Abstract
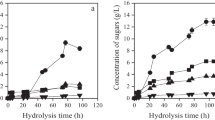
Utilized and waste jasmine flower contains a high portion of organic carbohydrate and other organic acids, making it a suitable substrate for bioethanol production. This study was designed to estimate the prospective of waste jasmine flower biomass applied with chemical (alkaline) and thermal pretreatment applied on samples through bioethanol production efficiencies. Therefore, pretreatment and enzymatic hydrolysis are directed to disrupt the complex cell wall layer and improve the accessibility towards polysaccharide fraction. Also, applying response surface methodology tools during fermentative bioethanol production to study the interactive effects of different bioprocess variables for higher bioethanol yield in batch small and large scale model is discussed. The immobilized yeast between jasmine found that jasmine sugar utilization was 50%. The jasmine flower's ethanol production was 6.54 g/L and after distillation of jasmine was 31.40 g/L at pH 4.5. Results showed that this immobilized yeast method could be successfully used for bioethanol production from waste jasmine flower.










Similar content being viewed by others
References
Azhar SHM, Abdulla R, Jambo SA, Marbawi H, Gansau JA, Faik AA, Rodrigues KF (2017) Yeasts in sustainable bioethanol production: a review. Biochem Biophys Rep 1(10):52–61. https://doi.org/10.1016/j.bbrep.2017.03.003
Balat M (2011) Production of bioethanol from lignocellulosic materials via the biochemical pathway: a review. Energy Convers Manag 52:858–875. https://doi.org/10.1016/j.enconman.2010.08.013
Behera S, Mohanty RC, Ray RC (2011) Ethanol production from mahula (Madhuca latifolia L.) flowers with immobilized cells of Saccharomyces cerevisiae in Luffa cylindrica L. sponge discs. Appl Energy 88(1):212–215. https://doi.org/10.1016/j.apenergy.2010.07.035
Chang YH, Chang KS, Chen CY, Hsu CL, Chang TC, Jang HD (2018) Enhancement of the efficiency of bioethanol production by Saccharomyces cerevisiae via gradually batch-wise and fed-batch increasing the glucose concentration. Fermentation 4(2):45. https://doi.org/10.3390/fermentation4020045
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28(3):350–356
Hu G, Heitmann JA, Rojas OJ (2008) Feedstock pretreatment strategies for producing ethanol from wood, bark and forest residues. BioResources 3:270–294
Mejica GFC, Unpaprom Y, Whangchai K, Ramaraj R (2021) Cellulosic-derived bioethanol from Limnocharis flava utilizing alkaline pretreatment. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-020-01218-7
Miller G (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428
Nguyen TVT, Unpaprom Y, Tandee K, Whangchai K, Ramaraj R (2020a) Physical pretreatment and algal enzyme hydrolysis of dried low-grade and waste longan fruits to enhance its fermentable sugar production. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-020-01176-0
Nguyen TV, Unpaprom Y, Manmai N, Whangchai K, Ramaraj R (2020b) Impact and significance of pretreatment on the fermentable sugar production from low-grade longan fruit wastes for bioethanol production. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-020-00977-7
Nong HT, Whangchai K, Unpaprom Y, Thararux C, Ramaraj R (2020a) Development of sustainable approaches for converting the agroweeds Ludwigia hyssopifolia to biogas production. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-020-01083-47
Nong HTT, Unpaprom Y, Whangchai K, Buochareon S, Ramaraj R (2020b) Assessment of the effects of anaerobic co-digestion of water primrose and cow dung with swine manure on biogas yield and biodegradability. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-020-01115-z
Periyasamy S, Venkatachalam S, Ramasamy S, Srinivasan V (2009) Production of bio-ethanol from sugar molasses using Saccharomyces cerevisiae. Mod Appl Sci 3(8):32–37. https://doi.org/10.3390/ijms9091621
Ramaraj R, Unpaprom Y (2019a) Optimization of pretreatment condition for ethanol production from Cyperus difformis by response surface methodology. 3 Biotech 9:218. https://doi.org/10.1007/s13205-019-1754-0
Ramaraj R, Unpaprom Y (2019b) Enzymatic hydrolysis of small-flowered nutsedge (Cyperus difformis) with alkaline pretreatment for bioethanol production. Maejo Int J Sci Technol 13(2):110–120
Ranjitha J, Vijayalakshmi S, Vijaya Kumar P, Nitin Ralph P (2014) Production of bio-gas from flowers and vegetable wastes using anaerobic digestion. Int J Res Eng Technol 3:279–283
Sabharwal S, Sudan S, Ranjan V (2013) Jasminum sambac linn (motia): a review. Int J Pharm Bio Sci 2(5):108–130
Sophanodorn K, Unpaprom Y, Whangchai K, Duangsuphasin A, Manmai N, Ramaraj R (2020) A biorefinery approach for the production of bioethanol from alkaline-pretreated, enzymatically hydrolyzed Nicotiana tabacum stalks as feedstock for the bio-based industry. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-020-01177-z
Taherzadeh MJ, Karimi K (2008) Pretreatment of lignocellulosic wastes to improve ethanol and biogas production: a review. Int J Mol Sci 9:1621–1651. https://doi.org/10.3390/ijms9091621
Unpaprom Y, Ramaraj R, Whangchai K (2017) A newly isolated green alga, Scenedesmus acuminatus, from Thailand with efficient hydrogen production. Chiang Mai J Sci 44:1270–1278
Vu PT, Unpaprom Y, Ramaraj R (2017) Evaluation of bioethanol production from rice field weed biomass. Emerg Life Sci Res 3:42–49. https://doi.org/10.7324/ELSR.2017.324249
Vu PT, Unpaprom Y, Ramaraj R (2018) Impact and significance of alkaline-oxidant pretreatment on the enzymatic digestibility of Sphenoclea zeylanica for bioethanol production. Bioresour Technol 247:125–130. https://doi.org/10.1016/j.biortech.2017.09.012
Wannapokin A, Ramaraj R, Unpaprom Y (2017) An investigation of biogas production potential from fallen teak leaves (Tectona grandis). Emer Life Sci Res 3:1–10. https://doi.org/10.7324/ELSR.2017.31110
Wannapokin A, Ramaraj R, Whangchai K (2018) Unpaprom Y (2018) Potential improvement of biogas production from fallen teak leaves with co-digestion of microalgae. 3 Biotech. 8(2):1–8. https://doi.org/10.1007/s13205-018-1084-7
Wolff D (2006) Nectar sugar composition and volumes of 47 species of Gentianales from a southern Ecuadorian Montane forest. Ann Bot 97(5):767–777. https://doi.org/10.1093/aob/mcl033
Yadav I, Juneja SK, Chauhan S (2015) Temple waste utilization and management: a review. Int J Eng Technol Sci Res 2:14–19
Acknowledgements
The authors would like to thank the program in Biotechology, School of Renewable Energy, Energy Research center, Maejo University, Thailand for providing the necessary facilities to complete the present work. The authors would also like to thank Mrs. Sawitree Tipanee for the laboratory management of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Rights and permissions
About this article
Cite this article
Khammee, P., Unpaprom, Y., Chaichompoo, C. et al. Appropriateness of waste jasmine flower for bioethanol conversion with enzymatic hydrolysis: sustainable development on green fuel production. 3 Biotech 11, 216 (2021). https://doi.org/10.1007/s13205-021-02776-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02776-x