinum) within Nectriaceae (Fig. 1). We therefore place S. taiwanensis in Nectriaceae. However, the relationship between S. taiwanensis and the two species of Acremonium (A. minutisporum and A. vitellinum) needs to be resolved.
ThelonectriaP. Chaverri & Salgado, in Chaverri, Salgado, Hirooka, Rossman & Samuels, Stud. Mycol. 68: 76 (2011).
Thelonectria was introduced to accommodate nectria-like sexual morph and a cylindrocarpon-like asexual morph (Brayford and Samuels 1993; Brayford et al. 2004; Chaverri et al. 2011; Salgado-Salazar et al. 2015, 2016). The genus is typified by Thelonectria discophora and is characterized by inconspicuous stromata, superficial, globose, subglobose, or pyriform to elongated and smooth or warty perithecia, a 2–3-layered perithecial wall and smooth, hyaline, 1-septate ascospores. The asexual morphs of Thelonectria are Cylindrocarpon-like and characterized by microconidia rare, sometimes seen on natural substrata; chlamydospores rare, abundant in one species; conidiophores arising laterally from hyphae, irregularly branched conidiophores or forming fascicles; phialides cylindrical; macroconidia curved, often broadest at upper third, with rounded apical cells and flattened or rounded basal cells, (3–)5–7(–9)- septate, with inconspicuous hilum (Chaverri et al. 2011).
Thelonectria species are commonly found in temperate, subtropical and tropical regions and the taxa occur as saprobes on decaying bark, roots, dead or dying trees and soil (Guu et al. 2007; Chaverri et al. 2011; Salgado-Salazar et al. 2012, 2015). Currently, about 50 species are included in the genus (Index Fungorum 2023). This study introduces two new species of Thelonectria based on phylogenetic and morphological analyses.
Thelonectria aquatica D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900270; Facesoffungi number: FoF 13934; Fig. 30
Thelonectria aquatica (MFLU 22–0254, holotype). a–b Appearance of ascomata on the host. c, d Section through ascoma. e Section of peridium in 3% KOH. f Section through peridium. g Paraphyses, h–i Asci. j Ascal apical ring. k–m Ascospores. n, o Culture on PDA. Scale bars: c–d = 100 μm, e = 50 μm, f, h, i = 30 μm, g, k–m = 10 μm, j = 15 μm
Etymology: Referring to the aquatic habitat of this fungus.
Holotype: MFLU 22–0254.
Saprobic on submerged decaying wood. Sexual morph: Ascomata 155–285(–305) × 260–356(–378) µm (x̄ = 218.8 × 308.4 μm, n = 5), perithecial, superficial or with base partially immersed in substratum on a minute stroma, solitary to gregarious, pyriform to subglobose, slightly papillate, yellow to orange, KOH-, with a darker red ostiolar area when dry, surface smooth to slightly roughened. Peridium 20–40 μm (x̄ = 31.7 μm, n = 11), comprises two layers, outer layer comprised orange cells of textura angularis to textura prismatica, sometimes with large textura globulosa cells; inner layer pale yellow to hyaline cells of textura prismatica. Paraphyses 4–8 μm (x̄ = 6.0 μm, n = 20), hyaline, septate, branched, smooth-walled. Asci (86–)88.5–105(–115) × 6–9 µm (x̄ = 96.7 × 7.5 μm, n = 20), unitunicate, cylindrical, 8-spored, with an apical ring. Ascospores 12–14 × 4–6 µm (x̄ = 12.6 × 4.9 μm, n = 30), uniseriate, ellipsoid to broadly ellipsoid, uniseptate, slightly constricted at the septum, hyaline, guttulate, smooth. Asexual morph: Undetermined.
Culture characteristics: Ascospore germinated on PDA within 24 h. Colonies growing on MEA, reaching 2–2.5 cm in one week at room temperature. Mycelium superficial, circular, with entire margin, flat, smooth, from above white, from below dark yellowish at centre, white at the edge.
Material examined: THAILAND, Nakhon Phanom Province, on submerged decaying wood in a stream, 12 November 2018, D.F. Bao, B-170 (MFLU 22–0254, holotype), ex-type culture, MFLUCC 22–0169.
GenBank numbers: ITS = OP876736, LSU = OP875057.
Notes: In the phylogenetic analysis, two new isolates Thelonectria aquatica and T. cylindricospora clustered as sister taxa (Fig. 31). The nucleotide comparison between these species revealed 15 bp (2.7%) differences in the ITS gene region. We therefore identified them as two distinct species in Thelonectria as recommended by Jeewon and Hyde (2016).
Phylogenetic tree based on RAxML analyses of a combined act, ITS, LSU, act and tef1-α dataset. Tree topology of the RAxML and MP analyses are similar. The tree is rooted to Nectria cinnabarina (A.R. 4477). The combined gene analysis included 55 strains with 2812 characters after aligned including gaps (ITS: 571 bp, LSU: 822 bp, act: 584 bp, tef1-α: 835 bp), of which 531 were parsimony-informative, 237 were parsimony-uninformative and 2044 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -14,241.228239. The matrix had 859 distinct alignment patterns, with 27.68% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.225983, C = 0.284387, G = 0.261256, T = 0.228374; substitution rates AC = 1.159947, AG = 2.128499, AT = 1.486238, CG = 1.120206, CT = 4.721893, GT = 1.000000; gamma distribution shape parameter α = 0.163203. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% are given at each node
Thelonectria aquatica is phylogenetically close to T. beijingensis (Fig. 31). Thelonectria aquatica is similar to the latter species in having superficial, subglobose to globose, orange red to red ascomata, two layers peridium, subcylindrical asci with an apical ring and uniseriate, fusiform-ellipsoid, uniseptate, hyaline, smooth ascospores. However, T. aquatica differs from T. beijingensis in having smaller ascomata (155–285(–305) × 260–356(–378) vs. 320–391 × 305–385 μm) and ascospores (12–14 × 4–6 vs. 13–17 × 4–7 μm) (Zeng and Zhuang 2013). In addition, phylogenetic analysis also showed that they are distinct (Fig. 31).
Thelonectria cylindricospora D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900271; Facesoffungi number: FoF 13935; Fig. 32
Etymology: Referring the cylindrical conidia of this fungus.
Holotype: KUN-HKAS 125812.
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Conidiomata stromatic, sporodochial, solitary, superficial, hyaline to pale brown. Conidiophores 10–22 × 2.5–3.5 µm (x̄ = 16 × 3 μm, n = 30), semi-macronematous, sometimes reduced to conidiogenous cells, branched, hyaline, septate, smooth-walled. Conidiogenous cells phialidic, terminal, hyaline. Conidia 41–49.5 × 5–7 µm (x̄ = 45.2 × 5.8 μm, n = 40), acrogenous, cylindrical with rounded ends, slightly curved, gradually become narrower towards both ends, hyaline, aseptate, guttulate, smooth-walled.
Culture characteristics: Conidia germinating on PDA within 24 h. Colonies growing slowly on PDA medium, surface velvety, with entire edge, white, dense; center dark yellow, color decreasing from the center to periphery from below.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in a stream, May 2021, H.W. Shen, H385 (KUN-HKAS 125812, holotype), ex-type culture, KUNCC22–12530 = CGMCC 3.24277.
GenBank number: ITS = OP876710, LSU = OP875066.
Notes: In our phylogenetic analysis, T. cylindricospora clustered with T. aquatica, T. beijingensis and T. guangdongensis in a monophyletic clade within the genus (Fig. 31). Thelonectria cylindricospora resembles T. beijingensis in having branched, septate, hyaline conidiophores and phialidic, terminal, hyaline conidiogenous cells and cylindrical, curved, hyaline conidia. However, T. cylindricospora differs from T. beijingensis by its reduced and shorter conidiophores (10–22 vs. (28–)30–77(–80) μm) and and shorter conidia (41–49.5 vs. (59–)60–93(–93.5) μm). In addition, conidia of T. beijingensis are 0–3-septate, while conidia of T. cylindricospora are aseptate (Zeng and Zhuang 2013). Thelonectria cylindricospora can be distinguished from T. guangdongensis in having aseptate and shorter and wider conidia (41–49.5 × 5–7 vs. 48–70 × 4.8–5.3 μm). While, conidia of T. guangdongensis are 2–5-septate (Zeng and Zhuang 2019).
Volutella Fr., Syst. mycol. (Lundae) 3(2): 458, 466 (1832)
Volutella was established by Fries (1832) with V. ciliata (Alb. & Schwein.) as the type species. Currently, 151 records of this genus are listed in the Index Fungorum (2023). However, few species have been revised and transferred to other genera such as Coccinonectria, Lectera, Koorchaloma, Pseudonectria, Scolecofusarium and Volutellonectria (Gräfenhan et al. 2011; Luo and Zhuang 2012; Cannon et al. 2012b; Lombard et al. 2015; Koukol et al. 2020; Crous et al. 2021b) and most species were seldom reported after their original descriptions; Hence the status of these species is still uncertain need to be revised (Gräfenhan et al. 2011). Only 11 species have sequence data in GenBank. Gräfenhan et al. (2011) revised the genus and accepted three species viz. Volutella ciliata, V. consors and V. citrinella in Volutella. Recently, seven additional species are introduced in the genus (Dubey and Pandey 2013; Zhang et al. 2017; Tibpromma et al. 2018; Perera et al. 2020; Lechat et al. 2022).
Luo and Zhuang (2012) established a sexual genus Volutellonectria (Vo.) with Vo. consors as the type and indicated that the asexual morph of this species is represented by Volutella (V.) minima. However, Gräfenhan et al. (2011) synonymised V. minima under Vo. Consors. In addition, they introduced a new species Vo. asiana and a new combination Vo. ciliata (= V. ciliata) in the genus. Lombard et al. (2015) pointed out that Volutellonectria is confusing in nomenclatural terms and should be replaced by Volutella, which has priority by date. And this treatment was accepted by later studies (Lechat et al. 2022; Perera et al. 2023). Most Volutella species are represented by their asexual morphs, only several species were known as sexual morphs: V. asiana, V. ciliata, V. citronella, V. consors, V. delonicis, V. minutissima, V. saulensis and V. thonneliana (Luo and Zhuang 2012; Perera et al. 2020, 2023; Lechat et al. 2022). The sexual morphs of Volutella are characterized by perithecial, solitary, superficial, obpyriform ascomata with an acute apex, turning dark red in 3% KOH and yellow in lactic acid, unitunicate, clavate asci with an apical ring and uniseptate, hyaline, often smooth-walled ascospores (Luo and Zhuang 2012; Lechat et al. 2022).The asexual morphs of Volutella are characterized by discoid sporodochia with marginal setae, simple to verticillate conidiophores, compact and phialidic conidiogenous cells, and 1-celled, ovoid to oblong conidia; synasexual morph present in some species with two or more whorls of conidiogenous cells (Gräfenhan et al. 2011; Lombard et al. 2015; Tibpromma et al. 2018).
In this study, three fresh collections were collected from freshwater habitats, and were identified as Volutella ciliata based on both phylogenetic and morphological analyses. Volutella ciliata is the only Volutella species reported from freshwater habitats.
Volutella ciliata (Alb. & Schwein.) Fr., Syst. Mycol. 3: 467. 1832.
Index Fungorum number: IF 208513; Facesoffungi number: FoF 11027; Fig. 33
Volutella ciliata (KUN-HKAS 125813). a, b Appearance of conidiomata on the host. c–f Squashed conidioma with setae. g–k Conidiophores, conidiogenous cells and developing conidia. l–r Conidia. s Surface view of culture on PDA. t Reverse view of culture on PDA. Scale bars: c = 200 μm, d = 200 μm, e–f = 80 μm, g–i = 20 μm, j, k = 10 μm, l = 30μ m, m–r = 5 μm
Saprobic on decaying wood submerged in a lake. Sexual morph: Undetermined. Asexual morph: Conidiomata 90–150 µm diam (x̄ = 119.2 μm, n = 5) superficial, solitary to gregarious, hyaline, with a white to buff slimy conidial mass in center, cupulate, setose. Conidiomatal setae 250–360 × 5–7.5 µm (x̄ = 304.9 × 6.4 μm, n = 25), cylindrical, subhyaline to hyaline, septate, unbranched, straight or slightly curved, tapering to apex, swollen terminally or intermediately, smooth-walled. Sporodochia sessile, globose, cream yellow, with several marginal setae. Conidiophores formed from the innermost layers of basal stroma, cylindrical, hyaline, branched or unbranched, septate, smooth-walled. Conidiogenous cells 9–13 × 1.5–2.5 µm (x̄ = 11.1 × 2.1 μm, n = 20), phialidic, cylindrical to subcylindrical, integrated, determinate, hyaline, gathered into a dense parallel layer, smooth-walled. Conidia 4–5.5 × 2–2.5 µm (x̄ = 4.8 × 2.4 μm, n = 50), forming slimy heads on sporodochium, ellipsoid with round ends, aseptate, hyaline, with small guttules.
Material examined: CHINA, Yunnan Province, Yiliang County, on submerged decaying wood in a stream, 16 May 2021, H.W. Shen, H318 (KUN-HKAS 125813), living culture, KUNCC 22–12532. CHINA, Yunnan Province, Yiliang County, on submerged decaying wood in a stream, 16 May 2021, H.W. Shen, H369 (KUN-HKAS 125816), living culture, KUNCC 22–12474. CHINA, Yunnan Province, Kunming City, on submerged decaying wood in a stream, 10 June 2021, X.G. Tian, L741 (KUN-HKAS 125801), living culture, KUNCC 22–12531.
GenBank numbers: KUNCC 22–12532: ITS = OP876706, LSU = OP875062, cmd = OQ025182, acl = OQ064513, rpb2 = OQ077574. KUNCC 22–12474: ITS = OP876708, LSU = OP875064, cmd = OQ025183, his3 = OQ064511, acl = OQ064514, rpb2 = OQ077575. KUNCC 22–12531: ITS = OP876730, LSU = OP875083, cmd = OQ025184, acl = OQ064515.
Notes: In our phylogenetic analysis, three new isolates (KUNCC 22–12532, KUNCC 22–12474 and KUNCC 22–12531) clustered with Volutella ciliata and two strains of V. roseola in a well-supported clade (Fig. 15). The two V. roseola strains (CBS 377.55 and CBS 128258) were provided by Vu et al. (2019) without detailed morphological descriptions. Thus, we doubt the two strains are misidentified based on phylogenetic analysis. However, we are unable to revise these two strains as the morphology of these two strains are not available. Hence, further studies are required to resolve this problem based on morphological and phylogenetic analyses.
Phylogenetic analysis showed that our three new isolates clustered within V. ciliata clade. Morphologically, our new isolates fit well with the description of V. ciliata (Gräfenhan et al. 2011). Therefore, we identified the three new isolates as V. ciliata, which is the first time of this species was collected from freshwater habitats.
Sarocladiaceae L. Lombard, in Crous et al., Persoonia 41: 343 (2018).
Sarocladiaceae was recently established by Crous et al. (2018). The family comprises two genera, Parasarocladium and Sarocladium, placed in Hypocreales (Crous et al. 2018; Hyde et al. 2020a). Phylogenetic analysis of Crous et al. (2018) showed that the family is sister to Bionectriaceae. While, Hyde et al. (2020a) showed that Sarocladiaceae is close to Flammocladiellaceae. In our phylogenetic analysis, Sarocladiaceae formed a sister clade to Myrotheciomycetaceae with 86% ML/ 1.00 PP support values (Fig. 1). In this study, Sarocladium kiliense was collected from freshwater habitats for the first time in China.
Sarocladium W. Gams & D. Hawksw., Kavaka 3: 57 (1976) [1975].
Sarocladium was established with S. oryzae as the type by Gams and Hawksworth (1975). The genus is characterized by cylindrical, phialidic conidiogenous cells and the phialides arise solitarily from undifferentiated hyphae or conidiophores that are sparsely to densely branched, with ellipsoidal conidia formed in false heads (Summerbell et al. 2011; Ou et al. 2020). Summerbell et al. (2011) reviewed the taxonomy of Sarocladium and included the species that belong to the A. bacillisporum and A. strictum clades and accepted eight species in the genus. Since then, several new species have been introduced to the genus (Yeh and Kirschner 2014; Giraldo et al. 2015; Liu et al. 2017b; Crous et al. 2018; Ou et al. 2020). Currently, 30 species are included in the genus (Index Fungroum 2023).
Species of Sarocladium are distributed in many countries e.g., Canada, China, Egypt, Germany, India, Islands, Kenya, Solomon, Sri Lanka and USA, (Summerbell et al. 2011; Giraldo et al. 2012, 2015; Liu et al. 2017b; Crous et al. 2018). They have been reported as plant pathogens, endophytes, mycoparasites (Rivera-Varas et al. 2007), saprobes and a few are opportunistic human pathogens (Rivera-Varas et al. 2007; Yeh and Kirschner 2014; Giraldo et al 2015; Liu et al. 2017b; Crous et al. 2018; Ou et al. 2020).
Sarocladium kiliense (Grütz) Summerb., in Summerbell, Gueidan, Schroers, Hoog, Starink, Arocha Rosete, Guarro & Scott, Stud. Mycol. 68: 158 (2011)
Index Fungorum number: IF 519592; Facesoffungi number: FoF 05816; Fig. 34
Saprobic on decaying wood submerged in a lake. Sexual morph: Undetermined. Asexual morph: Conidiomata sporodochial, superficial, solitary to gregarious, hairy, velvety, brown with white conidia on the conidiomata, setose. Setae up to 400 um, cylindrical, septate, unbranched, flexuous, brown to dark brown, paler towards the apex, smooth-walled. Sporodochia globose, hyaline to subhyaline, with several marginal setae. Conidiophores formed from the innermost layers of basal stroma, reduced to conidiogenous cells, cylindrical, branched or unbranched, hyaline, aseptate, smooth-walled. Conidiogenous cells 10–13 × 1–2 µm (x̄ = 11.7 × 1.6 μm, n = 30), phialidic, cylindrical to subcylindrical, determinate, integrated, hyaline, gathered into a dense parallel layer, smooth-walled. Conidia 5–7 × 1–2 µm (x̄ = 5.9 × 1.5 μm, n = 45), narrower cylindrical, clavate, with round ends, aseptate, hyaline, guttulate, smooth-walled.
Material examined: CHINA, Yunnan Province, Dali City, on submerged decaying wood in Haixihai Lake, 22 July 2021, S.P. Huang, L965 (KUN-HKAS 125792), living culture, KUNCC 22–12465.
GenBank numbers: ITS = OP876731, LSU = OP875084.
Notes: In our phylogenetic analysis, the new isolate KUNCC 22–12465 clustered with the ex-type strain of Sarocladium kiliense (Fig. 35). Sarocladium kiliense was originally described as Cephalosporium incoloratum by Sukapure and Thirumalachar (1965). It has been synonymized several times, as Acremonium incoloratum (Gams 1971) and Acremonium kiliense (Grütz 1925) based on morphological characters. Summerbell et al. (2011) initially provided the sequence data for this species and based on phylogenetic analysis, they transferred this species to Sarocladium as S. kiliense.
Phylogenetic tree based on RAxML analyses of a combined act, ITS and LSU dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Kiflimonium curvulum (CBS 430.66). The combined gene analysis included 33 strains with 2230 characters after aligned including gaps (act: 795 bp, ITS: 573 bp and LSU: 862 bp), of which 338 were parsimony-informative, 198 were parsimony-uninformative and 1694 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -10,154.610543. The matrix had 668 distinct alignment patterns, with 23.12% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.224695, C = 0.284995, G = 0.272715, T = 0.217594; substitution rates AC = 1.214019, AG = 2.132685, AT = 1.767746, CG = 0.545386, CT = 6.639778, GT = 1.000000; gamma distribution shape parameter α = 0.163723. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Sarocladium kiliense is ubiquitous, commonly found from soil, also isolated from man, cattle and maize (Sukapure and Thirumalachar 1965). In this study our new isolate was collected on submerged wood from freshwater habitats in China. Morphologically, the conidiogenous cells and conidia of our new isolate are identical to S. kiliense. We compared the base pairs differences for the ITS gene region which revealed 2 bp differences. We therefore identified our new isolate as S. kiliense, and it is a new record for China.
Stachybotryaceae L. Lombard & Crous, Persoonia 32: 283 (2014).
Stachybotryaceae was introduced by Crous et al. (2014) to accommodate Myrothecium, Peethamabra and Stachybotrys. The taxonomy of this family was revised by Lombard et al. (2016) based on morphological characters and multi-locus phylogenetic analysis. They accepted 33 genera in the family, including 21 new genera. While, Wijayawardene et al. (2018) accepted 36 genera. The last treatment of Stachybotryaceae was provided by Hyde et al. (2020a), and accepted 39 genera. Species in Stachybotryaceae are characterized by asexual morphs with mononematous to sporodochial to synnematous conidiomata, usually with phialidic conidiogenous cells that produce 0–1-septate conidia in dark green dry chains or slimy masses. Only three species of Stachybotryaceae (Koorchalomella salmonispora, Stachybotrys chartarum and S. chlorohalonata) have been reported from freshwater habitats. In this study, two Memnoniella species viz. Memnoniella echinate and M. oenanthes were collected from freshwater habitats for the first time. Stachybotrys chartarum and S. chlorohalonata were recollected from freshwater habitats in China.
Memnoniella Höhn., Centbl. Bakt. ParasitKde, Abt. II 60: 16 (1923) [1924].
Memnoniella was introduced by Von Höhnel (1924) based on M. aterrima. It is characterized by macronematous, mononematous, unbranched conidiophores, phialidic conidiogenous cells with conspicuous collarettes, and unicellular, aseptate, smooth to verrucose conidia arranged in dry chains or slimy masses. The morphology of Memnoniella and Stachybotrys are similar and several studies have treated them as congeneric. (Jong and Davies 1976; Castlebury et al. 2004; Wang et al. 2015). However, Lombard et al. (2016) showed that Memnoniella species forming a well-supported clade distant to the Stachybotrys clade, hence, Memnoniella was resurrected as a distinct genus in Stachybotryaceae which was accepted by later studies (Doilom et al. 2017; Hyde et al. 2020a; Mapook et al. 2020; Samarakoon et al. 2021). Currently, 24 records of Memnoniella are listed in Index Fungorum (2023), of which 10 species were transferred to other genera viz. Brevistachys and Stachybotrys. In this study, we introduce two new habitat and geographic records of Memnoniella.
Memnoniella echinata (Rivolta) Galloway, Trans. Br. mycol. Soc. 18(2): 165 (1933).
Index Fungorum number: IF 263706; Facesoffungi number: FoF 09367; Fig. 36
Saprobic on submerged decaying wood. Sexual morph: undetermined. Asexual morph: hyphomycetous. Colonies on the substrate superficial, gregarious, velvety, black, hairy. Mycelium mostly immersed in the substratum, composed branched, septate, pale brown to brown, smooth-walled hyphae. Conidiophores 65–100 × 3–5 µm (x̄ = 82.3 × 4.2 μm, n = 15), macronematous, mononematous, cylindrical, simple, erect, straight or slightly flexuous, unbranched or rarely branched, verrucose at surface, thick-walled, often becoming roughened, especially towards the apex, septate, hyaline at the base, olivaceous brown to dark brown at the apex, bearing a crown of phialides at the tip. Conidiogenous cells 7–9 × 3–5 µm (x̄ = 7.7 × 4.0 μm, n = 25), monophialidic, determinate, terminal, discrete, obovoid, forming a more or less compact head, with peripheral ones somewhat curved, smooth, sub-hyaline when young, olivaceous brown with age. Conidia 3–4 × 5–6 µm (x̄ = 3.6 × 5.3 μm, n = 30), catenate, often aggregated as large glistening heads in black, form in a long-chains, globose to subglobose, oval, verrucose at surface, aseptate, olivaceous brown to dark brown.
Culture characteristics: Colonies on PDA attaining 5 cm diam., within three weeks at room temperature, white in the beginning and brown to black with age, greyish white at middle, grey to brown at edge, circular, entire edge with raised on media surface, velvety.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Chenghai Lake, 28 May 2021, H.W. Shen, H406 (KUN-HKAS 125795), living culture, KUNCC 22–12529.
GenBank numbers: ITS = OP876714, LSU = OP875070, tub2 = OQ025191, tef1-α = OQ064519, rpb2 = OQ077577.
Notes: During our investigation of freshwater fungi from China, a stachybotrys-like taxon was collected and identified as Memnoniella echinata based on morphology and phylogeny. Our collection fits well with M. echinata in having macronematous, mononematous, erect conidiophores, monophialidic, discrete, determinate, terminal conidiogenous cells and globose to subglobose, olivaceous brown to dark brown, verrucose conidia (Lombard et al. 2016; Tennakoon et al. 2021). Phylogenetic analysis showed that the new collection clustered with four strains of M. echinata (Fig. 37). Memnoniella echinata has a worldwide distribution, commonly found in soil (Jarvis et al. 1998; Lombard et al. 2016; Tennakoon et al. 2021). Our collection was from submerged wood in freshwater habitats in China for the first time.
Phylogenetic tree based on RAxML analyses of a combined cmd, ITS, rpb2, tef1-α and tub2 dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Peethambara sundara (CBS 646.77 and CBS 521.96). The combined gene analysis included 81 strains with 2955 characters after aligned including gaps (cmd: 714 bp, ITS: 602 bp, rpb2: 721 bp, tef1-α: 545 bp, tub2: 368 bp), of which 1432 were parsimony-informative, 109 were parsimony-uninformative and 1414 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -39,090.972868. The matrix had 1736 distinct alignment patterns, with 31.48% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.232711, C = 0.287296, G = 0.257431, T = 0.222563; substitution rates AC = 1.239272, AG = 3.467193, AT = 1.311736, CG = 0.931445, CT = 5.548817, GT = 1.000000; gamma distribution shape parameter α = 0.286873. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Memnoniella oenanthes (M.B. Ellis) L. Lombard & Crous, in Lombard, Houbraken, Decock, Samson, Meijer, Réblová, Groenewald & Crous, Persoonia 36: 199 (2016).
Index Fungorum number: IF 816007; Facesoffungi number: FoF 13936; Fig. 38
Saprobic on submerged wood in a lake. Sexual morph: undetermined. Asexual morph: colonies on the substrate, superficial, scattered, black. Conidiophores 212–302 × 6.5–10.5 µm (x̄ = 257.3 × 8.4 μm, n = 10), cylindrical, solitary, erect, unbranched, straight or flexuous, septate, smooth, hyaline to subhyaline at the base, gradually becoming brown to dark brown towards the apex, bearing a crown of phialides at the tip. Conidiogenous cells 8.5–12.5 × 2–4 µm (x̄ = 10.5 × 3.0 μm, n = 20), in a single whorl at the apex of the conidiophore, monophialidic, determinate, terminal, ellipsoidal or obovoid, slightly curved, subhyaline to pale brown, smooth or slightly verrucose. Conidia 9–11 × 4–6 µm (x̄ = 10.1 × 5.4 μm, n = 30), solitary, reniform, ellipsoidal, curved, tapering at both ends, pale brown when young, dark brown to black at maturity, aseptate, verrucose.
Culture characters: Colonies on PDA slow growing, reaching 2.5–3.0 cm diam. after 2 weeks at room temperature, white to cream, or pale yellowish at the margins, cream at the centre, distinguished from the margin by white embossed hyphae with grey tufts in the centre; slightly radiating; reverse white cream at the margin, yellowish at the centre.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Chenghai Lake, 28 May 2021, H.W. Shen, H389 (KUN-HKAS 125794), living culture, KUNCC 22–12473.
GenBank numbers: ITS = OP876712, LSU = OP875068, tub2 = OQ025190, rpb2 = OQ077576.
Notes: Memnoniella oenanthes was reported as Stachybotrys oenanthes on dead stems of Oenanthe crocata by Ellis (1971). Lombard et al. (2016) re-evaluated the taxonomy of Stachybotriaceae based on phylogenetic analysis and morphological characters. The phylogenetic analysis showed that S. oenanthes clustered within Memnoniella. They, therefore, transferred S. oenanthes to Memnoniella as M. oenanthes.
Memnoniella oenanthes is characterized by erect, simple, 1–2 septate, solitary, smooth conidiophores, monophialidic, cylindrical or obovoid conidiogenous cells and reniform or ellipsoidal, smooth or verrucose conidia (Eills 1971, 1976; Wang et al. 2015). Our collection fits well with the description of M. oenanthes except the size of conidiophores and conidiogenous cells. The conidiophores of our collection are much longer (212–302 vs. 120–180 μm) and conidiogenous cells are smaller (8.5–12.5 × 2–4 vs.12–21 × 4–7 μm) than the holotype (Ellis 1971, 1976). Phylogenetic analysis showed that our new collection clustered with M. echinate with good support (Fig. 37). Thus, we identified our new collection as M. echinates based on phylogeny and morphology. Memnoniella echinate has been reported as saprobes on the stems of Euphorbia tirukalli and Oenanthe crocata from Guernsey and India. In this study, our new isolate was collected from freshwater habitats in China and it is a new record for China.
Stachybotrys Corda, Icon. fung. (Prague) 1: 21 (1837).
The asexual genus Stachybotrys is ubiquitous distributed worldwide. Species in the genus are common in soil, plant litter (hay, straw, cereal grains, and decaying plant debris) and air and a few species have been found from damp paper, cotton, linen, cellulose-based building materials water-damaged indoor buildings, and air ducts from both aquatic and terrestrial habitats (Izabel et al. 2010; Lombard et al. 2016; Hyde et al. 2020a). Stachybotrys was established by Corda (1837) with S. atrus as the type. The genus was revised by Lombard et al. (2016), their phylogenetic analysis showed that Stachybotrys s.l. is poloyphyletic and it can be segregated into ten genera, which is also supported by morphological observations. Hence, Lombard et al. (2016) refined the generic concept of Stachybotrys in a strict sense as conidiophores branching at the basal septum and the formation of thick-walled conidia sometimes bearing ornamentations.
Stachybotrys has been linked to the sexual genera Melanopsamma (Castlebury et al. 2004; Tang et al. 2007; Wang et al. 2015) and Ornatispora (Hyde and Goh 1999; Whitton et al. 2012; Wang et al. 2015). The sexual morph of M. pomiformis (type species) is linked to S. albipes and this was confirmed by ITS sequence analysis of Wang et al. (2015). Therefore, Wang et al. (2015) synonymized Melanopsamma under Stachybotrys. The sexual genus Ornatispora was introduced by Hyde and Goh (1999), they introduced O. taiwanensis with a Stachybotrys asexual state. In addition, Whitton et al. (2012) linked O. novaezelandiae to S. freycinetiae, and they found that O. nepalensis and O. taiwanensis have Stachybotrys asexual morphs. Morphologies of Ornatispora and Melanopsamma are similar, thus, Wang et al. (2015) synonymized Ornatispora under Stachybotrys. The sexual morphs of Stachybotrys are characterized by perithecial, stromatic, ksubglobose to obpyriform, black ascomata with setae irregularly arranged over the surface, clavate, 8-spored asci with a refractive apical ring and cylindrical, 1-septate, subhyaline, verrucose, ascospores with slightly tapering apices, surrounded by a thick mucoid sheath (Lombard et al. 2015).
There are 88 records of Stachybotrys on Species Fungorum of which 33 species have DNA sequence data in GenBank. Recently, Samarakoon et al. (2021) introduced a new species S. musae on Banana. Currently, 88 species are included in the genus, of which, only Stachybotrys chartarum and S. chlorohalonatus have been reported from freshwater habitats. In this study, we reported S. chartarum and S. chlorohalonatus from freshwater habitats again.
Stachybotrys chartarum (M.B. Ellis) L. Lombard & Crous, in Lombard, Houbraken, Decock, Samson, Meijer, Réblová, Groenewald & Crous, Persoonia 36: 199 (2016).
Index Fungorum number: IF 306362; Facesoffungi number: FOF01247; Fig. 39
Saprobic on decaying wood submerged in a lake. Sexual morph: Undetermined. Asexual morph: Colonies on the substrate, superficial, effuse, in groups, dark brown to black. Conidiophores 35–75.5 × 3–5 µm (x̄ = 55.2 × 3.7 μm, n = 25), macronematous, mononematous, single or in groups, verrucose, branched, straight to slightly flexuous, brown to olivaceous brown, septate, terminating with a crown of phialides at the apex. Conidiogenous cells 9.5–11 × 3–5 µm (x̄ = 10.2 × 3.9 μm, n = 15), monophialidic, terminal, discrete, in groups of 4–6 at the apex of each conidiophore, clavate to subclavate, reddish brown, with a minute collarette at the tip. Conidia 11–15 × 6.5–15 µm (x̄ = 8.5 × 13.5 μm, n = 30), acrogenous, globose to subglobose, verrucose, aseptate, thick-walled, dark brown to black.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Chenghai Lake, 26 May 2021, H.W. Shen, H421 (KUN-HKAS 125798), living culture, KUNCC 22–12469. CHINA, Yunnan Province, on submerged decaying wood in Xihu Lake, 8 May 2021, S.P. Huang, L419 (KUN-HKAS 125781), living culture, KUNCC 22–12470. CHINA, Yunnan Province, on submerged decaying wood in Erhai Lake, 1 April 2021, S.P. Huang, L228 (KUN-HKAS 125791), living culture, KUNCC 22–12471. CHINA, Yunnan Province, on submerged decaying wood in Xihu Lake, 8 May 2021, S.P. Huang, L420 (KUN-HKAS 125783), living culture, KUNCC 22–12472.
GenBank numbers: KUNCC 22–12469: ITS = OP876717, rpb2 = OQ077578. KUNCC 22–12470: ITS = OP876722, LSU = OP875075, rpb2 = OQ077581. KUNCC 22–12471: ITS = OP876718, rpb2 = OQ077579. KUNCC 22–12472: ITS = OP876723, LSU = OP875076, rpb2 = OQ077582.
Notes: In our phylogenetic analysis, four new isolates clustered with Stachybotrys chartarum with 100% ML/ 99% MP/ 1.00 PP support (Fig. 37). Morphologically, our isolates fit well with the description of S. chartarum, such as macronematous, mononematous, simple, erect, verrucose conidiophores, monophialidic, terminal, discrete conidiogenous cells and verrucose, aseptate conidia (Ellis 1971; Jong and Davis 1976; Matsushima 1975; Elidemir et al. 1999; Whitton et al. 2001; Wang et al. 2015). However, the conidia of our new isolates are globose to subglobose, while S. chartarum has ellipsoidal conidia (Elidemir et al. 1999).
Stachybotrys chartarum has a worldwide distribution and is often found in soil. It is reported as plant and human pathogens, saprobes and endophytes from aquatic and terrestrial habitats (Ellis 1971; Matsushima 1975; Jong and davis 1976; Elidemir et al. 1999; Whitton et al. 2001; Wang et al. 2015; Lombard et al. 2016). Luo et al. (2019) firstly reported S. chartarum from freshwater habitats. In this study, we recollected S. chartarum from freshwater habitats in China.
Stachybotrys chlorohalonata B. Andersen & Thrane [as 'chlorohalonata'], in Andersen, Nielsen, Thrane, Szaro, Taylor & Jarvis, Mycologia 95(6): 1228 (2003).
Index Fungorum number: IF 626958; Facesoffungi number: FoF 05447; Fig. 40
Saprobic on decaying wood submerged in a lake. Sexual morph: Undetermined. Asexual morph: Colonies on the substrate, superficial, effuse, dark brown to black. Conidiophores 36.5–68 × 3–4 µm (x̄ = 52.4 × 3.5 μm, n = 20), macronematous, mononematous, branched, verrucose, branched, straight to slightly flexuous, subhyaline to hyaline at the base, gradually becoming brown to dark brown towards the apes, smooth or slightly verrucose at maturity, granulate on the surface, septate, terminating with a crown of phialides at the apex. Conidiogenous cells 7–9 × 2–4 µm (x̄ = 7.9 × 3.2 μm, n = 20), monophialidic, discrete, determinate, terminal, clavate to subclavate, subhyaline when young, grey to greyish brown, smooth-walled, with conspicuous collarettes. Conidia 8–9 × 5–8 µm (x̄ = 8.6 × 6.4 μm, n = 50), acrogenous, ellipsoidal with round ends, verrucose, aseptate, guttulate, thick-walled, olive to dark brown or black.
Culture characteristics: Colonies on PDA reaching 3 cm diam, after 14 days at room temperature, white at first, irregular, raised, undulate, rough, after maturity, smooth at the margin, white from above, reverse cream to yellow at the margin, dark yellowish brown at centre.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Fuxian Lake, 12 July 2021, H.W. Shen, L496 (KUN-HKAS 125806), living culture, KUNCC 22–12467. CHINA, Yunnan Province, on submerged decaying wood in Fuxian Lake, 12 July 2021, H.W. Shen, L620 (KUN-HKAS 125805), living culture, KUNCC 22–12466. CHINA, Yunnan Province, on submerged decaying wood in Xihu Lake, 8 May 2021, S.P. Huang, L417 (KUN-HKAS 125784), living culture, KUNCC 22–12527. CHINA, Yunnan Province, on submerged decaying wood in Hixihai Lake, 22 July 2021, S.P. Huang, L713 (KUN-HKAS 125793), living culture, KUNCC 22–12468.
GenBank numbers: KUNCC 22–12467: LSU = OP875085, tef1-α = OQ064521, rpb2 = OQ077583. KUNCC 22–12466: ITS = OP876726, LSU = OP875079, tef1-α = OQ064523, rpb2 = OQ077585. KUNCC 22–12527: ITS = OP876721, LSU = OP875074, rpb2 = OQ077580. KUNCC 22–12468: ITS = OP876728, LSU = OP875081, tef1-α = OQ064524, rpb2 = OQ077586.
Notes: Stachybotrys chlorohalonata was introduced by Andersen et al. (2003) and isolated from cardboard on gypsum board. Stachybotrys chlorohalonata likely inhabits wet cellulose-containing materials such as fabric, hay, seaweed, grain, paper and soil and is found in Belgium, Denmark, Finland, Iraq, New Guinea, Spain and the USA. This study collected four fresh collections from freshwater habitats in China. Phylogenetic analysis showed that the four new isolates grouped with S. chlorohalonata (Fig. 37). With identical morphology to each, they were identified as S. chlorohalonata.
Hypocreales genera incertae sedis.
Emericellopsis J.F.H. Beyma, Antonie van Leeuwenhoek 6: 264 (1940) [1939].
Emericellopsis was described by Van Beyma (1940) with E. terricola as the type species. Since then, several species have been introduced worldwide in various habitats such as agricultural and forest soils, peat, rhizomes, prairies and freshwater-, estuarine- and marine-mud sediments (Stolk 1955; Gilman 1957; Mathur and Thirumalachar 1960, 1962; Backus and Orpurt 1961; Davidson and Christensen 1971; Zuccaro et al. 2004; Grum-Grzhimaylo et al. 2013). The placement of Emericellopsis has been revised several times by different authors. Emericellopsis was initially placed in Eurotiales (Van Beyma 1940). Phylogenetic analysis of Emericellopsis was firstly provided by Glenn et al. (1996) and placed in Hypocreales. Ogawa et al. (1997) set Emericellopsis in Hypocreaceae based on SSU and LSU sequence data. Subsequently, the genus was transferred to Bionectriaceae (Rossman et al. 1999, 2001; Grum-Grzhimaylo et al. 2013). Recently, Crous et al. (2018) transferred Emericellopsis to Myrotheciomycetaceae based on phylogenetic analysis. However, they did not include the type species of Emericellopsis (E. terricola) in their phylogenetic analysis.
In our phylogenetic analysis, Emericellopsis did not cluster within Myrotheciomycetaceae. It grouped with an Acremonium clade (including four species, A. alternatum, A. charticola, A. sclerotigenum and A. tubakii) and an unverified clade which comprises four species (A. fusidioides, Bulbithecium hyalosporum, Hapsidospora irregularis, and Leucosphaerina arxii) within Hypocreales with high support (95%ML/ 1.00 PP, Fig. 1). We, therefore, exclude Emericellopsis from Myrotheciomycetaceae. Our phylogenetic analysis showed that Bionectriaceae, Tilachlidiaceae, Emericellopsis, an Acremonium clade (including four species, A. alternatum, A. charticola, A. sclerotigenum and A. tubakii) and an unverified clade (A. fusidioides, B. hyalosporum, H. irregularis, and L. arxii) formed a monophyletic clade within Hypocreales (Fig. 1). Thus, this clade may need further revision as to whether they should be combined in Bionectriaceae. However, based on our current phylogenetic analysis, we place Emericellopsis in Hypocreales genera incertae sedis.
Microascales Luttr. ex Benny & R.K. Benj., Mycotaxon 12(1): 40 (1980).
The last treatment of Microascales was provided by Wijayawardene et al. (2022) comprising seven families viz. Ceratocystidaceae, Chadefaudiellaceae, Gondwanamycetaceae, Graphiaceae, Halosphaeriaceae, Microascaceae and Triadelphiaceae. Our phylogenetic analysis, includes all the families of Microascales except Chadefaudiellaceae which lacks sequence data. Our phylogenetic analysis showed that Ceratocystidaceae, Graphiaceae, Microascaceae and Triadelphiaceae formed well-supported monophyletic clades within Microascales (Fig. 1). While, Halosphaeriaceae is polyphyletic in our phylogenetic tree, the monotypic genera Nautosphaeria and Tubakiella are clustered out of Halosphaeriaceae (Fig. 1). Previous phylogenetic studies of Nautosphaeria and Tubakiella showed that the two genera always clustered together and were basal to Halosphaeriaceae (Sakayaroja et al. 2005a, b, 2011), while our phylogenetic analysis showed that Nautosphaeria and Tubakiella clustered together in an independent lineage within Microascales which is consistent with Hyde et al. (2020a). Hence, Nautosphaeria and Tubakiella are excluded from Halosphaeriaceae and placed in Microascales genera incertae sedis. In addition, our phylogenetic analysis showed that Cornuvesica species formed a monophyletic clade within Microascales (Fig. 1). Thus, a new family Cornuvesicaceae is introduced for Cornuvesica based on phylogenetic analysis, divergence time estimates and morphology.
Cornuvesicaceae D.F. Bao, K.D. Hyde & Z.L. Luo, fam. nov.
Index Fungorum number: IF 900272; Facesoffungi number: FoF 13937.
Etymology: Named after the type genus, Cornuvesica.
Type genus: Cornuvesica C.D. Viljoen, M.J. Wingf. & K. Jacobs.
On wood. Sexual morph: Ascomata superficial, scattered or in groups, dark, brown to black. Peridium firm, pseudoparenchymatous, textura epidermoidea to angularis. Ostiole hyphae convergent, compactly adhered to each other, pale brown to hyaline. Paraphyses terminating in obtuse apices, converging to form a narrow opening or slightly protruding beyond apical part of neck. Asci evanescent, deliquescing at an early stage. Ascospores falcate, straight or slightly curved, 1-septate, subhyaline, surrounded by hyaline sheath with both ends attenuated. Asexual morph: thielaviopsis-like with two distinct ranges of conidial dimensions. Micro-conidiophores abundant, straight, unbranched or branched, hyaline or rarely pale brown, smooth, septate. Macro-conidiophores less common than those of smaller size, formed on hyphae originated from perithecium, straight, unbranched or branched, pale brown to brown, smooth-walled, septate. Micro-conidiogenous cells phialidic, collarette in distinct, hyaline or occasionally pale brown, discrete or integrated, intercalary or terminal, cylindrical gradually tapering to the apex. Macro-conidiogenous cells phialidic, collarette indistinct, pale brown, discrete or integrated, intercalary or terminal, cylindrical gradually tapering to the apex. Micro-conidia hyaline, oblong with truncate ends, aseptate, smooth-walled, in chains, endogenous. Macro-conidia hyaline, doliiform, aseptate, smooth-walled, in chains, endogenous (Viljoen et al. 2000; Marincowitz et al. 2015).
Notes: Cornuvesica was introduced by Viljoen et al. (2000) to accommodate Ceratocystiopsis falcata, originally placed in Ceratocystis (Wright and Cain 1961). A phylogenetic analysis based on SSU sequence data showed that Cornuvesica clustered as a sister clade to Ceratocystis within Microascales (Hausner and Reid 2004). Therefore, Cornuvesica was placed in Ceratocystidaceae based on phylogenetic analysis (Réblová et al. 2011; de Beer et al. 2013a, 2014). Marincowitz et al. (2015) introduced three additional species in the genus. They reconstructed the phylogenetic analysis for Cornuvesica based on SSU and LSU sequence data and the result showed that Cornuvesica formed a monophyletic clade sister to Ceratocystidaceae within Microascales. Marincowitz et al. (2015) further placed Cornuvesica in Microascales genera incertae sedis. The placement of the genus was accepted by Hyde et al. (2020a) and Wijayawardene et al. (2020).
In our phylogenetic analysis, Cornuvesica formed a monophyletic clade grouping with Ceratocystidaceae, Graphiaceae, Gondwanamycetaceae and two strains of Sporendocladia bactrospora (Microascales genera incertae sedis) (Fig. 1). Morphologically, Cornuvesica is known by both sexual and asexual morphs. Cornuvesica is different from Ceratocystidaceae in having falcate, straight or slightly curved, 1-septate, subhyaline ascospores. While, ascospores of Ceratocystidaceae are aseptate, hyaline and varied in shape, hat-shaped, ellipsoidal or elongate to slightly curved, with rounded ends, oblong, cylindrical or narrowly fusiform to spindle-shaped. Graphiaceae is only known by the asexual morphs. It is different from Cornuvesica in having macronematous, synnematous conidiophores, conidiogenous cells in whorls of two to six, with annellidic extensions and cylindrical to obovoid conidia often with a distinct basal frill. While Cornuvesica has two types of conidiophores which are mononematous, conidiogenous cells of Cornuvesica are phialidic, collarette in distinct, discrete or integrated, intercalary or terminal and conidia are cylindrical to obovoid, often with a distinct basal frill. Gondwanamycetaceae is different from Cornuvesica in having hyaline, aseptate, fusiform to lunate or falcate or allantoid ascospores without sheath, mono-verticillate or penicillate conidiophores and cylindrical to allantoid, slimy conidia that are not in chains. Whereas, Cornuvesica has falcate or allantoid, 1-septate ascospores surrounded by hyaline sheath with both ends attenuated and Cornuvesica has two types of conidiophores which are mononematous, unbranched or branched and conidia are cylindrical to obovoid, often with a distinct basal frill.
In addition, Cornuvesica is phylogenetically distinct from Ceratocystidaceae, Graphiaceae, Gondwanamycetaceae (Fig. 1). The stem age of Cornuvesicaceae (209.88 MYA) falls within the family range (Hyde et al. 2017). Therefore, a new family Cornuvesicaceae is introduced to accommodate Cornuvesica based on morphology, phylogeny and divergence time estimates.
Halosphaeriaceae E. Müll. & Arx ex Kohlm., Can. J. Bot. 50: 1951 (1972).
Halosphaeriaceae (Microascales, Hypocrealmycetidae) comprises 64 genera (Pang 2002; Jones et al. 2009, 2015, 2017, 2019; Maharachchikumbura et al. 2015; Wijayawardene et al. 2017, 2018, 2022; Hyde et al. 2020a). Species in the family are commonly found in marine habitats and few species are from freshwater and terrestrial habitats (Jones et al. 2009, 2015, 2017, 2019; Hyde et al. 2020a). In our phylogenetic analysis, Halosphaeriaceae is polyphyletic and the genera Nautosphaeria and Tubakiella clustered out of Halosphaeriaceae (Fig. 1). Thus, Nautosphaeria and Tubakiella are excluded from Halosphaeriaceae and placed in Microascales genera incertae sedis based on phylogenetic analysis.
Ascosacculus J. Campb., J.L. Anderson & Shearer, Mycologia 95(3): 545 (2003).
Ascosacculus was introduced by Campbell et al. (2003) to accommodate two Halosarpheia species (A. aquaticus and A. heteroguttulatus) with A. aquaticus as the type species. The genus is known only by sexual moprs and it is characterized by immersed or superficial, globose to subglobose ascomata with long neck, 8-spored, thin-walled, early deliquescent asci, which lacking an apical pore and apical apparatus and fusiform to cylindrical, hyaline, 1-septate ascospores filled with many small guttules and having a hamate appendage at each apex that unfurls to form long, threadlike, sticky appendages (Campbell et al. 2003).
The genus currently comprises three species, and all three were reported from freshwater habitats (Wong et al. 1998; Campbell et al. 2003; Rosique-Gil et al. 2008; Luo et al. 2019). In this study, a new species A. thailandicus is introduced, which was collected from a freshwater habitat in Thailand.
Ascosacculus thailandicus D.F. Bao, K.D. Hyde & Z.L. Luo, sp. nov.
Index Fungorum number: IF 900273; Facesoffungi number: FoF 13938; Fig. 41
Etymology: Referring to Thailand, where species was collected.
Holotype – MFLU 22–0255.
Saprobic on decaying wood, submerged in freshwater habitats. Sexual morph: Ascomata 90–200 × 90–130 µm (x̄ = 144.3 × 109.3 μm, n = 5), immersed to semi-immersed, with a hyaline ostiole on the substrate, scattered, globose to subglobose or pyriform, papillate, hyaline to pale brown. Ostiole central, hyaline. Peridium 17–22 µm wide (x̄ = 19.5 μm, n = 30), comprises two layers, outer layers composed of dark brown cells of textura angularis, inner layer composed rectangular to subglobose hyaline cells. Paraphyses 6–10 µm (x̄ = 7.9 μm, n = 15), hyaline, septate, constricted at the septa, branched, smooth-walled. Asci 65–80 × 20–30 µm (x̄ = 72.1 × 25.3 μm, n = 20), 8-spored, unitunicate, obovoid to broadly clavate, sessile to short pedicellate, without an apical ring. Ascospores 25–30 × 8–10 µm (x̄ = 28.6 × 9.1 μm, n = 30), bi-seriate, hyaline, fusiform to ellipsoidal, straight, with a supra-median primary septum, slightly constrict at the septum, guttulate, smooth-walled, with or without a sheath and appendages. Asexual morph: Undetermined.
Material examined: THAILAND, Nakhon Phanom Province, on submerged decaying wood in a small river, 12 November 2018, D.F. Bao, B-165 (MFLU 22–0255, holotype), ex-type culture, MFLUCC 22–0170.
GenBank numbers: ITS = OP876735, LSU = OP875056, SSU = OP985137, tef1-α = OQ064525.
Notes: In the phylogenetic analysis, Ascosacculus thailandicus clustered with A. fusiformis (MFLUCC 14–0036) with 93% ML/ 82% MP/ 0.98 PP support (Fig. 42). The nucleotide comparison of ITS region between the two species revealed 5 bp differences. However, the two species differ. Ascosacculus fusiformis is characterized by unitunicate, cylindrical, pedicellate asci with a bilateral apical ring and uni-seriate, fusiform, 3-septate, hyaline, smooth-walled conidia with a thin mucilaginous sheath (Luo et al. 2019). While, A. thailandicus has obovoid to broadly clavate, sessile to short pedicellate asci and lacks an apical ring, ascospores are bi-seriate, hyaline, fusiform to ellipsoidal, uniseptate, and lack a mucilaginous sheath.
Phylogenetic tree based on RAxML analyses of a combined LSU, SSU and ITS dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Cylindrotrichum clavatum (CBS 125239) and C. gorii (DLUCC 0614). The combined gene analysis included 60 strains with 2607 characters after aligned including gaps (LSU: 1040 bp, SSU: 937 bp, ITS: 630 bp), of which 716 were parsimony-informative, 411 were parsimony-uninformative and 1480 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -19,573.549836. The matrix had 1232 distinct alignment patterns, with 37.17% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.252490, C = 0.226587, G = 0.285624, T = 0.235299; substitution rates AC = 1.161821, AG = 1.973334, AT = 1.190076, CG = 1.054753, CT = 5.721411, GT = 1.000000; gamma distribution shape parameter α = 0.286939. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Ascosacculus fusiformis differs from the morphological concepts of the genus, such as having cylindrical, pedicellate asci with an apical ring, and uniseriate, fusiform, 3-septate conidia. Asci of other Ascosacculus species are obovoid to broadly cylindrical lacking an apical ring and ascospores are bi-seriate and uniseptate. Thus, we doubt the reliability of the sequence data of A. fusiformis. Currently, only three species are introduced in the genus and with the few taxa sampled, a clear understanding of the characters of Ascosacculus cannot be provided. Hence, further fresh collections are required to better understand this genus.
Microascaceae Luttr. ex Malloch, Mycologia 62(4): 734 (1970).
Microascaceae was introduced by Luttrell (1951) to accommodate Microascus which was originally placed in Ophiostomataceae (Nannfeldt 1932) or Eurotiaceae (Emmonas and Dodge 1931; Moreau and Moreau 1953; Doguet 1957). Malloch (1970) validated the family and accepted Kernia, Lophotrichus and Petriellidium in the family. Sandoval-Denis et al. (2016a, b) revised Microascaceae and proposed several new taxa and combinations under Microascaceae based on multi-locus phylogenetic analysis. The last treatment of Microascaceae was provided by Hyde et al. (2020a), and 23 genera accepted. Species in this family have a worldwide distribution and most species are saprobes in soil, dung or on decaying plant materials (Seifert and Gams 2011; Sandoval-Denis et al. 2016a, b; Hyde et al. 2020a), while a few species are opportunistic pathogens of humans (de Hoog et al. 2011; Sandoval-Denis et al. 2013, 2016a, b; Lackner et al. 2014). In our phylogenetic analysis, Microascaceae clustered basal to Microascales, and the stem age of Microascaceae is around 153 MYA, which falls within the family range (Hyde et al. 2017).
Parascedosporium Gilgado, Gené, Cano & Guarro, Int. J. Syst. Evol. Microbiol. 57(9): 2176 (2007).
Notes: Parascedosporium was established by Gilgado et al. (2007) with P. tectonae as the type. Parascedosporium is a polymorphic genus with two asexual morphs, one is characterized by solitary conidiophores with sympodial conidia emerging from denticulate conidiogenous cells. The second one showed the features typical of Graphium wich has annellidic conidiogenous cells (Gilgado et al. 2007; Lackner and de Hoog 2011).
Lackner and de Hoog (2011) examined the ex-type culture (CBS 127.84) of Parascedosporium tectona and their phylogenetic analysis based on ITS sequence data showed that P. putredinis clustered with P. tectonae and there were maximally two base pairs differences within the clade. Therefore, they considered P. tectona as a synonym of P. putredinis. Later, de Beer et al. (2013b) examined the holotype of Parascedosporium tectona (IMI 95673d) and accepted the treatment of Lackner and de Hoog (2011). Zhang et al. (2021b) introduced another species P. sanyaense which was initially placed in Scedosporium. Zhang et al. (2021b) showed that Scedosporium sanyaense clustered with P. putredinis. They therefore transferred Scedosporium sanyaense to Parascedosporium as P. sanyaense. There are three Parascedosporium species listed in the Index Fungorum (2023). In this study, P. putredinis was collected from freshwater habitats in China.
Parascedosporium putredinis (Corda) Lackner & de Hoog, IMA Fungus 2(1): 44 (2011).
Index Fungorum number: IF 519652; Facesoffungi number: FoF 04557; Fig. 43
Parascedosporium putredinis (KUN-HKAS 125797). a, b Colonies on substrate. c–e Conidiophores and conidia. f–i Conidiogenous cells and conidia. j–n Conidia. o Surface view of culture on PDA. p Reverse view of culture on PDA. Scale bars: c–e = 300 μm, f = 30 μm, g, i, m, n = 20 μm, h = 10 μm, j–l = 5 μm
Saprobic on submerged decaying wood. Sexual morph: Undetermined. Asexual morph: Colonies effuse on natural substrate, scattered, dark brown. Mycelium partly immersed in substrate, composed of septate, branched, dark brown, smooth-walled hyphae. Conidiophores 670–918 × 13–25 μm (x̄ = 794 × 19.1 μm, n = 15), macronematous, synnematous, arising from a compact basal stroma, consisting of numerous individual conidiophores, erect, straight or flexuous, smooth-walled, dark brown, septate, with flaring conidiogenous head, containing a subhyaline to pale brown, mucoid conidial mass. Conidiogenous cells 16–24 × 1.5–2 μm (x̄ = 20 × 1.5 μm, n = 20), monoblastic, terminal, integrated, cylindrical, smooth-walled, hyaline to pale brown. Conidia 5–8 × 3–4 μm (x̄ = 6.3 × 3.2 μm, n = 30), acrogenous, solitary, subcylindrical to ellipsoidal, straight, rounded at both ends, aseptate, with small guttules, hyaline, smooth-walled.
Culture characteristics: Conidia germinated within 24 h on PDA, colonies grow rapidly on PDA at room temperature, reaching around 4 cm diam., after three weeks. Colonies on medium appear circular to irregular, medium dense, flat or effuse, with fimbriate edge, colonies from above and below white to greyish, white at middle, cream to pale grey at edge; reverse pale grey to pale yellowish grey.
Material examined: CHINA, Yunnan Province, on submerged decaying wood in Chenghai Lake, 28 May 2021, H.W Shen, H407 (KUN-HKAS 125797), living culture, KUNCC 22–12464.
GenBank numbers: ITS = OP876715, LSU = OP875071.
Notes: Parascedosporium putredinis was introduced by Lackner and de Hoog (2011). The species has a world-wide distribution and is reported from living plant leaves, seeds of Tectona grandis, other plant debris and soil or dung in Czech Republic, France, Jamaica, Japan, Thailand and the United Kingdom. In our phylogenetic analysis, our newly obtained strain clustered with 13 strains of P. putredinis with 100% ML/ 100% MP/ 1.00 PP support (Fig. 44). Morphology of our new isolate is quite similar to the holotype (Lackner and de Hoog 2011). Therefore, we identified our new isolate as P. putredinis and introduced as a new geographical and habitat record.
Phylogenetic tree based on RAxML analyses of a combined ITS and LSU dataset. Tree topology of the RAxML, MP and Bayesian analyses are similar. The tree is rooted to Trichoderma viride (DAOM JBT1003). The combined gene analysis included 35 strains with 1460 characters after aligned including gaps (ITS: 608 bp and LSU: 852 bp), of which 340 were parsimony-informative, 113 were parsimony-uninformative and 1007 characters were constant. The RAxML analysis of the combined dataset yielded the best scoring tree with a final ML optimization likelihood value of -7620.970690. The matrix had 567 distinct alignment patterns, with 39.27% undetermined characters or gaps. Estimated base frequencies were as follows: A = 0.240472, C = 0.261313, G = 0.275744, T = 0.222471; substitution rates AC = 1.665133, AG = 2.568340, AT = 2.153117, CG = 1.775983, CT = 5.631232, GT = 1.000000; gamma distribution shape parameter α = 0.208532. Bootstrap support values for RAxML (blue) and MP (red) greater than 70% and Bayesian posterior probabilities (black) greater than 0.95 are given at each node
Microascales genera incertae sedis
Gabarnaudia Samson & W. Gams, in Samson, Stud. Mycol. 6: 88 (1974).
Gabarnaudia was established by Samson (1974) with G. betae as the type species. Phylogenetic analysis of Hausner and Reid (2004) and de Beer et al. (2013b) showed that G. betae clustered within Sphaeronaemella (Melanconiellaceae, Diaporthomycetidae). Therefore, de Beer et al. (2013b, c) transferred G. betae to Sphaeronaemella. Gabarnaudia fimicola and G. humicola were also transferred to Sphaeronaemella (de Beer et al. 2013c). Currently, only G. cucumeris and G. tholispora are included in Gabarnaudia. Hyde et al. (2020a) and Wijayawardene et al. (2022) placed Gabarnaudia in Microascales genera incertae sedis. However, G. tholispora clustered with Calcarisporium arbuscula within the Hypocreales (de Beer et al. 2013c). Thus, de Beer et al. (2013b) doubted the reliability of sequence data of Gabarnaudia. In our phylogenetic analysis, G. tholispora clustered with Calcarisporium arbuscula within Hypocreales with 100%ML/1.00PP support (Fig. 1), consistent with de Beer et al. (2013b). Hence, the placement of G. tholispora needs further revision based on morphological characters and phylogenetic analysis. Gabarnaudia cucumeris was originally placed in Oospora, De Hoog et al. (1986) transferred it to Gabarnaudia based on morphological characters. However, de Beer et al. (2013c) mentioned that the status of this species is uncertain as the description of this species is not clear and lacks sequence data. Thus, the placement of Gabarnaudia needs revisions based on further morphological and phylogenetic analyses.
Nautosphaeria E.B.G. Jones, Transactions of the British Mycological Society 47 (1): 97 (1964).
Nautosphaeria was introduced by Jones (1964) with a single species N. cristaminuta. Nautosphaeria is characterized by spherical, hyaline to cream-colored ascomata, broadly clavate or ellipsoidal, pedunculate asci and one-celled, ellipsoidal, hyaline ascospores which possess a tuft of bristle-like appendages at each end and four tufts around the equator (Jones 1964). Previsous phylogenetic analysis showed that Nautosphaeria clustered with Tubakiella basal to Halosphaeriaceae (Sakayaroja et al. 2011). However, our phylogenetic analysis showed that N. cristaminuta clustered with Tubakiella galerita in a distinct clade within Microascales (Fig. 1). Hence, Nautosphaeria was excluded from Halosphaeriaceae. Divergence time estimates showed that the stem age of N. cristaminuta + T. galerita is around 181 MYA (Fig. 2). Both phylogenetic analysis and divergence time analysis suggested that N. cristaminuta and T. galerita can be transferred to a new family. However, the phylogenetic status of these two species is not stable. Hence, we place them in Microascales genera incertae sedis and further phylogenetic analysis is needed to clarify their placement.
Tubakiella Sakayaroj, K.L. Pang & E.B.G. Jones, Fungal Diversity 46: 97 (2011).
The monotypic genus Tubakiella was introduced with T. galerita by Sakayaroj et al. (2011), which was originally placed in Remispora. Sakayaroj et al. (2011) showed that two strains of T. galerita clustered with Nautosphaeria cristaminuta and Haligena elaterophora in a monophyletic clade basal to Halosphaeriaceae. Hence, Sakayaroj et al. (2011) established Tubakiella to accommodate T. galerita based on phylogenetic analysis. Hyde et al. (2020a) showed that T. galerita grouped with N. cristaminuta but clustered out of Halosphaeriaceae. Our phylogenetic analysis obtained a similar result (Fig. 1). Hence, Tubakiella is excluded from Halosphaeriaceae and placed in Microascales genera incertae sedis. Further phylogenetic studies are required to clarify the placement of Tubakiella.
Xenodactylariales D.F. Bao, K.D. Hyde & Z.L. Luo, ord. nov.
Index Fungorum number: IF 900274; Facesoffungi number: FoF 13939.
Type family: Xenodactylariaceae Crous, in Crous et al., Persoonia 41: 289 (2018).
Endophytic on plant tissue. Mycelium consists of smooth, hyaline, branched, septate, hyphae. Conidiophores reduced to conidiogenous cells on hyphae, erect to flexuous, hyaline, smooth-walled, with one to several denticulate apical loci. Conidia occurring in branched chains, hyaline, smooth-walled, subcylindrical, septate. (Adapted from Crous et al. 2018).
Notes: Xenodactylariaceae was introduced by Crous et al. (2018) with a single species X. thailandica which was collected on leaves of unidentified vine in Thailand. Phylogenetic analysis of Crous et al. (2018) showed that X. thailandica formed a distinct lineage within Hypocreales and close to Ophiocordycipitaceae. However, Crous et al. (2018) placed the family in Myrmecridiales, Diaporthomycetidae (Sordariomycetes). Hyde et al. (2020a) showed that Xenodactylariaceae grouped within Hypocreomycetidae and close to Torpedosporales. Recently, a phylogenetic analysis of Hyde et al. (2021) found that Xenodactylariaceae did not cluster with members of Diaporthomycetidae and suggested excluding this family from Diaporthomycetidae.
In our phylogenetic analysis, Xenodactylariaceae grouped within Hypocreomycetidae as a distinct lineage close to Torpedosporales (Fig. 1) which is consistent with the analysis of Hyde et al. (2020a). Xenodactylariaceae has subcylindrical, hyaline, septate conidia that are in branched chains. These characters are quite different from Torpedosporales; Conidia of Torpedosporales are solitary and helicoid e.g. Juncigenaceae has single, brown, helicoid conidia and conidia of Torpedosporaceae are solitary, irregularly helicoid and muriform (Hyde et al. 2020a). In addition, falls within the order range (Hyde et al. 2017). Thus, we transfer the family to Hypocreomycetidae in a newly introduced order Xenodactylariales based on the phylogenetic analysis and divergence time estimates.
Hypocreomycetidae families incertae sedis.
Campylosporaceae D.F. Bao, K.D. Hyde & Z.L. Luo, fam. nov.
Index Fungorum number: IF 900275; Facesoffungi number: FoF 13940.
Type genus: Campylospora Ranzoni, Farlowia 4(3): 373 (1953).
Saprobic on submerged leaves or endophytic in plants. Sexual morph: Undetermined. Asexual morph: Colonies hyaline to pale brown hyphae including variously shaped inflated cells. Conidiophores lateral or rarely terminal or intercalary, cylindrical or somewhat nodose, mostly simple or rarely sparsely branched. Conidiogenous cells integrated, typically proliferating sympodially. Conidia tetraradiate, hyaline, composed of two parts, deltoid and allantoid, both with two diverging branches at the ends; deltoid triangular to pyramidal, basal cells with ends rounded; apical cells of both parts rounded; branches aseptate. (Fiuza and Gusmão 2013; Marvanová and Laichmanová 2014).
Notes: The aquatic hyphomycetous genus Campylospora was introduced by Ranzoni (1953) with C. chaetocladia as the type species. Two additional species viz. C. filicladia and C. parvula were subsequently introduced in the genus (Kuzuha 1973; Nawawi 1974). Recently, C. brasiliensis and C. leptosome were introduced in Campylospora (Fiuza and Gusmão 2013; Marvanová and Laichmanová 2014). Currently, five species are accepted in the genus. All Campylospora species have been reported from freshwater habitats and are commonly found on submerged leaves or foams in stream. Campylospora chaetocladia and C. parvula have also been reported as endophytes from terrestrial habitats (Fisher and Petrini 1989; Sati and Belwal 2005). Campylospora species are widely distributed in worldwide e.g. Americas, Austria, Brazil, Ecuador, England, Hawaii, India, Malaysia, New Zealand, Peru, the South African Republic, Spain, Thailand, The USA and Venezuela (Nilsson 1962; Cowling 1963; Ranzoni 1979; Tubaki et al. 1983; Arambarri and Spinedi 1984; Aimer and Segedin 1985; Schoenlein-Crusius and Milanez 1990; Sridhar and Kaveriappa 1992; Matsushima 1993; Webster et al. 1994; Malosso 1995; Casas and Descals 1997; Schoenlein-Crusius 2002; Sakayaroj et al. 2005b; Cressa and Smits 2007; Smits et al. 2007; da Silva and Briedis 2009; Fiuza and Gusmão 2013).
Placement of Campylospora was unclear since it was established. Hyde et al. (2020a) and Wijayawardene (2022) place Campylospora in Hypocreomycetidae genera incertae sedis. In our phylogenetic analysis, Campylospora formed a monophyletic clade sister to Falcocladiaceae (the only family in Falcocladiales) within Hypocreomycetidae (Fig. 1). Our phylogenetic analysis suggests it can be introduced as a new family. In addition, the divergence time estimates showed that the stem age of Campylospora is 193.9 MYA which accords with the family level suggested by Hyde et al. (2017). Morphology of Campylospora is distinct in the subclass in having tetraradiate conidia, which are composed two parts of deltoid and allantoid, both with two diverging branches at the ends (Fiuza and Gusmão 2013; Marvanová and Laichmanová 2014). Hence, a new family Campylosporaceae is introduced to accommodate Campylospora based on phylogenetic analysis and divergence time estimates.
Excluded from Hypocreomycetidae
Faurelina Locq.-Lin., Revue Mycol., Paris 39(2): 127 (1975).
Faurelina was described by Locquin-Linard (1975) for several cleistothecial ascomycetes. The placement of Faurelina is controversial and has been changed many times by different authors (Locquin-Linard 1975; Parguey-Leduc and Locquin-Linard 1976; von Arx 1978; Cannon and Kirk 2007; Tang et al. 2007; Réblová et al. 2011; Maharachchikumbura et al. 2015; Wijayawardene et al. 2017; Hyde et al. 2020a). The last treatment of Faurelina was provided by Wijayawardene et al. (2022), and placed in Chadefaudiellaceae (Microascales).
Tang et al. (2007) firstly provided sequence data for a single strain of Faurelina indica (CBS 126.78) and this strain was similar to the type species of Ceratocystis (C. fimbriata). However, Réblová et al. (2011) doubted the reliability of this strain. They studied and sequenced two strains of F. indica including the ex-type strain (CBS 126.78), and confirmed that the strain of Tang et al. (2007) was based on different fungus. In addition, phylogenetic analysis of Réblová et al. (2011) showed that the two strains of F. indica have relationship with Didymellaceae (Pleosporales, Dothideomycetes). Therefore, we exclude Faurelina from Chadefaudiellaceae (Microascales). However, Hyde et al. (2020a) and Wijayawardene et al. (2022) placed Faurelina in Chadefaudiellaceae (Microascales). Our study is in agreement with Réblová et al. (2011) excluded Faurelina from Chadefaudiellaceae (Microascales). In our preliminary phylogenetic analysis, F. fimigena and F. indica clustered out of the Sordariomycetes (thus Faurelina was excluded from our phylogenetic analysis). The blast result showed that these two species have higher similarity to Dothideomycetes rather than Sordariomycetes. Our result is consistent with Réblová et al. (2011). Hence, we suggest to exclude Faurelina from Chadefaudiellaceae (Microascales). However, the placement of Faurelina need further studies based on phylogenetic analysis and morphological characteristics.
Discussion
Evolution of Hypocreomycetidae
The evolution of fungi is still a matter of debate (Calabon et al. 2021), and transitions from aquatic to terrestrial habitats have been commonly postulated (Hibbett and Binder 2001). The basal clade of the kingdom Fungi is Chytrids, also known as lower fungi, and are mainly found in the freshwater habitats (Raghukumar 2017). They developed uniflagellate zoospores, which helps to disperse in water (Raghukumar 2017). The unicellular group of fungi are thought to have changed their lifestyles several times and developed into multicellular organisms in the terrestrial environment, resulting in Ascomycota and Basidiomycota (Raghukumar 2017). The initial colonizer of terrestrial environment faced harsh physical environment thus, they underwent macroevolutionary jump (Selosse and Le Tacon 1998). The initial terrestrial colonization was formed by the association of fungus and phototrophs (Selosse and Le Tacon 1998). In some lineages, mutualism with fungi was an ancestral feature (Selosse and Le Tacon 1998). The colonization of fungi on land is thought to have started with the establishment of arbuscular mycorrhiza-like symbioses between fungi belonging to Glomeromycota and plant roots (Redecker et al. 2000; Raghukumar 2017). The fungi facilitated nutrient intake of these earlier plants (Bidartondo et al. 2011). Later studies revealed that the earliest mycorrhizal symbiosis was formed between Mucoromycota with land plants or dual Mucoromycotina-Glomeromycotina partnerships with land plants (Strullu-Derrien et al. 2014; Rimington et al. 2015; Feijen et al. 2018). Terrestrial fungi may be originated directly from marine or freshwater (Little 1990; Berbee et al. 2017). However, the transition from marine to freshwater is difficult, based on its limited frequency in phylogenies (del Campo and Ruiz-Trillo 2013; Berbee et al. 2017).
Marine Ascomycota have independently derived from terrestrial and freshwater ascomycetes (Spatafora et al. 1998). They clustered as sister clades to terrestrial or freshwater species and several genera of Ascomycota contain terrestrial and freshwater species together with marine species (Raghukumar 2017). Primary marine Ascomycota species are assumed to be derived from the marine environment, and secondary marine species are assumed to have a terrestrial ancestor (Spatafora et al. 1998; Raghukumar 2017). The phylogenetic analyses conducted by Vijaykrishna et al. (2006) showed that freshwater taxa have derived from terrestrial habitats several times independently. The freshwater taxa could be moved from terrestrial when the associated plants invaded water or ran off rainwater and sediments (Vijaykrishna et al. 2006). Evidently, some freshwater Ascomycota mainly occur on bamboo and in many countries, bamboo grows near river banks (Vijaykrishna et al. 2006). Freshwater Ascomycota shows various adaptations to aquatic environments, such as enhanced mechanisms for dispersal and subsequent attachment in freshwater (Vijaykrishna et al. 2006) and various environments (Ruisi et al. 2007). The freshwater Sordariomycetes reported in this study mainly occur on decayed wood in submerged lakes, rivers and streams. Vijaykrishna et al. (2006) also stated the potential of aquatic fungi to degrade submerged substrates which help the survival in water-logged conditions. Goh and Hyde (1996) proposed four artificial groups of hyphomycetes based on occurrences, namely 1) ingoldian fungi, 2) aero-aquatic hyphomycetes, 3) terrestrial-aquatic hyphomycetes and 4) submerged-aquatic hyphomycetes. Aero-aquatic hyphomycetes are mainly reported from ponds, ditches, or slow-running streams. They are characterized primarily by conspicuous conidiophores which occur in submerged leaves or woody substrates under semi-anaerobic conditions. Our new collections of Clonostachys rosea, Gliomastix masseei and Memnoniella echinate show visible larger conidiophores and are reported from submerged decaying wood near the lakes.
Despite being one of the largest classes, freshwater Sordariomycetes account for more than 60% of total freshwater ascomycetes (Shearer and Raja 2013). Samarakoon et al. (2016) and Hongsanan et al. (2017) studied several subclasses of unitunicate fungi based on divergence time estimate analysis. Hyde et al. (2021) recently reviewed the evolution of freshwater Diaporthomycetidae based on molecular clock analyses. In addition, evolutionary studies have been mostly conducted for Halosphaeriaceae and related orders such as Koralionastetales and Lulworthiales (Raghukumar 2017). In the study of Spatafora et al. (1998), marine Halosphaeriales (Microascales) species showed the independent evolution from terrestrial environment based on phylogenetic analyses. Further species of Halosphaeriaceae clustered as sister clades to terrestrial or freshwater species, and several genera of Ascomycota contain terrestrial and freshwater species together with marine species (Raghukumar 2017). As for accommodating freshwater, marine and terrestrial fungi, studying the evolution of Hypocreomycetidae are important for understanding the possible transition and evolution of aquatic and terrestrial ascomycetes.
In this study, we tried to address the possible evolutionary transitions of aquatic Hypocreomycetidae with broad taxon sampling. The result supports the transition of aquatic Hypocreomycetidae from terrestrial habitat to freshwater and marine habitats. Our finding concurred with the study of Vijaykrishna et al. (2006) who stated the evolution of freshwater ascomycetes form terrestrial habitats based on molecular phylogeny. Several molecular studies also stated the close association of several aquatic ascomycetes with terrestrial relatives (Liew et al. 2002). Shearer (1993) noted the evolution of aquatic hyphomycetes from terrestrial plant-associated or litter-associated fungi (Shearer 1993; Selosse et al. 2008), which was later supported in other ascomycetous fungi (Baschien et al. 2013). In the present study, the aquatic hyphomycetes, Clonostachys rosea, Gliomastix masseei and Memnoniella echinate were first reported from freshwater habitats. In contrast, previously they have been reported only in terrestrial habitats. However, we could not find a firm conclusion of the origin for these aquatic hyphomycetes. The early diverging clades of Hypocreomycetidae mostly comprise terrestrial fungi, while the marine and freshwater species show secondary independent evolution, represented by the order Glomerellales (Figs. 1, 2). The crown clade is represented by Nectriaceae (Hypocreales), which moderately comprises all three habitats with terrestrial ancestors. The following clades are represented by Coronophorales and Falcocladiales, which entirely include terrestrial fungi. The adjacent clade represents Torpedosporales which constitutes all three habitats, whereas a single species from the terrestrial habitat represents the next clade Xenodactylariales. Halosphaeriaceae (Microascales) clade mainly represents marine fungi, and only a few species show terrestrial and freshwater habitats that were secondarily derived. The representative families of Microascales, including Ceratocystidaceae, Cornuvesicaceae, Gondwanamycetaceae, Graphiaceae and Microascaceae, entirely accommodate terrestrial fungi, while Triadelphiaceae comprises a few freshwater fungi. The specific morphological adaptations observed among the representative species in various habitats were discussed under morphological adaptions of freshwater Hypocreomycetidae. However, no clear pattern in habitual transitions were observed within several lineages, and the absence of molecular data for several genera of the subclass renders the definitive conclusion. The details studies on fungal adaptation strategies, sexual and asexual connections, and their evolutionary changes can provide more information about their origin.
Divergence time estimates of Hypocreomycetidae
The divergence time estimates revealed that Hypocreomycetidae evolved during the early Permian (251–290 MYA) (Hyde et al. 2017, 2020a; Dayarathne et al. 2019). In this study, the estimated crown age of Hypocreomycetidae (290 MYA) is similar to previous studies and falls within the early Permian period (Hyde et al. 2017, 2020a; Dayarathne et al. 2019). However, previous studies used different methods to calibrate the evolution tree. Hyde et al. (2017) used fossil data (Paleoophiocordyceps coccophagus) and two secondary calibrations (the divergence time of Sordariomycetes and Leotiomycetes and crown age of Sordariomycetes). Dayarathne et al. (2019) used Paleoophiocordyceps coccophagus fossil data, and crown age of Sordariomycetes as secondary calibration and Hyde et al. (2020a) used two secondary calibrations (crown ages of Sordariomycetes and Dothideomycetes). In this study, we used Paleoophiocordyceps coccophagus fossil data and the crown age of Sordariomycetes as secondary calibration. The result showed that the stem and the crown age of Hypocreomycetidae are around 290 and 302 MYA, respectively. The crown ages of several orders in Hypocreomycetidae viz. Coronophorales (220 MYA), Glomerellales (218 MYA), Hypocreales (222 MYA), Microascales (249 MYA), and Torpedosporales (175 MYA) lie within the Jurassic period (251.9–201.3 MAY). The Jurassic period had a warm climate, which promoted the flourishing of gymnosperms and true ferns, and formed vast forests covering the world, which providing various hosts for fungi. In addition, during the mass extinction event, the death of animals or insect also provided nutrients for fungi to survive and diversify during this period.
Studies suggested that basal fungi originated from aquatic habitats and fungal territorialization occurred during the Cambrian, over 500 million years ago (MYA) (Taylor and Osborn 1996; Brundrett 2002; Wijayawardene et al. 2018). Ascomycota and Basidiomycota are divergent at a similar time with the fungal territorialization and evolved from terrestrial ancestors (Vijaykrishna et al. 2006; James et al. 2006; Liu et al. 2006; Lucking et al. 2009; Gueidan et al. 2011; Hyde et al. 2021). Based on the molecular clock and ancestral state analysis, freshwater taxa in Hypocreomycetidae have evolved from terrestrial ancestors (Fig. 2). They have divergent much later than the terrestrial taxa (Fig. 2). The Hypocreomycetidae may evolved from terrestrial to freshwater habitats in two pathways: 1) fungi may occur initially as pathogens, endophytes or saprobes on plants and they develop adaptation for freshwater when plants invaded freshwater habitats, 2) freshwater fungi may on the branches, stems or leaves in riparian vegetation and these substrates fallen into streams, the fungi colonized and adapted to the freshwater habitats (Shearer 1993; Hyde et al. 2021). Most freshwater Hypocreomycetidae have been reported on submerged wood or leaves, supporting Sherear (1993). The evolution of morphological characters in freshwater fungi along the molecular study can provide more information for evolutionary strategies.
Morphological adaptions of freshwater Hypocreomycetidae
Fungi show morphological adaptation when they transit from land to aquatic habitats. Sherer (1993) proposed several morphological adaptations of freshwater Ascomycota such as ascospores with appendages or sheaths, which help the ascospores attaching the substrate and remain connected as the water moves (Aniptodera, Ceriospora, Ceriosporopsis and Halosarpheia) (Shearer and Crane 1980; Shearer 1993). Filiform-like ascospores become sigmoid shape in the water which expands the area of the orthogonal projection, while the long filamentary shape enhances entanglement with the matrix (Webster and Davey 1984; Webster 1987). Few genera also developed deliquescent asci, which facilitate the dispersal of ascospores. Massive apical rings of asci also help the strong ejection of ascospores (Hyde and Goh 2003). The asexual freshwater species show branched, tetraradiate, sigmoid, helicosporous, and multicellular air trapping conidia which facilitate the dispersal and attachment of conidia to substrata shapes (Sherer 1993). However, it is hard to conclude that these characteristics have been developed as either pre-adaptation or convergent evolution since these characteristics are also found in terrestrial species (Vijaykrishna et al. 2006).
In our phylogenetic analysis, freshwater Hypocreomycetidae are mainly distributed in Campylosporaceae, Halosphaeriaceae, Nectriaceae and Reticulascaceae and therefore, our discussion is mainly focused to these families.
The asexual genus Campylospora (Campylosporaceae) entirely composed of freshwater species. The taxa are characterized by tetraradiate and hyaline conidia composed of two parts, deltoid and allantoid, and both show diverging branches at the ends. The presence of tetraradiate with diverging branches is a typical character of freshwater fungi, which helps the conidia to attach to the substrate and dispersal (Read et al. 1992; Vijaykrishna et al. 2006).
Halosphaeriaceae is a well-studied family with most marine species and few species from freshwater and terrestrial habitats (Pang 2002; Jones et al. 2009, 2015, 2017, 2019; Maharachchikumbura et al. 2015; Wijayawardene et al. 2017, 2018). Molecular data showed that Halosphaeriaceae has a marine ancestor (Fig. 2), which developed morphological adaptations to marine habitats. Based on morphological characters, Vijaykrishna et al. (2006) divided Halosphaeriaceae into two groups: (1) species with early deliquescing asci and appendaged ascospores and (2) species with persistent asci, often with an apical apparatus and mostly with ascospores with polar filamentous unfurling appendages. However, deliquescing asci may not be a good adaptation as deliquescing asci are rarely found in freshwater species (Hyde et al. 2000). The second group are common in freshwater and marine habitats. Persistent asci with an apical apparatus may have the ability to facilitate strong ejection of ascospores and ascospores with polar filamentous unfurling appendages, which is important for dispersal and subsequent attachment in freshwater habitats (Goh and Hyde 1996). Jones (2006) assumed the ancestral ascospore lacked appendages or spore wall ornamentation. Sakayaroj et al. (2011) suggested that the appendages were lost several times during the evolution based on the phylogenetic analysis. However, ascospores of few species lack appendages when they release from the ascus, yet when the ascospores mounted in seawater, which produce apical appendages, but not in freshwater (Savoryella appendiculata, Halosarpheia aquatica, Thalespora appendiculata) (Jones and Hyde 1992; Hyde 1992; Jones 2006). Our phylogenetic analysis showed that freshwater fungi clustered with marine taxa in four clades (Fig. 1). Most freshwater Halosphaeriaceae species have ascospores with appendages, and only a few species lack appendages (e.g. Nais inornata). Thus, we assume that the appendages of these species may be lost during their evolution, as proposed by Sakayaroj et al. (2011).
Nectriaceae is an ecologically diverse group with species that have been found as endophytes, saprobes and pathogens of plants, and some are entomogenous, a few species are human pathogens (Hyde et al. 2020a). Nectriaceae comprises the mostly freshwater taxa of Hypocreomycetidae (Luo et al. 2019). Several freshwater species have also been reported from terrestrial habitats (e.g., Aquanectria penicillioides, Chaetopsina penicillata, Gliomastix masseei, Neocosmospora brevis and Volutella ciliata). Only a few species (e.g. Atractium aquatica and Mariannaea aquaticola) are restricted to freshwater habitats (Luo et al. 2019; Calabon et al. 2022). However, there are no special morphological adaptations have been reported in the freshwater Nectriaceae species except for a few which show apical rings (e.g., A. aquatica, Atractium fusiformis and Thelonectria aquatica), that help the ascospores to release into the water (Vijaykrishna et al. 2006).
Reticulascaceae comprises four genera, two genera (Cylindrotrichum; Cylindrotrichum aquaticum, C. gorii, C. submersum and Kylindria; K. aquatica and K. chinensis) include freshwater species but also encompass terrestrial species (Luo et al. 2019). Shearer (1993) mentioned that freshwater fungi adapt to aquatic habitats by their asexual morphs (e.g. hyphomycetes). Cylindrotrichum and Kylindria were reported in their asexual morphs (Maharachchikumbura et al. 2018; Luo et al. 2019). No definite morphological adaptations were observed among the species except Kylindria aquatica, which has conidia with a slimy mucilaginous coating around it (Maharachchikumbura et al. 2018). This indicates the independent evolution of morphological adaption from freshwater to terrestrial habitats.
Diversity of freshwater Hypocreomycetidae
Fungi are ubiquitous and have been found in all habitats. The number of fungi has been estimated between 2.2 and 3.8 million; however, only about 150,600 species of fungi and fungus-like taxa have been so far described (Hyde et al. 2020c; Phukhamsakda et al. 2022; Wijayawardene et al. 2020). Freshwater fungi as a unique ecological group of highly diverse fungi; since Shearer (1993) reported 288 ascomycetes species found in watery settings, and this number has now expanded to 3077 (Calabon et al. 2022). However, many species are yet to be discovered (Bao et al. 2021; Calabon et al. 2022).
Hypocreomycetidae are an ecologically diverse group with species found in various environments. Hu et al. (2013) reported 57 Hypocreomycetidae species from freshwater habitats in China, and Luo et al. (2019) documented 76 species. Bao et al. (2021) provided a checklist of freshwater fungi in Yunnan Province during 2015–2020 and listed 21 freshwater Hypocreomycetidae species, and most species were collected from submerged decaying wood. In the latest study of Calabon et al. (2022), 156 Hypocreomycetidae species have been reported from freshwater habitats, with most documented from Lotic habitats (river and stream) (Calabon et al. 2022). In this study, 26 Hypocreomycetidae species have been reported from lentic freshwater habitats (lakes). This indicates that freshwater Hypocreomycetidae is highly diverse not only in lotic habitats but also in lentic habitats.
Update of Hypocreomycetidae based on the current study
In this section, we provide an updated classification of Hypocreomycetidae based on current study and recent publications (Hyde et al. 2020a; Huang et al. 2021; Wijayawardene et al. 2022; Xiao et al. 2023; Perera et al. 2023). Currently, Hypocreomycetidae comprises seven orders and 45 families. Herein, an updated outline of Hypocreomycetidae is provided.
Phylum ASCOMYCOTA Caval-Sm.
Subphylum PEZIZOMYCOTINA O.E. Erikss. & Winka.
Class Sordariomycetes O.E. Erikss. & Winka.
Subclass Hypocreomycetidae O.E. Erikss. & Winka
Coronophorales Nannf.
Bertiaceae Smyk.
Bertia De Not.
Gaillardiella Pat.
Ceratostomataceae G. Winter.
Arxiomyces P.F. Cannon & D. Hawksw.
Dactylidispora Y. Marín et al.
Echinusitheca Y. Marín et al.
Erythrocarpon Zukal.
Gonatobotrys Corda.
Harzia Costantin.
Melanospora Corda.
Microthecium Corda.
Neotrotteria Sacc.
Pseudomicrothecium Y. Marín et al.
Pustulipora P.F. Cannon.
Rhytidospora Jeng & Cain.
Scopinella Lév.
Setiferotheca Matsush.
Syspastospora P.F. Cannon & D. Hawksw.
Vittatispora P. Chaudhary et al.
Chaetosphaerellaceae Huhndorf et al.
Chaetosphaerella E. Müll. & C. Booth.
Crassochaeta Réblová.
Neochaetosphaerella Lar.N. Vassiljeva et al.
Spinulosphaeria Sivan.
Coronophoraceae Höhn.
Coronophora Fuckel.
Nitschkiaceae (Fitzp.) Nannf.
Acanthonitschkea Speg.
Biciliosporina Subram. & Sekar.
Botryola Bat. & J.L. Bezerra.
Fracchiaea Sacc.
Groenhiella Jørg. Koch et al.
Janannfeldtia Subram. & Sekar.
Lasiosphaeriopsis D. Hawksw. & Sivan.
Loranitschkia Lar.N. Vassiljeva.
Nitschkia G.H. Otth ex P. Karst.
Rhagadostoma Körb.
Rhagadostomella Etayo.
Tortulomyces Lar.N. Vassiljeva et al.
Parasympodiellaceae Hern.-Restr. et al.
Parasympodiella Ponnappa.
Scortechiniaceae Huhndorf et al.
Biciliospora Petr.
Coronophorella Höhn.
Euacanthe Theiss.
Neocryptosphaerella S.K. Huang & K.D. Hyde.
Neofracchiaea Teng.
Pseudocatenomycopsis Crous & L.A. Shuttlew.
Pseudocryptosphaerella S.K. Huang & K.D. Hyde.
Scortechinia Sacc.
Scortechiniella Arx & E. Müll.
Scortechiniellopsis Sivan.
Tympanopsis Starbäck.
Coronophorales genera incertae sedis.
Papulaspora Preuss.
Sphaerodes Clem.
Tengiomyces Réblová.
Falcocladiales R.H. Perera et al.
Falcocladiaceae Somrithipol et al.
Falcocladium S.F. Silveira et al.
Glomerellales Chadef. ex Réblová et al.
Ascocodinaceae D.F. Bao et al.
Ascocodina C.D. Viljoen et al.
Australiascaceae Réblová & W. Gams.
Monilochaetes Halst. ex Harter.
Glomerellaceae Locq. ex Seifert & W. Gams.
Colletotrichum Corda.
Malaysiascaceae Tibpromma & K.D. Hyde.
Malaysiasca Crous & M.J. Wingf.
Plectosphaerellaceae W. Gams et al.
Acremoniisimulans Tibpromma & K.D. Hyde.
Acrostalagmus Corda.
Brunneochlamydosporium Giraldo López & Crous.
Brunneomyces A. Giraldo, Gené & Guarro.
Chlamydosporiella Giraldo López & Crous.
Chordomyces Bilanenko et al.
Furcasterigmium Giraldo López & Crous.
Fuscohypha Giraldo López & Crous.
Gibellulopsis Bat. & H. Maia.
Lectera P.F. Cannon.
Longitudinalis Tibpromma & K.D. Hyde.
Musicillium Zare & W Gams.
Musidium Giraldo López & Crous.
Nigrocephalum Giraldo López & Crous.
Paragibellulopsis Giraldo López & Crous.
Paramusicillium Giraldo López & Crous.
Phialoparvum Giraldo López & Crous.
Plectosphaerella Kleb.
Sayamraella Giraldo López & Crous.
Sodiomyces A.A. Grum-Grzhim. et al.
Stachylidium Link.
Summerbellia Giraldo López & Crous.
Theobromium Giraldo López & Crous.
Verticillium Nees.
Xenoplectosphaerella Jayaward., Phukhams. & K.D. Hyde.
Reticulascaceae Réblová & W. Gams.
Blastophorum Matsush.
Cylindrotrichum Bonord.
Kylindria DiCosmo et al.
Sporoschismopsis Hol-Jech. & Hennebert.
Hypocreales Lindau.
Bionectriaceae Samuels & Rossman.
Acremonium Link.
Anthonectria Döbbeler.
Aphanotria Döbbeler.
Battarrina (Sacc.) Clem. & Shear.
Bryocentria Döbbeler.
Bryotria Döbbeler & P.G. Davison.
Chrysonectria Lechat & J. Fourn.
Clibanites (P. Karst.) P. Karst.
Clonostachys Corda.
Dimerosporiella Speg.
Fusariella Sacc.
Geonectria Lechat & J. Fourn.
Geosmithia Pitt.
Gliomastix Guég.
Gracilistilbella Seifert.
Halonectria E.B.G. Jones.
Heleococcum P.M. Jørg.
Hydropisphaera Dumort.
Laniatria Döbbeler & P.G. Davison.
Lasionectria (Sacc.) Cooke.
Lasionectriella Lechat & J. Fourn.
Mycoarachis Malloch & Cain.
Mycocitrus Möller.
Nectriella Nitschke ex Fuckel.
Nectriopsis Maire.
Nigrosabulum Malloch & Cain.
Ochronectria Rossman & Samuels.
Ovicillium Zare & W. Gams.
Ovicuculispora Etayo.
Paracylindrocarpon Crous et al.
Paranectria Sacc.
Periantria Döbbeler & P.G. Davison.
Peristomialis (W. Phillips) Boud.
Periantria Döbbeler & P.G. Davison.
Pronectria Clem.
Protocreopsis Yoshim Doi.
Pseudoacremonium Crous.
Roumegueriella Speg.
Selinia P. Karst.
Septofusidium W. Gams.
Stephanonectria Schroers & Samuels.
Stilbocrea Pat.
Synnemellisia N.K. Rao et al.
Verrucostoma Hirooka et al.
Calcarisporiaceae Jing Z. Sun et al.
Calcarisporium Preuss.
Verticimonosporium Matsush.
Clavicipitaceae (Lindau) Earle ex Rogerson.
Aciculosporium I. Miyake.
Aschersonia Mont.
Atkinsonella Diehl.
Balansia Speg.
Cavimalum Yoshim. Doi et al.
Claviceps Tul.
Collarina Giraldo et al.
Commelinaceomyces E. Tanaka.
Conoideocrella D. Johnson et al.
Corallocytostroma Y.N. Yu & Z.Y. Zhang.
Dussiella Pat.
Ephelis Fr.
Epichloë (Fr.) Tul. & C. Tul.
Epicrea Petr.
Helicocollum Luangsa-ard.
Helminthascus Tranzschel.
Heteroepichloë E. Tanaka et al.
Keithomyces Samson, Luangsa-ard & Houbraken.
Konradia Racib.
Loculistroma F. Patt & Charles.
Marquandomyces Samson, Houbraken & Luangsa-ard.
Metapochonia Kepler et al.
Metarhiziopsis D.W. Li et al.
Metarhizium Sorokīn.
Moelleriella Bres.
Mycomalus A. Möller.
Mycophilomyces Crous & M.J. Wingf.
Myriogenospora G.F. Atk.
Neobarya Lowen.
Neocordyceps Kobayasi.
Nigelia Luangsa-ard.
Nigrocornus Ryley & Langdon.
Orbiocrella D. Johnson et al.
Papiliomyces Luangsa-ard, Samson & Thanakitp.
Parepichloë J.F. White & P.V. Reddy.
Periglandula U. Steiner et al.
Petchia Thanakitp., Mongkols. & Luangsa-ard.
Pochonia Bat. & O.M. Fonseca.
Pseudomeria G.L. Barron.
Purpureomyces Luangsa-ard, Samson & Thanakitp.
Regiocrella Chaverri & K.T. Hodge.
Romanoa Thirum.
Rotiferophthora G.L. Barron.
Samuelsia Chaverri & K.T. Hodge.
Shimizuomyces Kobayasi.
Sphaerocordyceps Kobayasi.
Sungia Luangsa-ard, Samson & Thanakitp.
Tyrannicordyceps Kepler & Spatafora.
Ustilaginoidea Bref.
Yosiokobayasia Samson, Luangsa-ard & Thanakitp.
Cocoonihabitaceae W.Y. Zhuang & Z.Q. Zeng.
Cocoonihabitus W.Y. Zhuang & Z.Q. Zeng.
Cordycipitaceae Kreisel ex G.M. Sung et al.
Akanthomyces Lebert.
Amphichorda Fr.
Ascopolyporus Möller.
Beauveria Vuill.
Beejasamuha Subram. & Chandrash.
Blackwellomyces Spatafora & Luangsa-ard.
Cordyceps (Fr.) Link.
Coremiopsis Sizova & Suprun.
Engyodontium de Hoog.
Flavocillium H. Yu et al.
Gamszarea Z.F. Zhang & L. Cai.
Gibellula Cavara.
Hevansia Luangsa-ard et al.
Hyperdermium J. White et al.
Leptobacillium Zare & W. Gams.
Liangia H. Yu et al.
Neotorrubiella Tasan., Thanakitp. & Luangsa-ard.
Parengyodontium C.C. Tsang.
Pseudogibellula Samson & H.C. Evans.
Samsoniella Mongkols. et al.
Simplicillium W. Gams & Zare.
Flammocladiellaceae Crous et al.
Flammocladiella Crous et al.
Hypocreaceae De Not.
Arachnocrea Z. Moravec.
Dialhypocrea Speg.
Escovopsioides H.C. Evans & J.O. Augustin.
Escovopsis J.J. Muchovej & Della Lucia.
Hypocreopsis P. Karst.
Hypomyces (Fr.) Tul. & C. Tul.
= Cladobotryum Nees.
= Sibirina G.R.W. Arnold.
Kiflimonium Summerb., J.A. Scott, Guarro & Crous.
Lichenobarya Etayo et al.
Mycogone Link.
Protocrea Petch.
Rogersonia Samuels & Lodge.
Sepedonium Link.
Sphaerostilbella (Henn.) Sacc. & D. Sacc.
= Gliocladium Corda.
Sporophagomyces K. Põldmaa & Samuels.
Stephanoma Wallr.
Trichoderma Pers.
= Aphysiostroma Barrasa et al.
= Hypocrea Fr.
= Podocrea (Sacc.) Lindau.
= Podostroma P. Karst.
= Pseudohypocrea Yoshim. Doi.
= Sarawakus Lloyd.
Ijuhyaceae R.H. Perera, E.B.G. Jones, Maharachch., & K.D. Hyde.
Ijuhya Starbäck
= Peristomialis (W. Phillips) Boud.
Kallichroma Kohlm. & Volkm.-Kohlm.
Myrotheciomycetaceae Crous.
Leucosphaerina Arx.
Myrotheciomyces Crous.
Trichothecium Link.
Nectriaceae Tul. & C. Tul.
Albonectria Rossman & Samuels.
Allonectella Petr.
Aphanocladium W. Gams.
Aquanectria L. Lombard & Crous.
Atractium Link.
= Varicosporella Lechat & J. Fourn.
Baipadisphaeria Pinruan.
Bisifusarium L. Lombard et al.
Calonectria De Not.
Calostilbe Sacc. & Syd.
Campylocarpon Halleen et al.
Chaetonectrioides Matsush.
Chaetopsina Rambelli.
Chrysonectria Lechat & J. Fourn.
Cinnamomeonectria Salgado & P. Chaverri.
Coccinonectria Lombard & Crous.
Corallomycetella Henn.
Corallonectria C. Herrera & P. Chaverri.
Corinectria C. González & P. Chaverri.
Cosmospora Rabenh.
Cosmosporella S.K. Huang et al.
Curvicladiella Decock & Crous.
Cyanochyta Höhn.
Cyanonectria Samuels & Chaverri.
Cyanophomella Höhn.
Cylindrocladiella Boesew.
Cylindrodendrum Bonord.
Dacryoma Samuels.
Dactylonectria L. Lombard & Crous.
Dematiocladium Allegr. et al.
Fusarium Link.
Fusicolla Bonord.
Geejayessia Schroers et al.
Gliocephalotrichum J.J. Ellis & Hesselt.
Gliocladiopsis S.B. Saksena.
Globonectria Etayo.
Ilyonectria P. Chaverri & C. Salgado.
Longinectria O. Savary, M. Coton, E. Coton & J.L. Jany.
Luteonectria Sand.-Den., L. Lombard, Schroers & Rossman.
Macroconia (Wollenw.) Gräfenhan et al.
Macronectria Salgado & P. Chaverri.
Mariannaea G. Arnaud ex Samson.
Microcera Desm.
Nalanthamala Subram.
Nectria (Fr.) Fr.
Neocalonectria Crous.
Neocosmospora E.F. Sm.
Neonectria Wollenw.
Nothofusarium Crous, Sand.-Den. & L. Lombard.
Ophionectria Sacc.
Pandanaceomyces Tibpromma & K.D. Hyde.
Paracremonium L. Lombard & Crous.
Payosphaeria W.F. Leong.
Penicillifer Emden.
Persiciospora P.F. Cannon & D. Hawksw.
Pleiocarpon L. Lombard & D. Aiello.
Pleogibberella Sacc.
Pleurocolla Petr.
Pseudoachroiostachys Tibpromma & K.D. Hyde.
Pseudocosmospora C. Herrera & P. Chaverri.
Pseudonectria Seaver.
Rectifusarium Lombard et al.
Rugonectria P. Chaverri & Samuels.
Sarcopodium Ehrenb.
Scolecofusarium L. Lombard, Sand.-Den. & Crous.
Sedecimiella K.L. Pang et al.
Setofusarium (Nirenberg & Samuels) Crous & Sand.-Den.
Stylonectria Höhn.
Thelonectria P. Chaverri & C.G. Salgado.
= Neothyronectria Crous & Thangavel.
Thyronectria Sacc.
= Allantonectria Earle.
= Neothyronectria Crous & Thangavel.
= Pleonectria Sacc.
= Sulcatistroma A.W. Ramaley.
= Thyronectroidea.
Tumenectria Salgado & Rossman.
Varicosporellopsis Lechat & J. Fourn.
Vesicladiella Crous & M.J. Wingf.
Volutella Fr.
Xenoacremonium Lombard & Crous.
Xenocylindrocladium Decock et al.
Xenogliocladiopsis Crous & W.B. Kendr.
Xenoleptographium Marinc. et al.
Xenonectriella Weese.
Niessliaceae Kirschst.
Atronectria Etayo.
Circinoniesslia Samuels & M.E. Barr.
Cryptoniesslia Scheuer.
Eucasphaeria Crous.
Malmeomyces Starb.
Melchioria Penz. & Sacc.
Miyakeomyces Hara.
Myrmaeciella Lindau.
Myrtacremonium Crous.
Neoeucasphaeria Crous.
Niesslia Auersw.
= Hyaloseta A.W. Ramaley.
Nitschkiopsis Nannf. & R. Sant.
Paraniesslia K.M. Tsui et al.
Pseudohyaloseta Tibpromma & K.D. Hyde.
Pseudonectriella Petr.
Pseudorhynchia Höhn.
Rosasphaeria Jaklitsch & Voglmayr.
Taiwanascus Sivan & H.S. Chang.
Trichosphaerella E. Bommer et al.
Valetoniella Höhn.
Valetoniellopsis Samuels & M.E. Barr.
Polycephalomycetaceae Y.P. Xiao, Y.B Wang, T.C. Wen, H. Yu & K.D. Hyde.
Perennicordyceps Matočec & I. Kušan.
Pleurocordyceps Y.J. Yao et al.
Polycephalomyces Kobayasi.
Ophiocordycipitaceae G.H. Sung et al.
Drechmeria W. Gams & H.B. Jansson.
Hantamomyces Crous.
Harposporium Lohde.
Hirsutella Pat.
Hymenostilbe Petch.
Ophiocordyceps Petch.
Paraisaria Samson & B.L. Brady.
Purpureocillium Luangsa-ard et al.
Tolypocladium W. Gams.
Sarocladiaceae L. Lombard.
Parasarocladium Summerb. et al.
Sarocladium W. Gams & D. Hawksw.
Stachybotryaceae L. Lombard & Crous.
Achroiostachys L. Lombard & Crous.
Albifimbria L. Lombard & Crous.
Albosynnema E.F. Morris.
Alfaria Crous et al.
Alfariacladiella Crous & R.K. Schumach.
Brevistachys L. Lombard & Crous.
Capitofimbria L. Lombard & Crous.
Cymostachys L. Lombard & Crous.
Didymostilbe Henn.
Digitiseta Gordillo & Decock.
Dimorphiseta L. Lombard & Crous.
Globobotrys L. Lombard & Crous.
Grandibotrys L. Lombard & Crous.
Gregatothecium L. Lombard & Crous.
Hyalinostachys C.G. Lin & K.D. Hyde.
Inaequalispora L. Lombard & Crous.
Kastanostachys L. Lombard & Crous.
Koorchalomella Chona et al.
Melanopsamma Niessl.
Memnoniella Höhn.
Myrothecium Tode.
Myxospora L. Lombard & Crous.
Neomyrothecium L. Lombard & Crous.
Paramyrothecium L. Lombard & Crous.
Parasarcopodium Mel’nik et al.
Parvothecium L. Lombard & Crous.
Peethambara Subram. & Bhat.
Pseudoornatispora Tibpromma & K.D. Hyde.
Septomyrothecium Matsush.
Sirastachys L. Lombard & Crous.
Smaragdiniseta L. Lombard & Crous.
Stachybotrys Corda.
Striatibotrys L. Lombard & Crous.
Striaticonidium L. Lombard & Crous.
Tangerinosporium L. Lombard & Crous.
Virgatospora Finley.
Xenomyrothecium L. Lombard & Crous.
Xepicula Nag Raj.
Xepiculopsis Nag Raj.
Stromatonectriaceae R.H. Perera, E.B.G. Jones, Maharachch., & K.D. Hyde.
Stromatonectria Jaklitsch & Voglmayr.
Tilachlidiaceae Lombard & Crous.
Psychronectria J. Pawłowska et al.
Tilachlidium Preuss.
Xanthonectriaceae R.H. Perera, E.B.G. Jones, Maharachch., & K.D. Hyde.
Bullanockia Crous.
Xanthonectria J. Fourn. & P.-A. Moreau.
Hypocreales genera incertae sedis.
Acremoniopsis Giraldo et al.
Berkelella (Sacc.) Sacc.
Bryonectria Döbbeler.
Bulbithecium Udagawa & T Muroi.
Cephalosporiopsis Peyronel.
Chondronectria Etayo et al.
Cylindronectria Etayo.
Diploöspora Grove.
Emericellopsis J.F.H. Beym.
Gynonectria Döbbeler.
Hapsidospora Malloch & Cain.
Haptospora G.L. Barron.
Illosporiopsis D. Hawksw.
Illosporium Mart.
Leptobarya Etayo.
Lichenopenicillus Etayo.
Metadothella Henn.
Munkia Speg.
Neomunkia Petr.
Peloronectria Möller.
Pseudoidriella Crous & R.G. Shivas.
Pseudomeliola Speg.
Rodentomyces Doveri et al.
Roselliniella Vain.
Saksenamyces A.N. Rai & P.N. Singh.
Stanjemonium W. Gams et al.
Stilbella Lindau.
Ticonectria Döbbeler.
Trichonectria Kirschst.
Tilakidium Vaidya et al.
Microascales Luttr. ex Benny & Kimbr.
Ceratocystidaceae Locq. ex Réblová et al.
Ambrosiella Brader ex Arx & Hennebert.
Berkeleyomyces W.J. Nel et al.
Bretziella Z.W. de Beer et al.
Ceratocystis Ellis & Halst.
Chalaropsis Peyronel.
Davidsoniella Z.W. de Beer et al.
Endoconidiophora Münch.
Huntiella Z.W. de Beer et al.
Meredithiella McNew et al.
Phialophoropsis L.R. Batra emend. T.C. Harr.
Thielaviopsis Went.
Cornuvesicaceae D.F. Bao et al.
Cornuvesica C.D. Viljoen et al.
Chadefaudiellaceae Faurel & Schotter ex Benny & Kimbr.
Chadefaudiella Faurel & Schotter.
Gondwanamycetaceae Réblová et al.
Custingophora Stolk.
Knoxdaviesia M.J. Wingf et al.
Graphiaceae De Beer.
Graphium Corda.
Halosphaeriaceae E. Müll & Arx ex Kohlm.
Alisea J. Dupont & E.B.G. Jones.
Amphitrite S. Tibell.
Aniptodera Shearer & M. Miller.
Aniptosporopsis (K.D. Hyde) K.L. Pang.
Anisostagma K.R.L. Petersen & Jørg. Koch.
Antennospora Meyers.
Appendichordella R.G. Johnson et al.
Arenariomyces Höhnk.
Ascosacculus J. Campbell, J.L. Anderson & Shearer.
Bathyascus Kohlm.
Carbosphaerella I. Schmidt.
Ceriosporopsis Linder.
Chadefaudia Feldm.-Maz.
Corallicola Volkm.-Kohlm. & Kohlm.
Corollospora Werderm.
Cucullosporella K.D. Hyde & E.B.G. Jones.
Ebullia K.L. Pang.
Gesasha Abdel-Wahab & Nagah.
Haiyanga K.L. Pang & E.B.G. Jones.
Haligena Kohlm.
Halosarpheia Kohlm. & E. Kohlm.
Halosphaeria Linder.
Halosphaeriopsis T.W. Johnson.
Havispora K.L. Pang & Vrijmoed.
Iwilsoniella E.B.G. Jones.
Kitesporella Jheng & K.L. Pang.
Kochiella Sakay. et al.
Lautisporopsis E.B.G. Jones et al.
Lignincola Höhnk.
Limacospora Jørg. Koch & E.B.G. Jones.
Luttrellia Shearer.
Magnisphaera J. Campb. et al.
Marinospora A.R. Caval.
Moana Kohlm. & Volkm.-Kohlm.
Morakotiella Sakay.
Naïs Kohlm.
Natantispora J. Campb. et al.
Neptunella K.L. Pang & E.B.G. Jones.
Nereiospora E.B.G. Jones et al.
Nimbospora Jørg. Koch.
Nohea Kohlm. & Volkm.-Kohlm.
Oceanitis Kohlm.
Ocostaspora E.B.G. Jones et al.
Okeanomyces K.L. Pang & E.B.G. Jones.
Ondiniella E.B.G. Jones et al.
Ophiodeira Kohlm. & Volkm.-Kohlm.
Panorbis J. Campb. et al.
Paraaniptodera K.L. Pang et al.
Phaeonectriella R.A. Eaton & E.B.G. Jones.
Pileomyces K.L. Pang & Jheng.
Praelongicaulis Jones et al.
Pseudolignincola Chatmala & E.B.G. Jones.
Remispora Linder.
Saagaromyces K.L. Pang & E.B.G. Jones.
Sablicola E B.G. Jones et al.
Thalassogena Kohlm. & Volkm.-Kohlm.
Thalespora Chatmala & E.B.G. Jones.
Tinhaudeus K.L. Pang et al.
Tirispora E.B.G. Jones & Vrijmoed.
Toriella Sakay. et al.
Trailia G.K. Sutherl.
Trichomaris Hibbits et al.
Tunicatispora K.D. Hyde.
Microascaceae Luttr. ex Malloch.
Acaulium Sopp.
Brachyconidiellopsis Decock et al.
Canariomyces Arx.
Cephalotrichum Link.
Doratomyces Corda.
Echinobotryum Corda.
Enterocarpus Locq.-Lin.
Fairmania Sacc.
Gamsia M. Morelet.
Kernia Nieuwl.
Lomentospora Hennebert & B.G. Desai.
Lophotrichus R.K. Benj.
Microascus Zukal.
Parascedosporium Gilgado et al.
Petriella Curzi.
Pseudallescheria Negroni & I. Fisch.
Pseudoscopulariopsis M. Sandoval-Denis et al.
Rhinocladium Sacc. & Marchal.
Scedosporium Sacc. ex Castell. & Chalm.
Scopulariopsis Bainier.
Wardomyces F.T. Brooks & Hansf.
Wardomycopsis Udagawa & Furuya.
Yunnania H.Z. Kong.
Triadelphiaceae Y.Z. Lu et al.
Synnematotriadelphia Chuaseehar et al.
Triadelphia Shearer & J.L. Crane.
Microascales genera incertae sedis.
Bisporostilbella Brandsb. & E.F. Morris.
Cephalotrichiella Crous.
Cornuvesica C.D. Viljoen et al.
Gabarnaudia Samson & W. Gams.
Nautosphaeria E.B.G. Jones.
Tubakiella Sakayaroj, K.L. Pang & E.B.G. Jones.
Sporendocladia G. Arnaud ex Nag Raj & W.B. Kendr.
Torpedosporales E.B.G. Jones et al.
Etheirophoraceae Rungjindamai et al.
Etheirophora Kohlm. & Volkm.-Kohlm.
Swampomyces Kohlm. & Volkm.
Juncigenaceae E.B.G. Jones et al.
Elbamycella A. Poli.
Fulvocentrum E.B.G. Jones & Abdel-Wahab.
Juncigena Kohlm Kohlm. et al.
Khaleijomyces Abdel-Wahab.
Marinokulati E.B.G. Jones & K.L. Pang.
Torpedosporaceae E.B.G. Jones & K.L. Pang.
Torpedospora Meyer.
Xenodactylariales D.F. Bao et al.
Xenodactylariaceae Crous.
Xenodactylaria Crous.
Hypocreomycetidae families incertae sedis.
Campylosporaceae D.F. Bao et al.
Campylospora Ranzoni.
Hypocreomycetidae genera incertae sedis.
Dendroclathra Voglmayr & G. Delgado.
References
Agnihothrudu V (1959) Notes on fungi from north-east India. Trans Brit Mycol Soc 42:458–462
Aimer RD, Segedin BP (1985) Some aquatic hyphomycetes from New Zealand streams. N Z J Bot 23:273–299
Al-Bedak OA, Ismail MA, Mohamed RA (2019) Paracremonium moubasheri, a new species from an alkaline sediment of Lake Hamra in Wadi-El-Natron, Egypt with a key to the accepted species. Stud Mycol 4(1):216–222
Andersen B, Nielsen KF, Thrane U, Szaro T, Taylor JW, Jarvis BB (2003) Molecular and phenotypic descriptions of Stachybotrys chlorohalonata sp. nov. and two chemotypes of Stachybotrys chartarum found in water-damaged buildings. Mycologia 95(6):1227–1238
Arambarri AM, Spinedi HÁ (1984) Micoflora de lahojarasca de Nothofagus pumilio. III Darwiniana 25:321–330
Backus MP, Orpurt PA (1961) A new Emericellopsis from Wisconsin, with notes on other species. Mycologia 53(1):64–83
Bainier G (1907) Gliocladium roseum sp. nov. et Cephalosporium acremonium (Corda). Bull Soc Mycol France 23:111–114
Bao DF, Hyde KD, McKenzie EHC, Jeewon R, Su HY, Nalumpang S, Luo ZL (2021) Biodiversity of lignicolous freshwater hyphomycetes from China and Thailand and description of sixteen species. J Fungi 7:669
Barron GL (1968) The genera of hyphomycetes from soil. Williams & Wilkins, Baltimore
Baschien C, Tsui CKM, Gulis V, Szewzyk U, Marvanová L (2013) The molecular phylogeny of aquatic hyphomycetes with affinity to the Leotiomycetes. Fungal Biol 117(9):660–672
Beakes GW, Sekimoto S (2009) The evolutionary phylogeny of oomycetes—insights gained from studies of holocarpic parasites of algae and invertebrates. Wiley, New York, pp 1–24
Beakes GW, Glockling SL, Sekimoto S (2012) The evolutionary phylogeny of the oomycete “fungi.” Protoplasma 249:3–19
Beimforde C, Feldberg K, Nylinder S, Rikkinen J, Tuovila H, Dörfelt H, Gube M, Jackson DJ, Reitner J, Seyfullah LJ, Schmidt AR (2014) Estimating the Phanerozoic history of the Ascomycota lineages:combining fossil and molecular data. Mol Phylogenet Evol 78:386–398
Belliveau MJR, Bärlocher F (2005) Molecular evidence confirms multiple origins of aquatic hyphomycetes. Mycol Res 109(12):1407–1417
Berbee ML, James TY, Strullu-Derrien C (2017) Early diverging fungi: diversity and impact at the dawn of terrestrial life. Annu Rev Microbiol 71:41–60
Bidartondo MI, Read DJ, Trappe JM, Merckx V, Ligrone R, Duckett JG (2011) The dawn of symbiosis between plants and fungi. Biol Lett 7:574–577
Boekhout T, Aime MC, Begerow D, Gabaldón T, Heitman J, Kemler M, Khayhan K, Lachance MA, Louis EJ, Sun S, Vu D, Yurkov A (2021) The evolving species concepts used for yeasts: from phenotypes and genomes to speciation networks. Fungal Divers 109:27–55
Boonmee S, Wanasinghe DN, Calabon MS, Huanraluek N, Chandrasiri SKU, Jones EBG, Rossi W, Leonardi M, Singh SK, Rana S, Singh PN, Maurya DK, Lagashetti AC, Choudhary D, Dai YC, Zhao CL, Mu YH, Yuan HS, He SH, Phookamsak R, Jiang HB, Martín MP, Dueñas M, Telleria MT, Kałucka IL, Jagodziński AM, Liimatainen K, Pereira DS, Phillips AJL, Suwannarach N, Kumla J, Khuna S, Lumyong S, Potter TB, Shivas RG, Sparks AH, Vaghefi N, Abdel-Wahab MA, AbdelAziz FA, Li GJ, Lin WF, Singh U, Bhatt RP, Lee HB, Nguyen TTT, Kirk PM, Dutta AK, Acharya K, Sarma VV, Niranjan M, Rajeshkumar KC, Ashtekar N, Lad S, Wijayawardene NN, Bhat DJ, Xu RJ, Wijesinghe SN, Shen HW, Luo ZL, Zhang JY, Sysouphanthong P, Thongklang N, Bao DF, Aluthmuhandiram JVS, Abdollahzadeh J, Javadi A, Dovana F, Usman M, Khalid AN, Dissanayake AJ, Telagathoti A, Probst M, Peintner U, Garrido-Benavent I, Bóna L, Merényi Z, Boros L, Zoltán B, Stielow JB, Jiang N, Tian CM, Shams E, Dehghanizadeh F, Pordel A, Javan-Nikkhah M, Denchev TT, Denchev CM, Kemler M, Begerow D, Deng C-Y, Harrower E, Bozorov T, Kholmuradova T, Gafforov Y, Abdurazakov A, Xu JC, Mortimer PE, Ren GC, Jeewon R, Maharachchikumbura SSN, Phukhamsakda C, Mapook A, Hyde KD (2021) Fungal diversity notes 1387–1511: taxonomic and phylogenetic contributions on genera and species of fungal taxa. Fungal Divers 111:1–335
Boonyuen N, Chuaseeharonnachai C, Suetrng S, Sri-Indrasutdhi V, Sivichai S, Jones EBG, Pang KL (2011) Savoryellales (Hypocreomycetideae, Sordariomycetes): a novel lineage of aquatic ascomycetes inferred from multiple-gene phylogenies of the genera Ascotaiwania, Ascothailandia and Savoryella. Mycologia 103:1351–1371
Booth C (1959) Studies of Pyrenomycetes. IV. Nectria (part 1). Mycol Pap 73:1–115
Booth C (1966) The Genus Cylindrocarpon Mycol Pap 104:1–56
Brayford D, Samuels GJ (1993) Some didymosporous species of Nectria with nonmicroconid-ial Cylindrocarpon anamorphs. Mycologia 85:612–637
Brayford D, Honda BM, Mantiri FR, Samuels GJ (2004) Neonectria and Cylindrocarpon: the Nectria mammoidea group and species lacking macroconidia. Mycologia 96:572–597
Brundrett MC (2002) Coevolution of roots and mycorrhizas of land plants. New Phytol 154:275–304
Cai L, Zhang KQ, McKenzie EHC, Hyde KD (2003) Freshwater fungi from bamboo and wood submerged in the Liput River in the Philippines. Fungal Divers 13:1–12
Cai L, Kurniawati E, Hyde KD (2010) Morphological and molecular characterization of Mariannaea aquaticola sp. nov. collected from freshwater habitats. Mycol Prog 9:337–343
Calabon MS, Jones EBG, Boonmee S, Doilom M, Lumyong S, Hyde KD (2021) Five novel freshwater ascomycetes indicate high undiscovered diversity in lotic habitats in Thailand. J Fungi 7(2):117
Calabon MS, Hyde KD, Jones EBG, Luo ZL, Dong W, Hurdeal VG, Gentekaki E, Rossi W, Leonardi M, Thiyagaraja V, Lestari AS, Shen HW, Bao DF, Boonyuen N, Zeng M (2022) Freshwater fungal numbers. Fungal Divers 114:3–235
Campbell J, Anderson JL, Shearer CA (2003) Systematics of Halosarpheia based on morphological and molecular data. Mycologia 95:530–552
Cannon PF, Kirk PM (2007) Fungal families of the world. CABI, Wallingford
Cannon P, Buddie A, Bridge P, de Neergaard E, Lübeck M, Askar M (2012a) Lectera, a new genus of the Plectosphaerellaceae for the legume pathogen Volutella colletotrichoides. MycoKeys 3:23–36
Cannon PF, Damm U, Johnston PR, Weir BS (2012b) Colletotrichum current status and future directions. Stud Mycol 73:181–213
Carbone I, Kohn LM (1999) A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 91:553–556
Casas JJ, Descals E (1997) Aquatic hyphomycetes from mediterranean streams contrasting in chemistry and riparian canopy. Limnetica 13:35–55
Castlebury LA, Rossman AY, Sung GH, Hyten AS, Spatafora JW (2004) Multigene phylogeny reveals new lineage for Stachybotrys chartarum, the indoor air fungus. Mycol Res 108:864–872
Chaiwan N, Gomdola D, Wang S, Monkai J, Tibpromma S, Doilom M, Wanasinghe DN, Mortimer PE, Lumyong S, Hyde KD (2021) https://gmsmicrofungi.org: an online database providing updated information of microfungi in the Greater Mekong Subregion. Mycosphere 12:1513–1526
Chaverri P, Salgado C, Hirooka Y, Rossman AY, Samuels GJ (2011) Delimitation of Neonectria and Cylindrocarpon (Nectriaceae, Hypocreales, Ascomycota) and related genera with Cylindrocarpon-like anamorphs. Stud Mycol 68:57–78
Chethana KWT, Manawasinghe IS, Hurdeal VG, Bhunjun CS, Appadoo MA, Gentekaki E, Raspé O, Promputtha I, Hyde KD (2021) What are fungal species and how to delineate them? Fungal Divers 109:1–25
Corda A (1837) Icones fungorum hucusque cognitorum, vol 1. J.G. Calve, Prague
Costa LB, Rangel DE, Morandi MA, Bettiol W (2012) Impact of UV-B radiation on Clonostachys rosea germination and growth. World J Microbiol Biotechnol 28:2497–2504
Cowling SW (1963) The aquatic hyphomycetes of eastern Australia. Armidale. BSc Thesis, Australia
Crane JL, Schoknecht JD (1982) Hyphomycetes from freshwater swamps and hammocks. Canad J Bot 60(4):369–378
Cressa C, Smits G (2007) Aquatic hyphomycetes in two blackwater streams of Venezuela. Ecotropicos 20:82–85
Crous PW, Peerally A (1996) Gliocladiopsis irregularis sp. nov. and a note on Cylindrocladium spathyphylli. Mycotaxon 58:119–128
Crous PW, Wingfield MJ (1993) A re-evaluation of Cylindrocladiella, and a comparison with morphologically similar genera. Mycol Res 97:433–448
Crous PW, Kendick WB, Alfenas AC (1997) New species of hyphomycetes associated with Eucalyptus. S Afr J Bot 63:286–290
Crous PW, Groenewald JZ, Risede JM, Hywel-Jones NL (2004) Calonectria species and their Cylindrocladium anamorphs: species with sphaeropedunculate vesicles. Stud Mycol 50:415–429
Crous PW, Shivas RG, Quaedvlieg W, van der Bank M, Zhang Y, Summerell BA, Guarro J, Wingfield MJ, Wood AR, Alfenas AC, Braun U, Cano-Lira JF, García D, Marin-Felix Y, Alvarado P, Andrade JP, Armengol J, Assefa A, den Breeÿen A, Camele I, Cheewangkoon R, De Souza JT, Duong TA, Esteve-Raventós F, Fournier J, Frisullo S, García-Jiménez J, Gardiennet A, Gené J, Hernández-Restrepo M, Hirooka Y, Hospenthal DR, King A, Lechat C, Lombard L, Mang SM, Marbach PA, Marincowitz S, Marin-Felix Y, Montaño-Mata NJ, Moreno G, Perez CA, Pérez Sierra AM, Robertson JL, Roux J, Rubio E, Schumacher RK, Stchigel AM, Sutton DA, Tan YP, Thompson EH, van der Linde E, Walker AK, Walker DM, Wickes BL, Wong PT, Groenewald JZ (2014) Fungal planet description sheets: 214–280. Persoonia 32:184–306
Crous PW, Wingfield MJ, Burgess TI, Carnegie AJ, Hardy GESJ, Smith D, Summerell BA, Cano-Lira JF, Guarro J, Houbraken J, Lombard L, Martin MP, Sandoval-Denis M, Alexandrova AV, Barnes CW, Baseia IG, Bezerra JDP, Guarnaccia V, May TW, Hernandez-Restrepo M, Stchigel AM, Miller AN, Ordonez ME, Abreu VP, Accioly T, Agnello C, Agustin Colman A, Albuquerque CC, Alfredo DS, Alvarado P, Araujo-Magalhaes GR, Arauzo S, Atkinson T, Barili A, Barreto RW, Bezerra JL, Cabral TS, Camello Rodriguez F, Cruz RHSF, Daniels PP, da Silva BDB, de Almeida DAC, de Carvalho Junior AA, Decock CA, Delgat L, Denman S, Dimitrov RA, Edwards J, Fedosova AG, Ferreira RJ, Firmino AL, Flores JA, Garcia D, Gene J, Giraldo A, Gois JS, Gomes AAM, Goncalves CM, Gouliamova DE, Groenewald M, Gueorguiev BV, Guevara-Suarez M, Gusmao LFP, Hosaka K, Hubka V, Huhndorf SM, Jadan M, Jurjevic Z, Kraak B, Kucera V, Kumar TKA, Kusan I, Lacerda SR, Lamlertthon S, Lisboa WS, Loizides M, Luangsa-ard JJ, Lyskova P, Mac Cormack WP, Macedo DM, Machado AR, Malysheva EF, Marinho P, Matocec N, Meijer M, Mesic A, Mongkolsamrit S, Moreira KA, Morozova OV, Nair KU, Nakamura N, Noisripoom W, Olariaga I, Oliveira RJV, Paiva LM, Pawar P, Pereira OL, Peterson SW, Prieto M, Rodriguez-Andrade E, Rojo De Blas C, Roy M, Santos ES, Sharma R, Silva GA, Souza-Motta CM, Takeuchi-Kaneko Y, Tanaka C, Thakur A, Smith MT, Tkalcec Z, Valenzuela-Lopez N, van der Kleij P, Verbeken A, Viana MG, Wang XW, Groenewald JZ (2017) Fungal planet description sheets: 625–715. Persoonia 39:270–467
Crous PW, Luangsa-Ard JJ, Wingfield MJ, Carnegie AJ, Hernandez-Restrepo M, Lombard L, Roux J, Barreto RW, Baseia IG, Cano-Lira JF, Martín MP, Morozova OV, Stchigel AM, Summerell BA, Brandrud TE, Dima B, García D, Giraldo A, Guarro J, Gusmão LFP, Khamsuntorn P, Noordeloos ME, Nuankaew S, Pinruan U, Rodríguez-Andrade E, Souza-Motta CM, Thangavel R, van Iperen AL, Abreu VP, Accioly T, Alves JL, Andrade JP, Bahram M, Baral H-O, Barbier E, Barnes CW, Bendiksen E, Bernard E, Bezerra JDP, Bezerra JL, Bizio E, Blair JE, Bulyonkova TM, Cabral TS, Caiafa MV, Cantillo T, Colmán AA, Conceição LB, Cruz S, Cunha AOB, Darveaux BA, da Silva AL, da Silva GA, da Silva GM, da Silva RMF, de Oliveira RJV, Oliveira RL, De Souza JT, Dueñas M, Evans HC, Epifani F, Felipe MTC, Fernández-López J, Ferreira BW, Figueiredo CN, Filippova NV, Flores JA, Gené J, Ghorbani G, Gibertoni TB, Glushakova AM, Healy R, Huhndorf SM, Iturrieta-González I, Javan-Nikkhah M, Juciano RF, Jurjević Ž, Kachalkin AV, Keochanpheng K, Krisai Greilhuber I, Li Y-C, Lima AA, Machado AR, Madrid H, Magalhães OMC, Marbach PAS, Melanda GCS, Miller AN, Mongkolsamrit S, Nascimento RP, Oliveira TGL, Ordoñez ME, Orzes R, Palma MA, Pearce CJ, Pereira OL, Perrone G, Peterson SW, Pham THG, Piontelli E, Pordel A, Quijada L, Raja HA, Rosas de Paz E, Ryvarden L, Saitta A, Salcedo SS, Sandoval-Denis M, Santos TAB, Seifert KA, Silva BDB, Smith ME, Soares AM, Sommai S, Sousa JO, Suetrong S, Susca A, Tedersoo L, Telleria MT, Thanakitpipattana D, Valenzuela-Lopez N, Visagie CM, Zapata M, Groenewald JZ (2018) Fungal planet description sheets: 785–867. Persoonia 41:238–417
Crous PW, Carnegie AJ, Wingfield MJ, Sharma R, Mughini G, Noordeloos ME, Santini A, Shouche YS, Bezerra JDP, Dima B, Guarnaccia V, Imrefi I, Jurjević Ž, Knapp DG, Kovács GM, Magistà D, Perrone G, Rämä T, Rebriev YA, Shivas RG, Singh SM, Souza-Motta CM, Thangavel R, Adhapure NN, Alexandrova AV, Alfenas AC, Alfenas RF, Alvarado P, Alves AL, Andrade DA, Andrade JP, Barbosa RN, Barili A, Barnes CW, Baseia IG, Bellanger JM, Berlanas C, Bessette AE, Bessette AR, Biketova AY, Bomfim FS, Brandrud TE, Bransgrove K, Brito ACQ, CanoLira JF, Cantillo T, Cavalcanti AD, Cheewangkoon R, Chikowski RS, Conforto C, Cordeiro TRL, Craine JD, Cruz R, Damm U, de Oliveira RJV, de Souza JT, de Souza HG, Dearnaley JDW, Dimitrov RA, Dovana F, Erhard A, Esteve-Raventós F, Félix CR, Ferisin G, Fernandes RA, Ferreira RJ, Ferro LO, Figueiredo CN, Frank JL, Freire KTLS, García D, Gené J, Gęsiorska A, Gibertoni TB, Gondra RAG, Gouliamova DE, Gramaje D, Guard F, Gusmão LFP, Haitook S, Hirooka Y, Houbraken J, Hubka V, Inamdar A, Iturriaga T, Iturrieta-González I, Jadan M, Jiang N, Justo A, Kachalkin AV, Kapitonov VI, Karadelev M, Karakehian J, Kasuya T, Kautmanová I, Kruse J, Kušan I, Kuznetsova TA, Landell MF, Larsson K-H, Lee HB, Lima DX, Lira CRS, Machado AR, Madrid H, Magalhães OMC, Majerova H, Malysheva EF, Mapperson RR, Marbach PAS, Martín MP, MartínSanz A, Matočec N, McTaggart AR, Mello JF, Melo RFR, Mešić A, Michereff SJ, Miller AN, Minoshima A, Molinero-Ruiz L, Morozova OV, Mosoh D, Nabe M, Naik R, Nara K, Nascimento SS, Neves RP, Olariaga I, Oliveira RL, Oliveira TGL, Ono T, Ordoñez ME, Ottoni AM, Paiva LM, Pancorbo F, Pant B, Pawłowska J, Peterson SW, Raudabaugh DB, Rodríguez-Andrade E, Rubio E, Rusevska K, Santiago ALCMA, Santos ACS, Santos C, Sazanova NA, Shah S, Sharma J, Silva BDB, Siquier JL, Sonawane MS, Stchigel AM, Svetasheva T, Tamakeaw N, Telleria MT, Tiago PV, Tian CM, Tkalčec Z, Tomashevskaya MA, Truong HH, Vecherskii MV, Visagie CM, Vizzini A, Yilmaz N, Zmitrovich IV, Zvyagina EA, Boekhout T, Kehlet T, Læssøe T, Groenewald JZ (2019) Fungal planet description sheets: 868–950. Persoonia 42:291–473
Crous PW, Hernández-Restrepo M, Schumacher RK, Cowan DA, Maggs-Kölling G, Marais E, Wingfield MJ, Yilmaz N, Adan OCG, Akulov A, Duarte EÁ, Berraf-Tebbal A, Bulgakov TS, Carnegie AJ, de Beer ZW, Decock C, Dijksterhuis J, Duong TA, Eichmeier A, Hien LT, Houbraken JAMP, Khanh TN, Liem NV, Lombard L, Lutzoni FM, Miadlikowska JM, Nel WJ, Pascoe IG, Roets F, Roux J, Samson RA, Shen M, Spetik M, Thangavel R, Thanh HM, Thao LD, van Nieuwenhuijzen EJ, Zhang JQ, Zhang Y, Zhao LL, Groenewald JZ (2021a) New and interesting fungi. 4. Fungal Syst Evol 7:255–343
Crous PW, Lombard L, Sandoval-Denis M, Seifert KA, Schroers HJ, Chaverri P, Gené J, Guarro J, Hirooka Y, Bensch K, Kema GHJ, Lamprecht SC, Cai L, Rossman AY, Stadler M, Summerbell RC, Taylor JW, Ploch S, Visagie CM, Yilmaz N, Frisvad JC, Abdel-Azeem AM, Abdollahzadeh J, Abdolrasouli A, Akulov A, Alberts JF, Araújo JPM, Ariyawansa HA, Bakhshi M, Bendiksby M, Ben Hadj Amor A, Bezerra JDP, Boekhout T, Câmara MPS, Carbia M, Cardinali G, Castañeda-Ruiz RF, Celis A, Chaturvedi V, Collemare J, Croll D, Damm U, Decock CA, de Vries RP, Ezekiel CN, Fan XL, Fernández NB, Gaya E, González CD, Gramaje D, Groenewald JZ, Grube M, Guevara-Suarez M, Gupta VK, Guarnaccia V, Haddaji A, Hagen F, Haelewaters D, Hansen K, Hashimoto A, Hernández-Restrepo M, Houbraken J, Hubka V, Hyde KD, Iturriaga T, Jeewon R, Johnston PR, Jurjević Ž, Karalti İ, Korsten L, Kuramae EE, Kušan I, Labuda R, Lawrence DP, Lee HB, Lechat C, Li HY, Litovka YA, Maharachchikumbura SSN, Marin-Felix Y, Matio Kemkuignou B, Matočec N, McTaggart AR, Mlčoch P, Mugnai L, Nakashima C, Nilsson RH, Noumeur SR, Pavlov IN, Peralta MP, Phillips AJL, Pitt JI, Polizzi G, Quaedvlieg W, Rajeshkumar KC, Restrepo S, Rhaiem A, Robert J, Robert V, Rodrigues AM, Salgado-Salazar C, Samson RA, Santos ACS, Shivas RG, Souza-Motta CM, Sun GY, Swart WJ, Szoke S, Tan YP, Taylor JE, Taylor PWJ, Tiago PV, Váczy KZ, van de Wiele N, van der Merwe NA, Verkley GJM, Vieira WAS, Vizzini A, Weir BS, Wijayawardene NN, Xia JW, Yáñez-Morales MJ, Yurkov A, Zamora JC, Zare R, Zhang CL, Thines M (2021b) Fusarium: more than a node or a foot-shaped basal cell. Stud Mycol 98:100116
da Silva RF, Briedis GS (2009) Registro de la presencia de hifomicetos acuáticos em rios de La cordillera de la costa, Venezuela. Interciência 34:589–592
Dann EK, Cooke AW, Forsberg LI, Pegg KG, Tan YP, Shivas RG (2012) Pathogenicity studies in avocado with three nectriaceous fungi, Calonectria ilicicola, Gliocladiopsis sp. and Ilyonectria liriodendri. Plant Pathol 61:896–902
Dao HT, Beattie GAC, Rossman AY, Burgess LW, Holford P (2016) Four putative entomopathogenic fungi of armoured scale insects on Citrus in Australia. Mycol Prog 15:47
Davidson DE, Christensen M (1971) Emericellopsis stolkiae sp. nov. from saline soils in Wyoming. Trans Br Mycol Soc 57:385–391
Dayarathne MC, Maharachchikumbura SS, Jones EBG, Dong W, Devadatha B, Yang J, Ekanayaka AH, De Silva W, Sarma VV, Al-Sadi AM, Khongphinitbunjong K, Hyde KD, Zhao RL (2019) Phylogenetic revision of Savoryellaceae and evidence for its ranking as a subclass. Front Microbiol 10:840
de Beer ZW, Seifert KA, Wingfield MJ (2013a) The ophiostomatoid fungi: their dual position in the Sordariomycetes. In: Seifert KA, De Beer ZW, Wingfield MJ (eds), The Ophiostomatoid Fungi: expanding frontiers. CBS Biodiversity Series, vol. 12. CBS-KNAW Fungal Biodiversity Centre, Utrecht, The Netherlands, p. 1e19.
de Beer ZW, Seifert KA, Wingfield MJ (2013b) The ophiostomatoid fungi: their dual position in the Sordariomycetes. CBS Biodivers Ser 12:1–19
de Beer ZW, Seifert KA, Wingfield MJ (2013c) A nomenclator for ophiostomatoid genera and species in the Ophiostomatales and Microascales. Ophiostomatoid fungi: expanding frontiers. Utrecht, CBS-KNAW Fungal Biodiversity Centre. CBS Biodiversity Series 12:245–322
de Beer ZW, Duong TA, Barnes I, Wingfield BD, Wingfield MJ (2014) Redefining Ceratocystis and allied genera. Stud Mycol 79:1871–2219
de Hoog GS, de Smith MT, Guého E (1986) A revision of the genus Geotrichum and its teleomorphs. Stud Mycol 29:131
de Hoog GS, Guarro J, Gené J, Figueras MJ (2011) Atlas of clinical fungi. CD-ROM version 3.1. CBS-KNAW Fungal Biodiversity Centre, Utrecht, The Netherlands.
del Campo J, Ruiz-Trillo I (2013) Environmental survey meta-analysis reveals hidden diversity among unicellular opisthokonts. Mol Biol Evol 30(4):802–805
Dennis RWG (1960) British cup fungi and their allies. An introduction to the Ascomycetes. Bernard Quaritch Ltd., London, p 280
Doguet G (1957) Organogenie du Microascus stysanophorus (Matt.) Curzi. Bull Soc Mycol France 73:165–178
Doilom M, Dissanayake AJ, Wanasinghe DN, Boonmee S, Liu JK, Bhat DJ, Taylor JE, Bahkali AH, McKenzie EHC, Hyde KD (2017) Microfungi on Tectona grandis (Teak) in Northern Thailand. Fungal Divers 82:107–182
Domsch KH, Gams W, Anderson T (2007) Compendium of soil fungi, 2nd edn. IHW Verlag, Eching
Drummond AJ, Rambaut A (2007) BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7:214
Drummond AJ, Suchard MA, Xie D, Rambaut A (2012) Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol 29:1969–1973
Dubey R, Pandey AK (2013) Stachybotrys citri sp. nov. and Volutella rauvolfii sp. nov.—two novel dematiaceous hyphomycetes from India. Indian Phytopathol 66(2): 204–206
Elidemir O, Colasurdo GN, Rossmann SN, Fan LL (1999) Isolation of Stachybotrys from the lung of a child with pulmonary hemosiderosis. Pediatrics 104(4):964–996
Ellis MB (1971) Dematiaceous hyphomycetes. Commonwealth Mycological Institute, Kew
Ellis MB (1976) More dematiaceous hyphomycetes. Commonwealth Mycological Institute, Kew
Emmonas CW, Dodge BO (1931) The ascosporic stage of species of Scopulariopsis. Mycologia 23:313–331
Eriksson OE (2006) Outline of Ascomycota—2006. Myconet 12:1–82
Eriksson OE, Winka K (1997) Supraordinal taxa of Ascomycota. Myconet 1:1–16
Feijen FA, Vos RA, Nuytinck J, Merckx VS (2018) Evolutionary dynamics of mycorrhizal symbiosis in land plant diversification. Sci Rep 8(1):1–7
Fisher PJ, Petrini O (1989) Two aquatic hyphomycetes as endophytes in Alnus glutinosa roots. Mycol Res 92:367–368
Fiuza PO, Gusmão LFP (2013) Ingoldian fungi from semiarid Caatinga biome of Brazil. The Genus Campylospora Mycosphere 4(3):559–565
Fries EM (1832) Systema Mycologicum 3:261–524
Fryar SC, Booth W, Davies J, Hodgkiss JI, Hyde KD (2004) Distribution of fungi on wood in the Tutong River, Brunei. Fungal Divers 17:17–38
Gams W (1971) Cephalosporium-artige Schimmelpilze (Hyphomycetes). Gustav Fischer Verlag, Stuttgart, Germany
Gams W, Hawksworth DL (1975) Identity of Acrocylindrium oryzae Sawada and a similar fungus causing sheath-rot of rice. Kavaka 3:57–61
Gilgado F, Gené J, Cano J, Guarro J (2007) Reclassification of Graphium tectonae as Parascedosporium tectonae gen. nov., comb. nov., Pseudallescheria africana as Petriellopsis africana gen. nov., comb. nov. and Pseudallescheria fimeti as Lophotrichus fimeti comb. nov. Int J Syst Evol Microbiol 57:2171–2178
Gilman JC (1957) A manual of soil fungi, 2nd edn. Iowa State College Press, Ames
Giraldo A, Crous PW (2019) Inside Plectosphaerellaceae. Stud Mycol 92(1):227–286
Giraldo A, Gené J, Cano J, de Hoog S, Guarro J (2012) Two new species of Acremonium from Spanish soils. Mycologia 104:1456–1465
Giraldo A, Gené J, Sutton DA, Madrid H, de Hoog GS, Cano J, Decock C, Crous PW, Guarro J (2015) Phylogeny of Sarocladium (Hypocreales). Persoonia 34:10–24
Glass NL, Donaldson G (1995) Development of primer sets designed for use with PCR to amplify conserved genes from filamentous ascomycetes. Appl Environ Microbiol 61:1323–1330
Glenn AE, Bacon CW, Price R, Hanlin RT (1996) Molecular phylogeny of Acremonium and its taxonomic implications. Mycologia 88:369–383
Goh TK, Hyde KD (1996) Biodiversity of freshwater fungi. J Ind Microbiol 17:328–345
Gordillo A, Decock C (2019) Multigene phylogenetic and morphological evidence for seven new species of Aquanectria and Gliocladiopsis (Ascomycota, Hypocreales) from tropical areas. Mycologia 111(2):299–318
Gräfenhan T, Schroers HJ, Nirenberg HI, Seifert KA (2011) An overview of the taxonomy, phylogeny, and typification of nectriaceous fungi in Cosmospora, Acremonium, Fusarium, Stilbella, and Volutella. Stud Mycol 68:79–113
Grum-Grzhimaylo AA, Georgieva ML, Debets AJ, Bilanenko EN (2013) Are alkalitolerant fungi of the Emericellopsis lineage (Bionectriaceae) of marine origin? IMA Fungus 4(2):213–228
Grütz O (1925) Dermatologische Wochenschrift 80
Guarnaccia V, Van Niekerk J, Crous P, Sandoval-Denis M (2021) Neocosmospora spp. associated with dry root rot of citrus in South Africa. Phytopathol Mediterr 60(1):79–100
Guéguen F (1905) Gliomastix (Torula) chartarum n. gen. n. sp.; contribution á l’étude de la formation endogène des conidies. Bull Soc Mycol Fr 21:230–241
Gueidan C, Ruibal C, De Hoog GS, Schneider H (2011) Rock-inhabiting fungi originated during periods of dry climate in the late Devonian and middle Triassic. Fungal Biol 115:987–996
Guu JR, Ju YM, Hsieh HJ (2007) Nectriaceous fungi collected from forests in Taiwan. Bot Stud 48:187–203
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis programfor Windows 95/98/NT.
Halleen F, Schroers HJ, Groenewald JZ, Crous PW (2004) Novel species of Cylindrocarpon (Neonectria) and Campylocarpon gen. nov. associated with black foot disease of grapevines (Vitis spp.). Stud Mycol 50:431–455
Harter LL (1916) Sweet-potato scurf. J Agric Res 5:787–791
Hausner G, Reid J (2004) The nuclear small subunit ribosomal genes of Sphaeronaemella helvellae, Sphaeronaemella fimicola, Gabarnaudia betae, and Cornuvesica falcata: phylogenetic implications. Can J Bot 82:752–762
Hernández-Restrepo M, Gené J, Castañeda-Ruiz RF, Mena-Portales J (2017) Phylogeny of saprobic microfungi from Southern Europe. Stud Mycol 86:53–97
Herrera CS, Rossman AY, Samuels GJ, Pereira OL, Chaverri P (2015) Systematics of the Cosmospora viliuscula species complex. Mycologia 107:532–557
Hibbett DS, Binder M (2001) Evolution of marine mushrooms. Biol Bull 201(3):319–322
Hibbett DS, Binder M, Bischoff JF, Blackwell M, Cannon PF, Eriksson OE, Huhndorf S, James ST, Kirk PM, Lucking R, Lumbsch HT, Lutzoni F, Matheny PB, Mclaughlin DJ, Powell MJ, Redhed S, Schoch CL, Spatafora JF, Stalpers JA, Vilgalys R, Aime MC, Aptroot AA, Bauer R, Begerow D, Benny GL, Castelbury LA, Crous PW, Dai YC, Gams W, Geiser DM, Griffith GW, Gueidan C, Hawksworth DL, Hestmark G, Hosaka K, Humber RA, Hyde KD, Ironisde JE, Koljag U, Kurtzman CP, Larsson KH, Lichtwardt R, Longcore J, Miadlikowska J, Miller A, Moncalvo JM, Mozley-StandridgeS OF, Parmasto E, Reeb V, Rogers JD, Roux C, Ryvarden L, Sampaio JP, Schβler A, Sugiyama J, Thorn RG, Tibell L, Untereiner WA, Walker C, Wang Z, Weir A, Weiss M, White MM, Winka K, Yao YJ, Zhang N (2007) A higher–level phylogenetic classification of the fungi. Mycol Res 111:509–547
Hillis DM, Bull JJ (1993) An empirical test of bootstrapping as a method for assessing confidence in phylogenetic analysis. Syst Biol 42(2):182–192
Ho WH, Hyde KD (2000) A new family of freshwater ascomycetes. Fungal Divers 4:21–36
Holubova-Jechova V (1990) Problem s in the taxonomy of the Dematiaceous Hyphomycetes. Stud Mycol 32:41–48
Hongsanan S, Maharachchikumbura SSN, Hyde KD, Samarakoon MC, Jeewon R, Zhao Q, Al-Sadi AM, Bahkali AH (2017) An updated phylogeny of Sordariomycetes based on phylogenetic and molecular clock evidence. Fungal Divers 84:25–41
Hu DM, Liu F, Cai L (2013) Biodiversity of aquatic fungi in China. Mycology 4:125–168
Hu DM, Wang M, Cai L (2017) Phylogenetic assessment and taxonomic revision of Mariannaea. Mycol Prog 16(4):271–283
Huang SK, Jeewon R, Hyde KD, Bhat DJ, Wen TC (2018) Novel taxa within Nectriaceae: Cosmosporella gen. nov. and Aquanectria sp. nov. from freshwater habitats in China. Cryptogamie Mycol 39(2):169–192
Huang SK, Hyde KD, Maharachchikumbura SSN, McKenzie EHC, Wen TC (2021) Taxonomic studies of Coronophorales and Niessliaceae (Hypocreomycetidae). Mycosphere 12(1):875–992
Hughes SJ (1958) Revisiones Hyphomycetum aliquot cum appendice de nominibus rejiciendis. Can J Bot 36:727–836
Huhndorf SM, Iller ANM, Fernández FA (2004) Molecular systematics of the Sordariales: the order and the family Lasiosphaeriaceae redefined. Mycologia 96:368–387
Hyde KD (1992) Tropical Australian freshwater fungi IV. Halosarpheia aquatica sp. nov., Garethjonesia lacunosispora gen. et sp.nov. and Ophioceras dolichostomum (Ascomycetes). Aust Syst Bot 5:407–414
Hyde KD, Goh TK (1999) Fungi on submerged wood from River Coln, England. Mycol Res 103:1561–1574
Hyde KD, Goh TK (2003) Adaptations for dispersal in filamentous freshwater fungi. Fungal Divers 10:231–258
Hyde KD, Wong SW (2000) Didymella aptrooti sp. nov. from bamboo submerged in freshwater. Aust Mycol 18:54–59
Hyde KD, Sarma VV, Jones EBG (2000) Morphology and taxonomy of higher marine fungi. Fungal Divers 1:172–204
Hyde KD, Nilsson RH, Alias SA, Ariyawansa HA, Blair JE, Cai L, de Cock AWAM, Dissanayake AJ, Glockling SL, Goonasekara ID, Gorczak M, Hahn M, Jayawardena RS, van Kan JAL, Laurence MH, Le´vesque CA, Li X, Liu J-K, Maharachchikumbura SSN, Manamgoda DS, Martin FN, McKenzie EHC, McTaggart AR, Mortimer PE, Nair & Julia Pawłowska PVR, Rintoul TL, Shivas RG, Spies CFJ, Summerell BA, Taylor PWJ, Terhem RB, Udayanga D, Vaghefi N, Walther G, Wilk M, Wrzosek M, Xu J-C, Yan JY, Zhou N (2014) One stop shop: backbones trees for important phytopathogenic genera: I (2014). Fungal Divers 67:21–125
Hyde KD, Maharachchikumbura SS, Hongsanan S, Samarakoon MC, Lücking R, Pem D, Harishchandra D, Jeewon R, Zhao RL, Xu JC, Liu JK (2017) The ranking of fungi: a tribute to David L. Hawksworth on his 70th birthday. Fungal Divers 84:1–23
Hyde KD, Chaiwan N, Norphanphoun C, Boonmee S, Camporesi E, Chethana KWT, Dayarathne MC, de Silva NI, Dissanayake AJ, Ekanayaka AH, Hongsanan S, Huang SK, Jayasiri SC, Jayawardena RS, Jiang HB, Karunarathna A, Lin CG, Liu JK, Liu NG, Lu YZ, Luo ZL, Maharachchimbura SSN, Manawasinghe IS, Pem D, Perera RH, Phukhamsakda C, Samarakoon MC, Senwanna C, Shang Q, Tennakoon DS, Thambugala KM, Tibpromma S, Wanasinghe DN, Xiao YP, Yang J, Zeng XY, Zhang JF, Zhang SN, Bulgakov TS, Bhat DJ, Cheewangkoon R, Goh TK, Jones EBG, Kang JC, Jeewon R, Liu ZY, Lumyong S, Kuo CH, McKenzie EHC, Wen TC, Yan JY, Zhao Q (2018) Mycosphere notes 169–224. Mycosphere 9:271–430
Hyde KD, Dong Y, Phookamsak R, Jeewon R, Bhat DJ, Jones EBG, Liu NG, Abeywickrama PD, Mapook A, Wei DP, Perera RH, Manawasinghe IS, Pem D, Bundhun D, Karunarathna A, Ekanayaka AH, Bao DF, Li JF, Samarakoon MC, Chaiwan N, Lin CG, Phutthacharoen K, Zhang SN, Senanayake IC, Goonasekara ID, Thambugala KM, Phukhamsakda C, Tennakoon DS, Jiang HB, Yang J, Zeng M, Huanraluek N, Liu JK, Wijesinghe SN, Tian Q, Tibpromma S, Brahmanage RS, Boonmee S, Huang SK, Thiyagaraja V, Lu YZ, Jayawardena RS, Dong W, Yang EF, Singh SK, Singh SM, Rana S, Lad SS, Anand G, Devadatha B, Niranjan M, Sarma VV, Liimatainen K, Aguirre- Hudson B, Niskanen T, Overall A, Alvarenga RLM, Gibertoni TB, Pfliegler WP, Horváth E, Imre A, Alves AL, da Silva Santos AC, Tiago PV, Bulgakov TS, Wanasinghe DN, Bahkali AH, Doilom M, Elgorban AM, Maharachchikumbura SSN, Rajeshkumar KC, Haelewaters D, Mortimer PE, Zhao Q, Lumyong S, Xu JC, Sheng J (2020a) Fungal diversity notes 1151–1276: taxonomic and phylogenetic contributions on genera and species of fungal taxa. Fungal Divers 100:5–277
Hyde KD, Jeewon R, Chen YJ, Bhunjun CS, Calabon MS, Jiang HB, Lin CG, Norphanphoun C, Sysouphanthong P, Pem D, Tibpromma S, Zhang Q, Doilom M, Jayawardena RS, Liu JK, Maharachchikumbura SSN, Phukhamsakda C, Phookamsak R, Al-Sadi AM, Naritsada Thongklang N, Wang Y, Gafforov Y, Jones EBG, Lumyong S (2020b) The numbers of fungi: is the descriptive curve flattening? Fungal Divers 103:219–271
Hyde KD, Norphanphoun C, Maharachchikumbura SSN, Bhat DJ, Jones EBG, Bundhun D, Chen YJ, Bao DF, Boonmee S, Calabon MS, Chaiwan N, Chethana KWT, Dai DQ, Dayarathne MC, Devadatha B, Dissanayake AJ, Dissanayake LS, Doilom M, Dong W, Fan XL, Goonasekara ID, Hongsanan S, Huang SK, Jayawardena RS, Jeewon R, Karunarathna A, Konta S, Kumar V, Lin CG, Liu JK, Liu NG, Luangsa-ard J, Lumyong S, Luo ZL, Marasinghe DS, McKenzie EHC, Niego AGT, Niranjan M, Perera RH, Phukhamsakda C, Rathnayaka AR, Samarakoon MC, Samarakoon SMBC, Sarma VV, Senanayake IC, Shang QJ, Stadler M, Tibpromma S, Wanasinghe DN, Wei DP, Wijayawardene NN, Xiao YP, Yang J, Zeng XY, Zhang SN, Xiang MM (2020c) Refined families of sordariomycetes. Mycosphere 11:305–1059
Hyde KD, Bao DF, Hongsanan S, Chethana KWT, Yang J, Suwannarach N (2021) Evolution of freshwater Diaporthomycetidae (Sordariomycetes) provides evidence for five new orders and six new families. Fungal Divers 107:71–105
Ingold CT (1942) Aquatic hyphomycetes of decaying alder leaves. Trans Br Mycol Soc 25:339–417
Izabel TDSS, Cruz ACRD, Barbosa FR, Ferreira SML, Marques MFO, Gusmão LFP (2010) The genus Stachybotrys anamorphic fungi) in the semi-arid region of Brazil. Rev Bras Bot 33:479–487
James TY, Kauff F, Schoch CL, Matheny PB, Hofstetter V, Cox CJ, Celio G, Gueidan C, Fraker E, Miadlikowska J, Lumbsch HT, Rauhut A, Reeb V, Arnold AE, Amtoft A, Stajich JE, Hosaka K, Sung GH, Johnson D, O’Rourke B, Crockett M, Binder M, Curtis JM, Slot JC, Wang Z, Wilson AW, Schußler AW, Longcore JE, O’Donnell K, Mozley-Standridge S, Porter D, Letcher PM, Powell MJ, Taylor JW, Whit MM, Griffith GW, Davies DR, Humber RA, Morton JB, Sugiyama J, Rossman AY, Rogers JD, Pfister DH, Hewitt D, Hansen K, Hambleton S, Shoemaker RA, Kohlmeyer J, Volkmann-Kohlmeyer B, Spotts RA, Serdani M, Crous PW, Hughes KW, Matsuura K, Langer E, Langer G, Untereiner WA, Lücking R, Büdel B, Geiser DM, Aptroot A, Diederich P, Schmitt I, Schultz M, Yahr R, Hibbett DS, Lutzoni F, McLaughlin DJ, Spatafora JW, Vilgalys R (2006) Reconstructing the early evolution of Fungi using a six-gene phylogeny. Nature 443:818–822
Jarvis BB, Sorenson WG, Hintikka EL, Nikulin M, Zhou Y, Jiang J, Wang S, Hinkley S, Etzel RA, Dearborn D (1998) Study of toxin production by isolates of Stachybotrys chartarum and Memnoniella echinata isolated during a study of pulmonary hemosiderosis in infants. Appl Environ Microbiol 64(10):3620–3625
Jayasiri SC, Hyde KD, Ariyawansa HA, Bhat J, Buyck B, Cai L, Dai YC, Abd-Elsalam KA, Ertz D, Hidayat I, Jeewon R, Jones EBG, Bahkali AH, Karunarathna SC, Liu JK, Luangsa-Ard JJ, Lumbsch HT, Maharachchikumbura SSN, McKenzie EHC, Moncalvo JM, Ghobad-Nejhad M, Nilsson H, Pang KL, Pereira OL, Phillips AJL, Raspe O, Rollins AW, Romero AI, Etayo J, Selcuk F, Stephenson SL, Suetrong S, Taylor JE, Tsui CKM, Vizzini A, Abdel-Wahab MA, Wen TC, Boonmee S, Dai DQ, Daranagama DA, Dissanayake AJ, Ekanayaka AH, Fryar SC, Hongsanan S, Jayawardena RS, Li WJ, Perera RH, Phookamsak R, De Silva NI, Thambugala KM, Tian Q, Wijayawardene NN, Zhao RL, Zhao Q, Kang JC, Promputtha I (2015) The faces of fungi database: fungal names linked with morphology, phylogeny and human impacts. Fungal Divers 74:3–18
Jayawardena RS, Hyde KD, Damm U, Cai L, Liu M, Li XH, Zhang W, Zhao WS, Yan JY (2016) Notes on currently accepted species of Colletotrichum. Mycosphere 7:1192–1260
Jeewon R, Hyde KD (2016) Establishing species boundaries and new taxa among fungi: recommendations to resolve taxonomic ambiguities. Mycosphere 7:1669–1677
Jones EBG (1964) Nautosphaeria cristaminuta gen. et sp. nov., a marine pyrenomycete on submerged wood. Trans Br Mycol Soc 47:97–101
Jones EG (2006) Form and function of fungal spore appendages. Mycoscience 47(4):167–183
Jones EBG, Hyde KD (1992) Taxonomic studies on Savoryella Jones et Eaton (Ascomycotina). Bot Mar 35:83–91
Jones EBG, Sakayaroj J, Suetrong S, Somrithipol S (2009) Classification of marine Ascomycota, anamorphic taxa and Basidiomycota. Fungal Divers 35:1–187
Jones MDM, Forn I, Gadelha C, Egan MJ, Bass D, Massana R, Richards TA (2011) Discovery of novel intermediate forms redefines the fungal tree of life. Nature 474:200–203
Jones EBG, Hyde KD, Pang KL (2014) Freshwater fungi and fungallike organisms. De Gruyter, Germany
Jones EBG, Suetrong S, Sakayaroj J, Bahkali AH, Abdel-Waha MA, Boekhout T, Pang KL (2015) Classification of marine Ascomycota, Basidiomycota, Blastocladiomycota and Chytridiomycota. Fungal Divers 73:1–72
Jones EBG, Ju WT, Lu CL, Guo SY, Pang KL (2017) The Halosphaeriaceae revisited. Bot Mar 60(4):453–468
Jones EBG, Pang KL, Abdel-Wahab MA, Scholz B, Hyde KD, Boekhout T, Ebel R, Rateb ME, Henderson L, Sakayaroj J, Suetrong S, Dayarathne MC, Kumar V, Raghukumar S, Sridhar KR, Bahkali AHA, Gleason FH, Norphanphoun C (2019) An online resource for marine fungi. Fungal Divers 96(1):347–433
Jong SC, Davis EE (1976) Contributions to the knowledge of Stachybotrys and Memnoniella in culture. Mycotaxon 3:409–485
Katoh K, Standleym DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780
Katoh K, Rozewicki J, Yamada KD (2019) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinformatics 20(4):1160–1166
Kirk PM (1985) New or interesting microfung i. XIV. Dematiaceous Hyphomycetes from Mt Kenya Mycotaxon 23:305–352
Kiyuna T, Hyde KD, Kigawa R, Sano C, Miura S, Sugiyama J (2011) Molecular assessment of fungi in “black spots” that deface murals in the Takamatsuzuka and Kitora Tumuli in Japan: Acremonium sect. Gliomastix including Acremonium tumulicola sp. nov. and Acremonium felinum comb. nov. Mycoscience 52(1):1–17
Koukol O, Halasů V, Janošík L, Mlčoch P, Polhorský A, Šandová ZL (2020) Citizen science facilitates phylogenetic placement of old species of non-lichenised Pezizomycotina based on newly collected material. Czech Mycol 72(2):263–280
Krauss U, Martinez A, Hidalgo E, ten Hoopen M, Arroyo C (2002) Two-step liquid/solid state scaled-up production of Clonostachys rosea. Mycol Res 106:1449–1454
Kreisel H (1969) Grunzüge eines natürlichen Systems der Pilze. J. Cramer. Lehre 1–245
Kuzuha S (1973) Two new species of aquatic Hyphomycetes. J Jap Bot 48:220–224
Lackner M, de Hoog GS (2011) Parascedosporium and its relatives: phylogeny and ecological trends. IMA Fungus 2:39–48
Lackner M, de Hoog GS, Yang L, Moreno LF, Moreno LF, Ahmed SA, Andreas F, Kaltseis J, Nagl M, Lass-Flörl C, Risslegger B, Rambach G, Speth C, Robert V, Buzina W, Chen S, Bouchara JP, Cano-Lira JF, Guarro J, Gené J, Silva FF, Haido R, Haase G, Havlicek V, Garcia-Hermoso D, Meis JF, Hagen F, Kirchmair M, Rainer J, Schwabenbauer K, Zoderer M, Meyer W, Gilgado F, Schwabenbauer K, Vicente VA, Piecková E, Regenermel M, Rath PM, Steinmann J, de Alencar XW, Symoens F, Tintelnot K, Ulfig K, Velegraki A, Tortorano AM, Giraud S, Mina S, Rigler-Hohenwarter K, Hernando FL, Ramirez-Garcia A, Pellon A, Kaur J, Bergter EB, de Meirelles JV, da Silva ID, Delhaes L, Alastruey-Izquerdo A, Li RY, Lu QY, Moussa T, Almaghrabi O, Al-Zahrani H, Okada G, Deng S, Liao WQ, Zeng JS, Issakainen J, Lopes LCL (2014) Proposed nomenclature for Pseudallescheria, Scedosporium and related genera. Fungal Divers 67:1–10
Lechat C, Fournier J (2015) Varicosporella, a new aquatic genus in the Nectriaceae from France. Ascomycete.org 7(1):1–8
Lechat C, Fournier J (2018) Clonostachys spinulosispora (Hypocreales, Bionectriaceae), a new species on palm from French Guiana. Ascomycete.org 10:127–130
Lechat C, Fournier J (2019) Two new species of Chaetopsina (Nectriaceae) from Saül (French Guiana). Ascomycete.org 11(4):127–134
Lechat C, Fournier J (2020a) Two new species of Clonostachys (Bionectriaceae, Hypocreales) from Saül (French Guiana). Ascomycete.org 12:61–66
Lechat C, Fournier J (2020b) Chaetopsina pnagiana (Nectriaceae, Hypocreales), a new holomorphic species from Saül (French Guiana). Ascomycete.org 12(1):1–5
Lechat C, Fournier J (2021) Cosmospora xylariae (Nectriaceae), a new species from France, Germany and UK, with notes on C. berkeleyana, now Sphaerostilbella berkeleyana, and C. scruposae. Ascomycete.org 13(5):29
Lechat C, Farr DF, Hirooka Y, Minnis AM, Rossman AY (2010) A new species of Hydropisphaera, H. bambusicola, is the sexual state of Gliomastix fusigera. Mycotaxon 111:95–102
Lechat C, Fournier J, Chaduli D, Favel A (2022) Three new holomorphic species of Volutella (Nectriaceae, Hypocreales) from Saül (French Guiana). Ascomycete.org 14(3):11
Li J, Zhao J, Xu LJ, Li XL, Wang JG (2008) Endophytic fungi from rhizomes of Parispolyphylla var. yunnanensis. World J Microbiol Biotechnol 24:733–737
Liew ECY, Aptroot A, Hyde KD (2002) An evaluation of the monophyly of Massarina based on ribosomal DNA sequences. Mycologia 94:803–813
Link HF (1809) Observationes in ordines plantarum naturales. Dissertatio I. Ges. Naturf. Freunde Berlin Mag 3(1):3–42
Link JHF (1825) Caroli a Linne Species plantarum: exhibentes plantas rite cognitas, ad genera relatas, cum differentiis specificis, nominibus trivialibus, synonymis selectis, locis natalibus, secundum systema sexuale digestas. Impensis G. C. Nauk, Berolini 6(2):106
Little C (1990) The terrestrial invasion: an ecophysiological approach to the origins of land animals. CUP Archive.
Liu F, Cai L (2013) A novel species of Gliocladiopsis from freshwater habitat in China. Cryptogam Mycol 34:233–241
Liu YJ, Whelen S, Hall BD (1999) Phylogenetic relationships among ascomycetes: evidence from an RNA polymerse II subunit. Mol Biol Evol 16:1799–1808
Liu YJ, Hodson MC, Hall BD (2006) Loss of the flagellum happened only once in the fungal lineage: phylogenetic structure of kingdom Fungi inferred from RNA polymerase II subunit genes. BMC Evol Biol 6:74
Liu JK, Hyde KD, Jeewon R, Phillips AJL, Maharachchikumbura SSN, Ryberg M, Liu ZY, Zhao Q (2017a) Ranking higher taxa using divergence times: a case study in Dothideomycetes. Fungal Divers 84:75–99
Liu X, Guo Z, Huang G (2017b) Sarocladium brachiariae sp. nov., an endophytic fungus isolated from Brachiaria brizantha. Mycosphere 8:827–834
Locquin-Linard M (1975) Faurelina, nouveau genre Ascomycètes (Chadefaudiellaceae). Mycologie 39:125–129
Lombard L, Crous PW (2012) Phylogeny and taxonomy of the genus Gliocladiopsis. Persoonia 28:25–33
Lombard L, Van der Merwe NA, Groenewald JZ, Crous PW (2015) Generic concepts in Nectriaceae. Stud Mycol 80:189–245
Lombard L, Houbraken J, Decock C, Samson RA, Meijer M, Réblová M, Groenewald JZ, Crous PW (2016) Generic hyper-diversity in Stachybotriaceae. Persoonia 36(1):156–246
Lucking R, Pfister DH, Plata ER, Lumbsch HT (2009) Fungi evolved right on track. Mycologia 101:810–822
Lumbsch HT, Huhndorf SM (2007) Notes on ascomycete systematics. Nos. 4408–4750. Myconet 13:59–99
Lumbsch HT, Huhndorf SM (2010) Myconet volume 14. Part one. Outline of Ascomycota—2009. Part two. Notes on ascomycete systematics. Nos. 4751–5113. Fieldiana Life and Earth Sciences, 2010(1), 1–64
Luo J, Zhuang WY (2010a) Chaetopsinectria (Nectriaceae, Hypocreales), a new genus with Chaetopsina anamorphs. Mycologia 102(4):976–984
Luo J, Zhuang WY (2010b) Three new species of Neonectria (Nectriaceae, Hypocreales) with notes on their phylogenetic positions. Mycologia 102(1):142–152
Luo J, Zhuang WY (2012) Volutellonectria (Ascomycota, Fungi), a new genus with Volutella anamorphs. Phytotaxa 44:1–10
Luo ZL, Hyde KD, Liu JK, Bhat DJ, Bao DF, Li WL, Su HY (2018) Lignicolous freshwater fungi from China II: Novel Distoseptispora (Distoseptisporaceae) species from northwestern Yunnan Province and a suggested unified method for studying lignicolous freshwater fungi. Mycosphere 9:444–461
Luo ZL, Hyde KD, Liu JK, Maharachchikumbura SSN, Jeewon R, Bao DF, Bhat DJ, Lin CG, Li WL, Yang J, Liu NG, Lu YZ, Jayawardena RS, Li JF, Su HY (2019) Freshwater Sordariomycetes. Fungal Divers 99:451–660
Luttrell ES (1951) Taxonomy of the Pyrenomycetes. Uni Mo Stud 24:1–120
Lynch SC, Twizeyimana M, Mayorquin JS, Wang DH, Na F, Kayim M, Kasson MT, Thu PQ, Bateman C, Rugman-Jones P, Hulcr J, Stouthamer R, Eskalen A (2016) Identification, pathogenicity and abundance of Paracremonium pembeum sp. nov. and Graphium euwallaceae sp. nov.—two newly discovered mycangial associates of the polyphagous shot hole borer (Euwallacea sp.) in California. Mycologia 108(2):313–329
Maharachchikumbura SSN, Hyde KD, Jones EBG, McKenzie EHC, Huang SK, Abdel-Wahab MA, Daranagama DA, Dayarathne M, D’souza MJ, Goonasekara ID, Hongsanan S, Jayawardena RS, Kirk PM, Konta S, Liu JK, Liu ZY, Norphanphoun C, Pang KL, Perera RH, Senanayake IC, Shang QJ, Shenoy BD, Xiao YP, Bahkali AH, Kang JC, Somrothipol S, Suetrong S, Wen TC, Xu JC (2015) Towards a natural classification and backbone tree for Sordariomycetes. Fungal Divers 72:199–301
Maharachchikumbura SS, Hyde KD, Jones EG et al (2016) Families of Sordariomycetes. Fungal Divers 79:1–317
Maharachchikumbura SSN, Luo ZL, Su HY, Al-Sadi AM, Cheewangkoon R (2018) Reticulascaceae hyphomycetes from submerged wood in Yunnan, China. Phytotaxa 348(3):187–198
Maharachchikumbura SSN, Chen Y, Ariyawansa HA, Hyde KD, Haelewaters D, Perera RH, Samarakoon MC, Wanasinghe DW, Bustamante DE, Liu JK, Lawrence DP, Cheewangkoon R, Stadler M (2021) Integrative approaches for species delimitation in Ascomycota. Fungal Divers 109:155–179
Malloch D (1970) New concepts in the Microascaceae illustrated by two new species. Mycologia 62:727–740
Malosso E (1995) Ocorrência de Hyphomycetes (Fungi Imperfecti) e Fungos Zoospóricos em Ambientes Aquáticos (Rio do Monjolinho, São Carlos, SP). Universidade Federal de São Carlos, Brazil
Mamarabadi M, Jensen B, Jensen DF, L€ ubeck M (2008) Real-time RT-PCR expression analysis of chitinase and endoglucanase genes in the three-way interaction between the biocontrol strain Clonostachys rosea IK726, Botrytis cinerea and strawberry. FEMS Microbiol Lett 285:101–110
Mantiri FR, Samuels GJ, Rahe JE, Honda BM (2001) Phylogenetic relationship in Neonectria species having Cylindrocarpon anamorphs inferred from mitochondrial ribosomal DNA sequences. Canad J Bot 79:334–340
Mapook A, Hyde KD, McKenzie EHC, Gareth Jones EBG, Bhat DJ, Jeewon R, Stadler M, Samarakoon MC, Malaithong M, Tanunchai B, Buscot F, Wubet T, Purahong W (2020) Taxonomic and phylogenetic contributions to fungi associated with the invasive weed Chromolaena odorata (Siam weed). Fungal Divers 101:1–175
Marincowitz S, Duong TA, De Beer ZW, Wingfield MJ (2015) Cornuvesica: A little known mycophilic genus with a unique biology and unexpected new species. Fungal Biol 119(7):615–630
Marin-Felix Y, Groenewald JZ, Cai L, Chen Q, Marincowitz S, Barnes I, Bensch K, Braun U, Camporesi E, Damm U, de Beer ZW, Dissanayake A, Edwards J, Giraldo A, Hernández-Restrepo M, Hyde KD, Jayawardena RS, Lombard L, Luangsa-ard L, McTaggart AR, Rossman AY, Sandoval-Denis M, Shen M, Shivas RG, Tan YP, van der Linde EJ, Wingfield MJ, Wood AR, Zhang JQ, Zhang Y, Crous PW (2017) Genera of phytopathogenic fungi: GOPHY 1. Stud Mycol 86:99–216
Marsault E, Peterson ML (2017) Practical medicinal chemistry with Macrocycles: design, synthesis, and case studies. Wiley, Hoboken
Marvanová L, Laichmanová M (2014) Campylospora leptosoma sp. nov. and characteristics of Campylospora spp., based on morphology and on ITS sequences. Mycosphere 5(2):245–261
Mathur PN, Thirumalachar MJ (1960) A new Emericellopsis species with Stilbella-type of conidia. Mycologia 52:694–697
Mathur PN, Thirumalachar MJ (1962) Studies on some Indian soil fungi II. On some new or interesting Ascomycetes. Sydowia 16:1–6
Matsushima T (1975) Icones Microfungorum a Matsushima lectorum. Matsushima, Kobe, Japan
Matsushima T (1993) Matsushima mycological memoirs no. 7. Matsushima Fungus Collection, USA, p 75
Merli S (1992) Chaetopsina nimbae, a new species of dematiaceous Hyphomycetes. Mycotaxon 44:323–331
Miller MA, Pfeiffer W, Schwartz T (2010) Creating the CIPRES Science Gaeway for inference of large phylogenetic trees. In: Proceedings of the 2010 Gateway Computing Environments Workshop (GCE). New Orleans, Louisiana, pp 1–8.
Ming DQ, Luo LY, He XX, Wang MS, Fang WX, Chen SF, Chen WH, Han YF, Liang ZQ (2021) Paracremonium lepidopterorum, a new insect-associated fungus. Phytotaxa 524(2):85–91
Moreau F, Moreau M (1953) Etude du développement de quelques Aspergillacées. Rev Mycot 18:165–180
Moreira GM, Abreu LM, Carvalho VG, Schroers HJ, Pfenning LH (2016) Multi-locus phylogeny of Clonostachys subgenus Bionectria from Brazil and description of Clonostachys chloroleuca sp. nov. Mycol Prog 15:1031–1039
Morgan-Jones G (1979) Notes on Hyphomycetes. XXXI. Chaetopsina auburnensis sp.nov. Mycotaxon 8:411–416
Munk A (1957) Danish Pyrenomycetes, A preliminary flora. Dansk Botanisk Arkiv 17:1–491
Muvea AM, Meyhöfer R, Subramanian S, Poehling HM, Ekesi S, Maniania NK (2014) Colonization of onions by endophytic fungi and their impacts on the biology of Thrips tabaci. PLoS ONE 9:e108242
Nannfeldt JA (1932) Studien über die Morphologie und Systematik der nichtlichenisierten inoperculaten Discomyceten. Nova Acta Regiae Soc Sci Upsal Ser IV 8:1–368
Nawawi A (1974) A new Campylospora. Trans Br Mycol Soc 63:603–606
Nilsson S (1962) Some aquatic hyphomycetes from South America. Sven Bot Tidskr 56:351–361
Nonaka K, Kaneta T, Ōmura S, Masuma R (2015) Mariannaea macrochlamydospora, a new hyphomycete (Nectriaceae) from soil in the Bonin Islands. Japan Mycoscience 56(1):29–33
Nylander JAA (2004) MrModeltest v2. Program distributed by the author. Evolutionary Biology Centre, Uppsala University, Uppsala
O’Donnell K, Cigelnik E (1997) Two divergent intragenomic rDNA ITS2 types within a monophyletic lineage of the fungus Fusarium are nonorthologous. Mol Phylogenet Evol 7:103–116
O’Donnell K, Sutton DA, Rinaldi MG, Sarver BA, Balajee SA, Schroers HJ, Schroers RC, Schroers VARG, Crous PW, Crous N, Crous T, Jung K, Jung J, Lee YH, Kang S, Park B, Aoki T (2010) Internet-accessible DNA sequence database for identifying fusaria from human and animal infections. J Clin Microbiol 48:3708–3718
Ogawa H, Yoshimura A, Sugiyama J (1997) Polyphyletic origins of species of the anamorphic genus Geosmithia and the relationships of the cleistothecial genera: evidence from 18S, 5S and 28S rDNA sequence analyses. Mycologia 89:756–771
Ou JH, Lin GC, Chen CY (2020) Sarocladium species associated with rice in Taiwan. Mycol Prog 19(1):67–80
Pang KL (2002) Systematics of the Halosphaeriales: which morphological characters are important? In: Hyde KD (ed) Fungi in marine environments. Fungal Diversity Press, Hong Kong, pp 35–57
Pang KL, Alias SA, Chiang MW, Vrijmoed L, Jones EBG (2010) Sedecimiella taiwanensis gen. et sp. nov., a marine mangrove fungus in the Hypocreales (Hypocreomycetidae, Ascomycota). Bot Mar 53:493–498
Parguey-Leduc A, Locquin-Linard M (1976) L’ontogénie et la structures despérithèces de Faurelina fimigenes Locquin-Linard. Rev Mycol 40:161–175
Parkinson LE, Shivas RG, Dann EK (2017) Novel species of Gliocladiopsis (Nectriaceae, Hypocreales, Ascomycota) from avocado roots (Persea americana) in Australia. Mycoscience 58:95–102
Pem D, Jeewon R, Chethana KWT, Hongsanan S, Doilom M, Suwannarach N, Hyde KD (2021) Species concepts of Dothideomycetes: classification, phylogenetic inconsistencies and taxonomic standardization. Fungal Divers 109:283–319
Perera RH, Hyde KD, Maharachchikumbura SSN, Jones EBG, McKenzie EHC, Stadler M, Lee HB, Samarakoon MC, Ekanayaka AH, Camporesi E, Liu JK, Liu ZY (2020) Fungi on wild seeds and fruits. Mycosphere 11(1):2108–2480
Perera RH, Hyde KD, Jones EBG, Maharachchikumbura SSN, Bundhun D, Camporesi E, Akulov A, Liu JK, Liu ZY (2023) Profile of Bionectriaceae, Calcarisporiaceae, Hypocreaceae, Nectriaceae, Tilachlidiaceae, Ijuhyaceae fam. nov., Stromatonectriaceae fam. nov. and Xanthonectriaceae fam. nov. Fungal Divers 118:5–271
Petch T (1938) British Hypocreales. Trans Br Mycol Soc 21:243–301
Phukhamsakda C, Nilsson R, Bhunjun CS, de Farias ARG, Sun YR, Wijesinghe SN, Raza M, Bao DF, Lu L, Tibpromma S, Dong W, Tennakoon DS, Tian XG, Xiong YR, Karunarathna SC, Cai L, Luo ZL, Wang Y, Manawasinghe IS, Camporesi E, Kirk PM, Promputtha I, Kuo CS, Su HY, Doilom M, Li Y, Fu YP, Hyde KD (2022) The numbers of fungi: contributions from traditional taxonomic studies and challenges of metabarcoding. Fungal Divers 114:327–386
Prasher IB, Chauhan R (2017) Clonostachys indicus sp. nov. from India. Kavaka 48:22–26
Quandt CA, Kepler RM, Gams W, Araújo JPM, Ban S, Evans HC, Hughes D, Humber R, Hywel-Jones N, Li Z, Luangsa-ard JJ, Rehner SA, Sanjuan T, Sato H, Shrestha B, Sung GH, Yao YJ, Zare R, Spatafora JW (2014) Phylogenetic-based nomenclatural proposals for Ophiocordycipitaceae (Hypocreales) with new combinations in Tolypocladium. IMA Fungus 5:121–134
Rabenhorst L (1862) Cosmospora Coccinea Hedwigia 2:59
Raghukumar S (2017) Origin and evolution of marine fungi. Fungi in coastal and oceanic marine ecosystems. Springer, Cham, pp 307–321
Rambaut A (2014) FigTree 1.4.2. http://tree.bio.ed.ac.uk/software/figtr ee
Rambaut A, Suchard MA, Xie D, Drummond AJ (2013) Tracer version 1.6. http://tree.bio.ed.ac.uk/software/tracer
Rambelli A (1956) Chaetopsina nuovo genere di ifali demaziacei. Atti Accad Sci Ist Bologna 11:1–6
Rambelli A, Lunghini D (1976) Chaetopsina ivoriensis, a new species of dematiaceous Hyphomycetes. G Bot 110:253–258
Rannala B, Yang Z (1996) Probability distribution of molecular evolutionary trees: a new method of phylogenetic inference. J Mol Evol 43:304–311
Ranzoni FV (1953) The aquatic hyphomycetes of California. Farlowia 4:353–398
Ranzoni FV (1956) The perfect stage of Flagellospora penicillioides. Am J Bot 43:13–17
Ranzoni FV (1979) Aquatic hyphomycetes from Hawaii. Mycologia 71:786–795
Read SJ, Jones MST (1992) Attachment and germination of conidia. The ecology of aquatic hyphomycetes. Springer, Berlin, pp 135–151
Réblová M, Barr ME, Samuels GJ (1999) Chaetosphaeriaceae, a new family for Chaetosphaeria and its relatives. Sydowia 51:49–70
Réblová M, Gams W, Seifert KA (2011) Monilochaetes and allied genera of the Glomerellales, and a reconsideration of families in the Microascales. Stud Mycol 68:163–191
Redecker D, Kodner R, Graham LE (2000) Glomalean fungi from the Ordovician. Science 289:1920–1921
Rimington WR, Pressel S, Duckett JG, Bidartondo MI (2015) Fungal symbioses of basal vascular plants: reopening a closed book. New Phytol 205:1394–1398
Rivera-Varas VV, Freeman TA, Gudmestad NC, Secor GA (2007) Mycoparasitism of Helminthosporium solani by Acremonium strictum. Phytopathology 97:1331–1337
Rong IH, Gams W (2000) The hyphomycete genera Exochalara and Monilochaetes. Mycotaxon 76:451–462
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542
Rosique-Gil E, Gonzalez MC, Cifuentes J (2008) New records of three freshwater ascomycetes from an urban lagoon of Tabasco. Mexico Mycotaxon 105:249
Rossman AY (2000) Towards monophyletic genera in the holomorphic Hypocreales. Stud Mycol 45:27–34
Rossman AY (2014) Lessons learned from moving to one scientific name for fungi. IMA Fungus 5:81–89
Rossman AY, Samuels GJ, Rogerson CT, Lowen R (1999) Genera of Bionectriaceae, Hypocreaceae and Nectriaceae (Hypocreales, Ascomycetes). Stud Mycol 42:1–260
Rossman AY, McKemy JM, Pardo-Schultheiss RA, Schroers HJ (2001) Molecular studies of the Bionectriaceae using large subunit rDNA sequences. Mycologia 93:100–110
Rossman AY, Seifert KA, Samuels GJ, Minnis AM, Schroers HJ, Lombard L, Crous PW, Põldmaa K, Cannon PF, Summerbell RC, Geiser DM, Zhuang WY, Hirooka Y, Herrera C, Salgado-Salazar C, Chaverri P (2013) Genera in Bionectriaceae, Hypocreaceae, and Nectriaceae (Hypocreales) proposed for acceptance or rejection. IMA Fungus 4:41–51
Rossman AY, Allen WC, Braun U, Castlebury LA, Chaverri P, Crous PW, Hawksworth DL, Hyde KD, Johnston P, Lombard L, Romberg M, Samson RA, Seifert KA, Stone JK, Udayanga D, White JF (2016) Overlooked competing asexual and sexually typified generic names of Ascomycota with recommendations for their use or protection. IMA Fungus 7(2):289–308
Ruisi S, Barreca D, Selbmann L, Zucconi L, Onofri S (2007) Fungi in Antarctica. Rev Environ Sci Biotechnol 6:127–141
Sakayaroj J, Pang KL, Phongpaichit S, Jones EBG (2005a) A phylogenetic study of the genus Haligena (Halosphaeriales, Ascomycota). Mycologia 97:804–811
Sakayaroj J, Phongpaichit S, Jones EBG (2005b) Viability and biodiversity of freshwater hyphomycetes in foam at Ton Nga Chang Wildlife-Sanctuary, Songkhla, southern Thailand. Fungal Divers 18:135–145
Sakayaroj J, Pang KL, Jones EBG (2011) Multi-gene phylogeny of the Halosphaeriaceae: its ordinal status, relationships between genera and morphological character evolution. Fungal Divers 46(1):87–109
Saksena SB (1954) A new genus of Moniliaceae. Mycologia 46:660–666
Salgado-Salazar C, Rossman AY, Samuels GJ, Capdet M, Chaverri P (2012) Multigene phylogenetic analyses of the Thelonectria coronata and T. veuillotiana species complexes. Mycologia 104:1325–1350
Salgado-Salazar C, Rossman AY, Samuels GJ, Hirooka Y, Sanchez RM, Chaverri P (2015) Phylogeny and taxonomic revision of Thelonectria discophora (Ascomycota, Hypocreales, Nectriaceae) species complex. Fungal Divers 70:1–29
Salgado-Salazar C, Rossman AY, Chaverri P (2016) The genus Thelonectria (Nectriaceae, Hypocreales, Ascomycota) and closely related species with cylindrocarpon-like asexual states. Fungal Divers 80:411–455
Samarakoon MC, Hyde KD, Promputtha I, Hongsanan S, Ariyawansa HA, Maharachchikumbura SSN, Daranagama DA, Stadler M, Mapook A (2016) Evolution of Xylariomycetidae (Ascomycota: Sordariomycetes). Mycosphere 7:1746–1761
Samarakoon BC, Wanasinghe DN, Phookamsak R, Bhat DJ, Chomnunti P, Karunarathna SC, Lumyong S (2021) Stachybotrys musae sp. nov., S. microsporus, and Memnoniella levispora (Stachybotryaceae, Hypocreales) Found on Bananas in China and Thailand. Life 11(4):323
Samarakoon MC, Hyde KD, Maharachchikumbura SSM, Stadler M, Jones EBG, Promputtha I, Suwannarach N, Camporesi E, Bulgakov TS, Liu JK (2022) Taxonomy, phylogeny, molecular dating and ancestral state reconstruction of Xylariomycetidae (Sordariomycetes). Fungal Divers 112(1):1–88
Samson RA (1974) Paecilomyces and some allied Hyphomycetes. Stud Mycol 6:1–119
Samson RA, Bigg WL (1988) A new species of Mariannaea from California. Mycologia 80:131–134
Samuels GJ (1985) Four new species of Nectria and their Chaetopsina anamorphs. Mycotaxon 22:13–32
Samuels GJ (1989) Nectria and Penicillifer. Mycologia 81(3):347–355
Samuels GJ, Brayford D (1994) Species of Nectria (sensu lato) with red perithecia and striate ascospores. Sydowia 46:75–161
Samuels GJ, Seifert KA (1991) Two new species of Nectria with Stilbella and Mariannaea anamorphs. Sydowia 43:249–263
Samuels GJ, Candoussau F, Magni JF (1997) Fungicolous pyrenomycetes 2. Ascocodinaea gen. nov., and reconsideration of Litschaueria. Mycologia 89:156–162
Sandoval-Denis M, Sutton DA, Fothergill AW, Cano-Lira J, Gené J, Decock CA, de Hoog GS, Guarro J (2013) Scopulariopsis, a poorly known opportunistic fungus: spectrum of species in clinical samples and in vitro responses to antifungal drugs. J Clin Microbiol 51:3937–3943
Sandoval-Denis M, Gené J, Sutton DA, Cano-Lira JF, de Hoog GS, Decock CA, Wiederhold NP, Guarro J (2016a) Redefining Microascus, Scopulariopsis and allied genera. Persoonia 36:1–36
Sandoval-Denis M, Gené J, Sutton DA, Wiederhold NP, Cano-Lira JF, Guarro J (2016b) New species of Cladosporium associated with human and animal infections. Persoonia 36:281
Sandoval-Denis M, Lombard L, Crous PW (2019) Back to the roots: a reappraisal of Neocosmospora. Persoonia 43:90–185
Sati SC, Belwal M (2005) Aquatic hyphomycetes as endophytes of riparian plant roots. Mycologia 97(1):45–49
Schoch CL, Crous PW, Wingfield MJ, Wingfield BD (2000) Phylogeny of Calonectria and selected hypocrealean genera with cylindrical macroconidia. Stud Mycol 45:45–62
Schoenlein-Crusius IH (2002) Aquatic hyphomycetes from cerrado regions in the state of São Paulo, Brazil. Mycotaxon 81:457–462
Schoenlein-Crusius IH, Milanez AI (1990) Hyphomycetes aquáticos no Estado de São Paulo, Brasil. Rev Bras Bot 13:61–68
Schroers HJ (2001) A monograph of Bionectria (Ascomycota, Hypocreales, Bionectriaceae) and its Clonostachys anamorphs. Stud Mycol 46:1–214
Schroers HJ, Samuels GJ, Seifert KA, Gams W (1999) Classification of the mycoparasite Gliocladium roseum in Clonostachys as C. rosea, its relationship to Bionectria ochroleuca, and notes on other Gliocladium-like fungi. Mycologia 91:365–385
Schroers HJ, Gräfenhan T, Nirenberg HI, Seifert KA (2011) A revision of Cyanonectria and Geejayessia gen. nov., and related species with Fusarium-like anamorphs. Stud Mycol 68:115–138
Seifert KA (1985) Notes on some apocryphal genera of synnematal Hyphomycetes. Trans Br Mycol Soc 85:123–133
Seifert KA, Gams W (2011) The genera of hyphomycetes—2011 update. Persoonia 27:119–129
Seifert KA, Stanley SJ, Hyde KD (1995) Stilbella holubovae, a new synnematous hyphomycete species on driftwood from the Philippines and South Africa. Sydowia 47:258–262
Seifert KA, McMullen CR, Yee D, Reeleder RD, Dobinson KF (2003) Molecular differentiation and detection of ginseng-adapted isolates of the root rot fungus Cylindrocarpon destructans. Phytopathology 93:1533–1542
Selosse MA, Le Tacon F (1998) The land flora: a phototroph-fungus partnership? Trends Ecol Evol 13:15–20
Selosse MA, Vohník M, Chauvet E (2008) Out of the rivers: are some aquatic hyphomycetes plant endophytes? New Phytol 178:3–7
Senanayake IC, Rathnayaka AR, Marasinghe DS, Calabon MS, Gentekaki E, Lee HB, Hurdeal VG, Pem D, Dissanayake LS, Wijesinghe SN, Bundhun D, Nguyen TT, Goonasekara ID, Abeywickrama PD, Bhunjun CS, Jayawardena RS, Wanasinghe DN, Jeewon R, Bhat DJ, Xiang MM (2020) Morphological approaches in studying fungi: collection, examination, isolation, sporulation and preservation. Mycosphere 11:2678–2754
Shearer CA (1993) The freshwater ascomycetes. Nova Hedwigia 56:1–33
Shearer CA, Crane JL (1980) Aquatic ascomycetes with unfurling appendages. Bot Mar 23:607–615
Shearer CA, Raja HA (2013) Freshwater ascomycetes database: http://fungi.life.illinois.edu/. Accessed 11 May 2013
Shearer CA, Webster J (1991) Aquatic hyphomycete communities in the River Teign. IV Twig Colonization Mycol Res 95(4):413–420
Shearer CA, Raja HA, Miller AN, Nelson P, Tanaka K, Hirayama K, Marvanová L, Hyde KD (2009) The molecular phylogeny of freshwater Dothideomycetes. Stud Mycol 64:145–153
Sivichai S, Jones EBG, Hywel-Jones N (2002) Fungal colonisation of wood in a freshwater stream at Tad Ta Phu, Khao Yai National Park, Thailand. Fungal Divers 10:113–129
Smith EF (1899) Wilt disease of cotton, watermelon and cowpea (Neocosmospora nov. gen.). US Dep Agric Div Veg Physiol Pathol Bull 17:1–54
Smits G, Fernández R, Cressa C (2007) Preliminary study of aquatic Hyphomycetes from Venezuelan streams. Acta Bot Venez 30:345–355
Spatafora JW, Volkmann-Kohlmeyer B, Kohlmeyer J (1998) Independent terrestrial origins of the Halosphaeriales (marine Ascomycota). Am J Bot 85:569–1580
Sridhar KR, Kaveriappa KM (1992) Aquatic hyphomycetes of Western Ghat streams, India. Sydowia 44:66–77
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690
Stamatakis A, Hoover P, Rougemont J (2008) A rapid bootstrap algorithm for the RAxML web servers. Syst Biol 57:758–771
Stolk AC (1955) Emericellopsis minima sp. nov. and Westerdykella ornata gen. nov. sp. nov. Trans Br Mycol Soc 38:419–424
Strullu-Derrien C, Kenrick P, Pressel S, Duckett JG, Rioult JP, Strullu DG (2014) Fungal associations in Horneophyton ligneri from the Rhynie Chert (c. 407 million year old) closely resemble those in extant lower land plants: novel insights into ancestral plant–fungus symbioses. New Phytol 203:964–979
Sukapure RA, Thirumalachar MJ (1965) Studies on Cephalosporium species from India—III. Sydowia 19:171–175
Sukapure RS, Thirumalachar MJ (1966) Conspectus of species of Cephalosporium with particular reference to Indian species. Mycologia 58:351–361
Summerbell RC, Gueidan C, Schroers HJ, de Hoog GS, Starink M, Rosete YA, Guarro J, Scott JA (2011) Acremonium phylogenetic overview and revision of Gliomastix, Sarocladium, and Trichothecium. SIM 68:139–162
Sun ZB, Sun MH, Li SD (2015a) Draft genome sequence of mycoparasite Clonostachys rosea strain 67–1. Genome Announc 3:e00546-e615
Sun ZB, Sun MH, Li SD (2015b) Identification of mycoparasitism-related genes in Clonostachys rosea 67–1 active against Sclerotinia sclerotiorum. Sci Rep 5:18169
Sung GH, Hywel-Jones NL, Sung JM, Luangsa-Ard JJ, Shrestha B, Spatafora JW (2007) Phylogenetic classification of Cordyceps and the clavicipitaceous fungi. Stud Mycol 57:5–59
Sung GH, Poinar GO, Spatafora JW (2008) The oldest fossil evidence of animal parasitism by fungi supports a Cretaceous diversification of fungal-arthropod symbioses. Mol Phylogenet Evol 49:495–502
Sutton BC, Hodges CSJR (1976) Eucalyptus microfungi: some setose Hyphomycetes with phialides. Nova Hedwig 27:343–352
Swofford DL (2002) PAUP*: phylogenetic analysis using parsimony (*and other methods), version 4. Sinauer Associates, Sunderland
Tan YP, Shivas R (2022) 1Index of Australian Fungino. 1 (ISBN 978-0-6455911-0-1)
Tang AMC, Jeewon R, Hyde KD (2007) Phylogenetic utility of protein (RPB2, B-tubulin) and ribosomal (LSU, SSU) gene sequences in the systematics of Sordariomycetes (Ascomycota, Fungi). Antonie Leeuwenhoek 91(4):327–349
Taylor TN, Osborn JM (1996) The importance of fungi in shaping the paleoecosystem. Rev Palaeobot Palynol 90:249–262
Tennakoon DS, Kuo CH, Maharachchikumbura SSN, Thambugala KM, Gentekaki E, Phillips AJL, Bhat DJ, Wanasinghe DN, de Silva NI, Promputtha I, Hyde KD (2021) Taxonomic and phylogenetic contributions to Celtis formosana, Ficus ampelas, F. septica, Macaranga tanarius and Morus australis leaf litter inhabiting microfungi. Fungal Divers 108:1–215
Thiyagaraja V, Lücking R, Ertz D, Wanasinghe DN, Karunarathna SC, Camporesi E, Hyde KD (2020) Evolution of non-lichenized, saprotrophic species of Arthonia (Ascomycota, Arthoniales) and resurrection of Naevia, with notes on Mycoporum. Fungal Divers 102(1):205–224
Thiyagaraja V, Lücking R, Ertz D, Karunarathna SC, Wanasinghe DN, Lumyong S, Hyde KD (2021) The evolution of life modes in Stictidaceae, with three novel taxa. J Fungi 7(2):105
Tibpromma S, Hyde KD, McKenzie EH, Bhat DJ, Phillips AJ, Wanasinghe DN, Samarakoon MC, Jayawardena RS, Dissanayake AJ, Tennakoon DS, Doilom M, Phookamsak R, Tang AMC, Xu J, Mortimer PE, Promputtha I, Maharachchikumbura SSN, Khan S, Karunarathna SC (2018) Fungal diversity notes 840–928: microfungi associated with Pandanaceae. Fungal Divers 93:1–60
Toledo AV, Virla E, Humber RA, Paradell SL, Lastra CCL (2006) First record of Clonostachys rosea (Ascomycota: Hypocreales) as an entomopathogenic fungus of Oncometopia tucumana and Sonesimia grossa (Hemiptera: Cicadellidae) in Argentina. J Invertebr Pathol 92:7–10
Tubaki K, Watanabe K, Manoch L (1983) Aquatic hyphomycetes from Thailand. Trans Mycol Soc Japan 24:451–457
Van Beyma FH (1940) Beschreibung einiger neuer pilzarten aus dem Centraalbureau voor Schimmelcultures, Baarn (Nederland). Antonie Leeuwenhoek 6(1):263–290
Verdejo-Lucas S, Ornat C, Sorribas FJ, Stchiegel A (2002) Species of root-knot Nematodes and fungal egg parasites recovered from vegetables in almería and barcelona, Spain. J Nematol 34:405–408
Viccini G, Martinelli TR, Cognialli RC, de Faria RO, Carbonero ER, Sassaki GL, Mitchell DA (2009) Exopolysaccharide from surface-liquid culture of Clonostachys rosea originates from autolysis of the biomass. Arch Microbiol 191:369–378
Vijaykrishna D, Jeewon R, Hyde KD (2006) Molecular taxonomy, origins and evolution of freshwater ascomycetes. Fungal Divers 23:351–390
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol Res 172:4238–4246
Viljoen CD, Wingfield MJ, Jacobs K, Wingfield BD (2000) Cornuvesica, a new genus to accommodate Ceratocystiopsis falcata. Mycol Res 104(3):365–367
Voigt K, James TY, Kirk PM, Santiago ALDA, Waldman B, Griffith GW, Fu M, Radek R, Strassert JFH, Wurzbacher C, Jerônimo GH, Simmons DR, Seto K, Gentekaki E, Hurdeal VC, Hyde KD, Nguyen TTT, Lee BH (2021) Early-diverging fungal phyla: taxonomy, species concept, ecology, distribution, anthropogenic impact, and novel phylogenetic proposals. Fungal Divers 109:59–98
von Arx JA (1978) Notes on Microascaceae with the description of two species. Persoonia 10:23–31
Von Höhnel FV (1924) Studien über Hyphomyzeten. Zentralblatt Für Bakteriologie Und Parasitenkunde Abteilung 2(60):1–26
Vu D, Groenewald M, De Vries M, Gehrmann T, Stielow B, Eberhardt U, Al-Hatmi A, Groenewald JZ, Cardinali G, Houbraken J, Boekhout T, Crous PW, Robert V, Verkley GJM (2019) Large-scale generation and analysis of filamentous fungal DNA barcodes boosts coverage for kingdom fungi and reveals thresholds for fungal species and higher taxon delimitation. Stud Mycol 92:135–154
Wanasinghe DN, Jeewon R, Jones EBG, Boonmee S, Kaewchai S, Manawasinghe IS, Lumyong S, Hyde KD (2018) Novel palmicolous taxa within Pleosporales: multigene phylogeny and taxonomic circumscription. Mycol Prog 17:571–590
Wang Y, Hyde KD, McKenzie EHC, Jiang YL, Li W, Zhao DG (2015) Overview of Stachybotrys (Memnoniella) and current species status. Fungal Divers 70:17–83
Watanabe K, Hirose D (2021) A novel Mariannaea species isolated from decayed pine needles in Japan. Phytotaxa 522(3):211–220
Webster J (1987) Convergent evolution and the functional significance of spore shape in aquatic and semi-aquatic fungi. In: Rayner ADM, Brasier CM, Moore D (eds) Evolutionary biology of the fungi. Cambridge University Press, Cambridge, pp 191–201
Webster J (1992) Anamorph-teleomorph relationships. The ecology of aquatic hyphomycetes. Springer, Berlin, pp 99–117
Webster J, Davey RA (1984) Sigmoid conidial shape in aquatic fungi. Trans Br Mycol Soc 83:43–52
Webster J, Descals E (1979) The teleomorphs of waterborne Hyphomycetes from fresh water. The whole fungus, vol 2. National Museums of Canada, Ottawa, pp 419–447
Webster J, Marvanová L, Eicker A (1994) Spores from foam from South African Rivers. Nova Hedwig 57:379–398
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal rna genes for phylogenetics. PCR protocols. Elsevier, New York, pp 315–322
Whitton SR, McKenzie EHC, Hyde KD (2001) Microfungi on the pandanaceae: stachybotrys, with three new species. N Z J Bot 39(3):489–499
Whitton SR, McKenzie EHC, Hyde KD (2012) Fungi associated with pandanaceae. Fungal Divers 21:1–457
Wijayawardene DNN, McKenzie EHC, Hyde KD (2012) Towards incorporating anamorphic fungi in a natural classification—checklist and notes for 2011. Mycosphere 3:157–228
Wijayawardene NN, Hyde KD, Rajeshkumar KC, Hawksworth DL, Madrid H, Kirk PM, Braun U, Singh RV, Crous PW, Kukwa M, Lücking R, Kurtzman CP, Yurkov A, Haelewaters D, Apt- root A, Lumbsch HT, Timdal E, Ertz D, Etayo J, Phillips AJL, Groenewald JZ, Papizadeh M, Selbmann L, Dayarathne MC, Weerakoon G, Jones EBG, Suetrong S, Tian Q, Castañeda-Ruiz RF, Bahkali AH, Pang K-L, Tanaka K, Dai DQ, Sakayaroj J, Hujslová M, Lombard L, Shenoy BD, Suija A, Maharachchikumbura SSN, Thambugala KM, Wanasinghe DN, Sharma BO, Gaikwad S, Pandit G, Zucconi L, Onofri S, Egidi E, Raja HA, Kodsueb R, Cáceres MES, Pérez-Ortega S, Fiuza PO, Monteiro JS, Vasilyeva LN, Shivas RG, Prieto M, Wedin M, Olariaga I, Lateef AA, Agrawal Y, Fazeli SAS, Amoozegar MA, Zhao GZ, Pfliegler WP, Sharma G, Oset M, Abdel-Wahab MA, Takamatsu S, Bensch K, de Silva NI, De Kese A, Karunarathna A, Boonmee S, Pfister DH, Lu YZ, Luo ZL, Boonyuen N, Daranagama DA, Senanayake IC, Jayasiri SC, Samarakoon MC, Zeng XY, Doilom M, Quijada L, Rampadarath S, Heredia G, Dissanayake AJ, Jayawardana RS, Perera RH, Tang LZ, Phukhamsakda C, Hernández-Restrepo M, Ma X, Tibpromma S, Gusmao LFP, Weerahewa D, Karunarathna SC (2017) Notes for genera Ascomycota. Fungal Divers 86:1–594
Wijayawardene NN, Hyde KD, Lumbsch HT, Liu JK, Maharachchikumbura SS, Ekanayaka AH, Tian Q, Phookamsak R (2018) Outline of Ascomycota: 2017. Fungal Divers 88:167–263
Wijayawardene NN, Hyde KD, Al‑Ani LKT, Tedersoo L, Haelewaters D, Rajeshkumar KC, Zhao RL, Aptroot A, Leontyev DV, Saxena RK, Tokarev YS, Dai DQ, Letcher PM, Stephenson SL, Ertz D, Lumbsch HT, Kukwa M, Issi IV, Madrid H, Phillips AJL, Selbmann L, Pfliegler WP, Horváth E, Bensch K, Kirk PM, Kolaříková K, Raja HA, Radek R, Papp V, Dima B, Ma J, Malosso E, Takamatsu S, Rambold G, Gannibal PB, Triebel D, Gautam AK, Avasthi S, Suetrong S, Timdal E, Fryar SC, Delgado G, Réblová M, Doilom M, Dolatabadi S, Pawłowska J, Humber RA, Kodsueb R, Sánchez‑Castro I, Goto BT, Silva DKA, de Souza FA, Oehl F, da Silva GA, Silva IR, Błaszkowski J, Jobim K, Maia LC, Barbosa FR, Fiuza PO, Divakar PK, Shenoy BD, Castañeda‑Ruiz RF, Somrithipol S, Lateef AA, Karunarathna SC, Tibpromma S, Mortimer PE, Wanasinghe DN, Phookamsak R, Xu J, Wang Y, Tian F, Alvarado P, Li DW, Kušan I, Matočec N, Maharachchikumbura SSN, Papizadeh M, Heredia G, Wartchow F, Bakhshi M, Boehm E, Youssef N, Hustad VP, Lawrey JD, Santiago ALCMA, Bezerra JDP, Souza‑Motta CM, Firmino AL, Tian Q, Houbraken J, Hongsanan S, Tanaka K, Dissanayake AJ, Monteiro JS, Grossart HP, Suija A, Weerakoon G, Etayo J, Tsurykau A, Vázquez V, Mungai P, Damm U, Li QR, Zhang H, Boonmee S, Lu YZ, Becerra AG, Kendrick B, Brearley FQ, Motiejūnaitė J, Sharma B, Khare R, Gaikwad S, Wijesundara DSA, Tang LZ, He MQ, Flakus A, Rodriguez‑Flakus P, Zhurbenko MP, McKenzie EHC, Stadler M, Bhat DJ, Liu JK, Raza M, Jeewon R, Nassonova ES, Prieto M, Jayalal RGU, Erdoğdu M, Yurkov A, Schnittler M, Shchepin ON, Novozhilov YK, SilvaFilho AGS, Liu P, Cavender JC, Kang Y, Mohammad S, Zhang LF, Xu RF, Li YM, Dayarathne MC, Ekanayaka AH, Wen TC, Deng CY, Pereira OL, Navathe S, Hawksworth DL, Fan XL, Dissanayake LS, Kuhnert E, Grossart HP, Thines M (2020) Outline of Fungi and fungus-like taxa. Mycosphere 11:1060–1456
Wijayawardene NN, Hyde KD, Dai DQ, Sánchez-García M, Goto BT, Saxena RK, Erdoğdu M, Selçuk F, Rajeshkumar KC, Aptroot A, Błaszkowski J, Boonyuen N, da Silva GA, de Souza FA, Dong W, Ertz D, Haelewaters D, Jones EBG, Karunarathna SC, Kirk PM, Kukwa M, Kumla J, Leontyev DV, Lumbsch HT, Maharachchikumbura SSN, Marguno F, Martínez-Rodríguez P, Mešić A, Monteiro JS, Oehl F, Pawłowska J, Pem D, Pfliegler WP, Phillips AJL, Pošta A, He MQ, Li JX, Raza M, Sruthi OP, Suetrong S, Suwannarach N, Tedersoo L, Thiyagaraja V, Tibpromma S, Tkalčec Z, Tokarev YS, Wanasinghe DN, Wijesundara DSA, Wimalaseana SDMK, Madrid H, Zhang GQ, Gao Y, Sánchez-Castro I, Tang LZ, Stadler M, Yurkov A, Thines M (2022) Outline of Fungi and fungus-like taxa—2021. Mycosphere 13(1):53–453
Wingfield MJ (1987) A new name for Phialocephala illini. Mycotaxon 30:149–156
Wollenweber HW, Reinking OA (1935) Die Fusarien: ihre Beschreibung. Schadwirkung und Bekämpfung, Berlin, pp 1–355
Wong SW, Hyde KD, Jones EBG (1998) Halosarpheia heteroguttulata sp. nov. from submerged wood in streams. Can J Bot 76:1857–1862
Wright EF, Cain RF (1961) New species of the genus Ceratocystis. Can J Bot 39:1215–1230
Xiao YP, Wang YB, Hyde KD, Eleni G, Sun JZ, Yang Y, Meng J, Yu H, Wen TC (2023) Polycephalomycetaceae, a new family of clavicipitoid fungi segregates from Ophiocordycipitaceae. Fungal Divers 1–76
Yang J, Maharachchikumbura SSN, Bhat DJ, Hyde KD, McKenzie EHC, Jones EBG, Al-Sadi AM, Lumyong S (2016) Fuscosporellales, a new order of aquatic and terrestrial Hypocreomycetidae (Sordariomycetes). Cryptogam Mycol 37:449–475
Yang H, Wang GN, Zhang H (2021) Mariannaea submersa sp. nov., with a new habitat and geographic record of Mariannaea catenulata. Mycosystema 40(6):1286–1298
Yeh YH, Kirschner R (2014) Sarocladium spinificis, a new endophytic species from the coastal grass Spinifex littoreus in Taiwan. Bot Stud 55:25
Yu Y, Harris AJ, Blair C, He X (2015) RASP (Reconstruct Ancestral State in Phylogenies): a tool for historical biogeography. Mol Phyl Evol 87:46–49
Yu Y, Blair C, He X (2019) RASP 4: ancestral state reconstruction tool for multiple genes and characters. Mol Biol Evol 37:604–606
Zeng ZQ, Zhuang WY (2013) Four new taxa of Ilyonectria and Thelonectria (Nectriaceae) revealed by morphology and combined ITS and β-tubulin sequence data. Phytotaxa 85:15–25
Zeng ZQ, Zhuang WY (2014) A new holomorphic species of Mariannaea and epitypification of M. samuelsii. Mycol Prog 13(4):967–973
Zeng ZQ, Zhuang WY (2016) A new species of Cosmospora and the first record of sexual state of C. lavitskiae. Mycol Prog 15(6):1–7
Zeng ZQ, Zhuang WY (2019) The genera Rugonectria and Thelonectria (Hypocreales, Nectriaceae) in China. MycoKeys 55:101–120
Zeng ZQ, Zhuang WY (2022) Three new species of Clonostachys (Hypocreales, Ascomycota) from China. J Fungi 8(10):1027
Zhai N, Sun Z, Zhang Y, Zang R, Xu C, Geng Y, Zhang M (2019) Gliocladiopsis wuhanensis sp. nov. from China. Mycotaxon 134(2):313–319
Zhang N, Castlebury LA, Miller AN, Huhndorf SM, Schoch CL, Seifert KA, Rossman AY, Rogers JD, Kohlmeyer J, Volkmann-Kohlmeyer B, Sung GH (2006) An overview of the systematics of the Sordariomycetes based on a four-gene phylogeny. Mycologia 98(6):1076–1087
Zhang L, Yang J, Niu Q, Zhao X, Ye F, Liang L, Zhang KQ (2008) Investigation on the infection mechanism of the fungus Clonostachys rosea against nematodes using the green fluorescent protein. Appl Microbiol Biotechnol 78:983–990
Zhang ZF, Liu F, Zhou X, Liu XZ, Liu SJ, Cai L (2017) Culturable mycobiota from Karst caves in China, with descriptions of 20 new species. Persoonia 39(1):1–31
Zhang ZF, Zhou SY, Eurwilaichitr L, Ingsriswang S, Raza M, Chen Q, Zhao P, Liu F, Cai L (2021a) Culturable mycobiota from Karst caves in China II, with descriptions of 33 new species. Fungal Divers 106(1):29–136
Zhang ZY, Shao QY, Li X, Chen WH, Liang JD, Han YF, Huang JZ, Liang ZQ (2021b) Culturable fungi from urban soils in China I: description of 10 new taxa. Microbiol Spectr 37(4):449–475
Zhdanova NN (1966) Rare and new species of Dematiaceae isolated from maize rhizosphere of various climatic belts of the Ukrainian SSR. Mikrobiologicheskii Zhurnal, Kiev 28:36–40
Zhou SX, Qiao LJ, Kang JC, Hyde KD, Ma XY (2017) A new species of Monilochaetes from Nothapodytes pittosporoides. Phytotaxa 326:129–136
Zuccaro A, Summerbell RC, Gams W, Schroers HJ, Mitchell JI (2004) A new Acremonium species associated with Fucus spp., and its affinity with a phylogenetically distinct marine Emericellopsis clade. Stud Mycol 50:283–297
Zucconi L, Rambelli A (1993) A new species of Chaetopsina from tropical forest litter. Mycotaxon 48:5–12
Acknowledgements
We would like to thank the National Natural Science Foundation of China (Project ID: 32060005 and 31970021) for financial support. This study was also supported by the Yunnan Fundamental Research Project (Grant Nos. 202101AU070137, 202201AW070001) and Thailand research fund “Macrofungi diversity research from the Lancang-Mekong Watershed and Surrounding areas (Grant No. DBG6280009)”. Kevin D. Hyde thanks the National Research Council of Thailand (NRCT) grant “Total fungal diversity in a given forest area with implications towards species numbers, chemical diversity and biotechnology” (Grant No. N42A650547). We would like to thank Shaun Pennycook for checking the nomenclature. Dan-Feng Bao thanks Zheng-Quan Zhang, Long-Li Li, Si-Ping Huang, Xi Fu, Rui Gu, Liang Zhang and Jun He for their help with the sample collection, DNA extraction and PCR amplification. Thanks to Rong-Ju Xu and De-Ping Wei for their help with the specimen and culture preservation. Dhanushka Wanasinghe thanks CAS President’s International Fellowship Initiative (PIFI) for funding his postdoctoral research (Number 2021FYB0005), the National Science Foundation of China (NSFC) under the Project Code 32150410362, Yunnan Intelligence Union Program for Young Scientists (Number WGXZ2022086L) and the Postdoctoral Fund from Human Resources and Social Security Bureau of Yunnan Province.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Handling Editor: Nalin Wijayawardene.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bao, DF., Hyde, K.D., Maharachchikumbura, S.S.N. et al. Taxonomy, phylogeny and evolution of freshwater Hypocreomycetidae (Sordariomycetes). Fungal Diversity 121, 1–94 (2023). https://doi.org/10.1007/s13225-023-00521-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13225-023-00521-8